
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Yves SY Hsieh | + 2178 word(s) | 2178 | 2021-04-12 14:21:15 | | | |
| 2 | Peter Tang | Meta information modification | 2178 | 2021-06-09 10:43:51 | | |
Video Upload Options
(1,3;1,4)-β-D-Glucans, also named as mixed-linkage glucans, are unbranched non-cellulosic polysaccharides containing both (1,3)- and (1,4)-β-linkages. The linkage ratio varies depending upon species origin and has a significant impact on the physicochemical properties of the (1,3;1,4)-β-D-glucans.
1. Introduction
β-Glucans have been found to be highly abundant in plants, algae, fungi, and bacteria as one of the fundamental fibers in the cell walls. The polysaccharides are composed of D-glucopyranosyl units (Glcp) as building blocks. Depending on the glycosidic bonds between the glucose monomers, β-glucans can be classified into two sub-groups, cereal, and non-cereal β-glucans. For example, the yeast and fungi β-glucans contain mainly 1,3 and branching 1,6 linkages, whereas cereal β-glucans have linear glucan chains composed of 1,3 and 1,4 glycosidic linkages. (1,3;1,4)-β-D-Glucans, or mixed-linkage glucans (MLGs), have been found rich in rice, wheat, cereal grains, and oats, serving as important dietary fibers for our daily consumption, and also impact our metabolic activities by decreasing cholesterol and blood glucose [1].
Structurally, the (1,3;1,4)-β-D-glucan is a linear polymer, primarily containing (1,4)-linked β-D-glucose residues interspersed with a single (1,3)-β-linkage. The ratio of (1,3)- to (1,4)-β-linkages is a good indicator of the fine structure of (1,3;1,4)-β-D-glucan and determines its solubility and physicochemical properties [2]. The polysaccharide can be broken down by the specific polysaccharide hydrolase, the lichenase (EC 3.2.1.73), which cleaves at a (1,4)-β-d-glucopyranosyl linkage immediately adjacent to a (1,3)-β-D-glucopyranosyl residue, yielding oligosaccharides with distinctive structures such as the cellotriose Glcp-β-1,4-Glcp-β-1,3-Glcp (G4G4G3GR, with a degree of polymerization of three, namely DP3) and cellotetraose Glcp-β-1,4-Glcp-β-1,4-Glcp-β-1,3-Glcp (G4G4G4G3GR, DP4) (Figure 1) [3]. Since this discovery, the lichenase has been essential in the structural profiling of (1,3;1,4)-β-D-glucans. For example, the enzymatic digestion of (1,3;1,4)-β-D-glucans of cereal and grasses (family Poaceae), such as barley, results in a higher proportion of DP3 compared to DP4, with a ratio of 1.5–4.5 [4][5][6], whereas the (1,3;1,4)-β-D-glucans of horsetail (Equisetum spp.) of a monilophyte family Equisetaceae sensu stricto releases predominantly DP4 after lichenase digestion (Figure 1 and Table 1) [3][7][8]. The structure of (1,3;1,4)-β-d-glucans of the brown algae Ectocarpus sp. has been recently reported to have only DP3 repeating units (Table 1) [9]. In contrast, Glcp-β-1,3-Glcp (DP2) and DP3 are the most abundant oligomers in (1,3;1,4)-β-D-glucans from Sinorhizobium meliloti and Sarcina ventriculi, respectively [3][10]. Structural surveys reveal (1,3;1,4)-β-D-glucans display heterogeneous structures between different taxonomic groups (Table 1), which suggests differential functions in the cell walls of the different organisms.
