Your browser does not fully support modern features. Please upgrade for a smoother experience.

Submitted Successfully!
Thank you for your contribution! You can also upload a video entry or images related to this topic.
For video creation, please contact our Academic Video Service.
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Reem Khan | + 1755 word(s) | 1755 | 2021-05-18 08:04:05 |
Video Upload Options
We provide professional Academic Video Service to translate complex research into visually appealing presentations. Would you like to try it?
Cite
If you have any further questions, please contact Encyclopedia Editorial Office.
Khan, R. Electrochemical Biosensor. Encyclopedia. Available online: https://encyclopedia.pub/entry/12330 (accessed on 23 May 2026).
Khan R. Electrochemical Biosensor. Encyclopedia. Available at: https://encyclopedia.pub/entry/12330. Accessed May 23, 2026.
Khan, Reem. "Electrochemical Biosensor" Encyclopedia, https://encyclopedia.pub/entry/12330 (accessed May 23, 2026).
Khan, R. (2021, July 22). Electrochemical Biosensor. In Encyclopedia. https://encyclopedia.pub/entry/12330
Khan, Reem. "Electrochemical Biosensor." Encyclopedia. Web. 22 July, 2021.
Copy Citation
Electrochemical Biosensor can be used to detect low concentrations of analytes.
2D nanostructures
hierarchical structure
MXenes
graphene
biosensors
field analysis
TMDs
1. Introduction
The development of practical biosensors that can detect low concentrations of analytes inexpensively and rapidly has been the subject of intensive investigations with the goal to advance measurement sciences in fields such as clinical diagnostic, environmental monitoring, and food safety [1][2]. The success of biosensors as measurement tools relies on achieving the robustness and accuracy necessary to compete with conventional analytical tools for field analysis. The key requirement is to design an optimum sensing surface that can stabilize biological recognition molecules and be interfaced with physical transducers that convert the biorecognition into a quantifiable signal [3]. Therefore, the selection of an appropriate sensing material possessing both recognition and transduction functions is essential in achieving the needed performance. Varieties of materials have been used to create this interface including different forms of carbon, metals, and metal oxides [4]. Nanostructured materials introduced more than a decade ago have been combined with biological reagents to integrate the optical, electronic, and catalytic properties of nanomaterials with the biorecognition capabilities of the bio-entities, resulting in improved performance [5][6]. In most conventional designs, nanostructured materials are deposited in monolayered configurations, which limit the surface to the immobilization of a few biomolecules. The recent development of layered and precisely designed hierarchical nanostructures provides opportunities for creating multiscale structures with controlled functions and improved electrical, optical, and mechanical properties.
Research into elemental layered structures can be traced back to the 1930 with the pioneering work of Langmuir, who laid the foundations of surface science. Since then, surface scientists have studied the formation and properties of a large number of layered entities, some of which have shown potential in the sensing field [7]. To be used in sensing, these materials should possess high electronic, catalytic, and mechanical properties; they should be biocompatible and amenable to functionalization with biological molecules. In most cases, the as-synthesized structures need additional activation to generate the functional groups needed to attach biomolecules for achieving selective recognition and sensing. In some cases, the layered materials have the desired mechanical stability but lack sufficient electronic, optical, or surface properties necessary for field functional devices. Therefore, they have been interfaced with materials possessing these functions, creating a variety of more complex hybrid structures. The physicochemical properties and applications of hierarchical nanostructures in various fields have been reported in several recent reviews [8][9][10]; here, we focus primarily on their uses in the biosensing field.
Layered or multidimensional nanomaterials have gained significant attention in the biosensing field due to their high surface area, functionalized surface, and quantum size effect [11]. Their implementation in the biosensing field requires modification in order to impart selectivity and sensitivity for the detection of analytes within useful limits [12]. Other requirements are related to the challenge of scalable production of the structures themselves and the integration of sensing functions into the actual device [13]. The multifunctionality of 2D nanomaterials provides characteristics such as tunable structure and a large number of active sites along with an ultra-thin planar structure [14] that can be beneficial for detection and transduction. This ultra-thin assembly makes 2D nanomaterials more sensitive to external perturbations [15], and therefore, higher detection sensitivities can be achieved. Two-dimensional (2D) layered and multidimensional nanomaterials are commonly prepared by Top–Down Liquid Phase Exfoliation (TDLPE) or by Bottom–Up Surfactant Direct Growth (BUSD) methods where they can easily restack and lead to the formation of dense platforms [16]. To prevent uncontrollable restacking or aggregation, which can affect the biosensor’s performance, hierarchical structural architectures [17] can be designed by incorporating other dimensional entities [18] in between the layers. The resulting hybrid structures show not only the inherent characteristics of the parent 2D material but also functions such as porous structures with bulk void spaces for bioimmobilization or added catalytic sites for enhanced transduction [16]. A good hierarchical organization is expected to be mechanically adjustable and stable, permeable, having good pore volume, low structural density, and allow fast mass transport [19]. Thus, the ability to control these parameters is essential when using these materials for the design of field applicable biosensors.
