Your browser does not fully support modern features. Please upgrade for a smoother experience.

Submitted Successfully!
Thank you for your contribution! You can also upload a video entry or images related to this topic.
For video creation, please contact our Academic Video Service.
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | NESTOR ANDRES URBINA SUAREZ | + 1145 word(s) | 1145 | 2021-05-31 12:19:47 | | | |
| 2 | Dean Liu | -17 word(s) | 1128 | 2021-06-01 02:57:26 | | |
Video Upload Options
We provide professional Academic Video Service to translate complex research into visually appealing presentations. Would you like to try it?
Cite
If you have any further questions, please contact Encyclopedia Editorial Office.
Urbina Suarez, N. Tannery Wastewater. Encyclopedia. Available online: https://encyclopedia.pub/entry/10320 (accessed on 04 April 2026).
Urbina Suarez N. Tannery Wastewater. Encyclopedia. Available at: https://encyclopedia.pub/entry/10320. Accessed April 04, 2026.
Urbina Suarez, Nestor. "Tannery Wastewater" Encyclopedia, https://encyclopedia.pub/entry/10320 (accessed April 04, 2026).
Urbina Suarez, N. (2021, May 31). Tannery Wastewater. In Encyclopedia. https://encyclopedia.pub/entry/10320
Urbina Suarez, Nestor. "Tannery Wastewater." Encyclopedia. Web. 31 May, 2021.
Copy Citation
Las aguas residuales de las curtidurías son producto de un proceso de transformación de materia orgánica a materia no degradable, que requiere la adición de compuestos y aditivos que permitan dicha transformación, generando a su vez residuos altamente contaminantes no solo para la salud humana, sino también para el medio ambiente.
wastewater
AOPs
tannery
microalgae
biological process
leather industry
1. Introducción
Currently, there are various industrial processes that contribute to the deterioration of water quality; most require the use of highly polluting substances for terrestrial and aquatic ecosystems; therefore, it is imperative to apply a treatment before its discharge in different water bodies. The tanning sector allows the transformation of degradable animal leather into non-degradable leather by eliminating impurities and unnecessary materials [1]; however, this process generates different residues that represent a significant environmental problem, requiring specific treatments, which are expensive and inaccessible in many developing countries. [2]. In the global context, Asia together with Latin America and Europe are the main leather producers in the world [3][4] (Figure 1). It is estimated that the budget of this business in Europe is approximately 8 billion euros per year, due to the more than 3000 companies that employ 50,000 people, demonstrating their competitiveness in the global market [5]. In recent years, Latin America and Africa have increased the annual growth rate given the consumption of leather in these places [3]. Italy is the main leather producer on the European continent, owns 60% of the companies and exports more than 70% of the total production in Europe; it owns 15% of the world leather production, and in the European Union, it is the main producer with 65% of the total manufacture. [6]. In Latin America, Brazil and Argentina stand out in this sector; they have a significant number of emerging companies with great competitiveness in the market, exporting their products to countries such as China, Hong Kong, Vietnam and the United States. Globally, Colombia represents 5% of the tanning industry production, and in the Latin American context, it ranks 6th as a producer in this industry [5].
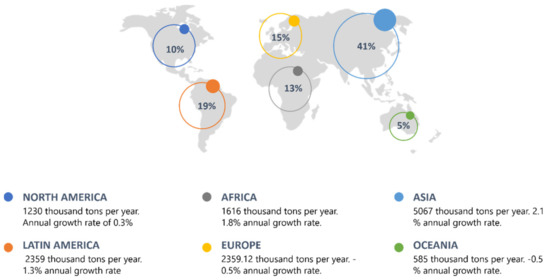
Figure 1. Global context of the leather industry.