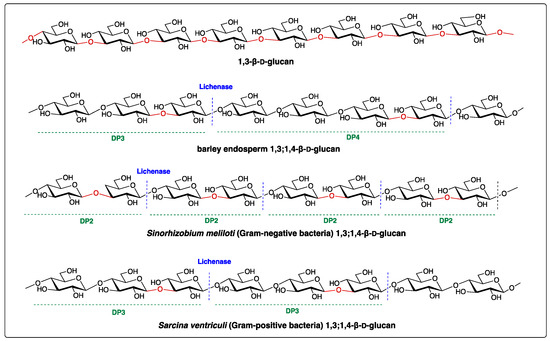
Figure 1. Structures of 1,3-β-D-glucan and (1,3;1,4)-β-D-glucans. Lichenase is an endo-hydrolase that hydrolyses the (1,3;1,4)-β-D-glucans into smaller oligomers, such as degree of polymerization 2 (DP2), DP3, and DP4 shown above. The blue dash lines represent the enzymatic cleavage sites. Oligosaccharide profiles in DPs vary depending on the taxonomic origin of (1,3;1,4)-β-D-glucans.
Table 1. The ratio of mixed-linkage glucan (MLG) oligosaccharides produced from different species after lichenase digestion.
|
Species |
DP2 |
DP3 |
DP4 |
References |
|---|---|---|---|---|
|
Hordeum vulgare |
0 |
1.8–3.5 |
1 |
[6] |
|
Triticum aestivum |
0 |
3.0–4.5 |
1 |
|
|
Avena sativa |
0 |
1.5–2.3 |
1 |
[6] |
|
Secale cereale |
0 |
1.9–3 |
1 |
[6] |
|
Equisetum arvense |
0 |
0.05–0.1 |
1 |
[3] |
|
Equisetum fluviatile |
0 |
0.1 |
1 |
[7] |
|
Cetraria islandica |
0 |
20.2–24.6 |
1 |
[6] |
|
Sinorhizobium meliloti |
1 |
0 |
0 |
[10] |
|
Sarcina ventriculi |
0 |
1 |
0 |
[13] |
|
Ectocarpus sp. |
0 |
1 |
0 |
[9] |
(1,3;1,4)-β-d-Glucans have been widely accepted as functional and bioactive ingredients in dietary fibers [14]. The polysaccharides are known to enhance the viscosity of the solution and form a matrix-like gel under suitable conditions [14][15][16]. Hence, (1,3;1,4)-β-D-glucans have been utilized in the industry as thickening agents for gravies, ice cream formulations, etc. [17], or as fat mimetics in low-calorie food [18]. Additionally, (1,3;1,4)-β-D-glucans extracted from human dietary fiber have been identified that can lower serum cholesterol, reduce the risk of type II diabetes and obesity, and provide other healthful benefits [19][20][21][22], sometimes they can also function as a source of metabolizable energy [23]. It is also observed that some bacteria contain MLG utilization loci (MLGULs), encoding genes that allow the bacteria to utilize MLGs as energy sources. Therefore, the MLGULs can serve as genetic markers for (1,3;1,4)-β-D-glucan catabolism in commensal gut bacteria [24].
2. Variation of (1,3;1,4)-β-d-Glucans in Different Taxa
2.1. Viridiplantae
(1,3;1,4)-β-D-Glucans have been found in the cell walls of the grasses and cereal family, also known as Poaceae, which consist of commercially important cereals. During the plant growth and development, the amount of (1,3;1,4)-β-D-glucans in the wall is found to increase proportionally to the cell elongation rate, reaching its maximum during the most rapid phase of cell growth, and is completely hydrolyzed when growth ceases. Not only functioning as structural elements, the (1,3;1,4)-β-D-glucans are present in high abundance in the walls of the aleurone layer surrounding the barley, rye, and oats starchy endosperms. These (1,3;1,4)-β-D-glucans are hydrolyzed by specific enzymes, the (1,3;1,4)-β-D-glucanases, when germination occurs, allowing mobilization of endosperm cell walls and also providing an extra carbon source that facilitates the germination process [25][26].
