Your browser does not fully support modern features. Please upgrade for a smoother experience.

Submitted Successfully!
Thank you for your contribution! You can also upload a video entry or images related to this topic.
For video creation, please contact our Academic Video Service.
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Sadanand Pandey | + 3012 word(s) | 3012 | 2021-05-26 05:19:52 | | | |
| 2 | Peter Tang | Meta information modification | 3012 | 2021-05-27 07:41:48 | | | | |
| 3 | Peter Tang | -2 word(s) | 3010 | 2021-06-01 03:55:46 | | | | |
| 4 | Conner Chen | Meta information modification | 3010 | 2021-09-22 03:06:30 | | |
Video Upload Options
We provide professional Academic Video Service to translate complex research into visually appealing presentations. Would you like to try it?
Cite
If you have any further questions, please contact Encyclopedia Editorial Office.
Pandey, S. Nanotechnology in Bladder Cancer. Encyclopedia. Available online: https://encyclopedia.pub/entry/10153 (accessed on 31 March 2026).
Pandey S. Nanotechnology in Bladder Cancer. Encyclopedia. Available at: https://encyclopedia.pub/entry/10153. Accessed March 31, 2026.
Pandey, Sadanand. "Nanotechnology in Bladder Cancer" Encyclopedia, https://encyclopedia.pub/entry/10153 (accessed March 31, 2026).
Pandey, S. (2021, May 27). Nanotechnology in Bladder Cancer. In Encyclopedia. https://encyclopedia.pub/entry/10153
Pandey, Sadanand. "Nanotechnology in Bladder Cancer." Encyclopedia. Web. 27 May, 2021.
Copy Citation
Bladder cancer (BC) is a type of cancer that occurs in the bladder's lining. The urinary system, which filters waste products from your blood and produces urine, involves the bladder. BC is the fourth most common cancer among men and the tenth most common cancer among women. Nanotechnology has been extensively developed for cancer management, including cancer diagnosis, detection, and treatment. Several nanoparticles (NP) can be used in in vitro cancer diagnostics, in vivo imaging enhancement, and drug loading techniques.
Bladder cancer
Nanoparticles
Therapy
Diagnosis
Nanotechnology
1. Introduction
The bladder is an essential organ of the urinary system having major roles in temporary urine storage via immense folded internal linings, as well as its expulsion due to bladder musculature contractions and relaxations [1][2]. The bladder is featured with the apex at the upper location, main body, triangular-shaped posterior fundus, as well as neck for convergence of fundus [3]. BC is the common cancer of the urinary tract, originating inside the linings of the bladder consisting of urothelial cells [4]. Urothelial cells are the source of connection between the kidney and bladder [5]. BC is the fourth most common cancer in men and tenth in women causing 15,000 deaths annually in the USA [6]. BC includes a variety of forms, depending on how it develops in the bladder’s specific cells, i.e., urothelial carcinoma, squamous cell carcinoma, and very rare adenocarcinoma in mucus-secreting cells of the bladder [7]. As far as symptoms of BC are concerned, it is often accompanied by hematuria, frequent painful urination, and pelvic pain [8]. The mechanism besides the propagation of BC lies in the resistant mutations in the tumor suppressor genes [9]. However, other risk factors include smoking, old age, chronic bladder inflammation, inheritance history, and exposure to certain chemicals and dyes [4][10][11][12][13]. As far as a generalized diagnosis of the BC is concerned, it can be initially determined through cystoscopy by inserting a cystoscope tube having a lens into the urethra to examine structural changes. Cystoscopy can be modified via attaching a specialized tool for collecting a sample for biopsy determination. Furthermore, the urine sample can also be opted to collect for analysis of the cancer cells presence [14][15]. After the confirmation of the presence of traces of cancer cells in urine confirming BC. Further preceded diagnostics can also be performed to examine the severity, and it includes CT scan, magnetic resonance imaging (MRI), positron emission tomography (PET), bone scan, and chest X-ray. The diagnosis of BC is challenging owing to the vast existence of gaps accompanying over-testing, over-diagnosis, over-treatment, non-specificity, and heterogeneous nature of malignant BC cells [16]. However, treatment is varied according to the type and severity based on low grade (benign) and high grade (metastatic) BC [8][17]. Therefore, the opted methods for BC therapy include surgery for removing cancer cells directly via transurethral resection of bladder tumor (TURBT), cystectomy, neobladder reconstruction, and ileal conduit [18][19]. Chemotherapy for BC includes the use of combinatorial chemotherapeutic agents in the form of gemcitabine and cisplatin (GC), cisplatin, methotrexate, and vinblastine (CMV), and gemcitabine and paclitaxel (GP) directly in the bladder via intravesical chemotherapy or systemic chemotherapy [17][20][21]. Radiation therapy uses beams of powerful energy, such as X-rays and protons, to destroy the cancer cells [22]. Immunotherapy is the most used treatment protocol for BC via intravesical and intravenous methods [23]. Intravesical immunotherapy can be done using the bacillus Calmette-Guerin (BCG) vaccine to mediate the immune system reaction that directs germ-fighting cells to the bladder [24]. However, intravenous immunotherapy can be performed through various immunotherapy drugs [25]. Nevertheless, all these modalities methods are associated with an increased economic burden, non-patient compliance, and need for targeted delivery, and most importantly with the involvement of virulent factors in tumor suppressor genes [26]. Chemotherapeutics agents utilized in chemotherapy are specifically associated with damaging cells in the bone marrow, intestinal mucosal linings and hair follicles, as well as developing severe infection and fatigue due to the depletion of leukocytes and red blood cells [27]. In terms of a generalized diagnosis of BC, it can be determined first by cystoscopy, which involves inserting a cystoscope tube with a lens into the urethra to investigate structural changes [28]. Cystoscopy can be modified via attaching specialized tool for collecting sample for biopsy determination [29][30]. Furthermore, urine sample can also be opted to collect for analysis of the cancer cells presence. After the confirmation of the presence of traces of cancer cells in urine confirming BC, further preceded diagnostics can also be performed to examine the severity and it includes CT scan, magnetic resonance imaging (MRI), positron emission tomography (PET), bone scan and chest X-ray [31][32][33]. The diagnosis of BC is challenging owing to the vast existence of gaps accompanying over-testing, over-diagnosis, over-treatment, non-specificity, and heterogeneous nature of malignant BC cells [34]. To overcome the limitations of conventional diagnostic methods for BC, a significant number of nanotechnology-based bioassays are highly encouraged [35]. In this regard, fluorescent cystoscopy has been developed using 5 -aminolevulinic acid (5-ALA) phostosensitizer followed by intravesical administration, and have the capability of proficient absorption of cancer cells by showing intense red color compared to surrounding normal tissues [36]. Similarly, ligand mediated approach-based nano-sensors are also of great interest as they can be developed by conjugating BC specified amino acid-based ligand PLZ4. PLZ4 functionalized nanomicelles preferentially enhance the uptake of only cancer cells under the mechanistic of photodynamic diagnosis [37]. Role of gold nanoparticles (GNP) in the diagnosis of BCs by inducing plasmon resonance is irreplaceable and resulting in colors of visible difference to be observed by naked eyes [38]. Cationic GNPs can be utilized with biocompatible anionic hyaluronic acid (HA) to produce visible blue color change [39]. Ultra-small particles of iron oxide (USPIO) ranging from 30 to 50 nm in diameter have the capability of uptake by reticuloendothelial system for ultra-sensitive diagnosis [40].