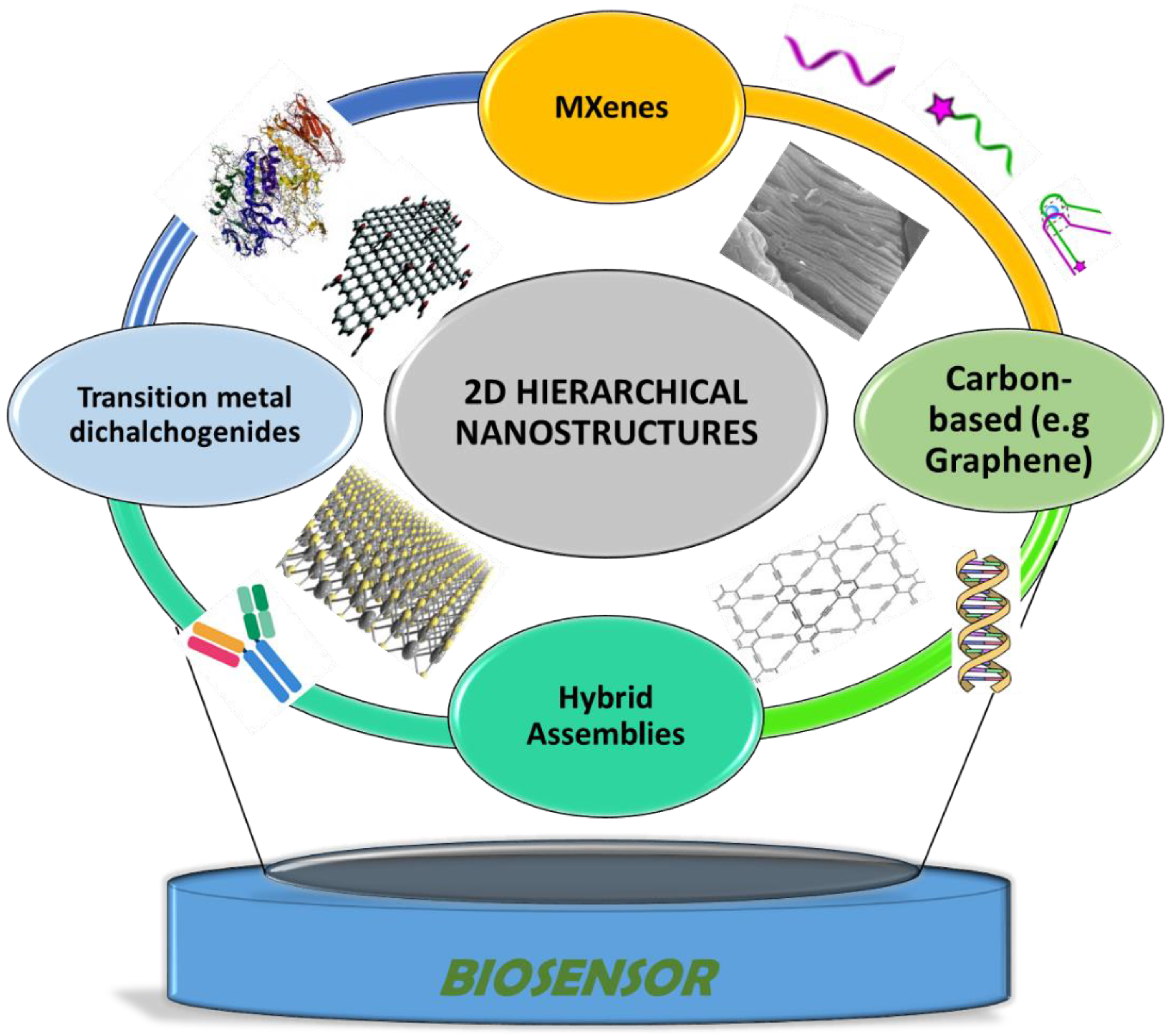 Figure 1. Hierarchical nanostructures used as electrode materials and bioimmobilization support for the construction of electrochemical biosensors discussed in this paper.