By enzymatic profiling and linkage analysis, the ratio of (1,4)- to (1,3)-β-linkages is defined within the range of 2.2–2.6:1 in (1,3;1,4)-β-D-glucans of cereals, and this translates to a ratio of DP3:DP4 units ranges from approximately 1.5:1 to 4.5:1, with the occasional occurrence of longer oligosaccharide units of up to DP > 13 [6][27][28]. So far, similar oligomeric distributions have been found within related species, which indicates that the (1,3)- and (1,4)-β-linkages are not randomly generated. On the other hand, whether the differences in ordered combinations of DP3 and DP4 constitutive blocks are species-dependent is yet to be solved. Among vascular plants, Equisetum species, also known as horsetails, are reported to have (1,3;1,4)-β-D-glucans in their cell walls with significantly lower DP3:DP4 ratio (0.05–0.1:1). A higher abundance of the DP4 repeat units may facilitate stronger interaction with the cellulosic microfibrils [3][7].
Chlorophyte green algae Ulva lactuca and the Bryophyte liverwort Lophocolea bidentata are reported to have (1,3;1,4)-β-D-glucan-like polysaccharides, but unlike the homopolymeric (1,3;1,4)-β-D-glucans in barley, their glucans tend to include xylose and arabinose, with a higher degree of polymerization in the latter [29]. (1,3;1,4)-β-D-arabinoglucan is a recently discovered polysaccharide in the walls of moss Physcomitrella patens [30]. It is a linear glucan-related polysaccharide with a structure similar to the (1,3;1,4)-β-D-glucan, except the three-linked Glcp is substituted with three-linked Araf residues. These studies showed that the non-vascular plants could also produce structurally related glucans. The function of (1,3;1,4)-β-D-arabinoglucan remains for further investigation.
(1,3;1,4)-β-D-Glucans are found in the walls of red algae and brown algae, and both are evolutionarily more distant to land plants. A sulfated (1,3;1,4)-β-D-glucan has been isolated from the red algae Kappaphycus alvarezii. This sulfated glucan has only 180 residues, with 64% sulfation on the (1,4)-β-linkages only, and the remaining (1,4)-β-linkages were sparse and unlikely to be in long sequences after one and another. The sulfated (1,4)-β-linkages likely link to the fibrillar cell wall polymers by their incompatible extraction behavior. It is also speculated that the sulfation helps control the construction and positioning of cellulose [31].
In recent years, the presence of (1,3;1,4)-β-D-glucans in algae species is further supported by their initial discovery in brown algae. Remarkably, a study by Salmeán et al. on brown algae, including six clades of main orders and contains 34 species in total, showed (1,3;1,4)-β-D-glucans were present in all as detected by antibody-based glycan array analysis. Even more astonishing is that, unlike the (1,3;1,4)-β-D-glucan in other species which is commonly composed of both DP3 and DP4 at different proportions, the (1,3;1,4)-β-D-glucan extracted from brown algae is exclusively built by DP3 as repeating units, as observed by high-performance anion-exchange chromatography with pulsed-amperometric detection (HPAEC-PAD) [9]. Other unusual freshwater algae, like Monodus subterraneus in Xanthophyceae and dinoflagellate Peridinium gatunense, have also been reported to have (1,3;1,4)-β-D-glucans, but the distribution DP3 and DP4 requires further investigation [32][33].
The presence of (1,3;1,4)-β-D-glucan in the cell walls of Equisetum arvense, Phaeophyceae (brown algae) in the phylum of Stramenopiles that is not closely related to any land plants and green algae, the non-conserved structural characteristics across lineages and the absence of (1,3;1,4)-β-D-glucans in some closely related families of grasses and cereal, suggest synthesis could be highly conserved, or a convergence-independent evolution of (1,3;1,4)-β-D-glucan synthase genes.
