Your browser does not fully support modern features. Please upgrade for a smoother experience.

Submitted Successfully!
Thank you for your contribution! You can also upload a video entry or images related to this topic.
For video creation, please contact our Academic Video Service.
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Yasmeen Siddiqui | + 3760 word(s) | 3760 | 2021-06-28 08:30:42 | | | |
| 2 | Camila Xu | Meta information modification | 3760 | 2021-06-29 06:16:43 | | |
Video Upload Options
We provide professional Academic Video Service to translate complex research into visually appealing presentations. Would you like to try it?
Cite
If you have any further questions, please contact Encyclopedia Editorial Office.
Siddiqui, Y. Fusarium oxysporum f. sp. niveum. Encyclopedia. Available online: https://encyclopedia.pub/entry/11426 (accessed on 10 May 2026).
Siddiqui Y. Fusarium oxysporum f. sp. niveum. Encyclopedia. Available at: https://encyclopedia.pub/entry/11426. Accessed May 10, 2026.
Siddiqui, Yasmeen. "Fusarium oxysporum f. sp. niveum" Encyclopedia, https://encyclopedia.pub/entry/11426 (accessed May 10, 2026).
Siddiqui, Y. (2021, June 29). Fusarium oxysporum f. sp. niveum. In Encyclopedia. https://encyclopedia.pub/entry/11426
Siddiqui, Yasmeen. "Fusarium oxysporum f. sp. niveum." Encyclopedia. Web. 29 June, 2021.
Copy Citation
Fusarium oxysporum f. sp. niveum (Fon) is the causative agent of Fusarium wilt disease of watermelon; it is the most serious soil-borne pathogen around the globe.
biology
disease control
Fusarium oxysporum
molecular markers
variability
1. Introduction
Fusarium is a complex genus and the most diverged species in the Eumycota for its worldwide distribution, causing diseases in plants, animals, and humans as well as the profuse presence of non-pathogenic Fusarium in the natural ecosystem [1]. Fusarium oxysporum species complex (Fosc) is an economically devastating species of Fusarium and is globally dispersed in various habitats, along with indoors, soil, and marine environments [2][3]. It is a crucial ubiquitous soil-borne phylogenetic diversified fungus with a vast host range including horticultural and grain crops that cause diseases like wilt, rot, and damping-off [4][5]. Members of this species complex are not the only source of uncontrollable vascular wilt diseases in various plants, but also the source of contagious diseases in humans and create a serious challenge to food security and public health [6]. In terms of the economic importance of the fungus, the pathogen was ranked fifth among the top 10 plant pathogenic fungi [7]. This seed and soil-borne plant pathogen cause serious detrimental effects on contaminated transplants showing symptoms like chlorosis, necrosis, immature leaf fall, vascular system browning, and finally wilting, which causes tremendous yield reduction. Additionally, if infection occurs earlier or during the harvesting period, some of them can produce mycotoxins in agricultural products [8][9]. Cereals and other food grains can be contaminated by Fusarium toxins and causes many diseases like feed refusal syndromes in mammals, mouldy sweet potato toxicity, and poisoning in bean hulls and different other living organisms [10].
In the early 1880s, Smith identified the wilt disease of watermelon from South Carolina and Georgia after that of cotton [11][12]. Watermelon wilt fungus was termed Fusarium niveum by Smith (1899) and also suggested that it was a variety of Neocosmospora vasinfectum var. niveum. After that, Wollenweber and Reinking (1935) gave a new name of watermelon Fusarium wilt of F. bulbigenum var. niveum Woll [13]. Based on this classification, Leach and Currence (1938) reflected that the Fusarium of watermelon and melon are various forms of F. bulbigenum var. niveum (form 1 and 2, correspondingly). Previously, Hansford (1926) first proposed that all species in the Fusarium unit assembled as a sole species, F. oxysporum [13]. Due to extreme host specificity exhibited by numerous pathogenic isolates of F. oxysporum, finally, Snyder and Hansen (1940) restated that all species within the unit Elegans be reflected a sole species, F. oxysporum, and proposed specialized forms (i.e., forma specialis (f. sp.)) that can distinguish particular virulence to one host or another [13]. Accordingly, F. oxysporum f. sp. niveum is named from the watermelon wilt form 1 and F. oxysporum f. sp. melonis from the melon wilt form 2. After that, the concept of formae speciales achieved widespread recognition, which led to the grouping of 10 species into a single unit with many pathogenic formae speciales, and further physiological races were derived from F. sp. [14][15].
Fusarium wilt pathogen is one of the most widely studied and devastating soil-borne pathogens around the world with both saprophytic and pathogenic members [13][16]. Non-pathogenic and pathogenic F. oxysporum strains remain in the soil, but the pathogenic strain causes severe vascular wilt disease in more than 150 economically major agricultural crop species. The most important crops that are likely to be infected by vascular wilt disease are banana, tomato, melon, watermelon, and cotton [17]. In the Cucurbitaceae family, eight various f. sp. have been identified; among them, F. oxysporum f. sp. cucumerium (Foc; cucumber), F. oxysporum f. sp. niveum (Fon; watermelon), and F. oxysporum f. sp. melonis (Fom; melon) are enormously important. Out of this, Fon is the most destructive pathogen of watermelon around the world [18]. The pathogen is responsible for yield losses of around 30–80% or even more [19][20], and presently is a major hindrance in watermelon cultivation. Moreover, difficulties faced by plant pathologists include reliable identification of the causal agents of the disease according to the epidemiological related parameters such as severity, levels of species, formae speciales, pathovars, biovars, and races. The properties of these characteristics would foster appropriate and relevant control measures [21].
2. Disease Cycle and Epidemiology
The Fon is a predominant, monocyclic, soil-borne, worldwide diversified fungus including saprophytic and pathogenic entities [4][5]. Fon is a host-specific to watermelon, it even cannot intimately infect concomitant cucurbits crops such as cucumber and cantaloupe with some limitation, which has been studied in greenhouse conditions [22]. The microorganism dispersing media are soil, plant debris, farm machinery [23], and seeds [24] and are known to survive more than 15 years without host plants [25]. Water and contaminated farm equipment can spread the pathogens over short distances, and for extensive areas, the spread of the disease has to be through contaminated soil, seeds, or seedlings. Normally, once a region becomes contaminated, it persists so emphatically [26]. The fungus infects the tissues of the plants as a germinating spore, and the growing hyphae penetrate the plant tissue through the wounds or openings near the site of elongation for root hair [27]. The fungal hyphae eventually penetrate the vascular tissue and produce microconidia [27]. The microconidia are then released into the xylem, which travels upward with the water and begins to colonize the watermelon’s vascular tissue of the plant [28]. Afterwards, there is an infection of the watermelon plant by the pathogen frequently inhabited within it, which remains until death or decay, as presented in Figure 1. At the last stages of the disease, the fungus produces thick mats of white mycelia and plenteous macroconidia. Under stressful environmental situations, chlamydospores develop from the aforementioned pathogen structures and conjoin into the soil. Fusarium wilt disease is generally achieved by spreading chlamydospores, which is the primary way for the pathogen's survival [26]. Chlamydospores are the lowest manageable attribute of Fusarium wilt infection and can live for more than 10–15 years. Fusarium wilt does not spread from plant to plant within season due to the absence of spore production above the ground in the field. Martyn and Vakalounakis (2017) indicated that it could also be spread by seeds [29]. Fon was first isolated from infected seed in 1928. Since then, several researchers have confirmed the seed-borne nature of Fon. Still, the spreading mechanism of the seed-borne nature of watermelon seed is mostly undiscovered and infection rates are normally less than 5% [13]. Another mechanism for the survival of pathogens is in the plant debris and the establishment of non-host plants that are alive [26]. At the advanced stages of infection, permanent wilt and yield losses on watermelon could reach around 30–80% or even more [19][20]. It becomes worse in sandy soils with a temperature range of 77° to 80 °F and a pH range of 5.5–6.5. At the anatomical level, the colonization procedure of pathogenic Fusarium species has been described by several researchers [30][31]. Disease development and symptom expression of host plants depend on the colonization of vessels by the pathogen [32].