Leather production consumes on average 10–25 m3 of water in its different stages and can generate on average 8 to 20 m3 of wastewater; this flow may vary depending on the technological development of each industry. According to Nagi et al. [2], this resource is used to transport chemical products from diffusion and in the phase of washing and extraction of undesirable compounds from the leather [7]. In this process, large amounts of wastewater are generated, significantly altering the quality of the water since they not only contain biodegradable compounds such as fats, proteins and carbohydrates, but also polluting compounds such as solvents, additives and toxic heavy metals typical of the process [8]. At least 90% of industries are reported to use basic chromium sulfate as a tanning agent [9][10], chromium (Cr) being one of the most toxic metals used in tanning and, without prior treatment, is released in effluents in toxic concentrations. The regulations of the World Health Organization (2020) recommend a reference value of 0.05 mg/L of total chromium in drinking water; above this concentration, it can exert a genotoxic and carcinogenic effect. High concentrations of chromium (VI) cause the deterioration of ecosystems and have an impact on human health as they cause kidney damage, liver damage, chronic bronchitis, nasal irritation, cancer, and DNA damage among other things [11][12]; these effects have led governments to apply stricter regulations and promote effective treatments to reduce the risk of contamination. Over the years, various techniques for treating tannery wastewater have been studied, focused on the removal of pollutants, particularly chromium. Some of these technologies are chemical coagulation, photodegradation, biodegradation, adsorption, ozonation, electrocoagulation and reverse osmosis [13]; however, the difficulty of implementing them is related to high energy consumption, use of large areas of land, high operation and maintenance costs [14]; in the same way, in some processes, other polluting byproducts are generated [15]. The foregoing has launched various biotechnological processes, including the use of microalgae, bacteria and fungi, as sustainable and economically affordable alternatives [16].
In recent years, microalgae and cyanobacteria have been proposed as a sustainable solution for the removal of nutrients and hazardous materials from wastewater [17][18][19][20]. In relation to the treatment of wastewater from tanneries, studies with various strains are still limited, the most common being Scenedesmus sp. and Chlorella sp., obtaining positive and promising results [2]. Microalgae have exhibited high tolerance to adapt in this environment, as well as an important role in reducing contaminants. The biomass of the microalgae has shown an absorbent capacity during the chromium elimination process, obtaining important results with Chlorella vulgaris, where a reduction of Cr(VI) to Cr(III) was also observed, going from a highly toxic compound to one less toxic, also attributing it to biological (enzymatic pathway) and non-biological mechanisms (glutathione releasing) [21]. Likewise, the use of residual biomass from the process is a way to maximize production of energy (biofuels) and generate byproducts of commercial interest (pigments, lipids, etc.), reducing the costs of the process [22][23][24][25][26][27][28][29]. Various studies have been carried out to identify the advantages of using microalgae in tannery waters. In India, one of the three largest leather producers after China and Italy, high concentrations of Pb, Cr, Zn and Cu were found, and an efficiency of 60–98% of removal of these metals under different conditions was demonstrated, specifically with microalgae [30]. In Brazil, progress has been made regarding the growth of microalgae consortia under different concentrations of tannery wastewater [16].
2. Technologies for the Treatment of Tannery Wastewater
The main technologies for the treatment of tannery wastewater focus only on certain parameters [31], as there is great difficulty in finding a treatment that completely reduces the pollutant load. Figure 2 presents a graphical description of the most common technologies. Since chromium is the main problem in this process, most treatments for these waters focus on the reduction and reuse of this element, followed by treatment for COD, BOD and TDS. These technologies include chemical coagulation processes [32][33] electrocoagulation [34], absorption, advanced oxidation processes [35] and biological processes such as phytoremediation [13][14].
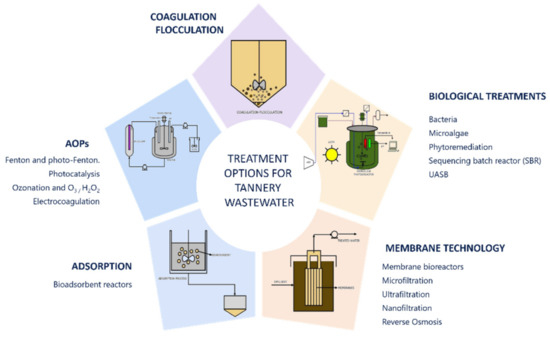
Figura 2. Opciones de tratamiento de las aguas residuales de las curtidurías.