Figure 1. Hierarchical nanostructures used as electrode materials and bioimmobilization support for the construction of electrochemical biosensors discussed in this paper.2. 2D-Based Biosensors for Wearable Devices
An emerging area of growth that can benefit from the implementation of 2D nanostructures is the development of portable and wearable biosensing devices in healthcare. These innovative platforms involve the integration of the biosensing components with wearables and require specific designs to achieve flexibility and wearability to the user for attaining health monitoring. The development of wearable and flexible bioelectronic devices is one of the most exciting and promising directions in the biosensing field. These devices are designed with the goal of complementing the capabilities of existing wearable sensors that are currently limited to tracking physical activities and vital signs. Significant effort is dedicated to developing devices that can monitor disease biomarkers, for example, for non-invasive monitoring glucose and lactate levels in sweat.
Due to their electronic and mechanical properties, 2D materials have the potential to serve as platforms for creating bending and flexible bioelectronic sensors that can be attached to skin or textiles. The recent advancements in nanopatterning and printing can facilitate the manufacturing of inexpensive components that integrate 2D nanomaterials and interface them with biomolecules for wearables. Recently, a MXene-based wearable biosensor was reported for the multi-component analysis of human sweat [20]. The developed flexible biosensor patch can be worn on the wrist with replaceable sensors. The biosensor was able to monitor the pH, glucose, and lactate levels in sweat. The replacement of the sensor is an effective strategy to prolong the service life of the biosensor patch and to deal with the unavoidable enzyme inactivation problem. Figure 10 provides an overview of the fabrication and working principle of the developed sensor patch. As shown in the figure, the patch has a flexible skin conforming design in which all three sensors (pH, glucose, and lactate) were placed in separate, replaceable compartments. Therefore, once any of the three sensors stopped working, it can easily be replaced with a new chip. The sensing chip is based on a Ti3C2 MXene Prussian blue (PB) composite with and the corresponding enzyme (GOx or lactase). The performance of this sensor was demonstrated by analyzing