
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Igor Buzalewicz | + 5032 word(s) | 5032 | 2021-05-28 04:46:30 | | | |
| 2 | Bruce Ren | -21 word(s) | 5011 | 2021-06-07 03:57:44 | | |
Video Upload Options
Quantifying changes in bacteria cells in the presence of antibacterial treatment is one of the main challenges facing contemporary medicine; it is a challenge that is relevant for tackling issues pertaining to bacterial biofilm formation that substantially decreases susceptibility to biocidal agents. Three-dimensional label-free imaging and quantitative analysis of bacteria–photosensitizer interactions, crucial for antimicrobial photodynamic therapy, is still limited due to the use of conventional imaging techniques. We present a new method for investigating the alterations in living cells and quantitatively analyzing the process of bacteria photodynamic inactivation. Digital holographic tomography (DHT) was used for in situ examination of the response of Escherichia coli and Staphylococcus aureus to the accumulation of the photosensitizers immobilized in the copolymer revealed by the changes in the 3D refractive index distributions of single cells. Obtained results were confirmed by confocal microscopy and statistical analysis.
1. Introduction
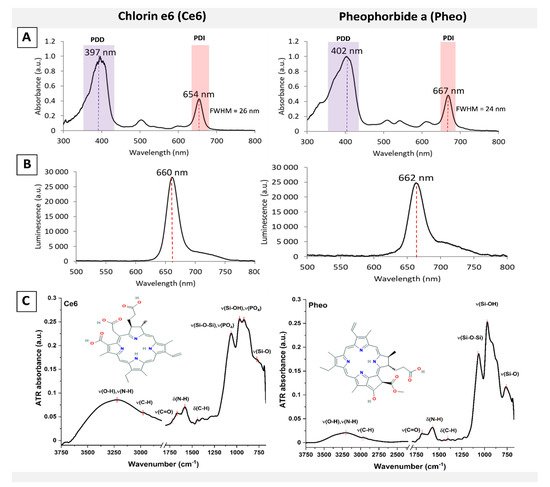
2. Photoactive Materials and Photosensitizers Characterization
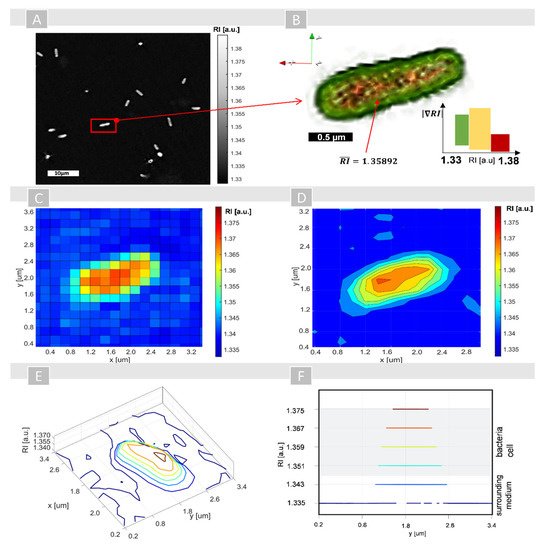
3. Qualitative and Quantitative Analysis of the Single Cells’ RI Variations Caused by Photosensitizer Penetration
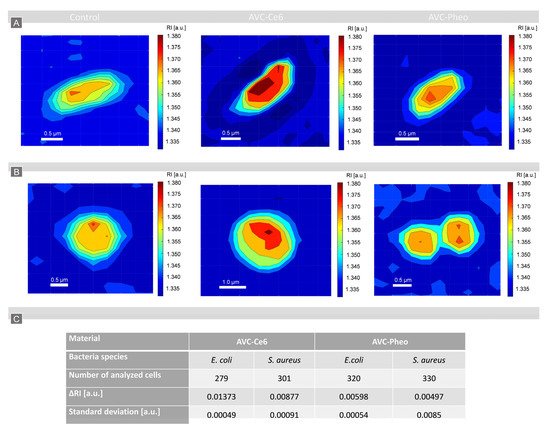
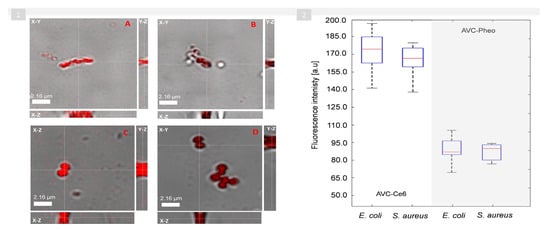
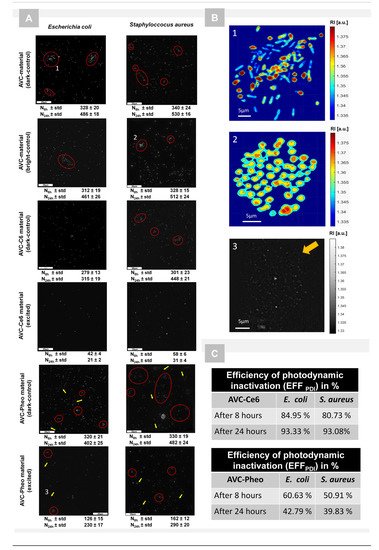
4. Photodynamic Inactivation of Bacteria Cells Revealed by DHT and Its Quantitative Analysis
References
- Jamal, M.A.; Garoge, K.; Rosenblatt, J.S.; Hachem, R.Y.; Raad, I.I. Development of Gendine-Coated Cannula for Continuous Subcutaneous Insulin Infusion for Extended Use. Antimicrob. Agents Chemother. 2015, 59, 4397–4402.
