
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Xavier Paredes | + 1661 word(s) | 1661 | 2021-06-04 11:03:29 | | | |
| 2 | Enzi Gong | -46 word(s) | 1615 | 2021-06-17 11:45:13 | | | | |
| 3 | Enzi Gong | + 65 word(s) | 1680 | 2021-06-29 09:57:36 | | |
Video Upload Options
Ionic liquids (more accurately room-temperature ionic liquids (RTILs)) can be defined as materials composed of organic or inorganic cations (like imidazolium or pyridinium) and anions (e.g., nitrate, acetate, tetrafluoroborate, dicyanamide, bis(trifluoromethylsulfonyl)imide and lactate) that are liquid at or below 100 °C. The number of possible combinations of a cation and anion is estimated to reach 106, making it theoretically possible to synthesize an IL targeted for a specific application or property.
1. Overview
Ionic liquids (ILs) were initially hailed as a green alternative to traditional solvents because of their almost non-existent vapor pressure as ecological replacement of most common volatile solvents in industrial processes for their damaging effects on the environment. It is common knowledge that they are not as green as desired, and more thought must be put into the biological consequences of their industrial use. Still, compared to the amount of research studying their physicochemical properties and potential applications in different areas, there is a scarcity of scientific papers regarding how these substances interact with different organisms. The intent of this review was to compile the information published in this area since 2015 to allow the reader to better understand how, for example, bacteria, plants, fish, etc., react to the presence of this family of liquids. In general, lipophilicity is one of the main drivers of toxicity and thus the type of cation. The anion tends to play a minor (but not negligible) role, but more research is needed since, owing to the very nature of ILs, except for the most common ones (imidazolium and ammonium-based), many of them are subject to only one or two articles.
2. Introduction
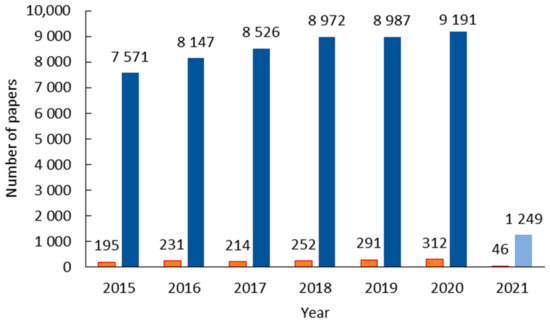
3. Conclusions
References
- Plechkova, N.V.; Seddon, K.R. Applications of Ionic Liquids in the Chemical Industry. Chem. Soc. Rev. 2008, 37, 123–150.
- Greer, A.J.; Jacquemin, J.; Hardacre, C. Industrial Applications of Ionic Liquids. Molecules 2020, 25, 5207.
- Seddon, K.R. Ionic Liquids for Clean Technology. J. Chem. Technol. Biotechnol. 1997, 68, 351–356.
- Feng, R.; Zhao, D.; Guo, Y. Revisiting Characteristics of Ionic Liquids: A Review for Further Application Development. J. Environ. Prot. 2010, 1, 95–104.
- Martins, V.L.; Torresi, R.M. Ionic Liquids in Electrochemical Energy Storage. Curr. Opin. Electrochem. 2018, 26–32.
- Rogers, R.D.; Seddon, K.R. Ionic Liquids-Solvents of the Future? Science 2003, 302, 792–793.
- Aparicio, S.; Atilhan, M.; Karadas, F. Thermophysical Properties of Pure Ionic Liquids: Review of Present Situation. Ind. Eng. Chem. Res. 2010, 49, 9580–9595.
- Gaune-Escard, M.; Seddon, K.R. (Eds.) Molten Salts and Ionic Liquids: Never the Twain? John Wiley: Hoboken, NJ, USA, 2010; ISBN 978-0-471-77392-4.
- Armand, M.; Endres, F.; MacFarlane, D.R.; Ohno, H.; Scrosati, B. Ionic-Liquid Materials for the Electrochemical Challenges of the Future. Nat. Mater. 2009, 8, 621–629.
- Welton, T.; Endres, F.; Abedin, S.Z.E.; Antonietti, M.; Smarsly, B.; Zhou, Y. Inorganic Synthesis. In Ionic Liquids in Synthesis; Wasserscheid, P., Welton, T., Eds.; John Wiley: Hoboken, NJ, USA, 2007; pp. 569–617. ISBN 978-3-527-62119-4.
- Malla, A.M.; Parveen, M.; Ahmad, F.; Azaz, S.; Alam, M. [Et3NH][HSO4]-Catalyzed Eco-Friendly and Expeditious Synthesis of Thiazolidine and Oxazolidine Derivatives. RSC Adv. 2015, 5, 19552–19569.