Nanotechnology advents the field of anti-cancer modalities via improving the drug loading by decorating the surface of nanoparticles with targeted receptor ligands, highly expressed on tumor surfaces [41][42][43][44][45][46][47][48]. Nanomaterials bypass the side effects of conventional therapy by improving the specificity and pharmacokinetics of anti-cancer drugs [49][50][51][52]. Anti-cancer NPs for treating BC are GNPs and they modified the acids and protein molecules for facilitating the rapid killing of cancer cells [53]. GNPs help in providing stability, as well as a strong affinity for attachment of ligands targeting BC [54]. Most effectively utilized nanocarriers in the treatment of BC include, polymeric nanoparticles because their synthesis is easy and cost-effective, provides superior viability and biodegradation [55]. Polymeric nanoparticles utilized in the treatment of BC are available in a wide range of natural and synthetic polymers, constituting macromolecules poly (lactide-coglycolide), poly (lactic acid), poly (caprolactone), and chitosan [55][56][57][58][59][60][61][62]. Similarly, the lipid-based nanoparticles are also imparting their role in treating BC by adapting formulation strategic forms of solid lipid nanoparticles (SLNs) and nanostructured lipid carriers (NLC) composed of phospholipids [63]. Lipid-based nanoparticles are highly advantageous in encapsulating anti-cancer drugs and their site-specific loading via overcoming their solubility issues [64]. Moreover, protein nanoparticles help in facilitating the targeted and controlled release of oral delivery using proteins such as albumin, gelatin, gliadin, and legumin [65].
2. Diagnosis of Bladder Cancer
2.1. Current Diagnosis Approaches of Bladder Cancer
Currently, different approaches such as cystoscopy, biopsy, urinary cytology, and imaging procedures are used to detect BC [66]. A thin, narrow tube (cystoscope) is placed through the urethra to perform cystoscopy. The cystoscope has a lens that helps to inspect inside of the urethra and bladder for signs of disease [67]. Urine cytology is a process in which a sample of urine is examined under a microscope to monitor for cancer problems [68]. Finally, imaging examinations such as computerized tomography (CT) urogram, magnetic resonance imaging (MRI), positron emission tomography (PET), bone scan, and chest X-ray can help with early diagnosis and influence care decisions [69].
Traditional methods have limited sensitivity and capacity to provide accurate and specific information on the condition, and they rely greatly on the size shift of lymph nodes and the presence of anatomic anomalies, which is typically the primary criteria used to determine the diagnosis [29][70]. Nanoparticles (NP) with a nanometric scale are novel materials that were first used to develop scanning probe microscopy and discover molecular structures, and since been used in a variety of diagnostic applications [71].
2.2. Current Biomarkers and Tests in Bladder Cancer Diagnosis
The identification of biomarkers in urine, tissue, and blood has been suggested as important components of precision medicine to address existing shortcomings in the diagnosis, treatment, and follow-up of BC. These modern molecular tests can aid in detecting disease earlier, risk-stratifying patients, improving oncological outcome prediction, and optimizing target therapies. We looked at the existing state of the art and the success of the most promising and accessible biomarkers. BTA stat®, BTA TRAK®, NMP22, and UroVysionTM are the only urinary diagnosis systems approved by the Food and Drug Administration (FDA) for BC diagnosis and follow-up. Meanwhile, ImmunoCytTM/uCyt+TM is only approved for BC follow-up [72]. Telomerase, nuclear matrix protein 22 (NMP22), cytokeratin 19, survivin, hyaluronidase (HAase), apolipoprotein A1 (ApoA1), miRNA-21, and galectin-1 protein, on the other hand, are effective biomarkers in the diagnosis of BC [73][74].
Despite some promising findings, particularly in terms of improved sensitivity, when compared to urinary cytology in the diagnostic environment. These molecules have yet to be incorporated into routine clinical practice, owing to the difficulty in deciding the required scenario for use and the lack of high-quality prospective trials. That resulting in a low level of proof. To define the position of these promising biomarkers, more prospective research and broad international collaborations are needed [75]. Nanotechnology has been extensively developed for cancer management, including cancer diagnosis, identification, and treatment. Nanoparticles for cancer treatment have a plethora of potential uses, but most are still in the preclinical stage. GNPs, for example, would be used in cancer screening in vitro studies, in vivo imaging improvement, and as potential drug loading strategies. Aside from that, different preparation methods alter particle sizes, shapes, and structures, reducing their clinical utility [76][77][78][79].
2.3. Nanomaterials for Imaging Approach
Current imaging methods, such as computed tomography and magnetic resonance imaging (MRI), rely heavily on the size shift of lymph nodes or the presence of anatomic anomalies as the primary criteria for diagnosis [80]. Nanoplatform of contrast agents has shown some promising results in imaging approaches to address this problem [81].
BC, as previously mentioned, has the highest recurrence rate of all cancers, owing in part to insufficient transurethral resection. The failure of cystoscopes to identify invisible lesions during the resection process results in inadequate resection [82]. Davis et al evaluated the ability of the endoscope approach and surface-enhanced Raman NPs to detect bladder tissue as cancerous or normal (Figure 1) [83]. After topically administration of NPs to human bladder tissue samples, both tissue permeability-based (passive) targeting and antibody-based (active) targeting was assayed. The receiver operating characteristic region under the curve (ROC AUC), for multiplexed molecular imaging of CD47 and Carbonic Anhydrase 9 tumor proteins was 0.93. (0.75, 1.00). Furthermore, with a ROC AUC of 0.93, passively targeted NPs allowed tissue classification. As compared to standard bladder urothelium, passively targeted nanoparticles penetrated five-time deeper and attached to tumor tissue at 3.3-time higher concentrations in cancer. Indicating that there is an improved surface permeation and retaining function in human BC.
