
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Tao Sun | + 3169 word(s) | 3169 | 2021-05-31 09:44:33 | | | |
| 2 | Karina Chen | Meta information modification | 3169 | 2021-06-15 09:58:35 | | | | |
| 3 | Conner Chen | Meta information modification | 3169 | 2021-10-11 09:54:57 | | |
Video Upload Options
Tissue engineering (TE) was initially designed to tackle clinical organ shortage problems. Although some engineered tissues have been successfully used for non-clinical applications, very few (e.g., reconstructed human skin) have been used for clinical purposes. As the current TE approach has not achieved much success regarding more broad and general clinical applications, organ shortage still remains a challenging issue. This very limited clinical application of TE can be attributed to the constraints in manufacturing fully functional tissues via the traditional top–down approach, where very limited cell types are seeded and cultured in scaffolds with equivalent sizes and morphologies as the target tissues. The newly proposed developmental engineering (DE) strategy towards the manufacture of fully functional tissues utilises a bottom–up approach to mimic developmental biology processes by implementing gradual tissue assembly alongside the growth of multiple cell types in modular scaffolds. This approach may overcome the constraints of the traditional top–down strategy as it can imitate in vivo-like tissue development processes.
1. Introduction
Tissue engineering (TE) had its breakthrough about 35 years ago when it was introduced by two pioneers of this research field, Dr. Joseph Vacanti (Children’s Hospital Boston, Boston, MS, USA) and Dr. Robert Langer (MIT, Cambridge, MS, USA) [1]. Vacanti and Langer [2] described TE as an interdisciplinary field that employs life science and engineering principles towards the development of biological substitutes to restore, maintain, and improve the functions of damaged tissues and organs. The initial motivation was to tackle the growing issue of organ shortages, which resulted in increasing deaths of patients on organ transplantation waiting lists [2][3] (pp. 3–4) [4] (p. 4). Another objective of TE was the manufacturing of assistive extracorporeal devices for defective organs, such as substitute living kidneys as alternatives to dialysis [5]. Apart from the clinical applications, the engineered biological substitutes were also intended to be used for non-clinical purposes, such as in vitro biology and physiology studies as well as drug testing [5][6][7][8][9][10][11][12][13][14].
The initiation of TE created high hopes of manufacturing and commercialising engineered tissues and organs for clinical applications, which led to the formation of several companies [15][16]. Thus, the private sector research and development (R&D) investments in TE compared to the federal R&D investments in TE were significantly higher with, for example, more than 3.7 billion USD difference in 2001 in the US [15]. Consequently, the private sector dominated TE research, which resulted in the development of a relatively practical focus [15]. During the past 35 years, great amounts of consistent worldwide research efforts have been attracted into TE, and significant progress has been achieved. Due to the advantages of three-dimensional (3D) tissue cultures over two-dimensional (2D) monolayer cell cultures [11], a variety of engineered tissues have been manufactured successfully for non-clinical purposes. For example, engineered small intestinal organoids have been used for drug screening, the investigation of intestinal diseases, and studies of host–pathogen interactions, organ function, and physiology [17] (pp. 273–297). Some biological substitutes with one to two cell types and relatively simple anatomic structures, such as engineered human skins, are also used successfully for clinical applications [18][19][20]. However, there have been very few successes achieved in terms of the manufacturing of fully functional tissues with more diverse cell types and complex anatomic structures for clinical purposes [15][16].
This very limited clinical application is mainly due to the fundamental obstacles associated with the traditional top–down tissue manufacturing approach [21]. As a consequence, an alternative bottom–up strategy, also called developmental engineering (DE), has been proposed to manufacture fully functional tissues and organs. This bottom–up approach may overcome the problems associated with the traditional top–down approach and has the potential to deliver the promised successes for clinical applications [21][22].
2. Top–Down Tissue Manufacturing Approach
TE is based on a biological triad of cells, signals, and scaffolds [15][21][23][24]. As the main TE components, live cells are strategically utilised for the creation of new tissues and their integration with existing host tissues. Different signals are either provided as growth factors, cytokines, or as specific mechanical or electric stimuli to regulate various cell behaviours. Scaffolds are utilised to hold the cells together to create the tissue’s physical structure; moreover, they have the same role as the extracellular matrix (ECM) of in vivo tissues [6][15][24]. As the functions of the ECM in natural tissues are complex and multivariate, the scaffolds are not only used to mechanically stabilise the cells by providing physical support for cell adhesion but also to regulate cell proliferation, migration, and differentiation [15][24]. Therefore, both signals and scaffolds are critical to provide the suitable environments for tissue regeneration.