- Römling, U.; Balsalobre, C. Biofilm infections, their resilience to therapy and innovative treatment strategies. J. Intern. Med. 2012, 272, 541–561.
- Vestby, L.K.; Grønseth, T.; Simm, R.; Nesse, L.L. Bacterial Biofilm and Its Role in the Pathogenesis of Disease. Antibiot. 2020, 9, 59.
- Del Pozo, J.L. Biofilm-related disease. Expert Rev. Anti-Infect. Ther. 2018, 16, 51–65.
- Jamal, M.; Ahmad, W.; Andleeb, S.; Jalil, F.; Imran, M.; Nawaz, M.A.; Hussain, T.; Ali, M.; Rafiq, M.; Kamil, M.A. Bacterial biofilm and associated infections. J. Chin. Med. Assoc. 2018, 81, 7–11.
- Chen, N.; Zhou, M.; Dong, X.; Qu, J.; Gong, F.; Han, Y.; Qiu, Y.; Wang, J.; Liu, Y.; Wei, Y.; et al. Epidemiological and clinical characteristics of 99 cases of 2019 novel coronavirus pneumonia in Wuhan, China: A descriptive study. Lancet 2020, 395, 507–513.
- Zhou, F.; Yu, T.; Du, R.; Fan, G.; Liu, Y.; Liu, Z.; Xiang, J.; Wang, Y.; Song, B.; Gu, X.; et al. Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: A retrospective cohort study. Lancet 2020, 395, 1054–1062.
- Póvoa, H.C.C.; Chianca, G.C.; Iorio, N.L.P.P. COVID-19: An Alert to Ventilator-Associated Bacterial Pneumonia. Infect. Dis. Ther. 2020, 9, 417–420.
- Bao, L.; Zhang, C.; Dong, J.; Zhao, L.; Li, Y.; Sun, J. Oral Microbiome and SARS-CoV-2: Beware of Lung Co-infection. Front. Microbiol. 2020, 11, 1840.
- Wickens, D.J.; West, G.; Kelly, P.J.; Verran, J.; Lynch, S.; Whitehead, K.A. Antimicrobial Activity of Nanocomposite Zirconium Nitride/Silver Coatings to Combat External Bone Fixation Pin Infections. Int. J. Artif. Organs 2012, 35, 817–825.
- Walker, J. Pin site infection in orthopaedic external fixation devices. Br. J. Nurs. 2012, 21, 148–151.
- Jennison, T.; McNally, M.; Pandit, H. Prevention of infection in external fixator pin sites. Acta Biomater. 2014, 10, 595–603.
