
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Andrew A. Gumbs | + 3567 word(s) | 3567 | 2021-06-21 11:49:51 | | | |
| 2 | Camila Xu | + 1382 word(s) | 4949 | 2021-06-22 10:19:07 | | |
Video Upload Options
Although there are currently no published RCTs on integrative medicine and pancreatic cancer, a significant amount of in vitro and animal studies, in addition, to case series and meta-analyses exist to help guide clinicians in making recommendations to patients with pancreatic cancer. When we use IM for our cancer patients often they are healing from complex digestive reconstructions and have to undergo adjuvant chemotherapy and sometimes radiation therapy before being completely recovered from major abdominal surgery. We have found that it is more realistic to not be too draconian with these patients.
1. Introduction
As opposed to other types of non-Western medicine, such as alternative medicine, complementary medicine, or holistic medicine, integrative medicine (IM) incorporates both Western and so-called Eastern or non-traditional medicine that has peer-reviewed published evidence done by medical doctors to back up its claims [1]. This difference is perhaps best appreciated by the fact that things like the Gerson diet and the Gonzalez regimen are not taught or advocated for in IM. This is because there is no adequate published literature to support the Gerson diet in the management of pancreatic cancer patients and there is peer-reviewed published research done by medical doctors that argue for worse outcomes in patients that are treated with the Gonzalez regimen as opposed to adjuvant chemotherapy [2].
Quite literally, IM “integrates” all “medicine” that is evidence-based. Because of this, IM completely supports and advocates for pancreatic resection and chemotherapy when indicated, whether neoadjuvant or adjuvant, for patients with pancreatic cancer. Despite the increasing public interest in IM, there is still a paucity of literature on IM recommendations for clinicians who deal with cancers. One of the other limitations is the difficulty in searching for specific IM recommendations because of the ubiquitous nature of the words “integrative” and “medicine”.
The growth of IM in the care of oncological patients is also hampered by surgeon’s reluctance to learn about this new field. Many patients with cancers that are principally cured through surgery are potentially being shepherded away from seeking IM consultation because of a palpable disdain of IM by their surgeons. In an effort to clarify what the actual evidence is, a review was done by a pancreatic surgeon who has also completed a 2-year IM Fellowship to better determine what if any peer-reviewed evidenced-based recommendations exist for patients to reduce their risk of developing recurrent pancreatic cancer.
2. Methods
A search was then done on Pubmed (https://pubmed.ncbi.nlm.nih.gov/, accessed on 25 May 2021) with the obtained words and combined with “pancreatic cancer/adenocarcinoma” for articles published over the last 10-years. All articles discussing complementary medicine (CM), functional medicine (FM), and holistic medicine were excluded. When available, this article reviewed the primary peer-reviewed published literature regarding recommendations for diet modification, lifestyle changes, and oral supplementation for patients with pancreatic cancer according to the principles of IM. The PRISMA checklist for abstract (Table 1) and manuscript design (Table 2) was used for the drafting of this manuscript [3].
| Section and Topic | Item # | Checklist Item | Reported (Yes/No) |
|---|---|---|---|
| Title | |||
| Title | 1 | Identify the report as a systematic review. | Y |
| Background | |||
| Objectives | 2 | Provide an explicit statement of the main objective(s) or question(s) the review addresses. | Y |
| Methods | |||
| Eligibility criteria | 3 | Specify the inclusion and exclusion criteria for the review. | Y |
| Information sources | 4 | Specify the information sources (e.g. databases, registers) used to identify studies and the date when each was last searched. | |
| Risk of bias | 5 | Specify the methods used to assess risk of bias in the included studies. | Y |
| Synthesis of results | 6 | Specify the methods used to present and synthesise results. | Y |
| Results | |||
| Included studies | 7 | Give the total number of included studies and participants and summarise relevant characteristics of studies. | Y |
| 8 | Present results for main outcomes, preferably indicating the number of included studies and participants for each. If meta-analysis was done, report the summary estimate and confidence/credible interval. If comparing groups, indicate the direction of the effect (i.e. which group is favoured). | Y | |
| Discussion | |||
| Limitations of evidence | 9 | Provide a brief summary of the limitations of the evidence included in the review (e.g. study risk of bias, inconsistency and imprecision). | Y |
| Interpretation | 10 | Provide a general interpretation of the results and important implications. | Y |
| Other | |||
| Funding | 11 | Specify the primary source of funding for the review. | Y |
| Registration | 12 | Provide the register name and registration number. | pending |
| Section and Topic | Item # | Checklist Item | Location Where Item Is Reported |
|---|---|---|---|
| Title | |||
| Title | 1 | Identify the report as a systematic review. | p.1 |
| Abstract | |||
| Abstract | 2 | See the PRISMA 2020 for Abstracts checklist. | Figure 2 |
| Introduction | |||
| Rationale | 3 | Describe the rationale for the review in the context of existing knowledge. | p.4 |
| Objectives | 4 | Provide an explicit statement of the objective(s) or question(s) the review addresses. | p. 4–5 |
| Methods | |||
| Eligibility criteria | 5 | Specify the inclusion and exclusion criteria for the review and how studies were grouped for the syntheses. | p. 5–6 |
| Information sources | 6 | Specify all databases, registers, websites, organisations, reference lists and other sources searched or consulted to identify studies. Specify the date when each source was last searched or consulted. | p. 5–6 |
| Search strategy | 7 | Present the full search strategies for all databases, registers and websites, including any filters and limits used. | p. 5–6 |
| Selection process | 8 | Specify the methods used to decide whether a study met the inclusion criteria of the review, including how many reviewers screened each record and each report retrieved, whether they worked independently, and if applicable, details of automation tools used in the process. | p. 5–6 |
| Data collection process | 9 | Specify the methods used to collect data from reports, including how many reviewers collected data from each report, whether they worked independently, any processes for obtaining or confirming data from study investigators, and if applicable, details of automation tools used in the process. | p. 5–6 |
| Data items | 10a | List and define all outcomes for which data were sought. Specify whether all results that were compatible with each outcome domain in each study were sought (e.g. for all measures, time points, analyses), and if not, the methods used to decide which results to collect. | p. 5–6 |