In order to replicate the necessary tissue structures and functions, and to avoid the immune rejection of the engineered tissues by the hosts in clinical applications, suitable cells and scaffold materials are firstly selected [5]. The underpinning fundamental biological interactions, including the cell–cell and cell–scaffold interactions, have to be systematically investigated and then used as specific guidance for subsequent tissue reconstructions [16][25][26]. However, this has been simplified or even neglected in the top–down TE approach, which is mainly based on one-stage seeding and culturing of a few main types of cells into 3D scaffolds that replicate the sizes and morphologies of the target tissues, as opposed to the complex natural tissue development processes (Figure 1) [15][21][23][24].
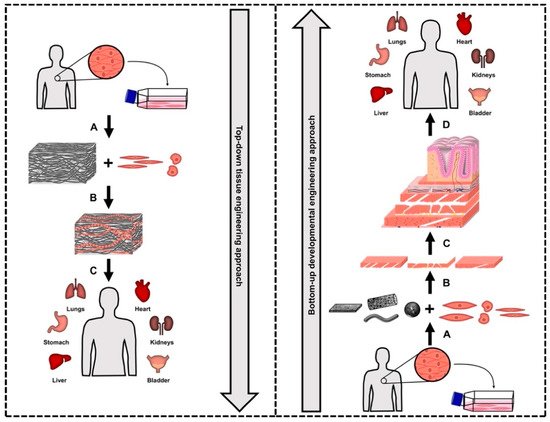
Figure 1. Comparison of the traditional top–down tissue engineering (left) and the bottom–up developmental engineering (right) approaches towards tissue reconstruction. The top–down tissue engineering approach (left) starts with (A) the isolation of the suitable dominant cell type(s) and the expansion of these isolated cells; the next step is (B) a one-stage process for seeding and culturing few dominant cell types of the target tissue in a three-dimensional scaffold with equivalent sizes and shapes as the target tissues/organs; (C) the reconstructed tissues can be used for clinical applications. The bottom–up developmental engineering approach (right) starts with (A) the isolation of multiple cell types and the expansion of these isolated cells separately; the next step is (B) the independent preparation of multiple modular tissue building blocks by culturing different cell types on corresponding modular scaffolds with specific sizes and morphologies; this is followed by (C) the gradual tissue reconstruction via layer-by-layer assembly of the modular tissue building blocks to reconstruct (D) the designated functional tissues/organs for clinical applications.
The advantages associated with this top–down TE strategy are apparent. Firstly, only the dominant cell types in the target tissues are isolated, expanded, and then utilised for tissue reconstruction, while other assistive cells, such as nerve and vascular cells, are not necessary. For example, only human dermal fibroblasts, or only human keratinocytes, or both human dermal fibroblasts and keratinocytes have been used as the dominant cell types for the reconstruction of engineered skin products, which have been used successfully for clinical purposes [16][27][28]. Chondrocytes have been employed as the single dominant cell type for the manufacture of functional cartilage products for clinical purposes, which have passed phase I or phase II of clinical trials, depending on the manufacturers [16][29][30]. Secondly, 3D scaffolds are usually fabricated to mainly replicate the overall sizes and morphologies but not the complex internal structures of the target tissues. Thirdly, a simple one-stage cell seeding and culturing in the scaffolds is adopted for the subsequent tissue reconstructions. Clearly, this top–down TE approach is suitable for the manufacturing of tissues with relatively few cell types and simple anatomic structures, such as skin and cartilage tissues [16][22][27][28][29][30].