- Rabin, N.; Zheng, Y.; Opoku-Temeng, C.; Du, Y.; Bonsu, E.; Sintim, H.O. Biofilm formation mechanisms and targets for developing antibiofilm agents. Futur. Med. Chem. 2015, 7, 493–512.
- Boudjemaa, R.; Steenkeste, K.; Canette, A.; Briandet, R.; Fontaine-Aupart, M.-P.; Marlière, C. Direct observation of the cell-wall remodeling in adhering Staphylococcus aureus 27217: An AFM study supported by SEM and TEM. Cell Surf. 2019, 5, 100018.
- Gutiérrez, D.; Hidalgo-Cantabrana, C.; Rodríguez, A.; García, P.; Ruas-Madiedo, P. Monitoring in Real Time the Formation and Removal of Biofilms from Clinical Related Pathogens Using an Impedance-Based Technology. PLoS ONE 2016, 11, e0163966.
- Wang, Y.; Reardon, C.P.; Read, N.; Thorpe, S.; Evans, A.; Todd, N.; Van Der Woude, M.; Krauss, T.F. Attachment and antibiotic response of early-stage biofilms studied using resonant hyperspectral imaging. npj Biofilms Microbiomes 2020, 6, 1–7.
- Harris, L.G.; Richards, R.G. Staphylococci and implant surfaces: A review. Injury 2006, 37, S3–S14.
- Rozbruch, S.R.; Kazmers, N.H.; Fragomen, A.T. Prevention of pin site infection in external fixation: A review of the literature. Strat. Trauma Limb Reconstr. 2016, 11, 75–85.
- Ogbemudia, O.A.; Bafor, A.; Edomwonyi, E.; Enemudo, R. Prevalence of pin tract infection: The role of combined silver sulphadiazine and chlorhexidine dressing. Niger. J. Clin. Pr. 2010, 13, 268–271.
- Lee, C.K.; Chua, Y.P.; Saw, A. Antimicrobial Gauze as a Dressing Reduces Pin Site Infection: A Randomized Controlled Trial. Clin. Orthop. Relat. Res. 2012, 470, 610–615.
- Sukakul, T.; Dahlin, J.; Pontén, A.; Antelmi, A.; Bruze, M.; Hamnerius, N.; Hauksson, I.; Isaksson, M.; Lejding, T.; Svedman, C. Contact allergy to polyhexamethylene biguanide (polyaminopropyl biguanide). Contact Dermat. 2020, 84, 326–331.
- Park, Y.; Depeursinge, C.; Popescu, G. Quantitative phase imaging in biomedicine. Nat. Photon. 2018, 12, 578–589.
- Kim, K.; Yoon, J.; Shin, S.; Lee, S.; Yang, S.-A.; Park, Y. Optical diffraction tomography techniques for the study of cell pathophysiology. J. Biomed. Photon. Eng. 2016, 2, 020201–1.
- Jung, J.; Hong, S.-J.; Kim, H.-B.; Kim, G.; Lee, M.; Shin, S.; Lee, S.; Kim, D.-J.; Lee, C.-G.; Park, Y. Label-free non-invasive quantitative measurement of lipid contents in individual microalgal cells using refractive index tomography. Sci. Rep. 2018, 8, 1–10.
- Kim, T.-K.; Lee, B.-W.; Fujii, F.; Kim, J.K.; Pack, C.-G. Physicochemical Properties of Nucleoli in Live Cells Analyzed by Label-Free Optical Diffraction Tomography. Cells 2019, 8, 699.
- Cotte, Y.; Toy, F.M.; Jourdain, P.; Pavillon, N.; Boss, D.; Magistretti, P.J.; Marquet, P.; Depeursinge, C. Marker-free phase nanoscopy. Nat. Photon. 2013, 7, 113–117.
- Lauer, V. New approach to optical diffraction tomography yielding a vector equation of diffraction tomography and a novel tomographic microscope. J. Microsc. 2002, 205, 165–176.
- Oh, J.; Ryu, J.S.; Lee, M.; Jung, J.-H.; Han, S.; Chung, H.J.; Park, Y. Three-dimensional label-free observation of individual bacteria upon antibiotic treatment using optical diffraction tomography. Biomed. Opt. Express 2020, 11, 1257–1267.