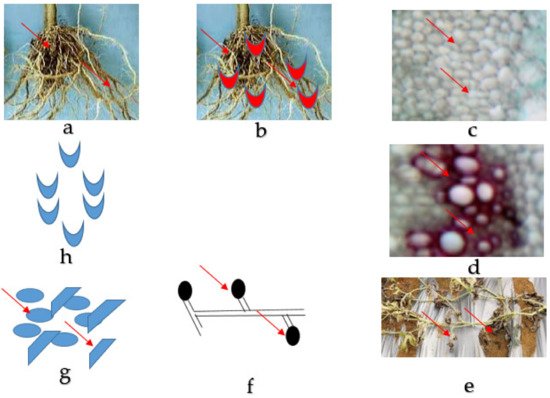
Figure 1. Fusarium wilt disease life cycle of watermelon caused by Fusarium oxysporum f. sp. niveum. (a) The healthy root of watermelon. (b) Germinating spore in contact with the root for penetration into the tissues. (c) Collapsed and distorted vessels in the xylem of the watermelon plant. (d) Gum produced by mycelia in the vessels. (e) The entire watermelon plant wilts and dies. (f) Spore formed by mycelia in the soil. (g) Micro and macroconidia present in the soil. (h) Germinating spore.
Initially, the infected watermelon plant showed symptoms like loss of pressure (turgor pressure) of vines and leaves, which could be recovered at night; exclusively single or some vines may be affected [33]. The progress of the disease in infected seedlings changed from dull green to yellow and finally necrotic [34]. When the fungus continues to colonize the xylem vessel, the plant forms more tyloses, finally limiting the water movement and the vine begins to wilt [26] (Figure 2).

Figure 2. Wilting disease symptoms of watermelon plants caused by Fusarium oxysporum f. sp. niveum under field condition. (a) Showing brown necrotic lesions at the base of the stem. (b) Showing a severe form of wilting, laterally the whole plant wilted and died. Pictures are from the personal collection of M. Z. Rahman collected during the field survey.
3. Races and Vegetative Compatibility Groups
Based on the aggressiveness of the pathogen or host cultivar’s resistance performance, Fon has been classified into four physiological races (0–3) (Table 1) [16][26]. Crall (1963) first reported Fon race 0 from Florida, USA, and stated that all modern varieties had resistant F0-1 gene to the race 0, so Fon race 0 loses its economic importance as a pathogen [13]. Fon race 1 is considered to be the widely prevalent race around the commercial watermelon-producing areas in the world. Race 1 was first identified by Smith (1894) from South Carolina, USA, and discrimination between races 0 and 1 could be more quantitative and not qualitative, and race 0 changed to race 1 based on aggressiveness [35]. At present, many diploid (seeded) and some triploid (seedless) cultivars have developed resistance toward Fon races 0 and 1 [13][26]. Later on, Fon race 2 was discovered in Israel and subsequently identified from the United States in 1981 [13]. The prevalence of race 2 was so high in some locations and showed more virulence than Fon race 1, which can infect commercial diploid seeded and triploid seedless varieties [22][36]. After 37 years later, another new race 3 was identified in Maryland, where it was reported to be the highest in terms of aggressiveness than races 0, 1, and 2 [16][37]. The main sources of Fon race 3 could be from contaminated seed or seedlings, selection, or mutation from races 0, 1, and 2 [16].
Table 1. Different watermelon varieties used to separate four races of Fon.
| Variety | Response of Disease | |||
|---|---|---|---|---|
| Race 0 | Race 1 | Race 2 | Race 3 | |
| All Sweet | R | R | S | S |
| Crimson sweet | R | S | S | S |
| Calhoun Gray | R | R | S | S |
| Sugar Baby | S | S | S | S |
| Black Diamond | S | S | S | S |
| Charleston Gray | R | S | S | S |
| PI-296341-FR | R | R | R | S |
R = Resistant, S = Susceptible.
Traditionally, F. oxysporum f. sp. niveum can be distinguished from other formae speciales and saprophytic strains of F. oxysporum only by its virulence on watermelon. Distinguishing races require the screening of pathogenic isolates on cultivars of varying levels of resistance. These tests are laborious and often inconsistent or inconclusive. Results can be greatly influenced by environmental factors, hosts age, inoculum level, and inoculation methods [38]. An alternative approach to the classification of strains of F. oxysporum is based on vegetative compatibility [39][40] where isolates are grouped into a specific phenotypic class [41]. Therefore, VCGs are useful for characterizing the genetic diversity within a formae speciales and, in some cases, for distinguishing pathogens from non-pathogens [42]
Genetic exchange and the sexual stage is absent in F. oxysporum, therefore it is limited to genetic transformation and the parasexual cycle. Asexual Fusarium oxysporum needs heterokaryosis for genetic exchange, which is regulated by a set of heterokaryon loci, and it helps in the formation of vegetative compatibility by the fusion of hypha and cell lysis [43]. Fusarium oxysporum with the capability to produce stable heterokaryon is in the same vegetative compatible group (VCG) or more likely genetically the same or clonal lineage [44][45].
Larkin et al. (1990) extensively studied 250 strains of Fon collected from five different states in the USA, Taiwan, and Australia and observed a significant correlation between vegetative compatibility group (VCG) and physiological races or virulence races. He reported three distinct VCGs (0080, 0081, and 0082) for Fon strains, in which race 1 and race 2 belong to VCG 0080 and VCG 0082, respectively [46], whereas VCG 0081 comprises only one Fon strain from Florida. He also demonstrated that the pathogenic strains of Fon were incompatible with non-pathogenic F. oxysporum; additionally, within the same race strains were compatible, but incompatibility was observed with the opposite race. Zhou and Everts (2007) described three VCGs: two were alike and earlier identified by Larkin et al. (1990) (i.e., VCG 0080 and VCG 0082), and the other one was distinct viz. VCG 0083 [47]. His results differed from those previously described by Larkin et al. (1990) and obtained an insignificant similarity between virulence race and VCG. VCG 0080 and VCG 0082 comprised all three race (0, 1, and 2) strains, and VCG 0083 consisted of only six isolates, which were classified as race 3 [16], but none of the strains was in VCG 0081. The newly identified race 3 is pathogenic to PI 296341-FR, previously stated to be resistant to race 2. Besides, race 3 isolates are compatible vegetatively with one another (VCG 0083) and incompatible with race 1 (VCG 0080) and race 2 (VCG 0082). Among these VCGs, VCG 0080 is considered the main diverged group followed by VCG 0082, VCG 0083, and 0081 distributed in a limited geographic area.