| 10b | List and define all other variables for which data were sought (e.g. participant and intervention characteristics, funding sources). Describe any assumptions made about any missing or unclear information. | p. 5–6 | |
| Study risk of bias assessment | 11 | Specify the methods used to assess risk of bias in the included studies, including details of the tool(s) used, how many reviewers assessed each study and whether they worked independently, and if applicable, details of automation tools used in the process. | p. 5–6 |
| Effect measures | 12 | Specify for each outcome the effect measure(s) (e.g. risk ratio, mean difference) used in the synthesis or presentation of results. | p. 5–6 |
| Synthesis methods | 13a | Describe the processes used to decide which studies were eligible for each synthesis (e.g. tabulating the study intervention characteristics and comparing against the planned groups for each synthesis (item #5)). | p. 5–6 |
| 13b | Describe any methods required to prepare the data for presentation or synthesis, such as handling of missing summary statistics, or data conversions. | p. 5–6 | |
| 13c | Describe any methods used to tabulate or visually display results of individual studies and syntheses. | p. 5–6 | |
| 13d | Describe any methods used to synthesize results and provide a rationale for the choice(s). If meta-analysis was performed, describe the model(s), method(s) to identify the presence and extent of statistical heterogeneity, and software package(s) used. | p. 5–6 | |
| 13e | Describe any methods used to explore possible causes of heterogeneity among study results (e.g. subgroup analysis, meta-regression). | p. 5–6 | |
| 13f | Describe any sensitivity analyses conducted to assess robustness of the synthesized results. | p. 5–6 | |
| Reporting bias assessment | 14 | Describe any methods used to assess risk of bias due to missing results in a synthesis (arising from reporting biases). | p. 5–6 |
| Certainty assessment | 15 | Describe any methods used to assess certainty (or confidence) in the body of evidence for an outcome. | p. 5–6 |
| Results | |||
| Study selection | 16a | Describe the results of the search and selection process, from the number of records identified in the search to the number of studies included in the review, ideally using a flow diagram. | p. 6–16 |
| 16b | Cite studies that might appear to meet the inclusion criteria, but which were excluded, and explain why they were excluded. | Figure 1 | |
| Study characteristics | 17 | Cite each included study and present its characteristics. | p. 6–16 |
| Risk of bias in studies | 18 | Present assessments of risk of bias for each included study. | p. 6–16 |
| Results of individual studies | 19 | For all outcomes, present, for each study: (a) summary statistics for each group (where appropriate) and (b) an effect estimate and its precision (e.g. confidence/credible interval), ideally using structured tables or plots. | p. 6–16 |
| Results of syntheses | 20a | For each synthesis, briefly summarise the characteristics and risk of bias among contributing studies. | p. 6–16 |
| 20b | Present results of all statistical syntheses conducted. If meta-analysis was done, present for each the summary estimate and its precision (e.g. confidence/credible interval) and measures of statistical heterogeneity. If comparing groups, describe the direction of the effect. | p. 6–16 | |
| 20c | Present results of all investigations of possible causes of heterogeneity among study results. | p. 6–16 | |
| 20d | Present results of all sensitivity analyses conducted to assess the robustness of the synthesized results. | p. 6–16 | |
| Reporting biases | 21 | Present assessments of risk of bias due to missing results (arising from reporting biases) for each synthesis assessed. | p. 6–16 |
| Certainty of evidence | 22 | Present assessments of certainty (or confidence) in the body of evidence for each outcome assessed. | p. 6–16 |
| Discussion | |||
| Discussion | 23a | Provide a general interpretation of the results in the context of other evidence. | p.16 |
| 23b | Discuss any limitations of the evidence included in the review. | p. 16 | |
| 23c | Discuss any limitations of the review processes used. | p. 16 | |
| 23d | Discuss implications of the results for practice, policy, and future research. | p. 16–24 | |
| Other Information | |||
| Registration and protocol | 24a | Provide registration information for the review, including register name and registration number, or state that the review was not registered. | pending |
| 24b | Indicate where the review protocol can be accessed, or state that a protocol was not prepared. | Prospero | |
| 24c | Describe and explain any amendments to information provided at registration or in the protocol. | NA | |
| Support | 25 | Describe sources of financial or non-financial support for the review, and the role of the funders or sponsors in the review. | p. 3 |
| Competing interests | 26 | Declare any competing interests of review authors. | NA |
| Availability of data, code and other materials | 27 | Report which of the following are publicly available and where they can be found: template data collection forms; data extracted from included studies; data used for all analyses; analytic code; any other materials used in the review. | Pending Prospero Registration |
Unlike IM, CM, and FM advocate for the use of pharmaceuticals such as non-steroidal anti-inflammatories for off-label uses in the belief that these medications can potentially reduce the risk of carcinogenesis. Furthermore, FM advocates for the use of intravenous vitamin therapy, because of this the review was restricted to peer-reviewed evidence for diet modification, lifestyle changes, and use of over the counter supplements that do not require a prescription or intravenous administration. Nonetheless, some evidence on the potential role of traditional pharmacologic agents was discussed to help guide the choice of medications to use when they are already indicated for recognized on-label uses. Furthermore, although therapies that did not meet the exclusion criteria may be mentioned for completeness sake, they will not be recommended.
Articles on traditional Chinese medicine and ayurvedic medicine were excluded because although IM Fellows are exposed to the basics of these practices, these therapies should only be administered by doctors licensed in these respective fields and not IM doctors. The search was then repeated on Google Scholar.
3. Results
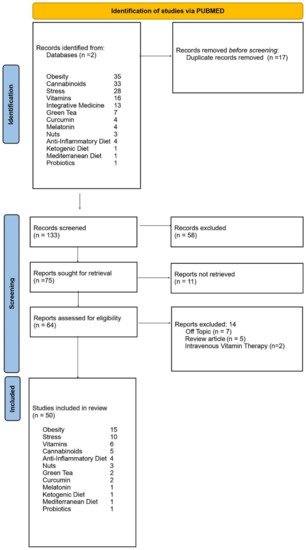
When a search for clinical trials, randomized controlled trials and meta-analyses was done for “pancreatic cancer” combined with “obesity”, “cannabinoids”, “stress”, “vitamins”, “integrative medicine”, “green tea”, “curcumin”, “melatonin”, “nuts”, “anti-inflammatory diet”, “ketogenic diet”, “Mediterranean diet”, and “probiotics” a total of 35; 33; 28; 16; 13; 7; 4; 4; 3; 2, 1, 1, and 1 articles were found, respectively (Figure 1) Clinical trials, randomized-controlled trials (RCT), meta-analyses, and case reports denote studies done on human subjects (Table 3). Clinical trials and RCTs were considered primary sources and meta-analyses and case reports considered secondary sources. When only studies older than 10 years old could be identified for a particular category, they were denoted in Table 3, but not Figure 1.