Since this artificial top–down strategy is fundamentally different from the natural tissue developmental processes in vivo, there are inherent limitations [15][22]. Firstly, very limited mass transfer in these heterogeneous liquid–solid tissue culture systems [21][31] (pp. 398–400, 471–475). The mass transfer between the 3D solid scaffolds and the surrounding liquids (e.g., cell suspensions or culture media) is mainly dependent on limited diffusion rather than more effective convection. Therefore, uniform cell seeding into the 3D scaffolds is a challenge, and most of the cells are only seeded onto the exterior surfaces of the scaffolds [21][31] (p. 397). Some cells might be introduced slightly deeper underneath the exterior surfaces; however, the survival of these cells is further endangered by the limited nutrient and oxygen supply, and metabolic waste removal within the scaffolds [21][31] (pp. 398–400, 471–475). Secondly, there is a lack of essential cells and tissues. From a technical perspective, the production of functional tissues requires the capability to introduce suitable cells into specific parts of a scaffold and to control the temporal–spatial organisation of different cell types to replicate the complex, ordered architectures of natural tissues [21]. Particularly, the introduction of the essential structures, such as nerves and blood vessels, is necessary [5][21][32]. Innervation plays a critical role not only in tissue and organ development, maturation, and regeneration but also in their functional and regulatory control, and modulation [32][33]. Moreover, due to the release of specific neuropeptides by autonomic and sensory nerves, the promotion of innervation can directly facilitate angiogenesis [32]. Blood vessels, similar to nerves, are also critical structures as they supply cells with oxygen and nutrients as well as remove their metabolic wastes and carbon dioxide [5][11]. The cell viability in vivo depends on the diffusion of oxygen, nutrients, and growth factors from the surrounding capillaries, and the diffusion distance is limited to only 100–200 microns [11][21]. In order to manufacture vascularised thick tissues or organs, specific cell types and proper angiogenic factors are required within the scaffolds [11]. However, in the top–down tissue manufacturing processes, apart from several dominant cell types, other essential cells, such as nerve and vascular cells, are usually not included [5][32]. In addition, the control over the temporal–spatial organisation of the seeded cells is very limited, which consequently restricts the structures and functions of the fabricated tissues [21][31] (p. 397). The resulting lack of the particularly essential nerves and blood vessels is a detrimental issue that hinders the reconstruction of fully functional tissues, and the subsequent proper performance and integration of the reconstructed tissues with the host [5][32]. For example, due to the lack of blood vessels, the mass transfer in the top–down TE processes has to rely on limited diffusion, which causes poor cell survival in the necrotic central regions [21][31] (pp. 398–400, 471–475). There have been various research efforts, especially towards the incorporation of blood vessels into some tissue constructs; however, no sufficient vasculatures have been constructed yet within the currently engineered tissues [11][21]. Thirdly, there is poor controllability. Due to the fundamental differences between the top–down TE approach and the natural tissue development processes, the comparability and the viability of rational process modifications of the top–down TE processes are minimised [15][22]. Therefore, most current top–down processes, being one-stage processes, are adapted more or less according to trial-and-error, resulting in poor controllability and reproducibility [15]. As a consequence, the development of standardised protocols for these top–down TE processes is obstructed [22].
Due to the aforementioned technical obstacles, up-scaling of these traditional tissue manufacturing processes as well as the production of complex organs, such as kidneys, livers, and hearts, is not supported by the top–down TE strategy [11][21][22]. The only successful TE product for clinical applications manufactured via the top–down approach is reconstructed human skin, which is comparably simple in its anatomic structure [16][22][27]. Even though various engineered skin products have been manufactured and used for clinical applications, they are still not fully functional. This is because only human keratinocytes and/or dermal fibroblasts are commonly utilised for the reconstruction of human skin, while other skin cells, such as melanocytes and vascular cells, are not included [28]. Thus, apart from the epidermal tissue with keratinocytes and/or the dermal tissue with fibroblasts, there is a lack of other structures, such as sweat glands, hair follicles, sensory nerve fibres, and vascular plexuses, in the currently reconstructed skins [28][34]. Other promising TE products for clinical purposes seem to be bioartificial cartilages, which have passed phase I or phase II clinical trials, depending on the producing company [16]. The attempts to apply other TE products, such as trachea, blood vessels, and bladders, for clinical purposes have not succeeded yet [16][21][35]. Therefore, several TE companies were only able to produce engineered tissues with very low profit margins while suffering from high R&D and production costs [16][22]. Hence, a number of high-profile bankruptcies occurred, such as the one of Advanced Tissue Sciences (ATS) in 2003, which resulted in the loss of about 300 million USD [16][36]. Accordingly, the scientific community raises criticism against the traditional top–down TE approach, which has not shown the initially promised potential for clinical applications, as most of the TE products just look like tissues, smell like tissues, and taste like tissues but do not fully function like the natural target tissues [22][37].