Globally collected Fon isolates were analyzed through mtDNA RFLP and the results indicated that no similarity was reported between geographic origin and race; RFLP was grouped into two common patterns that contained all three Fon races from various regions [48]. Some other forms of formae speciales (Table 2) like F. oxysporum f. spp. melonis [49], lycopersici [50], cubense [51], and asparagi [52] showed a complex relationship between genetic diversity and virulence. From these findings, it can be concluded that VCG and virulence (race or cross pathogenicity) were in a complex relationship among the strains of Fon [53]. Perhaps VCG cannot be used to differentiate the races of Fon; instead, it only helps to separate pathogenic strains from non-pathogenic strains of Fon and in the characterization of genetic variability among the Fon strains.
Table 2. Vegetative compatibility groups, formae speciales, and several races of Fo responsible for vascular wilt disease in many important crops.
| Host | Forma Specialis | VCG | Described Races | References |
|---|---|---|---|---|
| Watermelon | Niveum | 008- | 0, 1, 2, 3 | [11] |
| Melon | Melonis | 013- | 0, 1, 2, 1.2 Y. 1.2 W | [54] |
| Cucumber | Cucumerinum Radicis-cucumerinum |
018- 026- |
1, 2, 3 - |
[55] |
| Bitter gourd | Momordicae | - | - | [56] |
| Bottle gourd | lagenariae | 041- | - | [57] |
| Vegetable sponge | Luffae | - | - | [58] |
| Wax gourd | Benincasae | - | - | [13] |
| Tomato | lycopersici Radicis-lycopersici |
0030- 009- |
1, 2, 3 | [59] |
| Pisum sp | pisi | 007- | 1, 2, 5, 6 | [60] |
| Radish | raphani | 022- | - | [61] |
| Cotton | vasinfectum | 011- | 1, 2, 3, 4, 5, 6, 7, 8 | [62] |
| Banana | Cubense | 012- | 1, 2, 3, 4 | [63] |
| Bean | Phaseoli | 016- | 1, 2, 3, 4, 5, 6, 7 | [64] |
| Cabbage | conglutinans | 010- | 1, 2 | [65] |
| Chickpea | Ciceris | 028- | 1, 2, 3, 4 | [66] |
| Onion | Cepae | 0420- | - | [67][68] |
| Asparagus | Asparagi | 100- | - | [52] |
| Sweet potato | Batatas | 036- | 1, 2 | [69] |
| Sugar beet | Betae | 027- | - | [70] |
| Carnation | Dianthi | 002- | 1, 2, 3, 4 to 11 | [71] |
| Lettuce | Lactuace | 030- | 1, 2, 3 | [72] |
| Gladiolus | Gladioli | 034- | 2 | [73] |
| Tobacco | Nicotianae | 037- | 0, 1, 2, 3 | [74] |
| Alfalfa | Medicaginis | 004- | - | [75] |
| Potato | Tuberosi | 035- | - | [76] |
| Cyclamen | Cyclaminis | 015- | - | [77] |
| Chrysanthemum | Chrysanthemi | 005- | 1, 2, 3 | [78] |
4. Evolutionary Relationship between Races of Fon and VCGs
Restriction fragment length polymorphism (RFLP), isozyme analysis, analysis of intergenic spacer (IGS), and random amplified polymorphic DNA (RAPD) analysis were used to study genetic variability among several formae speciales of Fusarium oxysporum and demonstrated that isolates in the same VCG were similar in genetic makeup than the isolates in different VCG(s) [79]. The high similarities of DNA profiles among all three different races of Fon have been stated in previous works [48][80]. All these races were similar to mtDNA RFLP as well as some common sequences of chromosomal DNA. Generally, a mutation in the gene could cause a modification in developing a new race and its virulence. It was confirmed that the transformational mutagenesis of race 2 isolates of Fon caused a change to race 0 without a definite change in the VCG trait of that isolate [80]. Additionally, the recent instance of one race coming from the other race in a local community within the evolutionary lineage (VCG) was documented in Fo f. sp. lycopersici in California (USA) [81]. The spread of different races among the VCGs, especially the presence of race 2 in all three different VCGs, implies the probability of the development of a single VCG from the other because of the mutation in a vic gene. This mutation in the vic gene led to a modification from incompatibility to compatibility or vice versa and has been confirmed in some other fungi [82]. The mutation that alters the vegetative compatibility was also reported to describe the origin of two VCGs with similar virulence (race) in Fo f. sp. melonis [49]. The mtDNA polymorphism similarity between VCGs of Fo f. sp. niveum was discovered by Kim et al. (1992) and supported this model of evolution [48]. Finally, it can be concluded that the transformational mutagenesis of the Fon isolates could cause a change to race without a definite change in the VCG trait of that isolate.
5. Detection of Fon and Other Members of Formae Speciales
Rapid and precise diagnosis of the pathogen is a prerequisite for controlling disease and its management. Traditionally, researchers mainly depend on morphological study and molecular techniques [83]. Presently, disease assessment persists with the primary technique of discriminating host range and physiological races of an infective Fon isolate [13]. Differentiation of Fo races and formae speciales is habitually tested by using time-consuming and labour-intensive assessment of disease [84], therefore, formae-speciales-specific DNA sequencing for molecular screening technique is highly preferable [85].
To detect and identify various Fusarium wilt pathogens as well as the various studies among them, several reference genomes are available that can help in the diagnosis of plant pathogens rapidly, accurately, and cost-effectively [86]. Methods based on DNA polymorphisms, like amplified fragment length polymorphism (AFLP), simple sequence repeat (SSR), random amplified polymorphic DNA (RAPD), insertion-deletion (InDel), utilization of loop-mediated isothermal amplification (LAMP), single-nucleotide polymorphism (SNP), and using specific gene sequences like an internal transcribed spacer, β-tubulin, and calmodulin gene as well as elongation factor-1 alpha (EF-1α) have been reported to be reliable in this regard (Table 3) [87]. Diagnostic based genes like ribosomal intergenic spacer (IGS) or EF-1α help to differentiate between several species of fungi and sometimes subspecies separation, however, these may prove uncertain due to the presence of one or several clonal lineages of each forma specialis of F. oxysporum [86][88].
A close relationship in evolution was observed between the five formae speciales causing wilt disease of Cucurbitaceae based on mtDNA RFLPs and the isolates of a single forma specialis was grouped along with many other isolates, not of the same forma speciales, and it was observed that many formae speciales were formed in one branch. Within the five formae speciales, Fon appeared to be the most homogenous, but the most diverged group was F. f. sp. cucumerinum [48]. No polymorphisms were noticed among the 13 isolates of Fon including races 0, 1, and 2 collected from Israel and the USA using mtDNA RFLP analysis [89]. Additionally, the similarity was observed in the mtDNA RFLP map and the estimated size of the mtDNA between f. sp. melonis and F. f. sp. niveum (45.1 kb and 44.5 kb, respectively) [48][49].