2.2. Fungi and Lichens
Lichens are a combination of fungi and algae or cyanobacteria living in symbiosis, where fungi are recognized as the mycobiont and different algae or cyanobacteria as the photobiont in lichens. (1,3;1,4)-β-D-Glucans have been found in abundance in Parmeliaceae lichens, such as Cetraria, Evernia, Newropogon, Parmelia, Parmotrema, Rimelia, and Usnea [34]. These (1,3;1,4)-β-D-glucans isolated from lichen species from the family Parmeliaceae are also known as lichenin or lichenan. Specifically, the (1,3;1,4)-β-D-glucans extracted from the mycobiont cell wall of Cetraria islandica (Iceland moss) gave a higher ratio of DP3 to DP4 (20.2–24.6:1) compared barley and oats (1,3;1,4)-β-D-glucan (1.8–3.5:1) [35]. However, the ratio of (1,4)- to (1,3)-β-linkages in lichenin varies significantly among the lichens, ranging from 2.3:1 found in C. islandica, to 0.3:1 in oak moss Evernia prunastri [35]. The high proportion of three-linked Glcp in E. prunastri glucans raised the question of whether both linkages came from two separate glucans.
Besides the lichens, a non-lichen fungus, Aspergillus fumigatus, was found to have (1,3;1,4)-β-D-glucans in an alkali-insoluble fraction from its cell wall, which is suggested to be related to fungal cell wall rigidity [36]. The (1,3;1,4)-β-D-glucan was also detected in the cell wall of Neurospora crassa with monoclonal antibody [37]. A high percentage of (1,4)-linked glucose was reported in the glycosyl composition and linkage analysis of fungal cell wall from a filamentous fungus N. crassa [38]. It is highly likely that the occurrence of (1,3;1,4)-β-D-glucans are not restricted in the A. fumigatus and N. crassa. How the particular variation in the structures of (1,3;1,4)-β-D-glucans found may be related to their function is largely unknown. Other β-glucans, for example, the (1,3;1,6)-β-D-glucan, is common β-glucan found in the cell walls of fungal species and has shown to possess immunomodulatory activities in reducing SARS-CoV-2-induced cytokine storm in the infected patients [39]. A further β-glucan structural survey of taxa in other lichen and fungal families would be interesting to provide evidence in understanding structure-function relationships between β-glucan structures and their functions, and their potential implications for pharmaceutical research and development.
2.3. Bacteria
The presence of (1,3;1,4)-β-D-glucan in bacteria cell walls has been overlooked for many years until the recent discovery of (1,3;1,4)-β-D-glucans in the walls of Gram-negative bacteria Sinorhizobium meliloti [10]. Occurrence of (1,3;1,4)-β-D-glucans in the walls of microorganisms such as bacteria confirms that the (1,3;1,4)-β-D-glucan is not unique to planta and fungi, but further questions remain as to how the bacterial (1,3;1,4)-β-D-glucans structure and biosynthesis differ to the planta and fungal (1,3;1,4)-β-D-glucans.
The bacteria cell wall is a complex multilayered structure that protects the bacteria from an unpredictable and often hostile environment. Most bacteria, except mycoplasmas, have a complex cell wall composed of a mixture of polymers made of carbohydrates and amino acids. These are the peptidoglycan polymers that can provide cell rigidity and act as a physical barrier between the cell and its surrounding. In response to the different environments, some species of Gram-positive bacteria and Gram-negative bacteria secrete high concentrations of the exopolysaccharides (EXPs) deposited onto the bacterial surface (Figure 2). Their roles include basic functions such as maintaining structural integrity and preventing desiccation, to more complex activities of facilitating the interaction within bacterial communities. The bacterial EXPs have been found in many pathogenic bacteria and have a direct impact on human health because of their ability to form multicellular conglomerates called biofilms.