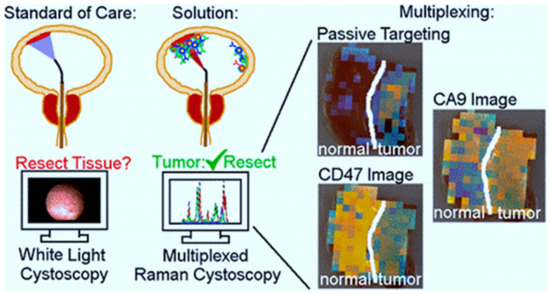
Figure 1. The use of surface-enhanced Raman scattering nanoparticle (NPs) for multiplexed imaging of bladder cancer tissue [83].
Quantum dots (QD) are fluorescent nanoparticles with superior optical properties compared to organic dyes, but in vivo use of them is constrained by the potential for systemic toxicity. Topical administration of targeted nanoparticles is appealing, because it allows for minimal exposure and dose reduction [84]. Pan et al. successfully indicated that topical (i.e., intravesical) administration of QD-conjugated anti-CD47 resulted in effective ex vivo endoscopic imaging of human BC. In addition, they investigated the biodistribution and toxicity of free QD, and anti-CD47-QD in mice in vivo [85]. Anti-CD47-QD biodistribution in vivo was measured using inductively coupled plasma mass spectrometry. While some mice showed extravesical biodistribution of QD, indicating a route for systemic exposure under some conditions, there was no substantial aggregation of QD outside of the bladder on average. Up to 7 days after injection, there were no signs of high toxicity. Intravesical administering of targeted NPs can minimize systemic exposure, but for clinical use, NPs with well-established bio - safety characteristics should be used to reduce long-term contamination in situations of drug toxicity.
The use of optical imaging technology in conjunction with cancer-specific molecular imaging agents may be an effective solution for promoting cancer detection and enabling image-guided surgical [86]. The most common endoscopic treatment for BC is white light cystoscopy, which has a low detection ability. Developing optical imaging innovations have a lot of promise for better diagnostic precision, but there are not any imaging agents for molecular specificity yet [87]. Pan et al demonstrated strong detection of BC with clinical-grade fluorescence imaging approaches, confocal endomicroscopy, and blue light cystoscopy in fresh surgically extracted human bladders using fluorescently labeled CD47 antibody (anti-CD47) as a molecular diagnostic agent [88]. The specificity and sensitivity for CD47-targeted imaging with blue light cystoscopy were 82.9% and 90.5%, respectively. Based on the results of this study, CD47-targeted molecular imaging could help with BC diagnosis and resection precision.
Despite numerous resections and long-term chemo and immunotherapy, most patients with non-muscle invasive BC undergo recurrence or worsening, necessitating cystectomy and a poor prognosis. Unfinished resection and reimplantation of cancer cells are potential causes, which could be avoided with better resection and adjuvant treatment [89]. Liang et al produced a guided drug for urothelial carcinoma identification, fluorescence-guided resection, and deep-penetrating adjuvant photodynamic therapy (UC) [90]. The method was based on up-conversion nanoparticles (UCNP), which can hold a photosensitizer and convert deep-penetrating near-infrared light into high-energy visible light, which is needed for tumor interpretation as well as the development of reactive oxygen species in the photosensitizer. The mean photoluminescence of cells in the targeted group was 5 to 8 times higher than in control groups as a result of the labeling, makes it for quick detection of positive cells with low background auto-fluorescence.
QDs are commonly used in biomedical fields as fluorescent labels. Yuan et al developed quantum dot (QD) fluorescent probes that were linked to a monoclonal antibody against prostate stem cell antigen (PSCA) (QD-PSCA). The targeted imaging of QD-PSCA probes in EJ human bladder urothelial cancer cells was investigated using this nanoplatform. They also looked into the efficacy of non-invasive tumor-targeted imaging using these QDs in vivo conditions. By chemical covalent linking, QDs with an emission wavelength of 605 nm (QD605) was covalently linked with PSCA to produce QD605-PSCA fluorescent probes. An ultraviolet spectrophotometer and a fluorescence spectrophotometer were used to test and determine the optical properties of the probes coupled and uncoupled with PSCA. To identify and interpret imaging of the probes for EJ cells, direct immune-fluorescent labeling was used. The findings showed that QD605-PSCA probes maintained QD605’s fluorescent properties, as well as the PSCA protein’s immunogenicity. The probes were able to identify the PSCA protein expressed in BC cells with high specificity, and the fluorescence was stable and long lasting. QD-PSCA fluorescent probes may be helpful for precise targeted labeling and imaging in bladder urothelial cancer cells, according to this report. The probes also have good optical durability, making them suitable for non-invasive targeted imaging, early detection, and targeted in vivo tumor therapy testing.
2.4. Nanomaterials in Biosensor Development
Researchers identified promising urinary biomarkers for the detection of BC as a result of their efforts to reduce the costs of BC diagnosis and improve patients’ quality of life by removing unnecessarily invasive diagnostic tests [91][92][93][94][95][96][97]. The potential biomarker for early diagnosis of BC is telomerase, nuclear matrix protein 22 (NMP22), cytokeratin 19, survivin, hyaluronidase (HAase), apolipoprotein A1 (ApoA1), miRNA-21, galectin-1 protein, etc. [98]. Biosensors for identifying urinary biomarkers have been established in tandem with their discovery, and they can give low detection limits, a large linear response range, high stability, and high accuracy [99].
3. Applications of Nanomaterials in Treatment of BC
As mentioned earlier, BC is considered one of the most recurrent urogenital cancers in the world [100]. BC is an epithelial carcinoma that unusual cells in the epithelial lining multiply without any control. The most frequent histological class of BC is transitional cell carcinoma (TCC), also named urothelial cell carcinoma (UCC) [41]. Several methods are generally used to treatments of patients who suffered from BC, including chemotherapy, surgery of tumor, radiotherapy, immune therapy, stem cell transformation, and radical cystectomy. However, as we know, these methods will have many side effects such as restricted bioavailability, toxicity, non-selectivity, fast clearance, and limitation in metastasis. Therefore, researchers are studying to find newer treatment methods with fewer side effects and more therapeutic effects [101]. Generally, BC is a very heterogeneous and complex disease with various biological subtypes. So, it has many challenges in grading, classification. Approximately, 70% of patients who suffered from bladder UCC show a superficial carcinoma named non-muscle-invasive BC (NMIBC), while the other patients (30%) progress a muscle-invasive carcinoma (MIBC) bearing the danger of the metastatic spread of the tumor [102]. The most common treatment for BC is surgery, but the findings have been shown that in approximately 80% of patients who underwent surgery, the tumor recurred after 5 years. Therefore, chemotherapy is still considered the main and important treatment in the inhibition of tumor recurrence and progression. Although chemotherapeutic agents, such as taxanes, cisplatin, gemcitabine, etc., facilitate prolong-term survival in many BC patients, the great recurrence rate of the tumor and serious adverse outcomes of therapeutic agents remain for BC therapy. Therefore, more effective and novel management is necessary to increase the quality and quantity of life of the BC patients [102].