3. Bottom–Up Tissue Manufacturing Approach
Although the TE challenges for clinical applications have already been recognised by scientists for many years, they are yet to be overcome [1][21][22]. This is mainly due to the inherent, insurmountable limitations of the top–down TE approach, which is fundamentally different from natural tissue development [22]. Therefore, an alternative bottom–up DE strategy (Figure 1 and Figure 2) has been proposed to mimic and exploit in vivo tissue development, structure, and function [25]. DE is closely related to systems biology and developmental biology [15][38].
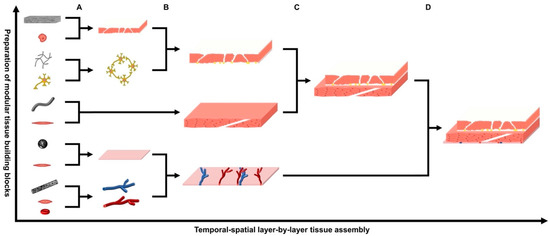
Figure 2. The gradual temporal–spatial layer-by-layer tissue assembly of functional tissues/organs via the bottom–up developmental engineering strategy. Firstly, (A) multiple modular tissue building blocks are prepared separately using different types of cells and their corresponding modular scaffolds with varying sizes and morphologies; (B–D) these modular tissue building blocks are gradually assembled layer-by-layer via specific temporal-spatial procedures to imitate the in vivo development processes as well as the anatomic structures of the target tissues/organs; this results in the formation of designated tissues/organs with increasing size and complexity.
Systems biology aims to investigate a whole biological system and the interactions of different components of the system at different levels [38]. Similarly, live tissues are integrated biological systems with closely related or integrated components (e.g., cells, ECM) (Figure 3) [25]. Therefore, in order to reconstruct fully functional tissues, the interactions among different cells and the surrounding ECM components or scaffolds must be coordinated and regulated as in the authentic target tissues [25]. To deliberately mimic these interactions, the bottom–up DE approach utilises various suitable biomaterials or scaffolds, and multiple cell types but not just several dominant cells. Most importantly, it also imitates the in vivo-like temporal–spatial coordination of these components in vitro [15][39]. Accordingly, the tenet of this bottom–up DE strategy is to replicate the integrated functional networks of cells and ECM components. As such, it is a step towards a systems biology approach for the reconstruction of fully functional tissues and organs.
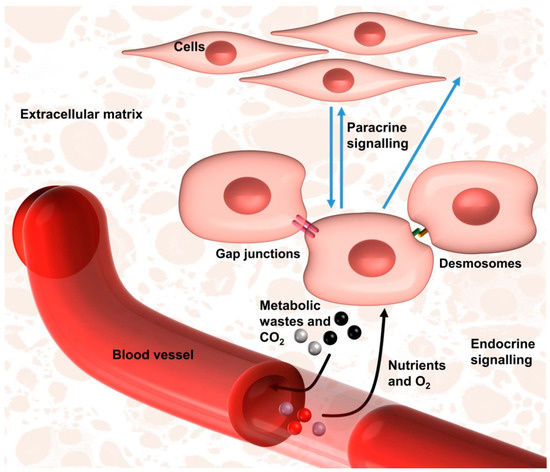
Figure 3. The native cell niche that is intended to be mimicked by developmental engineering. The cells are supplied with oxygen (O2) and nutrients, while their metabolic wastes and carbon dioxide (CO2) are removed via the enclosed vasculature. The cell behaviours are regulated by neighbouring cells via gap junctions or desmosomes, and by distant cells via paracrine signalling or vasculature-mediated endocrine signalling. The cells are also influenced by cell–extracellular matrix interactions.
Developmental biology is about the upgrowth of an organism from a fertilised ovum, which is a typical bottom–up process with a gradual increase of tissue size in parallel to cell growth [4] (p. 21) [15][21]. These natural development processes are intended to be imitated in DE by firstly preparing various modular tissue building blocks using different types of cells and corresponding modular scaffolds with smaller sizes and varying shapes, and then assembling these modular tissue building blocks layer by layer to gradually reconstruct the designated tissues or organs (Figure 1 and Figure 2) [15][21][22][31] (pp. 300–305).