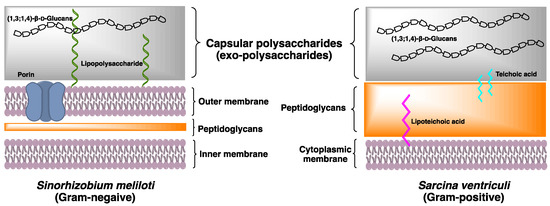
A recent study reported that the structure of (1,3;1,4)-β-D-glucan in Gram-negative bacteria S. meliloti is distinctive to the (1,3;1,4)-β-D-glucans of land plants and fungi, for its entire structure is composed of disaccharide repeating units Glcp-β-1,3-Glcp (DP2s) [10]. Methylation analysis showed that the partially methylated alditol acetates (PMAAs) derived from (1,3;1,4)-β-D-glucans of S. meliloti corresponded to (1,3)- and (1,4)-linked glucopyranosyl residues in a 1:1 ratio. Further structural investigation of (1,3;1,4)-β-D-glucans, using two-dimensional Nuclear Magnetic Resonance with Rotating-frame nuclear Overhauser Effect Spectroscopy (2D-NMR ROESY) spectrum, revealed the cross-peaks between H1 of (1,4)-linked Glcp and H3 of (1,3)-linked Glcp, and between H1 of (1,3)-linked Glcp and H4 of (1,4)-linked Glcp, corresponded to a -3)-β-D-Glcp-(1,4)-β-D-Glcp(1- repetitive polymeric structure. Lichenase digestion only releases DP2, confirming the presence of a novel (1,3;1,4)-β-D-glucan in S. meliloti.
Following the discovery of (1,3;1,4)-β-D-glucan in S. meliloti, we began to look into bacterial (1,3;1,4)-β-D-glucans that may have been overlooked in early literature. Gram-positive bacteria Sarcina ventriculi, a highly robust mesotrophic bacterial species commonly found in the soil and occasionally in human gastrointestinal tracts, can secrete and subsequently deposit an additional thick layer of EXPs onto the bacterial cellular surface [40]. An early X-ray diffraction study showed Sarcina EXPs to have a similar diffraction pattern to the (1,3;1,4)-β-D-glucans in grasses and cereals. Studies using lichenase digest have confirmed that the (1,3;1,4)-β-D-glucan of S. ventriculi is composed of DP3 G4G3GR, of which the oligosaccharide profiles are similar to the (1,3;1,4)-β-D-glucan recently isolated from the brown algae Ectocarpus sp. [9].
References
- Murphy, E.J.; Rezoagli, E.; Major, I.; Rowan, N.J.; Laffey, J.G. β-Glucan metabolic and immunomodulatory properties and potential for clinical application. J. Fungi 2020, 6, 356.
- Burton, R.A.; Gidley, M.J.; Fincher, G.B. Heterogeneity in the chemistry, structure and function of plant cell walls. Nat. Chem. Biol. 2010, 6, 724–732.
- Sørensen, I.; Pettolino, F.A.; Wilson, S.M.; Doblin, M.S.; Johansen, B.; Bacic, A.; Willats, W.G.T. Mixed-linkage (1→3),(1→4)-β-d-glucan is not unique to the Poales and is an abundant component of Equisetum arvense cell walls. Plant J. 2008, 54, 510–521.
- Burton, R.A.; Fincher, G.B. (1,3;1,4)-β-d-glucans in cell walls of the poaceae, lower plants, and fungi: A tale of two linkages. Mol. Plant 2009, 2, 873–882.
- Burton, R.A.; Fincher, G.B. Evolution and development of cell walls in cereal grains. Front. Plant Sci. 2014, 5, 456.
- Lazaridou, A.; Biliaderis, C.G. Molecular aspects of cereal β-glucan functionality: Physical properties, technological applications and physiological effects. J. Cereal Sci. 2007, 46, 101–118.
- Fry, S.C.; Nesselrode, B.H.W.A.; Miller, J.G.; Mewburn, B.R. Mixed-linkage (1→3,1→4)-β-d-glucan is a major hemicellulose of Equisetum (horsetail) cell walls. New Phytol. 2008, 179.
- Xue, X.; Fry, S.C. Evolution of mixed-linkage (1→3,1→4)-β-d-glucan (MLG) and xyloglucan in Equisetum (horsetails) and other monilophytes. Ann. Bot. 2012, 109, 873–886.