En Colombia se han realizado varios estudios de investigación para mitigar los impactos; entre ellos se encuentran la identificación y evaluación de contaminantes [31]; se ha informado del uso de la planta de Eichhornia crassipes en el tratamiento a escala piloto del agua de las curtidurías debido a su capacidad para acumular metales pesados y materia orgánica[32]; También se ha estudiado el efecto genotóxico de los efluentes de tenerías sobre cepas de Salmonella typhimurium , identificando un aumento mutagénico y la capacidad de generar daño en el ADN en linfocitos humanos[31][32][33]. Sin embargo, la aplicación de microalgas en aguas residuales de tenerías aún no se ha desarrollado en profundidad en este país.
References
- Suárez Escobar, A.F.; García Ubaque, C.A.; Vaca Bohórquez, M.L. Identificación y evaluación de la contaminación del agua por curtiembres en el municipio de Villapinzón. Tecnura 2012, 16, 185–193.
- Carreño, S.U.F. Diseño y evaluación de un biosistema de tratamiento a escala piloto de aguas de curtiembres a través de la Eichhornia crassipes. Rev. Colomb. Biotecnol. 2016, 18, 74–81.
- Quintero Salamanca, G.P.; Quijano Parra, A.; Melendez Gelvez, I. Efecto genotoxico del agua residual de la curtiembre San Faustino—Norte de Santander—Colombia. Rev. Colomb. Tecnol. Av. 2018, 2, 8–16.
- The Confederation of National Associations of Tanners and Dressers of the European Community (COTANCE). Available online: (accessed on 22 March 2021).
- Martinez, B.S.Y.; Romero, C.J.A. Revisión Del Estado Actual De La Industria De Las Curtiembres En Sus Procesos Y Productos: Un análisis De Su Competitividad. Rev. Fac. Cien. Econ. 2017, 26, 113–124.
- Lofrano, G.; Belgiorno, V.; Gallo, M.; Raimo, A.; Meriç, S. Toxicity reduction in leather tanning wastewater by improved coagulation flocculation process. Glob. NEST J. 2006, 8, 151–158.
- Pena, A.C.; Trierweiler, L.F.; Gutterres, M. Influence of photoperiod on biomass production and removal of nutrients from tannery effluents with microalgae consortium. In Proceedings of the XXXV IULTCS Congress 2019; Tegtmeyer, D., Meyer, M., Eds.; Association for Tannery Chemistry and Technology: Dresden, Germany, 2019; Volume 4, p. 19.
- Ouaissa, Y.A.; Chabani, M.; Amrane, A.; Bensmaili, A. Integration of Electro Coagulation and Adsorption for the Treatment of Tannery Wastewater–The Case of an Algerian Factory, Rouiba. Procedia Eng. 2012, 33, 98–101.
- Hansen, É.; Monteiro de Aquim, P.; Hansen, A.W.; Cardoso, J.K.; Ziulkoski, A.L.; Gutterres, M. Impact of Post-Tanning Chemicals on the Pollution Load of Tannery Wastewater. J. Environ. Manag. 2020, 269, 110787.
- Aravindhan, R.; Madhan, B.; Rao, J.R.; Nair, B.U.; Ramasami, T. Bioaccumulation of Chromium from Tannery Wastewater: An Approach for Chrome Recovery and Reuse. Environ. Sci. Technol. 2004, 38, 300–306.
- Chhikara, S.; Hooda, A.; Rana, L.; Dhankhar, R. Chromium (VI) Biosorption by Immobilized Aspergillus niger in Continuous Flow System with Special Reference to FTIR Analysis. J. Environ. Biol. 2010, 31, 561–566. Available online: (accessed on 22 March 2020).
- Daneshvar, E.; Zarrinmehr, M.J.; Kousha, M.; Hashtjin, A.M.; Saratale, G.D.; Maiti, A.; Vithanage, M.; Bhatnagar, A. Hexavalent Chromium Removal from Water by Microalgal-Based Materials: Adsorption, Desorption and Recovery Studies. Bioresour. Technol. 2019, 293, 122064.
- da Fontoura, J.T.; Rolim, G.S.; Farenzena, M.; Gutterres, M. Influence of Light Intensity and Tannery Wastewater Concentration on Biomass Production and Nutrient Removal by Microalgae Scenedesmus sp. Process Safe Environ. Prot. 2017, 111, 355–362.