Nowadays, nanotechnology has been dramatically helped us to diagnose and treat a wide variety of cancers like that BC. Recently, several nanoparticles (NP) such as polymeric NPs, lipid NPs, metallic NPs have been used to help BC therapy. Various forms of NPs can increase the solubility of drugs with weakly soluble, and multi-functional NPs have acceptable results against renal, bladder, prostate cancer. NPs are also utilized as a drug delivery system (DD) to increase effects and interactions between drugs and the urothelium. Furthermore, nanotechnology can also associate with other modern technologies to advance enhance effectively [103]. Here, we intend to focus on nanotechnologies with potential and useful applications in BC treatment, also we will investigate several nanomaterials as nanocarriers to reduce side effects and enhance the effect of chemotherapy drugs to improve treatment of BC.
References
- Compérat, E.; Varinot, J.; Moroch, J.; Eymerit-Morin, C.; Brimo, F. A practical guide to bladder cancer pathology. Nat. Rev. Urol. 2018, 15, 143–154.
- Sanli, O.; Dobruch, J.; Knowles, M.A.; Burger, M.; Alemozaffar, M.; Nielsen, M.E.; Lotan, Y. Bladder cancer. Nat. Rev. Dis. Primers 2017, 3, 1–19.
- Mossanen, M.; Macleod, L.C.; Chu, A.; Wright, J.L.; Dalkin, B.; Lin, D.W.; True, L.; Gore, J.L. Comparative effectiveness of a patient centered pathology report for bladder cancer care. J. Urol. 2016, 196, 1383–1389.
- Cumberbatch, M.G.K.; Jubber, I.; Black, P.C.; Esperto, F.; Figueroa, J.D.; Kamat, A.M.; Kiemeney, L.; Lotan, Y.; Pang, K.; Silverman, D.T. Epidemiology of bladder cancer: A systematic review and contemporary update of risk factors in 2018. Eur. Urol. 2018, 74, 784–795.
- Dolz, J.; Xu, X.; Rony, J.; Yuan, J.; Liu, Y.; Granger, E.; Desrosiers, C.; Zhang, X.; Ben Ayed, I.; Lu, H. Multiregion segmentation of bladder cancer structures in MRI with progressive dilated convolutional networks. Med Phys. 2018, 45, 5482–5493.
- Zhang, S.; Yu, Y.-H.; Zhang, Y.; Qu, W.; Li, J. Radiotherapy in muscle-invasive bladder cancer: The latest research progress and clinical application. Am J Cancer Res 2015, 5, 854–868.
- Zhao, X.; Qi, T.; Kong, C.; Hao, M.; Wang, Y.; Li, J.; Liu, B.; Gao, Y.; Jiang, J. Photothermal exposure of polydopamine-coated branched Au–Ag nanoparticles induces cell cycle arrest, apoptosis, and autophagy in human bladder cancer cells. Int. J. Nanomed. 2018, 13, 6413.
- Alanee, S.; Alvarado-Cabrero, I.; Murugan, P.; Kumar, R.; Nepple, K.G.; Paner, G.P.; Patel, M.I.; Raspollini, M.R.; Lopez-Beltran, A.; Konety, B.R. Update of the International Consultation on Urological Diseases on bladder cancer 2018: Non-urothelial cancers of the urinary bladder. World J. Urol. 2019, 37, 107–114.
- Joyce, C.; Rayi, A.; Kasi, A. Cancer, Tumor-Suppressor Genes. 2018. Available online: (accessed on 31 March 2021).
- Inamura, K. Bladder cancer: New insights into its molecular pathology. Cancers 2018, 10, 100.
- Roghani, A.; Panahi, S. Does Telemedicine Reduce health disparities? Longitudinal Evidence during the COVID-19 Pandemic in the US. medRxiv 2021.
- Roghani, A.; Panahi, S. The global distribution of COVID-19 vaccine: The role of macro-socioeconomics measures. medRxiv 2021.
- Roghani, A. The Influence of Covid-19 Vaccine on Daily Cases, Hospitalization, and Death Rate in Tennessee: A Case Study in the United States. medRxiv 2021.
- Wiener, H.; Mian, C.; Haitel, A.; Pycha, A.; Schatzl, G.; Marberger, M. Can urine bound diagnostic tests replace cystoscopy in the management of bladder cancer? J. Urol. 1998, 159, 1876–1880.
- Witjes, J.A.; Douglass, J. The role of hexaminolevulinate fluorescence cystoscopy in bladder cancer. Nat. Clin. Pract. Urol. 2007, 4, 542–549.
- Cheung, G.; Sahai, A.; Billia, M.; Dasgupta, P.; Khan, M.S. Recent advances in the diagnosis and treatment of bladder cancer. Bmc Med. 2013, 11, 1–8.
- Crabb, S.J.; Douglas, J. The latest treatment options for bladder cancer. Br. Med Bull. 2018, 128, 85–95.
- Li, K.; Xu, Y.; Tan, M.; Xia, S.; Xu, Z.; Xu, D. A retrospective comparison of thulium laser en bloc resection of bladder tumor and plasmakinetic transurethral resection of bladder tumor in primary non-muscle invasive bladder cancer. Lasers Med Sci. 2019, 34, 85–92.
- Gregg, J.R.; McCormick, B.; Wang, L.; Cohen, P.; Sun, D.; Penson, D.F.; Smith, J.A.; Clark, P.E.; Cookson, M.S.; Barocas, D.A. Short term complications from transurethral resection of bladder tumor. Can. J. Urol. 2016, 23, 8198–8203.
- Booth, C.M.; Karim, S.; Brennan, K.; Siemens, D.R.; Peng, Y.; Mackillop, W.J. Perioperative chemotherapy for bladder cancer in the general population: Are practice patterns finally changing? Urol. Oncol. 2018, 36, 89.e13-89.e20.