- Salmeán, A.A.; Duffieux, D.; Harholt, J.; Qin, F.; Michel, G.; Czjzek, M.; Willats, W.G.T.; Hervé, C. Insoluble (1→3),(1→4)-β-d-glucan is a component of cell walls in brown algae (Phaeophyceae) and is masked by alginates in tissues. Sci. Rep. 2017, 7.
- Pérez-Mendoza, D.; Rodríguez-Carvajal, M.Á.; Romero-Jiménez, L.; De Araujo Farias, G.; Lloret, J.; Gallegos, M.T.; Sanjuán, J. Novel mixed-linkage β-glucan activated by c-di-GMP in Sinorhizobium Meliloti. Proc. Natl. Acad. Sci. USA 2015, 112, E757–E765.
- Wood, P.J.; Weisz, J.; Blackwell, B.A. Molecular characterization of cereal beta-d-glucans. Structural analysis of oat beta-d-glucan and rapid structural evaluation of beta-d-glucans from different sources by high-performance liquid chromatography of oligosaccharides released by lichenase. Cereal Chem. 1991, 68, 31–39.
- Wood, P.J.; Braaten, J.T.; Scott, F.W.; Riedel, K.D.; Wolynetz, M.S.; Collins, M.W. Effect of dose and modification of viscous properties of oat gum on plasma glucose and insulin following an oral glucose load. Br. J. Nutr. 1994, 72, 731–743.
- Lee, J.; Hollingsworth, R.I. Oligosaccharide β-glucans with unusual linkages from Sarcina ventriculi. Carbohydr. Res. 1997, 304, 133–141.
- Cui, W.; Wood, P.J. Relationships between structural features, molecular weight and rheological properties of cereal β-d-glucans. Hydrocolloids 2000.
- Bohm, N.; Kulicke, W.M. Rheological studies of barley (1→3)(1→4)-β-glucan in concentrated solution: Mechanistic and kinetic investigation of the gel formation. Carbohydr. Res. 1999, 315, 302–331.
- Lazaridou, A.; Biliaderis, C.G.; Izydorczyk, M.S. Molecular size effects on rheological properties of oat β-glucans in solution and gels. Food Hydrocoll. 2003, 17, 693–712.
- Wood, P.J. Oat Beta-Glucan: Structure, Location, and Properties; American Association of Cereal Chemists, Inc.: St. Paul, MN, USA, 1986; pp. 121–152.
- Inglett, G.E. Hypocholesterolemic β-glucan-amylodextrins from oats as dietary fat-replacements. In ACS Book of Abstracts from 199th National Meeting; ACS publications: Washington, DC, USA, 1990.
- Morgan, K.R. Cereal β-glucans. In Handbook of Hydrocolloids; Philips, G., Williams, P., Eds.; Woodhead Publishing & CRC Press: Cambridge, UK, 2000.
- Brennan, C.S.; Cleary, L.J. The potential use of cereal (1→3,1→4)-β-d-glucans as functional food ingredients. J. Cereal Sci. 2005, 42, 1–13.
- Harris, P.J.; Smith, B.G. Plant cell walls and cell-wall polysaccharides: Structures, properties and uses in food products. Int. J. Food Sci. Technol. 2006, 41, 129–143.
- Othman, R.A.; Moghadasian, M.H.; Jones, P.J.H. Cholesterol-lowering effects of oat β-glucan. Nutr. Rev. 2011, 69, 299–309.
- Harris, P.J.; Fincher, G.B. Distribution, fine structure and function of (1,3;1,4)-β-Glucans in the grasses and other taxa. In Chemistry, Biochemistry, and Biology of 1-3 Beta Glucans and Related Polysaccharides; Elsevier Science Publishing Co. Inc.: San Diego, CA, USA, 2009.
- Tamura, K.; Hemsworth, G.R.; Déjean, G.; Rogers, T.E.; Pudlo, N.A.; Urs, K.; Jain, N.; Davies, G.J.; Martens, E.C.; Brumer, H. Molecular mechanism by which prominent human gut bacteroidetes utilize mixed-linkage beta-glucans, major health-promoting cereal polysaccharides. Cell Rep. 2017, 21, 417–430.