Table 3. PCR primers used to detect F. oxysporum, F. oxysporum formae speciales, and their races.
| Primers Name | Target Organism | Target Gene | References |
|---|---|---|---|
| β-tubulin | F. oxysporum | β-tubulin (TUB2) rDNA region | [90] |
| EF1 and EF2 | F. oxysporum | Translation elongation factor1-α coding region | [91][92] |
| ITS1 and ITS4 | F. oxysporum | ITS region of rDNA | [93] |
| NMS1 and NMS2 | F. oxysporum | The mitochondrial small rRNA subunit (mtSSU) region |
[94] |
| FIGS11 and FIGS12 | F. oxysporum | The intergenic spacer (IGS) large rRNA subunit gene region | [95] |
| Cal228F and CAL2Rd | F. oxysporum | calmodulin (cmdA) | [96][97] |
| 7cF and 11aR | F. oxysporum | RNA polymerase II second largest subunit (rpb2) |
[98] |
| Uni F and UniR | F. oxysporum | The endo-polyglacturonase gene (Pg1) | [99] |
| Fon-1and Fon-2 | F. oxysporum f. sp. niveum | Derived from the random amplified polymorphic DNA (RAPD) fragment |
[100] |
| FONSIX6-F and FONSIX6-R | F. oxysporum f. sp. niveum race 2 | The SIX6 (secreted in xylemprotein 6) | [101] |
| FNR3-F and FNR3-R | F. oxysporum f. sp. niveum race 3 | Pathogenicity chromosome | [102] |
| P12-F2B and P12-R1 | F. oxysporum f. sp. Lycopersici | Secreted in xylem 1 (SIX1) | [103][104] |
| SIX2-F2 and SIX2-R2 | F. oxysporum f. sp. Lycopersici | Secreted in xylem 2 (SIX2) | [103] |
| SIX5-F1 and SIX5-R1 | F. oxysporum f. sp. Lycopersici | Secreted in xylem 5 (SIX5) | [88] |
| SIX4-F1 and SIX4-R1 | F. oxysporum f. sp. lycopersici race 1 | Secreted in xylem 4 (SIX4) | [88] |
| SIX3-F1 and SIX3-R2 | F. oxysporum f. sp. lycopersici race 2 | Secreted in xylem 3 (SIX3) | [103] |
| SIX6b_210_F and SIX6b_210_R | F. oxysporum f. sp. cubense (Foc) race 1 | Secreted in xylem 6 (SIX6) | [105] |
| SIX8b_206_F and SIX8b_206_R | Foc subtropical race 4 | Secreted in xylem 8 (SIX8) | [105] |
| SIX1a_266_F and SIX1a_266_2_R | Foc tropical race 4 (TR4) | Secreted in xylem 1 (SIX1) | [105] |
The most important point for diagnostics is the genes that can encode proteins similar to virulence [106]. In the case of Fo, which attacks the seedlings of a tomato plant, some of the proteins were identified in secreted-in xylem (SIX) and xylem sap [104][107]. The proteins or other sets of molecules (and small RNAs and secondary metabolites) have been reported to be associated with pathogenesis during disease progression and colonization and are widely called effectors [106][108]. SIX genes were present across the formae speciales and the profile of the genes was used to distinguish the distinct formae speciales, isolates, and races [85]. The SIX gene profiling was used to differentiate the three races in F. f. sp. lycopersici [88]. The SIX genes (SIX1, SIX9, SIX4, and SIX8) were also present in another species of F. oxysporum [109][110], and SIX6 was present in F. f. sp. vasinfectum [111], which infects Brassica and Arabidopsis [112]. Moreover, SIX6 and SIX1 were present in F. f. sp. betae [84]; SIX7, SIX10, and SIX1 were present in F. f. sp. lini and canariensis [113]; and SIX7, SIX8, and SIX1 were reported to be present in F. f. sp. cubense [114], while SIX5, SIX7, and SIX3 were present in F. f. sp. cepae [110]. Additionally, SIX4 plays an important role in the virulence of F. f. sp. conglutinans, which caused a yellowish colour in the cabbage plant [115]. A further difference between races 0, 1, 2, and 3 of Fon could be possible because of the recent studies that have focused specifically on an effector gene (SIX6), which plays a vital role in initiating R-protein-mediated immunity [101][102]. The gene (SIX6) was reported to be effective in Fon-1 isolates that are called FonSIX6, and it could be the reason to initiate a resistance in some genotypes of Fon-1-resistance, whereas Fon race 2 isolates do not have the gene (FonSIX6 effector gene), which resulted in an escape from the higher disease severity and immune system of the plant [101]. Besides, F. f. sp. cubense and F. f. sp. lycopersici can be differentiated from other groups of formae speciales using PCR primers that are designed to identify the specific SIX effector genes [109].
Species-specific primer Fn-1/Fn-2 were synthesized from ITS sequences for accurate and rapid identification of pathogenic F. f. sp. niveum [116]. Primer set Fn-1/Fn-2 amplified only a single PCR band around 320 bp from Fon, but the primers were unsuccessful to amplify the DNA of several other fungi and formae speciales f. sp. cucumerinum. In contrast, primer set FON-1/FON-2 could amplify a single 174 bp DNA fragment, which could differentiate Fon from the other formae speciales and Fusarium spp, but could not amplify the DNA of other formae speciales infecting cucurbits such as cucumerinum, melons, momordicae, and luffae [117]. Additionally, secreted in xylem protein 6 (SIX6) (i.e., avirulence gene) was identified in Fon races 0, 1, and 3, but absent in race 2. As a result, Fon race 2 was capable of differentiation by using primer set FONSIX6-F/FONSIX6-R [102]. Finally, Fon race 3 could be distinguished by using primer set FNR3-F/FNR3-R, which amplified the pathogenicity chromosome region (511 bp) of the Fon genome [102], as presented in Figure 3.
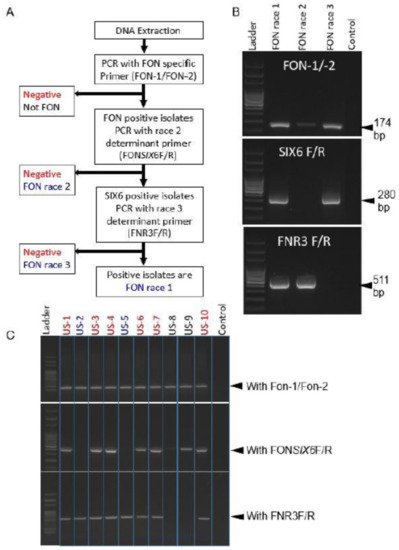
Figure 3. Flow diagram of three marker sets distinguishing Fon races 1, 2, and 3 (A); Example of differentiation of known Fon races (B); Identification of unknown Fon races 1, 2, and 3 (C). Source: [102].
References
- Gordon, T.R. Fusarium oxysporum and the Fusarium Wilt Syndrome. Annu. Rev. Phytopathol. 2017, 55, 23–39.
- Bell, B.P.; Khabbaz, R.F. Responding to the Outbreak of Invasive Fungal Infections. JAMA 2013, 309, 883.
- Brandt, M.E.; Park, B.J. Think Fungus—Prevention and Control of Fungal Infections. Emerg. Infect. Dis. 2013, 19, 1688–1689.
- Xiong, W.; Zhan, A. Testing clustering strategies for metabarcoding-based investigation of community-environment interactions. Mol. Ecol. Resour. 2018, 18, 1326–1338.
- LeBlanc, N.; Essarioui, A.; Kinkel, L.; Kistler, H.C. Phylogeny, Plant Species, and Plant Diversity Influence Carbon Use Phenotypes Among Fusarium Populations in the Rhizosphere Microbiome. Phytobiomes J. 2017, 1, 150–157.
- Zhang, Y.; Ma, L.-J. Deciphering Pathogenicity of Fusarium oxysporum from a Phylogenomics Perspective. Adv. Genet. 2017, 100, 179–209.