| Key Words | In Vitro | Animal Studies | Clinical Trials | RCTs | Meta-Analyses | Case Report |
|---|---|---|---|---|---|---|
| Obesity | x | x | ||||
| Stress | x | x | x | x (ongoing) |
x | |
| Vitamins | x | x | x | |||
| Cannabinoids | x | |||||
| Anti-Inflammatory Diet | x | x | ||||
| Nuts | x | |||||
| Green Tea | x * | x * | x | |||
| Curcumin | x | x | x | |||
| Melatonin | x | x | x * | |||
| Ketogenic Diet | x ^ | x ^ | ||||
| Mediterranean Diet | x | |||||
| Probiotics | x |
In the last 10 years, many articles have been published discussing the link between obesity and pancreatic carcinogenesis, and prior to this the first author even published 2 review articles on the topic [4][5]. Multiple clinical studies and meta-analyses have shown that in addition to obesity, body mass index (BMI), fasting glucose index, metabolic syndrome, high sweet beverage consumption, excess body weight, weight gain, and diabetes mellitus status all seem to be positively correlated with incidence of pancreatic cancer (Table 3) [6][7][8][9][10][11][12][13]. This pattern was also seen in an adolescent population in the Middle East, and adherence to a low fat diet seemed to be able to ameliorate this effect in post-menopausal women [14][15].
Once a patient has pancreatic adenocarcinoma, there does not seem to be a benefit to having a lower BMI, yet patients with a history of obesity do have a protective effect against developing pancreatic cancer after bariatric surgery [16][17]. In the European Prospective Investigation into Cancer and Nutrition (EPIC) trial where over 1100 patients were followed for 15 years, a healthy lifestyle was also found to have a protective effect against pancreatic cancer incidence [18]. Perhaps most compelling was the finding that a history of physical inactivity prior to being diagnosed with pancreatic adenocarcinoma was associated with an increased mortality rate and that obesity seems to confer a higher rate of chemotherapy resistance [19][20]. Clinical trials and meta-analyses regarding the link between obesity and the metabolic syndrome and pancreatic cancer, but no randomized-controlled trials exist.
The inflammatory effects of stress on carcinogenesis has been estimated to contribute to 30% of cancers with researchers even beginning to study the influence of problems related to the stress of working in this population [4][21]. The effects of depression in cancer patients has also been gaining importance over the years, with patients now routinely scheduled to see psychotherapists specialized in cancer patients after diagnosis. Depression has been shown to worsen quality of life in post-operative patients after surgery for pancreatic cancer and psychotherapy has been shown to be able to ameliorate the effects [22][23].
The role of stress has become such an important area of cancer research that a phase-II randomized controlled trial analyzing the potential benefit of pre-operative beta-blockade and cyclooxygenase inhibition to be continued 25 days post-operatively, called the PROSPER trial is currently underway in Germany (Table 3) [24]. The theory is that decreased stress mediators will be released during resection for cancers in the head of the pancreas. The plan is for 100 patients to be enrolled with the primary endpoints slated to be outcomes at 90 days [24].
Although conflicting reports have been published in other cancers, one study in vitro indicates that Fish Oil may have some preventative effects to carcinogenesis due to reduction in inflammation in vitro and in animal models [25]. In an effort to decrease inflammatory mediators, researchers have also studied several more traditional anti-inflammatory medications such as aspirin, sulindac, and even statins to ascertain whether or not they can influence the incidence of pancreatic cancer. In an animal model of mice given xenografts of pancreatic cancer cell lines, mice fed diets with sulindac, a non-steroidal anti-inflammatory drug, lived significantly longer than the control group [26]. Using the Surveillance, Epidemiology, and End Results (SEER) database, researchers identified 7813 patients with pancreatic ductal cancer and found a protective effect from pancreatic cancer in elderly patients on statin therapy [27].
A case-control study of patients in China with over 700 patients in each arm found a protective effect against pancreatic cancer for patients on aspirin [28]. This was again found to be the case in a meta-analysis of 12 studies of 4478 patients with pancreatic ductal cancer [29]. This is reiterated by the finding from a study of proton pump inhibitor (PPI) use that seems to lead to enhanced pancreatic cancer formation by almost 2-fold. Patients after duodenopancreatectomy are prone to marginal ulceration due to the gastro-intestinal reconstruction that is necessary after resection of the pancreatic head, as a result, it seems prudent to consider the H-2 receptor blocker over PPIs in patients that undergo this surgery for pancreatic cancer [30].
One found to look at Vitamin D levels and pancreatic cancer actually focused on milk consumption to see if dairy ingestion influenced the incidence of pancreatic cancer. A total of 14 studies with 2212 cases of pancreatic cancer out of over 862,000 people studied did not find conclusive evidence for dairy ingestion and pancreatic cancer risk [31]. On the contrary, a meta-analysis of 9 studies from the International Pancreatic Cancer Case–Control Consortium (PanC4) found a positive correlation with elevated serum vitamin D levels and pancreatic cancer [32]. For example, a major limitation in these studies is that sun exposure levels were not taken into consideration, a confounding factor that could easily and profoundly affect Vitamin D levels.
Another study that is difficult to interpret involved giving Vitamin E supplementation pre-operatively to patients scheduled to undergo pancreatectomy for pancreatic cancer [33]. In this phase I trial the safety of administering high doses of Vitamin E levels in 25 human patients with pancreatic ductal cancer was established, but without a control population, the conclusions that the authors attempt to draw are difficult to support. The authors state that apoptosis can be induced when Vitamin E given at doses ranging from 400-mg to 1600-mg daily are given, with 1 patient suffering diarrhea when the dose reached 3200 mg. Notably, an in vitro study has been published attempting to define the mechanism for this potential apoptotic effect [34].
Selenium levels initially appeared inversely related with the incidence of pancreatic cancer, but upon further analysis this was no longer found to be the case [35]. Two studies analyzing the effects of high dose intravenous Vitamin C have been published and suggest a potential survival benefit to patients with pancreatic cancer, but were excluded from analysis [36][37]. Although these 2 studies are from reputable authors, we cannot currently recommend these treatments outside of approved clinical trials.