The bottom–up DE strategy is different from the traditional top–down TE approach in several aspects. Firstly, more essential but not just several dominant cell types are used for tissue reconstruction. Secondly, multiple modular scaffolds with smaller sizes and varying shapes for different cell types are fabricated instead of a single scaffold with the same size and shape as the target tissue. Thirdly, functional tissues are reconstructed via carefully coordinated temporal and spatial processes rather than a simplified one-stage cell seeding and culturing procedure. Due to the use of smaller scaffolds for the preparation of modular tissue building materials and the gradual introduction of vascular systems within the reconstructed tissues, nutrient supply and metabolic waste removal are dependent on both convection and diffusion. Consequently, mass transfer within the progressively assembled tissues is not a limiting detrimental factor. As the bottom–up DE strategy is in vivo-like, it is more comparable to the imitated natural tissue development process than the conventional top–down TE approach [15][22]. Moreover, the bottom–up strategy with sequential stages, in contrast to the top–down approach with just a one-stage cell seeding and culturing procedure (Figure 1 and Figure 2), has improved observability and controllability [15][22]. Therefore, rational modifications to the temporal–spatial bottom–up processes are facilitated, rather than the empirically or trial-and-error-based modifications commonly adopted for the top–down procedures [15][22]. The consequentially enhanced reproducibility enables the design of standardised tissue manufacturing protocols, which is a major advantage of DE, making it significantly more efficient [15][22].
This layer-by-layer bottom-up DE approach is particularly advantageous to replicate the hierarchical architectures found in natural tissues [6][24][31] (pp. 300–305). This is mainly because the use of modular tissue building materials enables the imitation of the individual native cell niches (Figure 3) for different types of cells [5][16][26]. As previously mentioned, in order to reconstruct fully functional tissues, the whole system, as well as the complex interactions within diverse cell niches, have to be considered, including for example the cell–cell and cell–material interactions [16][25][26]. The scaffolds in TE are meant to replace the ECM in natural tissues; this has been approached by research on microgels, which mimic the fibrous structure, ECM-coated/embedded polymeric microcarriers, and ECM-derived microcarriers in cell culture and delivery applications [6][40][41][42][43]. However, the diverse ECM components fulfil more purposes than the provision of mechanical support and anchorage for the cells [15][24]. For example, they can regulate gene expression and even alter cell phenotypes via physical and/or biochemical influences, which are transduced by cell surface receptors through the cytoskeletons to the cell nuclei [5]. These cell–ECM interactions direct the tissue function and structural integrity and can even overwrite the initial cell programming [5][24]. The responses and adaptions of the cells to their niches or the surrounding matrix are exploited by DE to facilitate and regulate the designated in vivo-like tissue reconstructions [5][15][31] (pp. 227–242). Therefore, the imitation of native cell niches within the modular tissue building materials enables the bottom–up DE approach to reconstruct fully functional tissues with hierarchical architectures. Besides the layer-by-layer assembly of fully functional tissues or organs for clinical applications, the modular tissue building block materials can also be utilised as 3D tissue alternatives to 2D cell cultures or animal models for non-clinical purposes, such as drug testing.
In summary, the bottom–up DE strategy has the potential to overcome the challenges associated with the top–down TE approach and, therefore, to achieve the initially promised successes of TE, especially the manufacturing of fully functional tissues or organs to tackle the clinical organ shortage problems [21][22][31] (pp. 300–305).
References
- Vacanti, C.A. The History of Tissue Engineering. J. Cell. Mol. Med. 2006, 10, 569–576.
- Langer, R.; Vacanti, J.P. Tissue Engineering. Science 1993, 260, 920–926.
- Birla, R. Introduction to Tissue Engineering: Applications and Challenges, 1st ed.; Wiley: Hoboken, NJ, USA, 2014; pp. 3–4.
- Palsson, B.O.; Bhatia, S.N. Tissue Engineerig, Indian ed.; Pearson India Education: Chennai, India, 2016; pp. 4, 21.
- Grounds, M.D. Obstacles and Challenges for Tissue Engineering and Regenerative Medicine: Australian Nuances. Clin. Exp. Pharmacol. Physiol. 2017, 45, 390–400.
- Takezawa, T. A Strategy for the Development of Tissue Engineering Scaffolds That Regulate Cell Behavior. Biomaterials 2003, 24, 2267–2275.
- Sperling, L.E.; Reis, K.P.; Pranke, P.; Wendorff, J.H. Advantages and Challenges Offered by Biofunctional Core-Shell Fiber Systems for Tissue Engineering and Drug Delivery. Drug Discov. Today 2016, 21, 1243–1256.
- Cancedda, R.; Giannoni, P.; Mastrogiacomo, M. A Tissue Engineering Approach to Bone Repair in Large Animal Models and in Clinical Practice. Biomaterials 2007, 28, 4240–4250.