- Dean, R.; Van Kan, J.A.L.; Pretorius, Z.A.; Hammond-Kosack, K.E.; Di Pietro, A.; Spanu, P.; Rudd, J.; Dickman, M.; Kahmann, R.; Ellis, J.; et al. The Top 10 fungal pathogens in molecular plant pathology. Mol. Plant Pathol. 2012, 13, 414–430.
- Mudili, V.; Siddaih, C.N.; Nagesh, M.; Garapati, P.; Naveen Kumar, K.; Murali, H.S.; Yli Mattila, T.; Batra, H.V. Mould incidence and mycotoxin contamination in freshly harvested maize kernels originated from India. J. Sci. Food Agric. 2014, 94, 2674–2683.
- Chandra Nayaka, S.; Udaya Shankar, A.C.; Reddy, M.S.; Niranjana, S.R.; Prakash, H.S.; Shetty, H.S.; Mortensen, C.N. Control of Fusarium verticillioides, cause of ear rot of maize, by Pseudomonas fluorescens. Pest Manag. Sci. 2009, 65, 769–775.
- Kalagatur, N.K.; Kamasani, J.R.; Mudili, V. Assessment of Detoxification Efficacy of Irradiation on Zearalenone Mycotoxin in Various Fruit Juices by Response Surface Methodology and Elucidation of Its in-vitro Toxicity. Front. Microbiol. 2018, 9.
- Martyn, R.D. Fusarium wilt of watermelon: A historical review. In Proceedings of the Cucurbitaceae 2012, Xth EUCARPIA Meeting on Genetics and Breeding of Cucurbitaceae, Çukurova University, Antalya, Turkey, 15–18 October 2012.
- Ren, Y.; Jiao, D.; Gong, G.; Zhang, H.; Guo, S.; Zhang, J.; Xu, Y. Genetic analysis and chromosome mapping of resistance to Fusarium oxysporum f. sp. niveum (FON) race 1 and race 2 in watermelon (Citrullus lanatus L.). Mol. Breed. 2015, 35, 183.
- Martyn, R.D. Fusarium Wilt of Watermelon: 120 Years of Research. In Horticultural Reviews: Volume 42; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2014; pp. 349–442.
- Wamalwa, E.N.I.; Muoma, J.; Muyekho, F.N.; Wekesa, C.; Ajanga, S. Genetic Diversity of Fusarium oxysporum Races Associated with Cowpea Fields in Kakamega County. Fungal Genom. Biol. 2018, 8.
- Berrocal-Lobo, M.; Molina, A. Arabidopsis defense response against Fusarium oxysporum. Trends Plant Sci. 2008, 13, 145–150.
- Zhou, X.G.; Everts, K.L.; Bruton, B.D. Race 3, a New and Highly Virulent Race of Fusarium oxysporum f. sp. niveum Causing Fusarium Wilt in Watermelon. Plant Dis. 2010, 94, 92–98.
- Bertoldo, C.; Gilardi, G.; Spadaro, D.; Gullino, M.L.; Garibaldi, A. Genetic diversity and virulence of Italian strains of Fusarium oxysporum isolated from Eustoma grandiflorum. Eur. J. Plant Pathol. 2015, 141, 83–97.
- Keinath, A.P.; Hassell, R.L. Control of Fusarium Wilt of Watermelon by Grafting onto Bottlegourd or Interspecific Hybrid Squash Despite Colonization of Rootstocks by Fusarium. Plant Dis. 2014, 98, 255–266.
- Lü, G.; Guo, S.; Zhang, H.; Geng, L.; Song, F.; Fei, Z.; Xu, Y. Transcriptional profiling of watermelon during its incompatible interaction with Fusarium oxysporum f. sp. niveum. Eur. J. Plant Pathol. 2011, 131, 585–601.
- Martyn, R.D.; Netzer, D. Resistance to Races 0, 1, and 2 of Fusarium Wilt of Watermelon in Citrullus sp. PI-296341-FR. HortScience 1991, 26, 429–432.
- Strange, R.N.; Scott, P.R. Plant Disease: A Threat to Global Food Security. Annu. Rev. Phytopathol. 2005, 43, 83–116.
- Zhou, X.G.; Everts, K.L. Races and Inoculum Density of Fusarium oxysporum f. sp. niveum in Commercial Watermelon Fields in Maryland and Delaware. Plant Dis. 2003, 87, 692–698.
- Bruton, B.D.; Fish, W.W.; Zhou, X.G.; Everts, K.L.R.P. Fusarium wilt in seedless watermelons. In Proceedings of the 2007 Southeast Regional Vegetable Conference, Savannah, GA, USA, 5–7 January 2007; pp. 93–98.
- Boughalleb, N.; Mahjoub, M. El Frequency of Fusarium oxysporum F. sp. niveum and F. solani F. sp. Cucurbitae from Watermelon Seeds and Their Effect on Disease Incidence. Res. J. Parasitol. 2007, 2, 32–38.
- Zhang, M.; Xu, J.H.; Liu, G.; Yao, X.F.; Li, P.F.; Yang, X.P. Characterization of the watermelon seedling infection process by Fusarium oxysporum f. sp. niveum. Plant Pathol. 2015, 64, 1076–1084.
- Egel, D.S. and Martyn, R.D. Fusarium wilt of watermelon and other cucurbits. Plant Heal. Instr. 2013.
- Agrios, G. Plant Pathology, 5th ed.; Academic Press: Cambridge, MA, USA, 2005; ISBN 9780120445653/9780080473789.
- Di Pietro, A.; Madrid, M.P.; Caracuel, Z.; Delgado-Jarana, J.; Roncero, M.I.G. Fusarium oxysporum: Exploring the molecular arsenal of a vascular wilt fungus. Mol. Plant Pathol. 2003, 4, 315–325.
- Martyn, R.D.; Vakalounakis, D.J. Chapter 16: Fusarium Wilts of Greenhouse Cucurbits: Melon, Watermelon, and Cucumber. In Fusarium Wilts of Greenhouse Vegetable and Ornamental Crops; The American Phytopathological Society: St. Paul, MN, USA, 2017; pp. 159–174. ISBN 978-0-89054-482-2.
- Olivain, C.; Alabouvette, C. Process of tomato root colonization by a pathogenic strain of Fusarium oxysporum f. sp. lycopersici in comparison with a non-pathogenic strain. New Phytol. 1999, 141, 497–510.
- Zvirin, T.; Herman, R.; Brotman, Y.; Denisov, Y.; Belausov, E.; Freeman, S.; Perl-Treves, R. Differential colonization and defence responses of resistant and susceptible melon lines infected by Fusarium oxysporum race 1·2. Plant Pathol. 2010, 59, 576–585.
- Di, X.; Takken, F.L.W.; Tintor, N. How Phytohormones Shape Interactions between Plants and the Soil-Borne Fungus Fusarium oxysporum. Front. Plant Sci. 2016, 7.
- Kleczewski, N.M.; Egel, D.S. A Diagnostic Guide for Fusarium Wilt of Watermelon. Plant Health Prog. 2011, 12, 27.
- Keinath, A.P.; Wintermantel, W.M.; Zitter, T.A. (Eds.) Compendium of Cucurbit Diseases and Pests, 2nd ed.; The American Phytopathological Society: St. Paul, MN, USA, 2017; ISBN 978-0-89054-574-4.
- Martyn, R.D. Fusarium oxysporum f. sp. niveum Race 2: A Highly Aggressive Race New to the United States. Plant Dis. 1987, 71, 233.