Studies of resected pancreatic tumors have found a correlation between CB1 receptor activity and survival [38]. Subsequent studies on pancreatic cell lines treated with gemcitabine and cannabinoids subsequently found that autophagic cell death could be stimulated, and that cannabinoids could potentially make pancreatic cell lines more radio-sensitive [39][40]. Further studies also argue for an in vitro affect with cannabidiol (CBD) and tetrahydrocannabinol (THC) in its ability to inhibit pancreatic cell lines [41]. However, to date, no primary clinical evidence in humans exists to support the use of medical marijuana to improve survival in pancreatic cancer patients.
Although many studies have been done attempting to analyze the role of green tea in the incidence of pancreatic cancer, most studies are of questionable quality and not easily retrievable. Two meta-analyses looking at earlier data were able to be retrieved. One study analyzed 2317 cases from over 288,000 patients comprising 5 prospective studies and 3 case-controlled studies and did not conclusively find a correlation between incidence of pancreatic cancer and green tea consumption [42]. Another larger meta-analysis of 14 studies including almost 860,000 patients and over 8000 patients identified as having pancreatic cancer similarly found no correlation, however, this study did find a drop in the incidence of pancreatic cancer in Chinese patients aged over 60 [43].
In vitro and animal studies using MIA PaCa-2 cells suggest that Curcumin may inhibit angiogenesis and tumor growth in pancreatic cancer cells [44]. The pathway has been associated with a down regulation of NF-kB gene products and transcription factors. I/II study of 21 patients with gemcitabine-resistant pancreatic cancer was done and published 2 years earlier than this study, but it did not have a control arm and only evaluated safety of this treatment [45]. A notable point made by the authors is the relative ease with which patients can tolerate oral Curcumin supplementation with a median compliance rate reported as 100% (range = 79–100%).
A preclinical study from China using an animal model and xenografts with pancreatic cell MiaPaCa-2, found that melatonin supplementation may counter gemcitabine resistance. Furthermore, they identified NF-kB inhibition by melatonin administration in MiaPaCa-2, Panc-28, and AsPc-1 cell lines. They studied 5 mice in each arm and found that mice supplemented with melatonin had increased gemcitabine cytotoxicity and decreased invasion and cell proliferation resulting in smaller tumors [46]. The authors surmised that melatonin supplementation should be studied in patients with pancreatic cancer to see if it enhanced responsiveness to gemcitabine-based chemotherapy.
In a retrospective study of 25 patients with metastatic solid tumors treated with Tamoxifen and melatonin from 1996, the authors argue that this drug combination may have some potential survival benefit. Unfortunately, in this study, only 6 of the 25 patients had pancreatic cancer and survival ranged from 1–12+ months among them and they were not analyzed separately, as a result, the heterogenous nature of this study make any conclusions regarding the potential benefits of melatonin in patients with pancreatic cancer impossible. Furthermore, according to the design of this study, it is unclear whether melatonin alone or its combination with tamoxifen is necessary.
In perhaps an oversimplification of the complexities of diet, several large population-based studies have tried to ascertain the benefits if any of a diet rich in nut consumption with contradictory results. In the Nurses’ Health Study analyzing 75,680 women, researchers from Harvard University identified 466 females with pancreatic cancer and argued that a diet rich in nut consumption had an inverse relationship with the incidence of pancreatic cancer [47]. Culminating in the largest epidemiological study to look specifically at the role of nuts in pancreatic cancer incidence, the European Prospective Investigation into Cancer and Nutrition (EPIC) study, which identified 1283 patient with pancreatic ductal cancer in a population of over 400,000 people [48]. It is probable that dissecting diets out into specifics, such as level of nut consumption, is simply not useful and entire diets need to be studied.
In a study of 101,449 people aged from 52–78 followed for 8.5 years a total of 328 patients with pancreatic cancer were identified and no clear association with increased risk of pancreatic cancer risk was identified in patients determined to have a more pro-inflammatory diet when compared to an anti-inflammatory one [49]. Admittedly, the authors did acknowledge that results could have been affected by different follow-up times among the cohort. The study was then repeated by the same group after a median follow-up of 13.4 years with 2824 pancreatic cancers diagnosed, and the conclusion was the same [50]. Importantly, neither of these studies looked at patients already diagnosed with pancreatic cancer and only analyzed the risk of developing pancreatic cancer.
At the University of Iowa, mice with MIA PaCa-2 pancreatic cell line xenografts were fed a ketogenic diet and compared to a control group [51]. When mice were on a ketogenic diet and given ionizing radiation there appeared to be a significant survival benefit. As a result, a phase II trial was conducted, but only consisted of 2 patients with 1 patient dropping out of the trial due to the difficulty in maintaining the ketogenic diet. Although the animal model is interesting, the low number of patients enrolled in the trial and the difficulty in maintaining a ketogenic diet limit the ability to make recommendations regarding the efficacy of a ketogenic diet in patients with pancreatic cancer.
One paper analyzing 77,151 from Northern Sweden found a decreased incidence of pancreatic cancer in the population that adhered to a Mediterranean diet [52]. This protective benefit was quite small and appeared to be more related to alcohol consumption and resultant improved cardiovascular status.
These findings echoed the results of an Italian study of 20,000 cancer patients compared to 18,000 controls published in 2009 by a researcher in Milan who found that the Mediterranean diet had both a protective effect against cardiovascular disease, but also many cancers including pancreatic cancer. However, due to the fact that this study was more than 10 years old, it was not included inFigure 1[53].
An animal study published in 2020 found potential benefits to probiotic administration in inhibiting pancreatic cancer proliferation [54]. Tumors in mice that were treated with gemcitabine and probiotics had diminished pancreatic intraepithelial neoplasia (PanIN) changes and decreased expression of Ki-67 and vimentin when compared to mice treated with gemcitabine alone, indicating pancreatic carcinogenesis. Interestingly, these potential benefits were noted in mice that were treated with probiotics alone without the addition of gemcitabine. There currently are no published studies on humans with pancreatic cancer and probiotics.
4. Discussion
Although there are currently no published RCTs on integrative medicine and pancreatic cancer, a significant amount of in vitro and animal studies, in addition, to case series and meta-analyses exist to help guide clinicians in making recommendations to patients with pancreatic cancer. When we use IM for our cancer patients often they are healing from complex digestive reconstructions and have to undergo adjuvant chemotherapy and sometimes radiation therapy before being completely recovered from major abdominal surgery. We begin to introduce the principles of IM, but realize that diet and lifestyle modification may take months or even years to fully incorporate. The patient is provided with the information, and then we discuss adherence to recommendations on subsequent visits and review what the patient is doing well, and what perhaps needs more work.