- Mohd Udaiyappan, A.F.; Abu Hasan, H.; Takriff, M.S.; Sheikh Abdullah, S.R. A Review of the Potentials, Challenges and Current Status of Microalgae Biomass Applications in Industrial Wastewater Treatment. J. Water Process Eng. 2017, 20, 8–21.
- Crini, G.; Lichtfouse, E. Advantages and Disadvantages of Techniques Used for Wastewater Treatment. Environ. Chem. Lett. 2019, 17, 145–155.
- Pena, A.d.C.C.; Bertoldi, C.F.; da Fontoura, J.T.; Trierweiler, L.F.; Gutterres, M. Consortium of Microalgae for Tannery Effluent Treatment. Braz. Arch. Biol. Technol. 2019, 62.
- Quintero-Dallos, V.; García-Martínez, J.B.; Contreras-Ropero, J.E.; Barajas-Solano, A.F.; Barajas-Ferrerira, C.; Lavecchia, R.; Zuorro, A. Vinasse as a Sustainable Medium for the Production of Chlorella vulgaris UTEX 1803. Water 2019, 11, 1526.
- Guiza-Franco, L.; Orozco-Rojas, L.G.; Sánchez-Galvis, E.M.; García-Martínez, J.B.; Barajas-Ferreira, C.; Zuorro, A.; Barajas-Solano, A.F. Production of Chlorella vulgaris Biomass on Uv-Treated Wastewater as an Alternative for Environmental Sustainability on High-Mountain Fisheries. Chem. Eng. Trans. 2018, 64, 517–522.
- Garcia-Martinez, J.B.; Urbina-Suarez, N.A.; Zuorro, A.; Barajas-Solano, A.F.; Kafarov, V. Fisheries Wastewater as a Sustainable Media for the Production of Algae-Based Products. Chem. Eng. Trans. 2019, 76, 1339–1344.
- Estévez-Landazábal, L.L.; Barajas-Solano, A.F.; Barajas-Ferreira, C.; Kafarov, V. Improvement of lipid productivity on Chlorella vulgaris using waste glycerol and sodium acetate. CTF Cienc. Tecnol. Futuro 2013, 5, 113–126. Available online: (accessed on 22 March 2021).
- Yen, H.-W.; Chen, P.-W.; Hsu, C.-Y.; Lee, L. The Use of Autotrophic Chlorella vulgaris in Chromium (VI) Reduction under Different Reduction Conditions. J. Taiwan Inst. Chem. Eng. 2017, 74, 1–6.
- Garcia-Martinez, B.; Ayala-Torres, E.; Reyes-Gomez, O.; Zuorro, A.; Barajas-Solano, A.; Barajas-Ferreira, C. Evaluation of a Two-Phase Extraction System of Carbohydrates and Proteins from Chlorella vulgaris Utex 1803. Chem. Eng. Trans. 2016, 49, 355–360.
- Barajas-Solano, A.F.; Guzmán-Monsalve, A.; Kafarov, V. Effect of Carbon-Nitrogen Ratio for the Biomass Production, Hydrocarbons and Lipids on Botryoccus braunii UIS 003. Chem. Eng. Trans. 2016, 49, 247–252.
- Cuéllar-García, D.J.; Rangel-Basto, Y.A.; Urbina-Suarez, N.A.; Barajas-Solano, A.F.; Muñoz-Peñaloza, Y.A. Lipids production from Scenedesmus obliquus through carbon/nitrogen ratio optimization. J. Phys. Conf. Ser. 2019, 1388, 012043.
- Cuéllar-García, D.J.; Rangel-Basto, Y.A.; Barajas-Solano, A.F.; Muñoz-Peñaloza, Y.A.; Urbina-Suarez, N.A. Towards the production of microalgae biofuels: The effect of the culture medium on lipid deposition. BioTechnologia 2019, 100, 273–278.
- Zuorro, A.; Leal-Jerez, A.G.; Morales-Rivas, L.K.; Mogollón-Londoño, S.O.; Sanchez-Galvis, E.M.; García-Martínez, J.B.; Barajas-Solano, A.F. Enhancement of Phycobiliprotein Accumulation in Thermotolerant Oscillatoria sp. through Media Optimization. ACS Omega 2021, 6, 10527–10536.