- Kuś, A.; Krauze, W.; Makowski, P.L.; Kujawińska, M. Holographic tomography: Hardware and software solutions for 3D quantitative biomedical imaging (Invited paper). ETRI J. 2019, 41, 61–72.
- Kuś, A.; Krauze, W.; Kujawinska, M. Limited-angle holographic tomography with optically controlled projection generation. In Proceedings of the Three-Dimensional and Multidimensional Microscopy: Image Acquisition and Processing XXII, San Francisco, CA, USA, 9 March 2015; Volume 9330, p. 933007.
- Kus, A.; Krauze, W.; Kujawinska, M. Active limited-angle tomographic phase microscope. J. Biomed. Opt. 2015, 20, 111216.
- Kim, T.-K.; Lee, B.-W.; Fujii, F.; Lee, K.-H.; Lee, S.; Park, Y.; Kim, J.K.; Lee, S.-W.; Pack, C.-G. Mitotic Chromosomes in Live Cells Characterized Using High-Speed and Label-Free Optical Diffraction Tomography. Cells 2019, 8, 1368.
- Koshi, E.; Mohan, A.; Rajesh, S.; Philip, K. Antimicrobial photodynamic therapy: An overview. J. Indian Soc. Periodontol. 2011, 15, 323–327.
- Gouveia, T.H.N.; De Souza, D.F.S.; Aguiar, F.H.B.; Ambrosano, G.M.B.; Lima, D.A.N.L. Effect of ammonium acryloyldimethyltaurate copolymer on the physical and chemical properties of bleached dental enamel. Clin. Oral Investig. 2019, 24, 2701–2711.
- Daneluz, J.; Favero, J.D.S.; Dos Santos, V.; Weiss-Angeli, V.; Gomes, L.B.; Mexias, A.S.; Bergmann, C.P. The Influence of Different Concentrations of a Natural Clay Material as Active Principle in Cosmetic Formulations. Mater. Res. 2020, 23, 23.
- Nigro, F.; Cerqueira, C.; Rossi, A.; Cardoso, V.; Vermelho, A.B.; Ricci-Júnior, E.; Dos Santos, E.P.; Mansur, C.R.E. Development, characterization and in vitro toxicity evaluation of nanoemulsion-loaded hydrogel based on copaiba oil and coenzyme Q10. Colloids Surf. A Physicochem. Eng. Asp. 2020, 586, 124132.
- Marta, K.; Iwona, H.; Agnieszka, U.-J.; Igor, B.; Halina, P. Sol-gel Coated Fiberoptic Applicator for Photodynamic Medicine-Optical and AFM Characterization. Biocybern. Biomed. Eng. 2012, 32, 41–50.
- Calori, I.R.; Caetano, W.; Tedesco, A.C.; Hioka, N. Self-aggregation of verteporfin in glioblastoma multiforme cells: A static and time-resolved fluorescence study. Dye. Pigment. 2020, 182, 108598.
- Wawrzyńska, M.; Duda, M.; Hołowacz, I.; Kaczorowska, A.; Ulatowska-Jarża, A.; Buzalewicz, I.; Kałas, W.; Wysokińska, E.; Biały, D.; Podbielska, H.; et al. Photoactive Pore Matrix for In Situ Delivery of a Photosensitizer in Vascular Smooth Muscle Cells Selective PDT. Materials 2019, 12, 4110.
- Yue, C.; Zhang, C.; Alfranca, G.; Yang, Y.; Jiang, X.; Yang, Y.; Pan, F.; De La Fuente, J.M.; Cui, D. Near-Infrared Light Triggered ROS-activated Theranostic Platform based on Ce6-CPT-UCNPs for Simultaneous Fluorescence Imaging and Chemo-Photodynamic Combined Therapy. Theranostics 2016, 6, 456–469.
- Gladkova, O.; Parkhats, M.; Gorbachova, A.; Terekhov, S. FTIR spectra and normal-mode analysis of chlorin e6 and its degradation-induced impurities. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2010, 76, 388–394.
- Sato, H.; Ozaki, Y.; Uehara, K.; Araki, T.; Iriyama, K. ATR/FT-IR Study of a Monolayer Film of Chlorophyll-a on a Germanium Plate. Appl. Spectrosc. 1993, 47, 1509–1512.