- Park, K. Facing the Truth about Nanotechnology in Drug Delivery. ACS Nano 2013, 7, 7442–7447.
- Holmes, A.M.; Solari, R.; Holgate, S.T. Animal Models of Asthma: Value, Limitations and Opportunities for Alternative Approaches. Drug Discov. Today 2011, 16, 659–670.
- Naderi, H.; Matin, M.M.; Bahrami, A.R. Review Paper: Critical Issues in Tissue Engineering: Biomaterials, Cell Sources, Angiogenesis, and Drug Delivery Systems. J. Biomater. Appl. 2011, 26, 383–417.
- Muschler, G.F.; Raut, V.P.; Patterson, T.E.; Wenke, J.C.; Hollinger, J.O. The Design and Use of Animal Models for Translational Research in Bone Tissue Engineering and Regenerative Medicine. Tissue Eng. Part. B Rev. 2010, 16, 123–145.
- Mathur, A.; Loskill, P.; Shao, K.; Huebsch, N.; Hong, S.; Marcus, S.G.; Marks, N.; Mandegar, M.; Conklin, B.R.; Lee, L.P.; et al. Human IPSC-Based Cardiac Microphysiological System For Drug Screening Applications. Sci. Rep. 2015, 5, 8883:1–8883:7.
- Cook, J.L.; Hung, C.T.; Kuroki, K.; Stoker, A.M.; Cook, C.R.; Pfeiffer, F.M.; Sherman, S.L.; Stannard, J.P. Animal Models of Cartilage Repair. Bone Joint Res. 2014, 3, 89–94.
- Lenas, P.; Moos, M.; Luyten, F.P. Developmental Engineering: A New Paradigm for the Design and Manufacturing of Cell-Based Products. Part I: From Three-Dimensional Cell Growth to Biomimetics of In vivo Development. Tissue Eng. Part. B Rev. 2009, 15, 381–394.
- Williams, D.F. Challenges With the Development of Biomaterials for Sustainable Tissue Engineering. Front. Bioeng. Biotechnol. 2019, 7, 127:1–127:10.
- Däullary, T.; Fey, C.; Berger, C.; Metzger, M.; Zdzieblo, D. Bioartificial gut—Current state of small intestinal tissue engineering. In Biomaterials for Organ and Tissue Regeneration, 1st ed.; Vrana, N., Knopf-Marques, H., Barthes, J., Eds.; Woodhead Publishing: Duxford, UK, 2020; pp. 273–297.
- Pham, C.; Greenwood, J.; Cleland, H.; Woodruff, P.; Maddern, G. Bioengineered Skin Substitutes for the Management of Burns: A Systematic Review. Burns 2007, 33, 946–957.
- Nicoletti, G.; Brenta, F.; Bleve, M.; Pellegatta, T.; Malovini, A.; Faga, A.; Perugini, P. Long-Term in vivo Assessment of Bioengineered Skin Substitutes: A Clinical Study. J. Tissue Eng. Regen. Med. 2015, 9, 460–468.
- Savoji, H.; Godau, B.; Hassani, M.S.; Akbari, M. Skin Tissue Substitutes and Biomaterial Risk Assessment and Testing. Front. Bioeng. Biotechnol. 2018, 6, 86:1–86:18.
- Tiruvannamalai-Annamalai, R.; Armant, D.R.; Matthew, H.W.T. A Glycosaminoglycan Based, Modular Tissue Scaffold System for Rapid Assembly of Perfusable, High Cell Density, Engineered Tissues. PLoS ONE 2014, 9, e84287:1–e84287:15.
- Lenas, P. Developmental Biology in Bioartificial Tissue Design: Manufacturing and Regulatory Considerations. Regen. Med. 2018, 13, 7–11.
- Caddeo, S.; Boffito, M.; Sartori, S. Tissue Engineering Approaches in the Design of Healthy and Pathological In vitro Tissue Models. Front. Bioeng. Biotechnol. 2017, 5, 40:1–40:22.
- Marga, F.; Neagu, A.; Kosztin, I.; Forgacs, G. Developmental Biology and Tissue Engineering. Birth Defects Res. Part. C Embryo Today Rev. 2007, 81, 320–328.
- Lenas, P.; Ikonomou, L. Developmental Engineering: Design of Clinically Efficacious Bioartificial Tissues through Developmental and Systems Biology. Sci. China Life Sci. 2018, 61, 978–981.