- Rouprêt, M.; Neuzillet, Y.; Masson-Lecomte, A.; Colin, P.; Compérat, E.; Dubosq, F.; Houede, N.; Larre, S.; Pignot, G.; Puech, P. CCAFU french national guidelines 2016-2018 on bladder cancer. Prog. Urol. 2016, 27, S67–S91.
- Tree, A.C.; Jones, K.; Hafeez, S.; Sharabiani, M.T.A.; Harrington, K.J.; Lalondrelle, S.; Ahmed, M.; Huddart, R.A. Dose-limiting urinary toxicity with pembrolizumab combined with weekly hypofractionated radiation therapy in bladder cancer. Int. J. Radiat. Oncol. Biol. Phys. 2018, 101, 1168–1171.
- Song, D.; Powles, T.; Shi, L.; Zhang, L.; Ingersoll, M.A.; Lu, Y.J. Bladder cancer, a unique model to understand cancer immunity and develop immunotherapy approaches. J. Pathol. 2019, 249, 151–165.
- Pettenati, C.; Ingersoll, M.A. Mechanisms of BCG immunotherapy and its outlook for bladder cancer. Nat. Rev. Urol. 2018, 15, 615–625.
- Wołącewicz, M.; Hrynkiewicz, R.; Grywalska, E.; Suchojad, T.; Leksowski, T.; Roliński, J.; Niedźwiedzka-Rystwej, P. Immunotherapy in bladder cancer: Current methods and future perspectives. Cancers 2020, 12, 1181.
- Wu, E.; Hadjiiski, L.M.; Samala, R.K.; Chan, H.-P.; Cha, K.H.; Richter, C.; Cohan, R.H.; Caoili, E.M.; Paramagul, C.; Alva, A. Deep learning approach for assessment of bladder cancer treatment response. Tomography 2019, 5, 201–208.
- Oun, R.; Moussa, Y.E.; Wheate, N.J. The side effects of platinum-based chemotherapy drugs: A review for chemists. Dalton Trans. 2018, 47, 6645–6653.
- Lotan, Y.; Bivalacqua, T.J.; Downs, T.; Huang, W.; Jones, J.; Kamat, A.M.; Konety, B.; Malmström, P.-U.; McKiernan, J.; O’Donnell, M. Blue light flexible cystoscopy with hexaminolevulinate in non-muscle-invasive bladder cancer: Review of the clinical evidence and consensus statement on optimal use in the USA—update 2018. Nat. Rev. Urol. 2019, 16, 377–386.
- Lodewijk, I.; Dueñas, M.; Rubio, C.; Munera-Maravilla, E.; Segovia, C.; Bernardini, A.; Teijeira, A.; Paramio, J.M.; Suárez-Cabrera, C. Liquid biopsy biomarkers in bladder cancer: A current need for patient diagnosis and monitoring. Int. J. Mol. Sci. 2018, 19, 2514.
- Santoni, G.; Morelli, M.B.; Amantini, C.; Battelli, N. Urinary markers in bladder cancer: An update. Front. Oncol. 2018, 8, 362.
- Todenhöfer, T.; Struss, W.J.; Seiler, R.; Wyatt, A.W.; Black, P.C. Liquid biopsy-analysis of circulating tumor DNA (ctDNA) in bladder cancer. Bladder Cancer 2018, 4, 19–29.
- Van Der Pol, C.B.; Chung, A.; Lim, C.; Gandhi, N.; Tu, W.; McInnes, M.D.; Schieda, N. Update on multiparametric MRI of urinary bladder cancer. J. Magn. Reson. Imaging 2018, 48, 882–896.
- Wu, S.; Zheng, J.; Li, Y.; Wu, Z.; Shi, S.; Huang, M.; Yu, H.; Dong, W.; Huang, J.; Lin, T. Development and validation of an MRI-based radiomics signature for the preoperative prediction of lymph node metastasis in bladder cancer. EBioMedicine 2018, 34, 76–84.
- Faba, O.R.; Tyson, M.D.; Artibani, W.; Bochner, B.H.; Burkhard, F.; Gilbert, S.M.; Kälble, T.; Madersbacher, S.; Seiler, R.; Skinner, E.C. Update of the ICUD–SIU International Consultation on Bladder Cancer 2018: Urinary diversion. World J. Urol. 2019, 37, 85–93.
- Arshad, R.; Pal, K.; Sabir, F.; Rahdar, A.; Bilal, M.; Shahnaz, G.; Kyzas, G.Z. A Review of the nanomaterials use for the diagnosis and therapy of Salmonella typhi. J. Mol. Struct. 2021, 129928.
- Grossman, H.B.; Stenzl, A.; Fradet, Y.; Mynderse, L.A.; Kriegmair, M.; Witjes, J.A.; Soloway, M.S.; Karl, A.; Burger, M. Long-term decrease in bladder cancer recurrence with hexaminolevulinate enabled fluorescence cystoscopy. J. Urol. 2012, 188, 58–62.
- Lin, T.-Y.; Zhang, H.; Luo, J.; Li, Y.; Gao, T.; Lara Jr, P.N.; de Vere White, R.; Lam, K.S.; Pan, C.-X. Multifunctional targeting micelle nanocarriers with both imaging and therapeutic potential for bladder cancer. Int. J. Nanomed. 2012, 7, 2793–2804.
- Jazayeri, M.H.; Aghaie, T.; Nedaeinia, R.; Manian, M.; Nickho, H. Rapid noninvasive detection of bladder cancer using survivin antibody-conjugated gold nanoparticles (GNPs) based on localized surface plasmon resonance (LSPR). Cancer Immunol. Immunother. 2020, 69, 1833–1840.
- Liu, R.; Xiao, W.; Hu, C.; Xie, R.; Gao, H. Theranostic size-reducible and no donor conjugated gold nanocluster fabricated hyaluronic acid nanoparticle with optimal size for combinational treatment of breast cancer and lung metastasis. J. Control. Release 2018, 278, 127–139.
- Merinopoulos, I.; Gunawardena, T.; Stirrat, C.; Cameron, D.; Eccleshall, S.C.; Dweck, M.R.; Newby, D.E.; Vassiliou, V.S. Diagnostic Applications of Ultrasmall Superparamagnetic Particles of Iron Oxide for Imaging Myocardial and Vascular Inflammation. JACC Cardiovasc. Imaging 2020.
- Chen, C.-H.; Chan, T.-M.; Wu, Y.-J.; Chen, J.-J. Review: Application of Nanoparticles in Urothelial Cancer of the Urinary Bladder. J. Med Biol. Eng. 2015, 35, 419–427.