- Yu, J.; Wang, G.; Wang, X.; Xu, Y.; Chen, S.; Wang, X.; Jiang, L. Improving the freeze-thaw stability of soy protein emulsions via combing limited hydrolysis and Maillard-induced glycation. LWT 2018, 91, 63–69.
- Kizil, R.; Irudayaraj, J.; Seetharaman, K. Characterization of Irradiated Starches by Using FT-Raman and FTIR Spectroscopy. J. Agric. Food Chem. 2002, 50, 3912–3918.
- Popescu, G.; Park, Y.; Lue, N.; Best-Popescu, C.; DeFlores, L.; Dasari, R.R.; Feld, M.S.; Badizadegan, K. Optical imaging of cell mass and growth dynamics. Am. J. Physiol. Physiol. 2008, 295, C538–C544.
- Lagage, V.; Uphoff, S. Filming flagella and pili in action. Nat. Rev. Genet. 2018, 16, 584.
- Salton, M.R.J.; Kim, K.S. Structure-Medical Microbiology-NCBI Bookshelf. Available online: (accessed on 26 November 2020).
- De Aldecoa, A.L.I.; Zafra, O.; González-Pastor, J.E. Mechanisms and Regulation of Extracellular DNA Release and Its Biological Roles in Microbial Communities. Front. Microbiol. 2017, 8, 1390.
- Marquis, R.E. Immersion Refractometry of Isolated Bacterial Cell Walls. J. Bacteriol. 1973, 116, 1273–1279.
- Valkenburg, J.A.; Woldringh, C.L. Phase separation between nucleoid and cytoplasm in Escherichia coli as defined by immersive refractometry. J. Bacteriol. 1984, 160, 1151–1157.
- Liu, P.; Chin, L.; Ser, W.; Ayi, T.; Yap, P.; Bourouina, T.; Leprince-Wang, Y. Real-time Measurement of Single Bacterium’s Refractive Index Using Optofluidic Immersion Refractometry. Procedia Eng. 2014, 87, 356–359.
- Pour, P.A.; Kenney, M.C.; Kheradvar, A. Bioenergetics Consequences of Mitochondrial Transplantation in Cardiomyocytes. J. Am. Hear. Assoc. 2020, 9, e014501.
- Baczewska, M.; Eder, K.; Ketelhut, S.; Kemper, B.; Kujawińska, M. Refractive Index Changes of Cells and Cellular Compartments Upon Paraformaldehyde Fixation Acquired by Tomographic Phase Microscopy. Cytom. Part. A 2020, 99, 388–398.
- George, S.; Hamblin, M.R.; Kishen, A. Uptake pathways of anionic and cationic photosensitizers into bacteria. Photochem. Photobiol. Sci. 2009, 8, 788–795.
- Zechini, B.; Versace, I. Inhibitors of multidrug resistant efflux systems in bacteria. Recent Pat. Anti-Infect. Drug Discov. 2009, 4, 37–50.
- Pathania, R.; Sharma, A.; Gupta, V.K. Efflux pump inhibitors for bacterial pathogens: From bench to bedside. Indian J. Med. Res. 2019, 149, 129–145.
- Preuß, A.; Zeugner, L.; Hackbarth, S.; Faustino, M.; Neves, M.; Cavaleiro, J.; Roeder, B. Photoinactivation of Escherichia coli (SURE2) without intracellular uptake of the photosensitizer. J. Appl. Microbiol. 2012, 114, 36–43.
- Kowal, K.; Cronin, P.; Dworniczek, E.; Żegliński, J.; Tiernan, P.; Wawrzyńska, M.; Podbielska, H.; Tofail, S.A.M. Biocidal effect and durability of nano-TiO2 coated textiles to combat hospital acquired infections. RSC Adv. 2014, 4, 19945.
- Roeder, B.; Wabnitz, H. Time-resolved fluorescence spectroscopy of hematoporphyrin, mesoporphyrin, pheophorbide a and chlorin e6 in ethanol and aqueous solution. J. Photochem. Photobiol. B Biol. 1987, 1, 103–113.