- Petkar, A.; Harris-Shultz, K.; Wang, H.; Brewer, M.T.; Sumabat, L.; Ji, P. Genetic and phenotypic diversity of Fusarium oxysporum f. sp. niveum populations from watermelon in the southeastern United States. PLoS ONE 2019, 14, e0219821.
- Amaradasa, B.S.; Beckham, K.; Dufault, N.; Sanchez, T.; Ertek, T.S.; Iriarte, F.; Paret, M.; Ji, P. First Report of Fusarium oxysporum f. sp. niveum Race 3 Causing Wilt of Watermelon in Florida, U.S.A. Plant Dis. 2018, 102, 1029.
- Ploetz, R.C. Fusarium wilt of banana. Phytopathology 2015, 105, 1512–1521.
- Correll, J.C.; Klittich, C.J.R.; Leslie, J.F. Nitrate nonutilizing mutants of Fusarium oxysporum and their use in vegetative compatibility tests. Phytopathology 1987, 77, 1640–1646.
- Puhalla, J.E. Classification of strains of Fusarium oxysporum on the basis of vegetative compatibility. Can. J. Bot. 1985, 63, 179–183.
- Gordon, T.R.; Okamoto, D. Variation within and between populations of Fusarium oxysporum based on vegetative compatibility and mitochondrial DNA. Can. J. Bot. 1992, 70, 1211–1217.
- Lievens, B.; Rep, M.; Thomma, B.P.H.J. Recent developments in the molecular discrimination of formae speciales of Fusarium oxysporum. Pest Manag. Sci. Former. Pestic. Sci. 2008, 64, 781–788.
- Shahi, S.; Beerens, B.; Bosch, M.; Linmans, J.; Rep, M. Nuclear dynamics and genetic rearrangement in heterokaryotic colonies of Fusarium oxysporum. Fungal Genet. Biol. 2016, 91, 20–31.
- Kistler, H.C. Genetic diversity in the plant-pathogenic fungus Fusarium oxysporum. Phytopathology 1997, 87, 474–479.
- Strom, N.B.; Bushley, K.E. Two genomes are better than one: History, genetics, and biotechnological applications of fungal heterokaryons. Fungal Biol. Biotechnol. 2016, 3, 4.
- Larkin, R.P.; Hopkins, D.L.; Martin, F.N. Vegetative compatibility within Fusarium oxysporum f.sp. niveum and its relationship to virulence, aggressiveness, and race. Can. J. Microbiol. 1990, 36, 352–358.
- Everts, K.L.; Egel, D.S.; Langston, D.; Zhou, X.-G. Chemical management of Fusarium wilt of watermelon. Crop Prot. 2014, 66, 114–119.
- Kim, D.H.; Martyn, R.D.; Magill, C.W. Restriction fragment length polymorphism groups and physical map of mitochondrial DNA from Fusarium oxysporum f. sp. niveum. Phytopathology 1992, 82, 346–353.
- Jacobson, D.J.; Gordon, T.R. Fusarium oxysporum f. sp. melonis: A case study of diversity within a forma specialis. Phytopathology 1991, 81, 1064–1067.
- Elias, K.S.; Schneider, R.W. Vegetative compatibility groups in Fusarium oxysporum f. sp. lycopersici. Phytopathology 1991, 81, 159–162.
- Bentley, S.; Pegg, K.G.; Moore, N.Y.; Davis, R.D.; Buddenhagen, I.W. Genetic Variation Among Vegetative Compatibility Groups of Fusarium oxysporum f. sp. cubense Analyzed by DNA Fingerprinting. Phytopathology 1998, 88, 1283–1293.
- Elmer, W.H. Classification of Fusarium oxysporum f. sp. asparagi into Vegetatively Compatible Groups. Phytopathology 1989, 79, 88.
- Zhou, X.G.; Everts, K.L. Characterization of a Regional Population of Fusarium oxysporum f. sp. niveum by Race, Cross Pathogenicity, and Vegetative Compatibility. Phytopathology 2007, 97, 461–469.
- Risser, G. A Proposed Nomenclature of Fusarium oxysporum f. sp. melonis Races and Resistance Genes in Cucumis melo. Phytopathology 1976, 66, 1105.
- Vakalounakis, D.J. Allelism of the Fcu-1 and Foc genes conferring resistance to Fusarium wilt in cucumber. Eur. J. Plant Pathol. 1996, 102, 855–858.
- Sun, S.K. A New Fusarium Wilt of Bitter Gourd in Taiwan. Plant Dis. 1983, 67, 226.
- Matuo, T.; Yamamoto, I. On Fusarium oxysporum f. sp. lagenariae nf causing wilt of Lagenaria vulgaris var. hispida. Trans. Mycol. Soc. Japan 1967, 8, 61–63.
- Kawai, I.; Suzuki, H.; Kawai, K. On the pathogenicity of wilt Fusarium of the cucurbitaceous plants and their forms. Shizuoka Agric. Exp. Stn. Bull 1958, 3, 49–68.
- Inami, K.; Yoshioka-Akiyama, C.; Morita, Y.; Yamasaki, M.; Teraoka, T.; Arie, T. A genetic mechanism for emergence of races in Fusarium oxysporum f. sp. lycopersici: Inactivation of avirulence gene AVR1 by transposon insertion. PLoS ONE 2012, 7, e44101.
- Merzoug, A.; BeLABid, L.; Youcef-BenkAdA, M.; Benfreha, F.; Bayaa, B. Pea Fusarium wilt races in western Algeria. Plant Prot. Sci. 2014, 50, 70–77.
- Kim, H.; Hwang, S.-M.; Lee, J.H.; Oh, M.; Han, J.W.; Choi, G.J. Specific PCR detection of Fusarium oxysporum f. sp. raphani: A causal agent of Fusarium wilt on radish plants. Lett. Appl. Microbiol. 2017, 65, 133–140.
- Skovgaard, K.; Nirenberg, H.I.; O’Donnell, K.; Rosendahl, S. Evolution of Fusarium oxysporum f. sp. vasinfectum Races Inferred from Multigene Genealogies. Phytopathology 2001, 91, 1231–1237.
- Warman, N.M.; Aitken, E.A.B. The Movement of Fusarium oxysporum f.sp. cubense (Sub-Tropical Race 4) in Susceptible Cultivars of Banana. Front. Plant Sci. 2018, 9.
- Gutiérrez Salgado, A.; Gepts, P.; Debouck, D.G. Evidence for two gene pools of the Lima bean, Phaseolus lunatus L., in the Americas. Genet. Resour. Crop Evol. 1995, 42, 15–28.
- Liu, X.; Xing, M.; Kong, C.; Fang, Z.; Yang, L.; Zhang, Y.; Wang, Y.; Ling, J.; Yang, Y.; Lv, H. Genetic Diversity, Virulence, Race Profiling, and Comparative Genomic Analysis of the Fusarium oxysporum f. sp. conglutinans Strains Infecting Cabbages in China. Front. Microbiol. 2019, 10.
- Haware, M.P. Races of Fusarium oxysporum f. sp. ciceri. Plant Dis. 1982, 66, 809.
- Bayraktar, H. Genetic diversity and population structure of Fusarium oxysporum f. sp. cepae, the causal agent of Fusarium basal plate rot on onion, using RAPD markers. J. Agric. Sci. 2010, 16.