Patients often ask about the Cornell China Study, which advocated for a Vegan diet. Although the New York Times termed the study the “Grand-Prix of epidemiology”, only 6500 people were actually enrolled in the study and this cohort was followed for 20 years. The main conclusions of the study were reported in a best-selling book and essentially advocate for a plant-based diet to reduce a host of chronic illnesses ranging from coronary artery disease, auto-immune illness, and even cancer, it was not possible to find many peer-reviewed journal articles discussing the findings of this study [55]. Regardless if it can be tolerated, a Vegan diet with Vitamin B12 supplementation certainly does not seem to be a problem [56].
Although one study in an animal model exists advocating for potential benefits of a ketogenic diet does exist, the absence of adequate human studies and difficulty in adhering to this regimen in patients already suffering from pancreatic cancer make recommending this diet difficult [51]. A ketogenic diet is essentially a high protein diet, which most people adhere to by eating large amounts of animal proteins, which goes against studies arguing for plant-based diets. If a ketogenic diet is to be followed, it seems like a plant-based version could be interesting, but potentially unrealistic.
In the Nurses’ health study, a prospective study on 75,680 women published in the New England Journal of Medicine in 2013, women who ate a 28 g of nuts, at least 2 times a week, were found to have a decreased risk of pancreatic cancer [57]. This could be due to better glycemic control, decreased ingestion of animal products or something inherent in the nuts [47]. Nuts have also long been known to reduce the risk of diabetes mellitus and cardiovascular disease, 2 factors that have a strong influence on mortality and overall survival.
Alcohol consumption has recently been found to increase the risk of developing many cancers. This was recently shown to be the case for pancreatic cancer in a European study published in 2018 [58]. In addition, alcohol consumption increases the risk for the development of pancreatitis, which could be an even more significant problem in a patient who has undergone pancreatic resection in the past. Alcohol consumption should be limited to no more than 1 glass of wine/day and ideally less or even complete abstinence.
Tobacco smoking is the only clear risk factor for the development of pancreatic cancer. Total abstinence is paramount for everybody, but in particular, patients with pancreatic cancer or risk factors for it. On the contrary, marijuana smoking itself does not have a known increased risk of developing pancreatic cancer. As a result, CBD oil with or without THC should probably be considered in patients with pancreatic cancer being treated with gemcitabine-based chemotherapy.
Although earlier exposure to pesticides and herbicides cannot be changed, patients are informed that they can stop any further exposure to these chemicals due to gardening or work in the outdoors. They are reminded to wash all produce thoroughly and to avoid pesticides and herbicides and to peel non-organic vegetables and fruits and, if economically feasible, buy organic. Additionally, they are instructed to preserve and cook all foods in glass containers instead of plastic, and reminded to never microwave food in plastic containers. For motivated patients, we also direct them to the Environmental Working Group website (www.EWG.org), which discussed endocrine disruptors and how to avoid them in their environment and body care products.
It is well-known that exercise is key to maintaining a normal BMI. The researchers found that people with pancreatic cancer tended to exercise less than people did not develop pancreatic cancer. Notably, the exercise had to be “leisure-time” exercise and not work-related exercise [59]. Indicating that stress reduction may also influence carcinogenesis and recurrence rates.
There are good data that mindful based stress reduction with a plant-based diet can improve quality of life, tumor markers and telomere length in a number of cancers [60][61][62][63]. John Kabat Zinn has wonderful work on Mindful Based Stress Reduction meditation. Belle Ruth Naprastek also has wonderful CD’s and tapes for stress reduction. Although pancreatic cancer in particular has not been studied yet, this modality certainly seems worthwhile.
References
- Gorski:, D.H.; Novella, S.P. Clinical trials of integrative medicine: Testing whether magic works? Trends Mol. Med. 2014, 20, 473–476.
- Chabot, J.A.; Tsai, W.-Y.; Fine, R.L.; Chen, C.; Kumah, C.K.; Antman, K.A.; Grann, V.R. Pancreatic Proteolytic Enzyme Therapy Compared With Gemcitabine-Based Chemotherapy for the Treatment of Pancreatic Cancer. J. Clin. Oncol. 2010, 28, 2058–2063.
- Page, M.J.; McKenzie, J.; Bossuyt, P.; Boutron, I.; Hoffmann, T.; Mulrow, C.D.; Shamseer, L.; Tetzlaff, J.M.; Akl, E.A.; Brennan, S.E.; et al. The PRISMA 2020 statement: An updated guideline for reporting systematic reviews. Br. Med J. 2021, 372, 105906.
- Gumbs, A.A. Obesity, Pancreatitis, and Pancreatic Cancer. Obes. Surg. 2008, 18, 1183–1187.
- Gumbs, A.A.; Bessler, M.; Milone, L.; Schrope, B.; Chabot, J. Contribution of obesity to pancreatic carcinogenesis. Surg. Obes. Relat. Dis. 2008, 4, 186–193.
- Navarrete-Muñoz, E.M.; Wark, A.P.; Romaguera, D.; Bhoo-Pathy, N.; Michaud, D.; Molina-Montes, E.; Tjonneland, A.; Olsen, A.; Overvad, K.; Boutron-Ruault, M.-C.; et al. Sweet-beverage consumption and risk of pancreatic cancer in the European Prospective Investigation into Cancer and Nutrition (EPIC). Am. J. Clin. Nutr. 2016, 104, 760–768.
- Zheng, Z.; Zheng, R.; He, Y.; Sun, X.; Wang, N.; Chen, T.; Chen, W. Risk Factors for Pancreatic Cancer in China: A Multicenter Case-Control Study. J. Epidemiol. 2016, 26, 64–70.
- Andersen, D.K.; Korc, M.; Petersen, G.M.; Eibl, G.; Li, D.; Rickels, M.; Chari, S.T.; Abbruzzese, J.L. Diabetes, Pancreatogenic Diabetes, and Pancreatic Cancer. Diabetes 2017, 66, 1103–1110.
- Carreras-Torres, R.; Johansson, M.; Gaborieau, V.; Haycock, P.C.; Wade, K.; Relton, C.L.; Martin, R.M.; Smith, G.D.; Brennan, P. The Role of Obesity, Type 2 Diabetes, and Metabolic Factors in Pancreatic Cancer: A Mendelian Randomization Study. J. Natl. Cancer Inst. 2017, 109.