the level of glucose and lactate in artificial sweat. Later, the sensor patch was further tested on human subjects. The sensor displayed satisfactory performance with high selectivity for the targeted analytes. The idea behind the sensing patch has the potential to generate marketable products that can be used for the noninvasive detection of biomarkers at early stages.
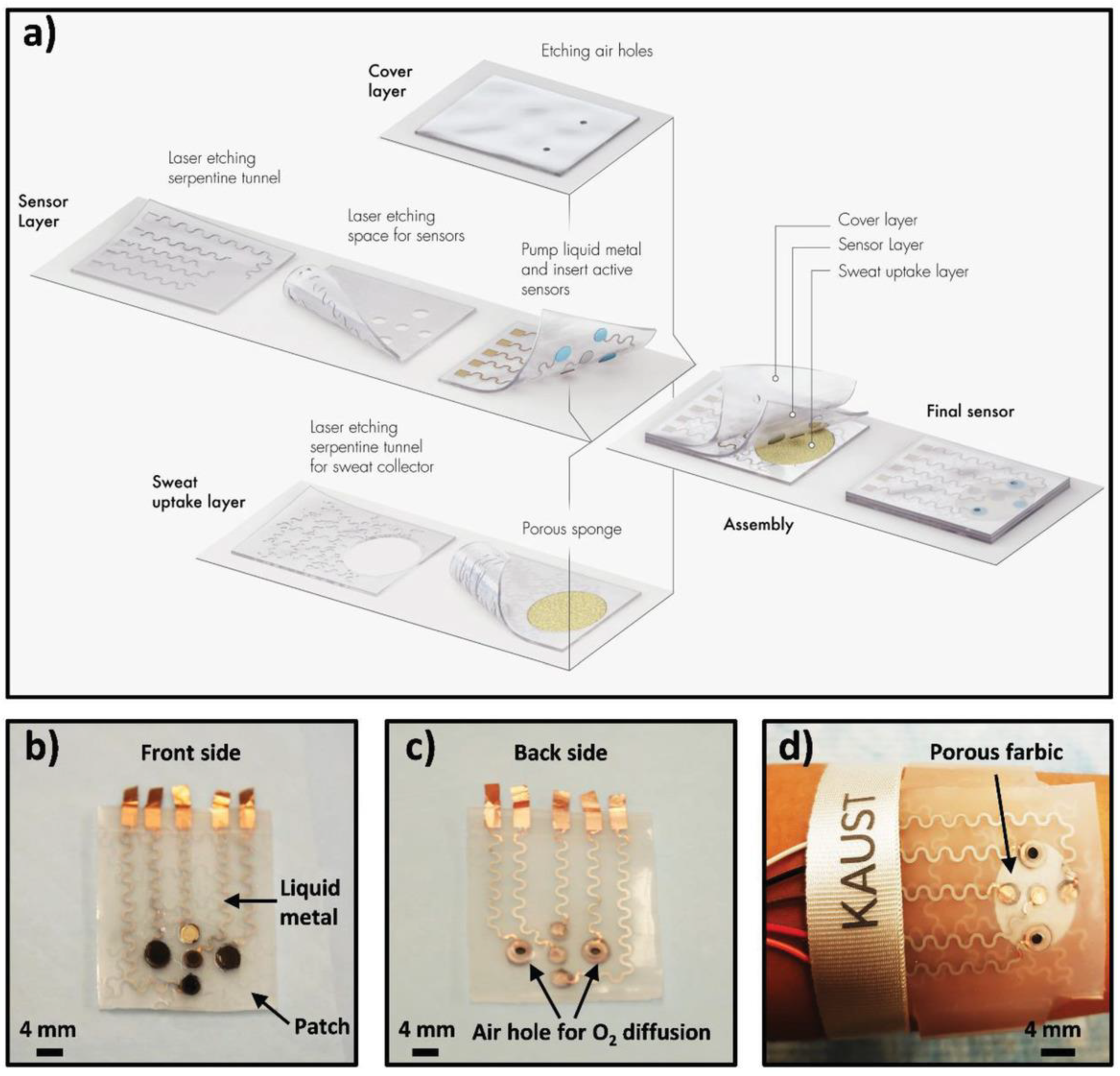 Figure 10. Schematic drawings and corresponding images of the wearable sweat sensor (a) components of the sensing system including a sweat-uptake layer, a sensor layer, and a cover layer. (b) Front side of the patch, (c) Back side of the sensor, (d) Images of the sensor wristband laminated on human skin. Reprinted with permission from [20].
Figure 10. Schematic drawings and corresponding images of the wearable sweat sensor (a) components of the sensing system including a sweat-uptake layer, a sensor layer, and a cover layer. (b) Front side of the patch, (c) Back side of the sensor, (d) Images of the sensor wristband laminated on human skin. Reprinted with permission from [20].3. Conclusions and Future Outlook
In summary, 2D materials and hybrid configurations show potential as candidate platforms for the development of next-generation electrochemical biosensing devices, including flexible and wearable user-interactive sensors. This paper provided an overview of the 2D nanostructures, their properties, and their applicability as electrode materials, modifiers, and support for the immobilization of biomolecules. Several examples of applications in the food, clinical, and environmental fields have been discussed and their performances summarized in extensive application-related tables. As compared to previously developed nanomaterials, engineered 2D nanostructures provide some advanced capabilities such as a single-layered or stacked layered structure providing a very large surface area, organized and tailorable morphology, flexibility in design, and superior conductivity. Their surface can be decorated with other materials, such as Au or Pt NPs, and their terminal groups can be easily functionalized with biomolecules, creating hybrid structures with the enhanced electrical, catalytic, or optical performance and detection capabilities for targeted biomolecular recognition. These physicochemical properties of 2D nanostructures make them a suitable candidate for biosensing, particularly for the rapidly growing flexible and wearable devices.