- Ziółkowska, K.; Kwapiszewski, R.; Brzózka, Z. Microfluidic Devices as Tools for Mimicking the in vivo Environment. New J. Chem. 2011, 35, 979–990.
- Ter Horst, B.; Chouhan, G.; Moiemen, N.S.; Grover, L.M. Advances in Keratinocyte Delivery in Burn Wound Care. Adv. Drug Deliv. Rev. 2018, 123, 18–32.
- Vig, K.; Chaudhari, A.; Tripathi, S.; Dixit, S.; Sahu, R.; Pillai, S.; Dennis, V.A.; Singh, S.R. Advances in Skin Regeneration Using Tissue Engineering. Int. J. Mol. Sci. 2017, 18, 789.
- Camarero-Espinosa, S.; Rothen-Rutishauser, B.; Foster, E.J.; Weder, C. Articular Cartilage: From Formation to Tissue Engineering. Biomater. Sci. 2016, 4, 734–767.
- Keeney, M.; Lai, J.H.; Yang, F. Recent Progress in Cartilage Tissue Engineering. Curr. Opin. Biotechnol. 2011, 22, 734–740.
- Van Blitterswijk, C.A.; de Boer, J. Tissue Engineering, 2nd ed.; Academic Press: London, UK, 2015; pp. 227–242, 261, 300–305, 397–400, 471–475.
- Das, S.; Gordián-Vélez, W.J.; Ledebur, H.C.; Mourkioti, F.; Rompolas, P.; Chen, H.I.; Serruya, M.D.; Cullen, D.K. Innervation: The Missing Link for Biofabricated Tissues and Organs. NPJ Regen. Med. 2020, 5, 11:1–11:19.
- Kumar, A.; Brockes, J.P. Nerve Dependence in Tissue, Organ, and Appendage Regeneration. Trends Neurosci. 2012, 35, 691–699.
- Moniz, T.; Costa Lima, S.A.; Reis, S. Human Skin Models: From Healthy to Disease-Mimetic Systems; Characteristics and Applications. Br. J. Pharmacol. 2020, 177, 4314–4329.
- Paolo Macchiarini: A Surgeon’s Downfall. Available online: (accessed on 19 October 2020).
- Pangarkar, N.; Pharoah, M.; Nigam, A.; Hutmacher, D.W.; Champ, S. Advanced Tissue Sciences Inc.: Learning from the Past, a Case Study for Regenerative Medicine. Regen. Med. 2010, 5, 823–835.
- Polykandriotis, E.; Popescu, L.M.; Horch, R.E. Regenerative Medicine: Then and Now-an Update of Recent History into Future Possibilities. J. Cell. Mol. Med. 2010, 14, 2350–2358.
- Lenas, P.; Moos, M.; Luyten, F.P. Developmental Engineering: A New Paradigm for the Design and Manufacturing of Cell-Based Products. Part II. From Genes to Networks: Tissue Engineering from the Viewpoint of Systems Biology and Network Science. Tissue Eng. Part. B Rev. 2009, 15, 395–422.
- Mironov, V.; Visconti, R.P.; Kasyanov, V.; Forgacs, G.; Drake, C.J.; Markwald, R.R. Organ Printing: Tissue Spheroids as Building Blocks. Biomaterials 2009, 30, 2164–2174.
- Newsom, J.P.; Payne, K.A.; Krebs, M.D. Microgels: Modular, Tunable Constructs for Tissue Regeneration. Acta Biomater. 2019, 88, 32–41.
- Kumar Meena, L.; Rather, H.; Kedaria, D.; Vasita, R. Polymeric Microgels for Bone Tissue Engineering Applications—A Review. Int. J. Polym. Mater. Polym. Biomater. 2020, 69, 381–397.
- Shekaran, A.; Lam, A.; Sim, E.; Jialing, L.; Jian, L.; Wen, J.T.P.; Chan, J.K.Y.; Choolani, M.; Reuveny, S.; Birch, W.; et al. Biodegradable ECM-Coated PCL Microcarriers Support Scalable Human Early MSC Expansion and in vivo Bone Formation. Cytotherapy 2016, 18, 1332–1344.
- Kornmuller, A.; Brown, C.F.C.; Yu, C.; Flynn, L.E. Fabrication of Extracellular Matrix-Derived Foams and Microcarriers as Tissue-Specific Cell Culture and Delivery Platforms. J. Vis. Exp. 2017, e55436:1–e55436:11.