- Gordon-Dseagu, V.; Devesa, S.S.; Goggins, M.; Stolzenberg-Solomon, R. Pancreatic cancer incidence trends: Evidence from the Surveillance, Epidemiology and End Results (SEER) population-based data. Int. J. Epidemiol. 2018, 47, 427–439.
- Da Silva, M.; Weiderpass, E.; Licaj, I.; Lissner, L.; Rylander, C. Excess body weight, weight gain and obesity-related cancer risk in women in Norway: The Norwegian Women and Cancer study. Br. J. Cancer 2018, 119, 646–656.
- Chung, K.M.; Singh, J.; Lawres, L.; Dorans, K.J.; Garcia, C.; Burkhardt, D.; Robbins, R.; Bhutkar, A.; Cardone, R.; Zhao, X.; et al. Endocrine-Exocrine Signaling Drives Obesity-Associated Pancreatic Ductal Adenocarcinoma. Cell 2020, 181, 832–847.e18.
- Park, S.K.; Oh, C.-M.; Kim, M.-H.; Ha, E.; Choi, Y.-S.; Ryoo, J.-H. Metabolic syndrome, metabolic components, and their relation to the risk of pancreatic cancer. Cancer 2020, 126, 1979–1986.
- Jiao, L.; Chen, L.; White, D.L.; Tinker, L.; Chlebowski, R.T.; Van Horn, L.V.; Richardson, P.; Lane, D.; Sangi-Haghpeykar, H.; El-Serag, H.B. Low-fat Dietary Pattern and Pancreatic Cancer Risk in the Women’s Health Initiative Dietary Modification Randomized Controlled Trial. J. Natl. Cancer Inst. 2018, 110, 49–56.
- Levi, Z.; Rottenberg, Y.; Twig, G.; Katz, L.; Leiba, A.; Derazne, E.; Tzur, D.; Eizenstein, S.; Keinan-Boker, L.; Afek, A.; et al. Adolescent overweight and obesity and the risk for pancreatic cancer among men and women: A nationwide study of 1.79 million Israeli adolescents. Cancer 2019, 125, 118–126.
- Jiang, Q.-L.; Wang, C.-F.; Tian, Y.-T.; Huang, H.; Zhang, S.-S.; Zhao, D.-B.; Ma, J.; Yuan, W.; Sun, Y.-M.; Che, X.; et al. Body mass index does not affect the survival of pancreatic cancer patients. World J. Gastroenterol. 2017, 23, 6287–6293.
- Schauer, D.P.; Feigelson, H.S.; Koebnick, C.; Caan, B.; Weinmann, S.; Leonard, A.C.; Powers, J.D.; Yenumula, P.R.; Arterburn, D.E. Bariatric Surgery and the Risk of Cancer in a Large Multisite Cohort. Ann. Surg. 2019, 269, 95–101.
- Naudin, S.; Viallon, V.; Hashim, D.; Freisling, H.; Jenab, M.; Weiderpass, E.; Perrier, F.; McKenzie, F.; Bueno-De-Mesquita, H.B.; Olsen, A.; et al. Healthy lifestyle and the risk of pancreatic cancer in the EPIC study. Eur. J. Epidemiol. 2019, 35, 1–12.
- Incio, J.; Liu, H.; Suboj, P.; Chin, S.M.; Chen, I.X.; Pinter, M.; Ng, M.R.; Nia, H.T.; Grahovac, J.; Kao, S.; et al. Obesity-Induced Inflammation and Desmoplasia Promote Pancreatic Cancer Progression and Resistance to Chemotherapy. Cancer Discov. 2016, 6, 852–869.
- Pratapwar, M.; Stenzel, A.E.; Joseph, J.M.; Fountzilas, C.; Etter, J.L.; Mongiovi, J.M.; Cannioto, R.; Moysich, K.B. Physical Inactivity and Pancreatic Cancer Mortality. J. Gastrointest. Cancer 2020, 51, 1088–1093.
- De Boer, A.G.E.M.; Bruinvels, D.; Tytgat, K.M.A.J.; Schoorlemmer, A.; Klinkenbijl, J.H.G.; Frings-Dresen, M.H.W. Employment status and work-related problems of gastrointestinal cancer patients at diagnosis: A cross-sectional study. BMJ Open 2011, 1, e000190.
- Ryu, J.S.; Choi, S.W.; Yun, S.S.; Koo, B.H.; Choi, I.S.; Kim, S.J.; Park, J.S.; Seok, J.-H.; Yoon, D.-S. Preliminary Findings on the Effectiveness of Meaning-Centered Psychotherapy in Patients with Pancreatobiliary Cancer. Yonsei Med. J. 2018, 59, 1107–1114.
- Sato, N.; Hasegawa, Y.; Saito, A.; Motoi, F.; Ariake, K.; Katayose, Y.; Nakagawa, K.; Kawaguchi, K.; Fukudo, S.; Unno, M.; et al. Association between chronological depressive changes and physical symptoms in postoperative pancreatic cancer patients. Biopsychosoc. Med. 2018, 12, 13.
- Hüttner, F.J.; Rooman, I.; Bouche, G.; Knebel, P.; Hüsing, J.; Mihaljevic, A.L.; Hackert, T.; Strobel, O.; Büchler, M.W.; Diener, M.K. Pancreatic resection with perioperative drug repurposing of propranolol and etodolac: Trial protocol of the phase-II randomised placebo controlled PROSPER trial. BMJ Open 2020, 10, e040406.
- Ding, Y.; Mullapudi, B.; Torres, C.; Mascariñas, E.; Mancinelli, G.; Diaz, A.M.; McKinney, R.; Barron, M.; Schultz, M.; Heiferman, M.; et al. Omega-3 Fatty Acids Prevent Early Pancreatic Carcinogenesis via Repression of the AKT Pathway. Nutrients 2018, 10, 1289.
- Li, H.; Yang, A.L.; Chung, Y.T.; Zhang, W.; Liao, J.; Yang, G.-Y. Sulindac inhibits pancreatic carcinogenesis in LSL-KrasG12D-LSL-Trp53R172H-Pdx-1-Cre mice via suppressing aldo-keto reductase family 1B10 (AKR1B10). Carcinogenesis 2013, 34, 2090–2098.