In addition to the properties of 2D nanomaterials discussed here, there are still some challenges that need to be addressed to exploit their full potential and achieve the practical realization of biosensing devices. Most demonstration of sensing capabilities of the 2D nanomaterials is still restricted to lab-based research. In order to move this concept to the market, the simple integration of the hierarchical materials with the biomolecules and measuring of the signal is not sufficient. Following the initial concept, there is a need to further develop and integrate the biosensing system with electronic circuitry and data analysis tools, deploy, and fully validate these devices in realistic environments. The scalable fabrication and large-scale manufacturing of these devices also require developing methods that will enable the automatic production of these nano assemblies and their deposition on flexible supports through approaches such as printing. In addition to the deposition of the 2D nanostructure or the in situ growth of the nanostructure directly on the sensing platform, efforts should be dedicated to the immobilization of the bioreceptor, e.g., enzymes onto the printed layers to incorporate both the transducer and the biomolecule components for biorecognition. Improvements in manufacturing and device fabrication are needed to lower fabrication costs, improve reproducibility, and facilitate large-scale production of units, which are all essential steps toward commercialization. In addition, issues related to connectivity with smartphones, Bluetooth, and other widely used communication devices, and data transmission features are needed to develop a user-interactive communication and data analysis. Further, special attention should be paid to the mechanical deformation of the wearable sensors and how this affects the performance over time. Moreover, such systems still need to be studied to address the consideration of strain tolerance of not only the 2D nanomaterial but their interactions with the surrounding environment. Second to the structural modifications, the environmental stability of the developed 2D materials and methodologies to mitigate their degradability must be taken into account. Third, developing structures with predictable and rationally designed characteristics should be sought rather than pursuing exploratory trials to find the ideal configurations. For example, how the properties of 2D nanomaterials affect performance of the sensing device should be systematically studied, and a structure–property relationship should be developed through combined theoretical and experimental investigations. Such developments require an integrated interdisciplinary approach and close collaborations of analytical chemists, materials scientists, biochemists, physicists, electrical engineering experts, and practitioners in the fields of use. These efforts can lead to portable diagnostic devices that are particularly useful for field analysis and widely distributed low-cost screening.
References
- Lou, Z.; Wang, L.; Jiang, K.; Wei, Z.; Shen, G. Reviews of wearable healthcare systems: Materials, devices and system integration. Mater. Sci. Eng. R Rep. 2020, 140, 100523.
- Wang, L.; Chen, D.; Jiang, K.; Shen, G. New insights and perspectives into biological materials for flexible electronics. Chem. Soc. Rev. 2017, 46, 6764–6815.
- Wang, L.; Lou, Z.; Shen, G. 2D Nanomaterials with Hierarchical Architecture for Flexible Sensor Application. ACS Symp. Ser. 2020, 1353, 93–116.
- Keshavarz, M.; Chowdhury, A.R.H.; Kassanos, P.; Tan, B.; Venkatakrishnan, K. Self-assembled N-doped Q-dot carbon nanostructures as a SERS-active biosensor with selective therapeutic functionality. Sens. Actuators B Chem. 2020, 323, 128703.
- Othman, A.; Karimi, A.; Andreescu, S. Functional nanostructures for enzyme based biosensors: Properties, fabrication and applications. J. Mater. Chem. B 2016, 4, 7178–7203.
- Khan, R.; Sherazi, T.A.; Catanante, G.; Rasheed, S.; Marty, J.L.; Hayat, A. Switchable fluorescence sensor toward PAT via CA-MWCNTs quenched aptamer-tagged carboxyfluorescein. Food Chem. 2020, 312, 126048.
- Yuhara, J.; He, B.; Matsunami, N.; Nakatake, M.; le Lay, G. Graphene’s Latest Cousin: Plumbene Epitaxial Growth on a “Nano WaterCube”. Adv. Mater. 2019, 31, 1901017.
- Wang, J.; Tang, J.; Ding, B.; Malgras, V.; Chang, Z.; Hao, X.; Wang, Y.; Dou, H.; Zhang, X.; Yamauchi, Y. Hierarchical porous carbons with layer-by-layer motif architectures from confined soft-template self-assembly in layered materials. Nat. Commun. 2017, 8, 15717.
- Lee, W.-K.; Odom, T.W. Designing Hierarchical Nanostructures from Conformable and Deformable Thin Materials. Acs Nano 2019, 13, 6170–6177.