- Barani, M.; Bilal, M.; Rahdar, A.; Arshad, R.; Kumar, A.; Hamishekar, H.; Kyzas, G.Z. Nanodiagnosis and nanotreatment of colorectal cancer: An overview. J. Nanoparticle Res. 2021, 23, 1–25.
- Barani, M.; Bilal, M.; Sabir, F.; Rahdar, A.; Kyzas, G.Z. Nanotechnology in ovarian cancer: Diagnosis and treatment. Life Sci. 2020, 118914.
- Barani, M.; Mirzaei, M.; Mahani, M.T.; Nematollahi, M.H. Lawsone-loaded Niosome and its Antitumor Activity in MCF-7 Breast Cancer Cell Line: A Nano-herbal Treatment for Cancer. Daru J. Pharm. Sci. 2018, 1–7.
- Barani, M.; Mirzaei, M.; Torkzadeh-Mahani, M.; Adeli-Sardou, M. Evaluation of carum-loaded niosomes on breast cancer cells: Physicochemical properties, in vitro cytotoxicity, flow cytometric, DNA fragmentation and cell migration assay. Sci. Rep. 2019, 9, 1–10.
- Barani, M.; Mirzaei, M.; Torkzadeh-Mahani, M.; Lohrasbi-Nejad, A.; Nematollahi, M.H. A new formulation of hydrophobin-coated niosome as a drug carrier to cancer cells. Mater. Sci. Eng. C Mater. Biol. Appl. 2020, 113, 110975.
- Barani, M.; Mukhtar, M.; Rahdar, A.; Sargazi, G.; Thysiadou, A.; Kyzas, G.Z. Progress in the Application of Nanoparticles and Graphene as Drug Carriers and on the Diagnosis of Brain Infections. Molecules 2021, 26, 186.
- Barani, M.; Mukhtar, M.; Rahdar, A.; Sargazi, S.; Pandey, S.; Kang, M. Recent Advances in Nanotechnology-Based Diagnosis and Treatments of Human Osteosarcoma. Biosensors 2021, 11, 55.
- Seddighi, N.S.; Salari, S.; Izadi, A.R. Evaluation of antifungal effect of iron-oxide nanoparticles against different Candida species. IET Nanobiotechnol. 2017, 11, 883–888.
- Poor, M.H.S.; Khatami, M.; Azizi, H.; Abazari, Y. Cytotoxic activity of biosynthesized Ag nanoparticles by Plantago major towards a human breast cancer cell line. Rend. Lincei 2017, 28, 693–699.
- Yaghoubi, S.; Karimi, M.H.; Lotfinia, M.; Gharibi, T.; Mahi-Birjand, M.; Kavi, E.; Hosseini, F.; Sineh Sepehr, K.; Khatami, M.; Bagheri, N. Potential drugs used in the antibody–drug conjugate (ADC) architecture for cancer therapy. J. Cell. Physiol. 2020, 235, 31–64.
- Sharifi, F.; Sharififar, F.; Sharifi, I.; Alijani, H.Q.; Khatami, M. Cytotoxicity, leishmanicidal, and antioxidant activity of biosynthesised zinc sulphide nanoparticles using Phoenix dactylifera. IET Nanobiotechnol. 2017, 12, 264–269.
- Sztandera, K.; Gorzkiewicz, M.; Klajnert-Maculewicz, B. Gold Nanoparticles in Cancer Treatment. Mol. Pharm. 2019, 16, 1–23.
- Vines, J.B.; Yoon, J.-H.; Ryu, N.-E.; Lim, D.-J.; Park, H. Gold Nanoparticles for Photothermal Cancer Therapy. Front. Chem. 2019, 7.
- Loras, A.; Suárez-Cabrera, C.; Martínez-Bisbal, M.C.; Quintás, G.; Paramio, J.M.; Martínez-Máñez, R.; Gil, S.; Ruiz-Cerdá, J.L. Integrative metabolomic and transcriptomic analysis for the study of bladder cancer. Cancers 2019, 11, 686.
- Barani, M.; Nematollahi, M.H.; Zaboli, M.; Mirzaei, M.; Torkzadeh-Mahani, M.; Pardakhty, A.; Karam, G.A. In silico and in vitro study of magnetic niosomes for gene delivery: The effect of ergosterol and cholesterol. Mater. Sci. Eng. C Mater. Biol. Appl. 2019, 94, 234–246.
- Barani, M.; Sabir, F.; Rahdar, A.; Arshad, R.; Z Kyzas, G. Nanotreatment and Nanodiagnosis of Prostate Cancer: Recent Updates. Nanomaterials 2020, 10, 1696.
- Barani, M.; Torkzadeh-Mahani, M.; Mirzaei, M.; Nematollahi, M.H. Comprehensive evaluation of gene expression in negative and positive trigger-based targeting niosomes in HEK-293 cell line. Iran. J. Pharm. Res. 2020, 19, 166.
- Bilal, M.; Barani, M.; Sabir, F.; Rahdar, A.; Kyzas, G.Z. Nanomaterials for the treatment and diagnosis of Alzheimer’s disease: An overview. NanoImpact 2020, 100251.
- Das, S.S.; Bharadwaj, P.; Bilal, M.; Barani, M.; Rahdar, A.; Taboada, P.; Bungau, S.; Kyzas, G.Z. Stimuli-responsive polymeric nanocarriers for drug delivery, imaging, and theragnosis. Polymers 2020, 12, 1397.
- Davarpanah, F.; Yazdi, A.K.; Barani, M.; Mirzaei, M.; Torkzadeh-Mahani, M. Magnetic delivery of antitumor carboplatin by using PEGylated-Niosomes. Daru J. Pharm. Sci. 2018, 26, 57–64.
- Ebrahimi, A.K.; Barani, M.; Sheikhshoaie, I. Fabrication of a new superparamagnetic metal-organic framework with core-shell nanocomposite structures: Characterization, biocompatibility, and drug release study. Mater. Sci. Eng. C Mater. Biol. Appl 2018, 92, 349–355.
- Obeid, M.A.; Tate, R.J.; Mullen, A.B.; Ferro, V.A. Chapter 8 - Lipid-based nanoparticles for cancer treatment. In Lipid Nanocarriers for Drug Targeting; Grumezescu, A.M., Ed.; William Andrew Publishing: Oxford, UK, 2018; pp. 313–359.
- Mizrahy, S.; Hazan-Halevy, I.; Landesman-Milo, D.; Peer, D. Advanced strategies in immune modulation of cancer using lipid-based nanoparticles. Front. Immunol. 2017, 8, 69.