- Jeon, C.Y.; Pandol, S.J.; Wu, B.; Cook-Wiens, G.; Gottlieb, R.A.; Merz, C.N.B.; Goodman, M.T. The Association of Statin Use after Cancer Diagnosis with Survival in Pancreatic Cancer Patients: A SEER-Medicare Analysis. PLoS ONE 2015, 10, e0121783.
- Risch, H.A.; Lu, L.; Streicher, S.A.; Wang, J.; Zhang, W.; Ni, Q.; Kidd, M.S.; Yu, H.; Gao, Y.-T. Aspirin Use and Reduced Risk of Pancreatic Cancer. Cancer Epidemiol. Biomark. Prev. 2017, 26, 68–74.
- Sun, J.; Li, Y.; Liu, L.; Jiang, Z.; Liu, G. Aspirin use and pancreatic cancer risk. Medicine 2019, 98, e18033.
- Alkhushaym, N.; Almutairi, A.R.; AlThagafi, A.; Fallatah, S.B.; Oh, M.; Martin, J.R.; Babiker, H.M.; McBride, A.; Abraham, I. Exposure to proton pump inhibitors and risk of pancreatic cancer: A meta-analysis. Expert Opin. Drug Saf. 2020, 19, 327–334.
- Genkinger, J.M.; Wang, M.; Li, R.; Albanes, D.; Anderson, K.E.; Bernstein, L.; Brandt, P.V.D.; English, D.; Freudenheim, J.L.; Fuchs, C.S.; et al. Dairy products and pancreatic cancer risk: A pooled analysis of 14 cohort studies. Ann. Oncol. 2014, 25, 1106–1115.
- Waterhouse, M.; Risch, H.A.; Bosetti, C.; Anderson, K.E.; Petersen, G.M.; Bamlet, W.; Cotterchio, M.; Cleary, S.P.; Ibiebele, T.I.; La Vecchia, C.; et al. Vitamin D and pancreatic cancer: A pooled analysis from the Pancreatic Cancer Case—Control Consortium. Ann. Oncol. 2015, 26, 1776–1783.
- Springett, G.M.; Husain, K.; Neuger, A.; Centeno, B.; Chen, D.-T.; Hutchinson, T.Z.; Lush, R.M.; Sebti, S.; Malafa, M.P. A Phase I Safety, Pharmacokinetic, and Pharmacodynamic Presurgical Trial of Vitamin E δ-tocotrienol in Patients with Pancreatic Ductal Neoplasia. EBioMedicine 2015, 2, 1987–1995.
- Francois, R.; Zhang, A.; Husain, K.; Wang, C.; Hutchinson, S.; Kongnyuy, M.; Batra, S.K.; Coppola, D.; Sebti, S.M.; Malafa, M.P. Vitamin E δ-tocotrienol sensitizes human pancreatic cancer cells to TRAIL-induced apoptosis through proteasome-mediated down-regulation of c-FLIPs. Cancer Cell Int. 2019, 19, 189.
- Han, X.; Li, J.; Brasky, T.M.; Xun, P.; Stevens, J.; White, E.; Gammon, M.D.; He, K. Antioxidant intake and pancreatic cancer risk. Cancer 2012, 119, 1314–1320.
- Monti, D.A.; Mitchell, E.; Bazzan, A.J.; Littman, S.; Zabrecky, G.; Yeo, C.J.; Pillai, M.V.; Newberg, A.B.; Deshmukh, S.; Levine, M. Phase I Evaluation of Intravenous Ascorbic Acid in Combination with Gemcitabine and Erlotinib in Patients with Metastatic Pancreatic Cancer. PLoS ONE 2012, 7, e29794.
- Polireddy, K.; Dong, R.; Reed, G.; Yu, J.; Chen, P.; Williamson, S.; Violet, P.-C.; Pessetto, Z.; Godwin, A.K.; Fan, F.; et al. High Dose Parenteral Ascorbate Inhibited Pancreatic Cancer Growth and Metastasis: Mechanisms and a Phase I/IIa study. Sci. Rep. 2017, 7, 17188.
- Michalski, C.W.; Oti, F.E.; Erkan, M.; Sauliunaite, D.; Bergmann, F.; Pacher, P.; Batkai, S.; Müller, M.W.; Giese, N.A.; Friess, H.; et al. Cannabinoids in pancreatic cancer: Correlation with survival and pain. Int. J. Cancer 2008, 122, 742–750.
- Donadelli, M.; Dando, I.; Zaniboni, T.; Costanzo, C.; Pozza, E.D.; Scupoli, M.T.; Scarpa, A.; Zappavigna, S.; Marra, M.; Abbruzzese, A.; et al. Gemcitabine/cannabinoid combination triggers autophagy in pancreatic cancer cells through a ROS-mediated mechanism. Cell Death Dis. 2011, 2, e152.
- Yasmin-Karim, S.; Moreau, M.; Mueller, R.; Sinha, N.; Dabney, R.; Herman, A.; Ngwa, W. Enhancing the Therapeutic Efficacy of Cancer Treatment With Cannabinoids. Front. Oncol. 2018, 8, 114.
- Yang, Y.; Huynh, N.; Dumesny, C.; Wang, K.; He, H.; Nikfarjam, M. Cannabinoids Inhibited Pancreatic Cancer via P-21 Activated Kinase 1 Mediated Pathway. Int. J. Mol. Sci. 2020, 21, 8035.
- Zeng, J.-L.; Li, Z.-H.; Wang, Z.-C.; Zhang, H.-L. Green Tea Consumption and Risk of Pancreatic Cancer: A Meta-analysis. Nutrients 2014, 6, 4640–4650.
- Chen, K.; Zhang, Q.; Peng, M.; Shen, Y.; Wan, P.; Xie, G. Relationship between tea consumption and pancreatic cancer risk. Eur. J. Cancer Prev. 2014, 23, 353–360.
- Bimonte, S.; Barbieri, A.; Palma, G.; Luciano, A.; Rea, D.; Arra, C. Curcumin Inhibits Tumor Growth and Angiogenesis in an Orthotopic Mouse Model of Human Pancreatic Cancer. BioMed Res. Int. 2013, 2013, 1–8.