- Swift, C.E.; Wickliffe, E.R.; Schwartz, H.F. Vegetative Compatibility Groups of Fusarium oxysporum f. sp. cepae from Onion in Colorado. Plant Dis. 2002, 86, 606–610.
- Brayford, D. IMI descriptions of fungi and bacteria, set 112, nos 1111-1120. Mycopathologia 1992, 118, 37–64.
- Harveson, R.M.; Rush, C.M. Genetic Variation Among Fusarium oxysporum Isolates from Sugar Beet as Determined by Vegetative Compatibility. Plant Dis. 1997, 81, 85–88.
- Baayen, R.P.; Van Dreven, F.; Krijger, M.C.; Waalwijk, C. Genetic diversity in Fusarium oxysporum f. sp. dianthi and Fusarium redolens f. sp. dianthi. Eur. J. Plant Pathol. 1997, 103, 395–408.
- Pasquali, M.; Dematheis, F.; Gilardi, G.; Gullino, M.L.; Garibaldi, A. Vegetative Compatibility Groups of Fusarium oxysporum f. sp. lactucae from Lettuce. Plant Dis. 2005, 89, 237–240.
- Roebroeck, E.J.A.; Groen, N.P.A.; Mes, J.J. Detection of latent Fusarium oxysporum in gladiolus corms. Acta Hortic. 1990, 469–476.
- LaMondia, J.A. Fusarium wilt of tobacco. Crop Prot. 2015, 73, 73–77.
- Katan, T.; Di Primo, P. Current status of vegetative compatibility groups in Fusarium oxysporum: Supplement (1999). Phytoparasitica 1999, 27, 273–277.
- Venter, S.L.; Theron, D.J.; Steyn, P.J.; Ferreira, D.I.; Eicker, A. Relationship between vegetative compatibility and pathogenicity of isolates of Fusarium oxysporum f. sp. tuberosi from potato. Phytopathology 1992, 82, 858–862.
- Lori, G.A.; Petiet, P.M.; Malbrán, I.; Mourelos, C.A.; Wright, E.R.; Rivera, M.C. Fusarium wilt of cyclamen: Pathogenicity and vegetative compatibility groups structure of the pathogen in Argentina. Crop Prot. 2012, 36, 43–48.
- Matić, S.; Gilardi, G.; Gullino, M.L.; Garibaldi, A. Evidence for an expanded host range of Fusarium oxysporum f. sp. chrysanthemi. J. Plant Pathol. 2018, 100, 97–104.
- Chen, W.-Q.; Swart, W.J. Genetic Variation Among Fusarium oxysporum Isolates Associated with Root Rot of Amaranthus hybridus in South Africa. Plant Dis. 2001, 85, 1076–1080.
- Kim, D.H.; Martyn, R.D.; Magill, C.W. Chromosomal polymorphism in Fusarium oxysporum f. sp. niveum. Phytopathol. N. Y. Balt. St. Paul 1993, 83, 1209.
- Cai, G.; Gale, L.R.; Schneider, R.W.; Kistler, H.C.; Davis, R.M.; Elias, K.S.; Miyao, E.M. Origin of Race 3 of Fusarium oxysporum f. sp. lycopersici at a Single Site in California. Phytopathology 2003, 93, 1014–1022.
- Puhalla, J.E.; Spieth, P.T. Heterokaryosis in Fusarium moniliforme. Exp. Mycol. 1983, 7, 328–335.
- Bosland, P.W.; Williams, P.H. An evaluation of Fusarium oxysporum from crucifers based on pathogenicity, isozyme polymorphism, vegetative compatibility, and geographic origin. Can. J. Bot. 1987, 65, 2067–2073.
- Covey, P.A.; Kuwitzky, B.; Hanson, M.; Webb, K.M. Multilocus Analysis Using Putative Fungal Effectors to Describe a Population of Fusarium oxysporum from Sugar Beet. Phytopathology 2014, 104, 886–896.
- Van Dam, P.; Fokkens, L.; Schmidt, S.M.; Linmans, J.H.J.; Kistler, H.C.; Ma, L.-J.; Rep, M. Effector profiles distinguish formae speciales of Fusarium oxysporum. Environ. Microbiol. 2016, 18, 4087–4102.
- Haegi, A.; Catalano, V.; Luongo, L.; Vitale, S.; Scotton, M.; Ficcadenti, N.; Belisario, A. A Newly Developed Real-Time PCR Assay for Detection and Quantification of Fusarium oxysporum and Its Use in Compatible and Incompatible Interactions with Grafted Melon Genotypes. Phytopathology 2013, 103, 802–810.
- Xia, J.W.; Sandoval-Denis, M.; Crous, P.W.; Zhang, X.G.; Lombard, L. Numbers to names - restyling the Fusarium incarnatum-equiseti species complex. Persoonia Mol. Phylogeny Evol. Fungi 2019, 43, 186–221.
- Lievens, B.; Houterman, P.M.; Rep, M. Effector gene screening allows unambiguous identification of Fusarium oxysporum f. sp. lycopersici races and discrimination from other formae speciales. FEMS Microbiol. Lett. 2009, 300, 201–215.
- Kim, D.H.; Martyn, R.D.; Magill, C.W. Mitochondrial DNA restriction fragment length polymorphisms in Fusarium oxysporum f. sp. niveum. Phytoparasitica 1991, 19, 211–223.
- O’Donnell, K.; Cigelnik, E. Two Divergent Intragenomic rDNA ITS2 Types within a Monophyletic Lineage of the Fungus Fusarium Are Nonorthologous. Mol. Phylogenet. Evol. 1997, 7, 103–116.
- Geiser, D.M.; del Mar Jiménez-Gasco, M.; Kang, S.; Makalowska, I.; Veeraraghavan, N.; Ward, T.J.; Zhang, N.; Kuldau, G.A.; O’donnell, K. FUSARIUM-ID v. 1.0: A DNA sequence database for identifying Fusarium. Eur. J. Plant Pathol. 2004, 110, 473–479.
- O’Donnell, K.; Ward, T.J.; Robert, V.A.R.G.; Crous, P.W.; Geiser, D.M.; Kang, S. DNA sequence-based identification of Fusarium: Current status and future directions. Phytoparasitica 2015, 43, 583–595.
- White, T.J.; Bruns, T.; Lee, S.; Taylor, J. Amplification and direct sequencing of fungal ribosomal RNA genes for phylogenetics. PCR Protoc. Guid. Methods Appl. 1990, 18, 315–322.
- Li, K.N.; Rouse, D.I.; German, T.L. PCR primers that allow intergeneric differentiation of ascomycetes and their application to Verticillium spp. Appl. Environ. Microbiol. 1994, 60, 4324–4331.
- Appel, D.J.; Gordon, T.R. Relationships among pathogenic and nonpathogenic isolates of Fusarium oxysporum based on the partial sequence of the intergenic spacer region of the ribosomal DNA. MPMI Mol. Plant Microbe Interact. 1996, 9, 125–138.
- Carbone, I.; Kohn, L.M. A method for designing primer sets for speciation studies in filamentous ascomycetes. Mycologia 1999, 91, 553–556.
- Groenewald, J.Z.; Nakashima, C.; Nishikawa, J.; Shin, H.-D.; Park, J.-H.; Jama, A.N.; Groenewald, M.; Braun, U.; Crous, P.W. Species concepts in Cercospora: Spotting the weeds among the roses. Stud. Mycol. 2013, 75, 115–170.