- Liang, Y.; Wang, Y.; Wang, L.; Liang, Z.; Li, D.; Xu, X.; Chen, Y.; Yang, X.; Zhang, H.; Niu, H. Self-crosslinkable chitosan-hyaluronic acid dialdehyde nanoparticles for CD44-targeted siRNA delivery to treat bladder cancer. Bioact. Mater. 2021, 6, 433–446.
- Daneshmand, S.; Bazargani, S.T.; Bivalacqua, T.J.; Holzbeierlein, J.M.; Willard, B.; Taylor, J.M.; Liao, J.C.; Pohar, K.; Tierney, J.; Konety, B. Blue light cystoscopy for the diagnosis of bladder cancer: Results from the US prospective multicenter registry. Urol. Oncol. 36, pp. 361.e1–361.e6.
- Nayyar, R.; Dadhwal, R.; Kapil, A.; Pandey, R.M.; Dogra, P. Urethral instillation of povidone-iodine reduces post-cystoscopy urinary tract infection in males: A randomized controlled trial. Sci. Rep. 2020, 10, 1–7.
- Lavery, H.J.; Zaharieva, B.; McFaddin, A.; Heerema, N.; Pohar, K.S. A prospective comparison of UroVysion FISH and urine cytology in bladder cancer detection. BMC Cancer 2017, 17, 1–7.
- Huang, L.; Kong, Q.; Liu, Z.; Wang, J.; Kang, Z.; Zhu, Y. The diagnostic value of MR imaging in differentiating T staging of bladder cancer: A meta-analysis. Radiology 2018, 286, 502–511.
- Masoumnezhad, M.; Rajabi, M.; Chapnevis, A.; Dorofeev, A.; Shateyi, S.; Kargar, N.S.; Nik, H.S. An Approach for the Global Stability of Mathematical Model of an Infectious Disease. Symmetry 2020, 12, 1778.
- Khandan, F.M.; Afzali, D.; Sargazi, G.; Gordan, M. Novel uranyl-curcumin-MOF photocatalysts with highly performance photocatalytic activity toward the degradation of phenol red from aqueous solution: Effective synthesis route, design and a controllable systematic study. J. Mater. Sci. Mater. Electron. 2018, 29, 18600–18613.
- Miyake, M.; Owari, T.; Hori, S.; Nakai, Y.; Fujimoto, K. Emerging biomarkers for the diagnosis and monitoring of urothelial carcinoma. Res. Rep. Urol. 2018, 10, 251.
- Oeyen, E.; Hoekx, L.; De Wachter, S.; Baldewijns, M.; Ameye, F.; Mertens, I. Bladder cancer diagnosis and follow-up: The current status and possible role of extracellular vesicles. Int. J. Mol. Sci. 2019, 20, 821.
- Miyamoto, D.T.; Mouw, K.W.; Feng, F.Y.; Shipley, W.U.; Efstathiou, J.A. Molecular biomarkers in bladder preservation therapy for muscle-invasive bladder cancer. Lancet Oncol. 2018, 19, e683–e695.
- Ye, F.; Zhao, Y.; El-Sayed, R.; Muhammed, M.; Hassan, M. Advances in nanotechnology for cancer biomarkers. Nano Today 2018, 18, 103–123.
- Singh, R. Nanotechnology based therapeutic application in cancer diagnosis and therapy. 3 Biotech 2019, 9, 1–29.
- Jin, C.; Wang, K.; Oppong-Gyebi, A.; Hu, J. Application of Nanotechnology in Cancer Diagnosis and Therapy-A Mini-Review. Int. J. Med Sci. 2020, 17, 2964.
- Zeraati, M.; Kazemzadeh, P.; Barani, M.; Sargazi, G. Selecting the Appropriate Carbon Source in the Synthesis of SiC Nano-Powders Using an Optimized Fuzzy Model. Silicon 2021. Available online: (accessed on 31 March 2021).
- Mirhosseini, H.; Mostafavi, A.; Shamspur, T.; Sargazi, G. Preparation of novel ternary gC 3 N 4/WO 3/ZnO nanocomposite adsorbent with highly effective imidacloprid removal: Optimization design and a controllable systematic study. J. Mater. Sci. Mater. Electron. 2020, 31, 17903–17920.
- Galgano, S.J.; McDonald, A.M.; Rais-Bahrami, S.; Porter, K.K.; Choudhary, G.; Burgan, C.; Bhambhvani, P.; Nix, J.W.; Morgan, D.E.; Li, Y. Utility of 18F-Fluciclovine PET/MRI for Staging Newly Diagnosed High-Risk Prostate Cancer and Evaluating Response to Initial Androgen Deprivation Therapy: A Prospective Single-Arm Pilot Study. Am. J. Roentgenol. 2020.
- Duan, Y.; Xu, Y.; Mao, D.; Liew, W.H.; Guo, B.; Wang, S.; Cai, X.; Thakor, N.; Yao, K.; Zhang, C.J. Photoacoustic and magnetic resonance imaging bimodal contrast agent displaying amplified photoacoustic signal. Small 2018, 14, 1800652.
- Loras, A.; Trassierra, M.; Sanjuan-Herráez, D.; Martínez-Bisbal, M.; Castell, J.; Quintás, G.; Ruiz-Cerdá, J. Bladder cancer recurrence surveillance by urine metabolomics analysis. Sci. Rep. 2018, 8, 1–10.
- Davis, R.M.; Kiss, B.; Trivedi, D.R.; Metzner, T.J.; Liao, J.C.; Gambhir, S.S. Surface-enhanced Raman scattering nanoparticles for multiplexed imaging of bladder cancer tissue permeability and molecular phenotype. Acs Nano 2018, 12, 9669–9679.
- Bruna, N.; Collao, B.; Tello, A.; Caravantes, P.; Díaz-Silva, N.; Monrás, J.; Órdenes-Aenishanslins, N.; Flores, M.; Espinoza-Gonzalez, R.; Bravo, D. Synthesis of salt-stable fluorescent nanoparticles (quantum dots) by polyextremophile halophilic bacteria. Sci. Rep. 2019, 9, 1–13.
- Pan, Y.; Chang, T.; Marcq, G.; Liu, C.; Kiss, B.; Rouse, R.; Mach, K.E.; Cheng, Z.; Liao, J.C. In vivo biodistribution and toxicity of intravesical administration of quantum dots for optical molecular imaging of bladder cancer. Sci. Rep. 2017, 7, 1–9.