- Kanai, M.; Yoshimura, K.; Asada, M.; Imaizumi, A.; Suzuki, C.; Matsumoto, S.; Nishimura, T.; Mori, Y.; Masui, T.; Kawaguchi, Y.; et al. A phase I/II study of gemcitabine-based chemotherapy plus curcumin for patients with gemcitabine-resistant pancreatic cancer. Cancer Chemother. Pharmacol. 2010, 68, 157–164.
- Ju, H.Q.; Li, H.; Tian, T.; Lu, Y.X.; Bal, L.; Chen, L.Z.; Sheng, H.; Mo, H.Y.; Zeng, J.B.; Deng, W.; et al. Melatonin overcomes gemcitabine resistance in pancreatic ductal adenocarcinoma by abrogating nuclear factor-jB activation. J. Pineal Res. 2016, 60, 27–38.
- Bao, Y.; Hu, F.B.; Giovannucci, E.L.; Wolpin, B.M.; Stampfer, M.J.; Willett, W.C.; Fuchs, C.S. Nut consumption and risk of pancreatic cancer in women. Br. J. Cancer 2013, 109, 2911–2916.
- Obón-Santacana, M.; Luján-Barroso, L.; Freisling, H.; Naudin, S.; Boutron-Ruault, M.; Mancini, F.R.; Rebours, V.; Kühn, T.; Katzke, V.; Boeing, H.; et al. Consumption of nuts and seeds and pancreatic ductal adenocarcinoma risk in the European Prospective Investigation into Cancer and Nutrition. Int. J. Cancer 2020, 146, 76–84.
- Zheng, J.; Merchant, A.T.; Wirth, M.D.; Zhang, J.; Antwi, S.O.; Shoaibi, A.; Shivappa, N.; Stolzenberg-Solomon, R.Z.; Hebert, J.R.; Steck, S.E. Inflammatory potential of diet and risk of pancreatic cancer in the Prostate, Lung, Colorectal and Ovarian (PLCO) Cancer Screening Trial. Int. J. Cancer 2018, 142, 2461–2470.
- Zheng, J.; Wirth, M.D.; Merchant, A.T.; Zhang, J.; Shivappa, N.; Stolzenberg-Solomon, R.Z.; Hebert, J.R.; Steck, S.E. Inflammatory Potential of Diet, Inflammation-Related Lifestyle Factors, and Risk of Pancreatic Cancer: Results from the NIH-AARP Diet and Health Study. Cancer Epidemiol. Biomark. Prev. 2019, 28, 1266–1270.
- Zahra, A.; Fath, M.; Opat, E.; Mapuskar, K.A.; Bhatia, S.K.; Rodman, S.; Iii, S.N.R.; Snyders, T.P.; Chenard, C.A.; Eichenberger-Gilmore, J.M.; et al. Consuming a Ketogenic Diet while Receiving Radiation and Chemotherapy for Locally Advanced Lung Cancer and Pancreatic Cancer: The University of Iowa Experience of Two Phase 1 Clinical Trials. Radiat. Res. 2017, 187, 743–754.
- Tognon, G.; Nilsson, L.M.; Lissner, L.; Johansson, I.; Hallmans, G.; Lindahl, B.; Winkvist, A. The Mediterranean Diet Score and Mortality Are Inversely Associated in Adults Living in the Subarctic Region. J. Nutr. 2012, 142, 1547–1553.
- La Vecchia, C. Association between Mediterranean dietary patterns and cancer risk. Nutr. Rev. 2009, 67, S126–S129.
- Chen, S.-M.; Chieng, W.-W.; Huang, S.-W.; Hsu, L.-J.; Jan, M.-S. The synergistic tumor growth-inhibitory effect of probiotic Lactobacillus on transgenic mouse model of pancreatic cancer treated with gemcitabine. Sci. Rep. 2020, 10, 1–12.
- Campbell, T.; Parpia, B.; Chen, J. Diet, lifestyle, and the etiology of coronary artery disease: The Cornell China Study. Am. J. Cardiol. 1998, 82, 18–21.
- Selinger, E.; Kühn, T.; Procházková, M.; Anděl, M.; Gojda, J. Vitamin B12 Deficiency Is Prevalent Among Czech Vegans Who Do Not Use Vitamin B12 Supplements. Nutrients 2019, 11, 3019.
- Bao, Y.; Han, J.; Hu, F.B.; Giovannucci, E.L.; Stampfer, M.J.; Willett, W.C.; Fuchs, C.S. Association of Nut Consumption with Total and Cause-Specific Mortality. N. Engl. J. Med. 2013, 369, 2001–2011.
- Naudin, S.; Li, K.; Jaouen, T.; Assi, N.; Kyrø, C.; Tjønneland, A.; Overvad, K.; Boutron-Ruault, M.-C.; Rebours, V.; Védie, A.-L.; et al. Lifetime and baseline alcohol intakes and risk of pancreatic cancer in the European Prospective Investigation into Cancer and Nutrition study. Int. J. Cancer 2018, 143, 801–812.
- Kollarova, H.; Azeem, K.; Tomaskova, H.; Horakova, D.; Prochazka, V.; Martinek, A.; Shonova, O.; Sevcikova, J.; Sevcikova, V.; Janout, V. Is physical activity a protective factor against pancreatic cancer? Bratislavske Lekarske Listy 2014, 115, 474–478.
- Saxe, G.A.; Hebert, J.R.; Carmody, J.F.; Kabat-Zinn, J.; Rosenzweig, P.H.; Jarzobski, D.; Reed, G.W.; Blute, R.D. Can diet in conjunction with stress reduction affect the rate of increase in prostate specific antigen after biochemical recurrence of prostate cancer? J. Urol. 2001, 166, 2202–2207.
- Cramer, H.; Lauche, R.; Paul, A.; Dobos, G. Mindfulness-Based Stress Reduction for Breast Cancer—A Systematic Review and Meta-Analysis. Curr. Oncol. 2012, 19, 343–352.
- Lengacher, C.A.; Reich, R.R.; Kip, K.E.; Barta, M.; Ramesar, S.; Paterson, C.L.; Moscoso, M.S.; Carranza, I.; Budhrani, P.H.; Kim, S.J.; et al. Influence of mindfulness-based stress reduction (MBSR) on telomerase activity in women with breast cancer (BC). Biol. Res. Nurs. 2014, 16, 438–447.
- Carlson, L.; Rouleau, C.R.; Garland, S. The impact of mindfulness-based interventions on symptom burden, positive psychological outcomes, and biomarkers in cancer patients. Cancer Manag. Res. 2015, 7, 121–131.