- Meier, H.; Reid, J.S.G. Reserve polysaccharides other than starch in higher plants. In Plant Carbohydrates I. Intracellular Carbohydrates; Springer Science & Business Media: Berlin, Germany, 1982.
- Kim, J.B.; Olek, A.T.; Carpita, N.C. Cell wall and membrane-associated exo-β-d-glucanases from developing maize seedlings. Plant Physiol. 2000, 123, 471–485.
- Fincher, G.B.; Stone, B.A. CEREALS|Chemistry of Nonstarch Polysaccharides. In Encyclopedia of Grain Science; Wrigley, C., Ed.; Elsevier: Amsterdam, The Netherlands, 2004; pp. 206–223.
- Li, W.; Cui, S.W.; Kakuda, Y. Extraction, fractionation, structural and physical characterization of wheat β-d-glucans. Carbohydr. Polym. 2006, 63, 408–416.
- Popper, Z.A.; Fry, S.C. Primary cell wall composition of bryophytes and charophytes. Ann. Bot. 2003, 91, 1–12.
- Eder, M.; Tenhaken, R.; Driouich, A.; Lütz-Meindl, U. Occurrence and characterization of arabinogalactan-like proteins and hemicelluloses in Micrasterias (Streptophyta). J. Phycol. 2008, 44, 1221–1234.
- Lechat, H.; Amat, M.; Mazoyer, J.; Buléon, A.; Lahaye, M. Structure and distribution of glucomannan and sulfated glucan in the cell walls of the red alga Kappaphycus alvarezii (Gigartinales, Rhodophyta). J. Phycol. 2000, 36, 891–902.
- Ford, C.W.; Percival, E. 551. Polysaccharides synthesised by Monodus subterraneus. Part II. The cell-wall glucan. J. Chem. Soc. 1965.
- Nevo, Z.; Sharon, N. The cell wall of Peridinium westii, a non cellulosic glucan. BBA Biomembr. 1969, 173, 161–175.
- Olafsdottir, E.S.; Ingólfsdottir, K. Polysaccharides from lichens: Structural characteristics and biological activity. Planta Med. 2001, 67, 199–208.
- Honegger, R.; Haisch, A. Immunocytochemical location of the (1→3) (1→4)-β-glucan lichenin in the lichen-forming ascomycete Cetraria islandica (Icelandic moss). New Phytol. 2001, 150, 739–746.
- Fontaine, T.; Simenel, C.; Dubreucq, G.; Adam, O.; Delepierre, M.; Lemoine, J.; Vorgias, C.E.; Diaquin, M.; Latgé, J.P. Molecular organization of the alkali-insoluble fraction of Aspergillus fumigatus cell wall. J. Biol. Chem. 2000, 275, 27594–27607.
- Ao, J.; Free, S.J. Genetic and biochemical characterization of the GH72 family of cell wall transglycosylases in Neurospora Crassa. Fungal Genet. Biol. 2017, 101, 46–54.
- Maddi, A.; Free, S.J. α-1,6-mannosylation of N-linked oligosaccharide present on cell wall proteins is required for their incorporation into the cell wall in the filamentous fungus Neurospora crassa. Eukaryot. Cell 2010, 9, 1766–1775.
- Murphy, E.J.; Masterson, C.; Rezoagli, E.; O’Toole, D.; Major, I.; Stack, G.D.; Lynch, M.; Laffey, J.G.; Rowan, N.J. β-Glucan extracts from the same edible shiitake mushroom Lentinus edodes produce differential in-vitro immunomodulatory and pulmonary cytoprotective effects—Implications for coronavirus disease (COVID-19) immunotherapies. Sci. Total Environ. 2020, 732, 139330.
- Canale-Parola, E. Biology of the sugar-fermenting Sarcinae. Bacteriol. Rev. 1970, 34, 82–97.