- Tummers, W.S.; Warram, J.M.; Tipirneni, K.E.; Fengler, J.; Jacobs, P.; Shankar, L.; Henderson, L.; Ballard, B.; Pfefer, T.J.; Pogue, B.W. Regulatory aspects of optical methods and exogenous targets for cancer detection. Cancer Res. 2017, 77, 2197–2206.
- Mari, A.; Abufaraj, M.; Gust, K.M.; Shariat, S.F. Novel endoscopic visualization techniques for bladder cancer detection: A review of the contemporary literature. Curr. Opin. Urol. 2018, 28, 214–218.
- Pan, Y.; Volkmer, J.-P.; Mach, K.E.; Rouse, R.V.; Liu, J.-J.; Sahoo, D.; Chang, T.C.; Metzner, T.J.; Kang, L.; Van De Rijn, M. Endoscopic molecular imaging of human bladder cancer using a CD47 antibody. Sci. Transl. Med. 2014, 6, ra148–ra260.
- Bellmunt, J.; Kim, J.; Reardon, B.; Perera-Bel, J.; Orsola, A.; Rodriguez-Vida, A.; Wankowicz, S.A.; Bowden, M.; Barletta, J.A.; Morote, J. Genomic predictors of good outcome, recurrence, or progression in high-grade t1 non–muscle-invasive bladder cancer. Cancer Res. 2020, 80, 4476–4486.
- Liang, L.; Care, A.; Sunna, A.; Campbell, D.; Walsh, B.; Zvyagin, A.; Gillatt, D.; Polikarpov, D. Abstract B27: Photoluminescent nanoconjugates for molecular imaging of bladder cancer. AACR 2020.
- Ghazy, E.; Kumar, A.; Barani, M.; Kaur, I.; Rahdar, A.; Behl, T. Scrutinizing the Therapeutic and Diagnostic Potential of Nanotechnology in Thyroid Cancer: Edifying drug targeting by nano-oncotherapeutics. J. Drug Deliv. Sci. Technol. 2020, 102221.
- Ghazy, E.; Rahdar, A.; Barani, M.; Kyzas, G.Z. Nanomaterials for Parkinson disease: Recent progress. J. Mol. Struct. 2020, 129698.
- Hajizadeh, M.R.; Maleki, H.; Barani, M.; Fahmidehkar, M.A.; Mahmoodi, M.; Torkzadeh-Mahani, M. In vitro cytotoxicity assay of D-limonene niosomes: An efficient nano-carrier for enhancing solubility of plant-extracted agents. Res. Pharm. Sci. 2019, 14, 448.
- Hajizadeh, M.R.; Parvaz, N.; Barani, M.; Khoshdel, A.; Fahmidehkar, M.A.; Mahmoodi, M.; Torkzadeh-Mahani, M. Diosgenin-loaded niosome as an effective phytochemical nanocarrier: Physicochemical characterization, loading efficiency, and cytotoxicity assay. Daru J. Pharm. Sci. 2019, 27, 329–339.
- Hasanein, P.; Rahdar, A.; Barani, M.; Baino, F.; Yari, S. Oil-in-water microemulsion encapsulation of antagonist drugs prevents renal ischemia-reperfusion injury in rats. Appl. Sci. 2021, 11, 1264.
- Mukhtar, M.; Bilal, M.; Rahdar, A.; Barani, M.; Arshad, R.; Behl, T.; Brisc, C.; Banica, F.; Bungau, S. Nanomaterials for Diagnosis and Treatment of Brain Cancer: Recent Updates. Chemosensors 2020, 8, 117.
- Nikazar, S.; Barani, M.; Rahdar, A.; Zoghi, M.; Kyzas, G.Z. Photo-and Magnetothermally Responsive Nanomaterials for Therapy, Controlled Drug Delivery and Imaging Applications. ChemistrySelect 2020, 5, 12590–12609.
- Zvereva, M.; Pisarev, E.; Hosen, I.; Kisil, O.; Matskeplishvili, S.; Kubareva, E.; Kamalov, D.; Tivtikyan, A.; Manel, A.; Vian, E. Activating telomerase TERT promoter mutations and their application for the detection of bladder cancer. Int. J. Mol. Sci. 2020, 21, 6034.
- Yang, Y.; Zeng, B.; Li, Y.; Liang, H.; Yang, Y.; Yuan, Q. Construction of MoS 2 field effect transistor sensor array for the detection of bladder cancer biomarkers. Sci. China Chem. 2020, 63, 997–1003.
- Xiong, Q.; Liu, A.; Ren, Q.; Xue, Y.; Yu, X.; Ying, Y.; Gao, H.; Tan, H.; Zhang, Z.; Li, W. Cuprous oxide nanoparticles trigger reactive oxygen species-induced apoptosis through activation of erk-dependent autophagy in bladder cancer. Cell Death Dis. 2020, 11, 1–13.
- Miyata, Y.; Matsuo, T.; Ohba, K.; Mitsunari, K.; Mukae, Y.; Otsubo, A.; Harada, J.; Matsuda, T.; Kondo, T.; Sakai, H. Present Status, Limitations and Future Directions of Treatment Strategies Using Fucoidan-Based Therapies in Bladder Cancer. Cancers 2020, 12, 3776.
- Yu, K.; Liu, M.; Dai, H.; Huang, X. Targeted drug delivery systems for bladder cancer therapy. J. Drug Deliv. Sci. Technol. 2020, 56, 101535.
- He, M.-H.; Chen, L.; Zheng, T.; Tu, Y.; He, Q.; Fu, H.-L.; Lin, J.-C.; Zhang, W.; Shu, G.; He, L. Potential applications of nanotechnology in urological cancer. Front. Pharmacol. 2018, 9, 745.
More
Information
Subjects:
Oncology
Contributor
MDPI registered users' name will be linked to their SciProfiles pages. To register with us, please refer to https://encyclopedia.pub/register
:
View Times:
820
Revisions:
4 times
(View History)
Update Date:
22 Sep 2021
Notice
You are not a member of the advisory board for this topic. If you want to update advisory board member profile, please contact office@encyclopedia.pub.
OK
Confirm
Only members of the Encyclopedia advisory board for this topic are allowed to note entries. Would you like to become an advisory board member of the Encyclopedia?
Yes
No
${ textCharacter }/${ maxCharacter }
Submit
Cancel
Back
Comments
${ item }
|
More
No more~
There is no comment~
${ textCharacter }/${ maxCharacter }
Submit
Cancel
${ selectedItem.replyTextCharacter }/${ selectedItem.replyMaxCharacter }
Submit
Cancel
Confirm
Are you sure to Delete?
Yes
No