- Zhang, Q.; Shreeve, J.M. Energetic Ionic Liquids as Explosives and Propellant Fuels: A New Journey of Ionic Liquid Chemistry. Chem. Rev. 2014, 114, 10527–10574.
- Ribeiro, A.P.C.; Vieira, S.I.C.; França, J.M.; Queirós, C.S.; Langa, E.; Lourenço, M.J.V.; Murshed, S.M.S.; de Castro, C.A.N. Thermal Properties of Ionic Liquids and Ionanofluids. In Ionic Liquids: Theory, Properties, New Approaches; IntechOpen: Rijeka, Croatia, 2011.
- Prado, R.; Weber, C.C. Chapter 1—Applications of Ionic Liquids. In Application, Purification, and Recovery of Ionic Liquids; Kuzmina, O., Hallett, J.P., Eds.; Elsevier: Amsterdam, The Netherlands, 2016; pp. 1–58. ISBN 978-0-444-63713-0.
- Zhang, Z.; Song, J.; Han, B. Catalytic Transformation of Lignocellulose into Chemicals and Fuel Products in Ionic Liquids. Chem. Rev. 2017, 117, 6834–6880.
- Brandt, A.; Gräsvik, J.; Hallett, J.P.; Welton, T. Deconstruction of Lignocellulosic Biomass with Ionic Liquids. Green Chem. 2013, 15, 550–583.
- Marrucho, I.M.; Branco, L.C.; Rebelo, L.P.N. Ionic Liquids in Pharmaceutical Applications. Annu. Rev. Chem. Biomol. Eng. 2014, 5, 527–546.
- Earle, M.J.; Seddon, K.R. Ionic Liquids. Green Solvents for the Future. Pure Appl. Chem. 2000, 72, 1391–1398.
- Cvjetko Bubalo, M.; Hanousek, K.; Radošević, K.; Gaurina Srček, V.; Jakovljević, T.; Radojčić Redovniković, I. Imidiazolium Based Ionic Liquids: Effects of Different Anions and Alkyl Chains Lengths on the Barley Seedlings. Ecotoxicol. Environ. Saf. 2014, 101, 116–123.
- Neumann, J.; Steudte, S.; Cho, C.-W.; Thöming, J.; Stolte, S. Biodegradability of 27 Pyrrolidinium, Morpholinium, Piperidinium, Imidazolium and Pyridinium Ionic Liquid Cations under Aerobic Conditions. Green Chem. 2014, 16, 2174–2184.
- Pawłowska, B.; Telesiński, A.; Biczak, R. Phytotoxicity of Ionic Liquids. Chemosphere 2019, 237, 124436.
- Tang, S.L.Y.; Smith, R.L.; Poliakoff, M. Principles of Green Chemistry: PRODUCTIVELY. Green Chem. 2005, 7, 761–762.
- Anastas, P.; Eghbali, N. Green Chemistry: Principles and Practice. Chem. Soc. Rev. 2009, 39, 301–312.
- Regulation (EC) No 1907/2006 of the European Parliament and of the Council of 18 December 2006 Concerning the Registration, Evaluation, Authorisation and Restriction of Chemicals (REACH), Establishing a European Chemicals Agency, Amending Directive 1999/45/EC and Repealing Council Regulation (EEC) No 793/93 and Commission Regulation (EC) No 1488/94 as Well as Council Directive 76/769/EEC and Commission Directives 91/155/EEC, 93/67/EEC, 93/105/EC and 2000/21/EC. Volume 396. 2006. Available online: (accessed on 12 February 2021).
- Pernak, J.; Rogoża, J.; Mirska, I. Synthesis and Antimicrobial Activities of New Pyridinium and Benzimidazolium Chlorides. Eur. J. Med. Chem. 2001, 36, 313–320.
- Pernak, J.; Kalewska, J.; Ksycińska, H.; Cybulski, J. Synthesis and Anti-Microbial Activities of Some Pyridinium Salts with Alkoxymethyl Hydrophobic Group. Eur. J. Med. Chem. 2001, 36, 899–907.
- Amde, M.; Liu, J.-F.; Pang, L. Environmental Application, Fate, Effects, and Concerns of Ionic Liquids: A Review. Environ. Sci. Technol. 2015, 49, 12611–12627.
- Heckenbach, M.E.; Romero, F.N.; Green, M.D.; Halden, R.U. Meta-Analysis of Ionic Liquid Literature and Toxicology. Chemosphere 2016, 150, 266–274.
- Weyhing-Zerrer, N.; Gundolf, T.; Kalb, R.; Oßmer, R.; Rossmanith, P.; Mester, P. Predictability of Ionic Liquid Toxicity from a SAR Study on Different Systematic Levels of Pathogenic Bacteria. Ecotoxicol. Environ. Saf. 2017, 139, 394–403.