- Rangel-Basto, Y.A.; García-Ochoa, I.E.; Suarez-Gelvez, J.H.; Zuorro, A.; Barajas-Solano, A.F.; Urbina-Suarez, N.A. The Effect of Temperature and Enzyme Concentration in the Transesterification Process of Synthetic Microalgae Oil. Chem. Eng. Trans. 2018, 64, 331–336.
- Barajas-Solano, A.F.; Gonzalez-Delgado, A.D.; Kafarov, V. Effect of Thermal Pre-Treatment on Fermentable Sugar Production of Chlorella vulgaris. Chem. Eng. Trans. 2014, 37, 655–660.
- Zuorro, A.; García-Martínez, J.B.; Barajas-Solano, A.F. The Application of Catalytic Processes on the Production of Algae-Based Biofuels: A Review. Catalysts 2021, 11, 22.
- Ajayan, K.V.; Selvaraju, M.; Unnikannan, P.; Sruthi, P. Phycoremediation of Tannery Wastewater Using Microalgae Scenedesmus Species. Int. J. Phytoremed. 2015, 17, 907–916.
- Diaz-Angulo, J.; Porras, J.; Mueses, M.; Torres-Palma, R.A.; Hernandez-Ramirez, A.; Machuca-Martinez, F. Coupling of heterogeneous photocatalysis and photosensitized oxidation for diclofenac degradation: Role of the oxidant species. J. Photochem. Photobiol. A Chem. 2019, 383, 112015.
- Song, Z.; Williams, C.J.; Edyvean, R.G.J. Treatment of Tannery Wastewater by Chemical Coagulation. Desalination 2004, 164, 249–259.
- Achouri, O.; Panico, A.; Bencheikh-Lehocine, M.; Derbal, K.; Pirozzi, F. Effect of Chemical Coagulation Pretreatment on Anaerobic Digestion of Tannery Wastewater. J. Environ. Eng. 2017, 143, 4017039.
- Donneys-Victoria, D.; Bermúdez-Rubio, D.; Torralba-Ramírez, B.; Marriaga-Cabrales, N.; Machuca-Martínez, F. Removal of indigo carmine dye by electrocoagulation using magnesium anodes with polarity change. Environ. Sci. Pollut. Res. 2019, 26, 7164–7176.
- Lara-Ramos, J.A.; Saez, C.; Machuca-Martínez, F.; Rodrigo, M.A. Electro-ozonizers: A new approach for an old problem. Sep. Purif. Technol. 2020, 241.
- Suárez Escobar, A.F.; García Ubaque, C.A.; Vaca Bohórquez, M.L. Identificación y evaluación de la contaminación del agua por curtiembres en el municipio de Villapinzón. Tecnura 2012, 16, 185–193.
- Carreño, S.U.F. Diseño y evaluación de un biosistema de tratamiento a escala piloto de aguas de curtiembres a través de la Eichhornia crassipes. Rev. Colomb. Biotecnol. 2016, 18, 74–81.
- Quintero Salamanca, G.P.; Quijano Parra, A.; Melendez Gelvez, I. Efecto genotoxico del agua residual de la curtiembre San Faustino—Norte de Santander—Colombia. Rev. Colomb. Tecnol. Av. 2018, 2, 8–16.
More
Information
Subjects:
Cell Biology
Contributor
MDPI registered users' name will be linked to their SciProfiles pages. To register with us, please refer to https://encyclopedia.pub/register
:
View Times:
1.1K
Revisions:
2 times
(View History)
Update Date:
01 Jun 2021
Notice
You are not a member of the advisory board for this topic. If you want to update advisory board member profile, please contact office@encyclopedia.pub.
OK
Confirm
Only members of the Encyclopedia advisory board for this topic are allowed to note entries. Would you like to become an advisory board member of the Encyclopedia?
Yes
No
${ textCharacter }/${ maxCharacter }
Submit
Cancel
Back
Comments
${ item }
|
More
No more~
There is no comment~
${ textCharacter }/${ maxCharacter }
Submit
Cancel
${ selectedItem.replyTextCharacter }/${ selectedItem.replyMaxCharacter }
Submit
Cancel
Confirm
Are you sure to Delete?
Yes
No