- O’Donnell, K.; Sarver, B.A.J.; Brandt, M.; Chang, D.C.; Noble-Wang, J.; Park, B.J.; Sutton, D.A.; Benjamin, L.; Lindsley, M.; Padhye, A.; et al. Phylogenetic Diversity and Microsphere Array-Based Genotyping of Human Pathogenic Fusaria, Including Isolates from the Multistate Contact Lens-Associated U.S. Keratitis Outbreaks of 2005 and 2006. J. Clin. Microbiol. 2007, 45, 2235–2248.
- Hirano, Y.; Arie, T. PCR-based differentiation of Fusarium oxysporum ff. sp. lycopersici and radicis-lycopersici and races of F. oxysporum f. sp. lycopersici. J. Gen. Plant Pathol. 2006, 72, 273–283.
- Lin, Y.-H.; Chen, K.-S.; Chang, J.-Y.; Wan, Y.-L.; Hsu, C.-C.; Huang, J.-W.; Chang, P.-F.L. Development of the molecular methods for rapid detection and differentiation of Fusarium oxysporum and F. oxysporum f. sp. niveum in Taiwan. N. Biotechnol. 2010, 27, 409–418.
- Niu, J.; Arentshorst, M.; Seelinger, F.; Ram, A.F.J.; Ouedraogo, J.P. A set of isogenic auxotrophic strains for constructing multiple gene deletion mutants and parasexual crossings in Aspergillus niger. Arch. Microbiol. 2016, 198, 861–868.
- Hudson, O.; Waliullah, S.; Fulton, J.C.; Ji, P.; Dufault, N.S.; Keinath, A.; Ali, M.E. Marker Development for Differentiation of Fusarium oxysporum f. sp. Niveum Race 3 from Races 1 and 2. Int. J. Mol. Sci. 2021, 22, 822.
- Van Der Does, H.C.; Lievens, B.; Claes, L.; Houterman, P.M.; Cornelissen, B.J.C.; Rep, M. The presence of a virulence locus discriminates Fusarium oxysporum isolates causing tomato wilt from other isolates. Environ. Microbiol. 2008, 10, 1475–1485.
- Rep, M.; Van Der Does, H.C.; Meijer, M.; Van Wijk, R.; Houterman, P.M.; Dekker, H.L.; De Koster, C.G.; Cornelissen, B.J.C. A small, cysteine-rich protein secreted by Fusarium oxysporum during colonization of xylem vessels is required for I-3-mediated resistance in tomato. Mol. Microbiol. 2004, 53, 1373–1383.
- Carvalhais, L.C.; Henderson, J.; Rincon-Florez, V.A.; O’Dwyer, C.; Czislowski, E.; Aitken, E.A.B.; Drenth, A. Molecular Diagnostics of Banana Fusarium Wilt Targeting Secreted-in-Xylem Genes. Front. Plant Sci. 2019, 10.
- De Sain, M.; Rep, M. The Role of Pathogen-Secreted Proteins in Fungal Vascular Wilt Diseases. Int. J. Mol. Sci. 2015, 16, 23970–23993.
- Gawehns, F.; Houterman, P.M.; Ichou, F.A.; Michielse, C.B.; Hijdra, M.; Cornelissen, B.J.C.; Rep, M.; Takken, F.L.W. The Fusarium oxysporum Effector Six6 Contributes to Virulence and Suppresses I-2-Mediated Cell Death. Mol. Plant-Microbe Interact. 2014, 27, 336–348.
- Weiberg, A.; Wang, M.; Lin, F.-M.; Zhao, H.; Zhang, Z.; Kaloshian, I.; Huang, H.-D.; Jin, H. Fungal Small RNAs Suppress Plant Immunity by Hijacking Host RNA Interference Pathways. Science (80-) 2013, 342, 118–123.
- Fraser-Smith, S.; Czislowski, E.; Meldrum, R.A.; Zander, M.; O’Neill, W.; Balali, G.R.; Aitken, E.A.B. Sequence variation in the putative effector gene SIX8 facilitates molecular differentiation of Fusarium oxysporum f. sp. cubense. Plant Pathol. 2014, 63, 1044–1052.
- Sasaki, K.; Nakahara, K.; Tanaka, S.; Shigyo, M.; Ito, S. Genetic and Pathogenic Variability of Fusarium oxysporum f. sp. cepae Isolated from Onion and Welsh Onion in Japan. Phytopathology 2015, 105, 525–532.
- Chakrabarti, A.; Rep, M.; Wang, B.; Ashton, A.; Dodds, P.; Ellis, J. Variation in potential effector genes distinguishing Australian and non-Australian isolates of the cotton wilt pathogen Fusarium oxysporum f.sp. vasinfectum. Plant Pathol. 2011, 60, 232–243.
- Thatcher, L.F.; Gardiner, D.M.; Kazan, K.; Manners, J.M. A Highly Conserved Effector in Fusarium oxysporum Is Required for Full Virulence on Arabidopsis. Mol. Plant-Microbe Interact. 2012, 25, 180–190.
- Laurence, M.H.; Summerell, B.A.; Liew, E.C.Y. Fusarium oxysporum f. sp. canariensis: Evidence for horizontal gene transfer of putative pathogenicity genes. Plant Pathol. 2015, 64, 1068–1075.
- Meldrum, R.A.; Fraser-Smith, S.; Tran-Nguyen, L.T.T.; Daly, A.M.; Aitken, E.A.B. Presence of putative pathogenicity genes in isolates of Fusarium oxysporum f. sp. cubense from Australia. Australas. Plant Pathol. 2012, 41, 551–557.
- Kashiwa, T.; Inami, K.; Fujinaga, M.; Ogiso, H.; Yoshida, T.; Teraoka, T.; Arie, T. An avirulence gene homologue in the tomato wilt fungus Fusarium oxysporum f. sp. lycopersici race 1 functions as a virulence gene in the cabbage yellows fungus F. oxysporum f. sp. conglutinans. J. Gen. Plant Pathol. 2013, 79, 412–421.
- Zhang, Z.; Zhang, J.; Wang, Y.; Zheng, X. Molecular detection of Fusarium oxysporum f. sp. niveum and Mycosphaerella melonis in infected plant tissues and soil. FEMS Microbiol. Lett. 2005, 249, 39–47.
- Lin, Y.-H.; Chen, K.-S.; Liou, T.-D.; Huang, J.-W.; Chang, P.-F.L. Development of a molecular method for rapid differentiation of watermelon lines resistant to Fusarium oxysporum f. sp. niveum. Bot. Stud. 2009, 50, 273–280.
More
Information
Contributor
MDPI registered users' name will be linked to their SciProfiles pages. To register with us, please refer to https://encyclopedia.pub/register
:
View Times:
2.1K
Revisions:
2 times
(View History)
Update Date:
29 Jun 2021
Notice
You are not a member of the advisory board for this topic. If you want to update advisory board member profile, please contact office@encyclopedia.pub.
OK
Confirm
Only members of the Encyclopedia advisory board for this topic are allowed to note entries. Would you like to become an advisory board member of the Encyclopedia?
Yes
No
${ textCharacter }/${ maxCharacter }
Submit
Cancel
Back
Comments
${ item }
|
More
No more~
There is no comment~
${ textCharacter }/${ maxCharacter }
Submit
Cancel
${ selectedItem.replyTextCharacter }/${ selectedItem.replyMaxCharacter }
Submit
Cancel
Confirm
Are you sure to Delete?
Yes
No