- Egea, J.; Fabregat, I.; Frapart, Y.M.; Ghezzi, P.; Görlach, A.; Kietzmann, T.; Kubaichuk, K.; Knaus, U.G.; Lopez, M.G.; Olaso-Gonzalez, G.; et al. Corrigendum to European contribution to the study of ROS: A summary of the findings and prospects for the future from the COST action BM1203 (EU-ROS) [Redox Biol. 13 (2017) 94–162]. Redox Biol. 2018, 14, 694–696.
- Cheng, M.M.-C.; Cuda, G.; Bunimovich, Y.L.; Gaspari, M.; Heath, J.R.; Hill, H.D.; Mirkin, C.A.; Nijdam, A.J.; Terracciano, R.; Thundat, T. Nanotechnologies for biomolecular detection and medical diagnostics. Curr. Opin. Chem. Biol. 2006, 10, 11–19.
- Zhu, W.; Park, S.; Yogeesh, M.N.; McNicholas, K.M.; Bank, S.R.; Akinwande, D. Black Phosphorus Flexible Thin Film Transistors at Gighertz Frequencies. Nano Lett. 2016, 16, 2301–2306.
- Joshi, N.; Hayasaka, T.; Liu, Y.; Liu, H.; Oliveira, O.N.; Lin, L. A review on chemiresistive room temperature gas sensors based on metal oxide nanostructures, graphene and 2D transition metal dichalcogenides. Microchim. Acta 2018, 185, 213.
- Gan, X.; Zhao, H.; Wong, K.-Y.; Lei, D.Y.; Zhang, Y.; Quan, X. Covalent functionalization of MoS2 nanosheets synthesized by liquid phase exfoliation to construct electrochemical sensors for Cd (II) detection. Talanta 2018, 182, 38–48.
- Wang, K.; Lou, Z.; Wang, L.; Zhao, L.; Zhao, S.; Wang, D.; Han, W.; Jiang, K.; Shen, G. Bioinspired Interlocked Structure-Induced High Deformability for Two-Dimensional Titanium Carbide (MXene)/Natural Microcapsule-Based Flexible Pressure Sensors. Acs Nano 2019, 13, 9139–9147.
- Cheng, L.; Wang, X.; Gong, F.; Liu, T.; Liu, Z. 2D Nanomaterials for Cancer Theranostic Applications. Adv. Mater. 2020, 32, e1902333.
- Lou, Z.; Wang, L.; Jiang, K.; Shen, G. Programmable three-dimensional advanced materials based on nanostructures as building blocks for flexible sensors. Nano Today 2019, 26, 176–198.
- Yang, T.; Wang, W.; Huang, Y.; Jiang, X.; Zhao, X. Accurate Monitoring of Small Strain for Timbre Recognition via Ductile Fragmentation of Functionalized Graphene Multilayers. Acs Appl. Mater. Interfaces 2020, 12, 57352–57361.
- Bolotsky, A.; Butler, D.; Dong, C.; Gerace, K.; Glavin, N.R.; Muratore, C.; Robinson, J.A.; Ebrahimi, A. Two-Dimensional Materials in Biosensing and Healthcare: From In Vitro Diagnostics to Optogenetics and Beyond. Acs Nano 2019, 13, 9781–9810.
- Zhang, C.; Wang, X.; Hou, M.; Li, X.; Wu, X.; Ge, J. Immobilization on Metal–Organic Framework Engenders High Sensitivity for Enzymatic Electrochemical Detection. Acs Appl. Mater. Interfaces 2017, 9, 13831–13836.
More
Information
Subjects:
Materials Science, Biomaterials
Contributor
MDPI registered users' name will be linked to their SciProfiles pages. To register with us, please refer to https://encyclopedia.pub/register
:
View Times:
1.7K
Revision:
1 time
(View History)
Update Date:
22 Jul 2021
Notice
You are not a member of the advisory board for this topic. If you want to update advisory board member profile, please contact office@encyclopedia.pub.
OK
Confirm
Only members of the Encyclopedia advisory board for this topic are allowed to note entries. Would you like to become an advisory board member of the Encyclopedia?
Yes
No
${ textCharacter }/${ maxCharacter }
Submit
Cancel
Back
Comments
${ item }
|
More
No more~
There is no comment~
${ textCharacter }/${ maxCharacter }
Submit
Cancel
${ selectedItem.replyTextCharacter }/${ selectedItem.replyMaxCharacter }
Submit
Cancel
Confirm
Are you sure to Delete?
Yes
No





