1000/1000
Hot
Most Recent

Codeine is derived from morphine, an opioid analgesic, and has weaker analgesic and sedative effects than the parent molecule. This weak opioid is commonly used in combination with other drugs for over-the-counter cough relief medication. Due to the psychoactive properties of opioid drugs, the easily obtained codeine often becomes subject to misuse. Codeine misuse has emerged as a concerning public health issue due to its associated adverse effects such as headache, nausea, vomiting, and hemorrhage. Thus, it is very important to develop reliable analytical techniques to detect codeine for both quality control of pharmaceutical formulations and identifying drug misuse in the community. This review aims to provide critical outlooks on analytical methods napplicable to the determination of codeine.
Codeine (3-methylmorphine) is an alkaloid prepared from the methylation of morphine derived from poppy seeds (Papaver somniferum) [1]. It is used to manage mild to moderate pain, including chronic cancer pain, and has antitussive, antistress, and antidiarrheal properties [2][3][4]. In the body, a small amount of codeine is metabolized to form morphine (its active metabolite). Until now, the precise mechanism of action of codeine is not known; however, similar to morphine, codeine can bind to opioid receptors in the brain and induce signaling processes throughout the brain and the rest of the body. Codeine also has a sedative effect which helps reduce the pain sensation [1]. The drug has also been used in combination with acetaminophen or aspirin for more effective pain relief [1].
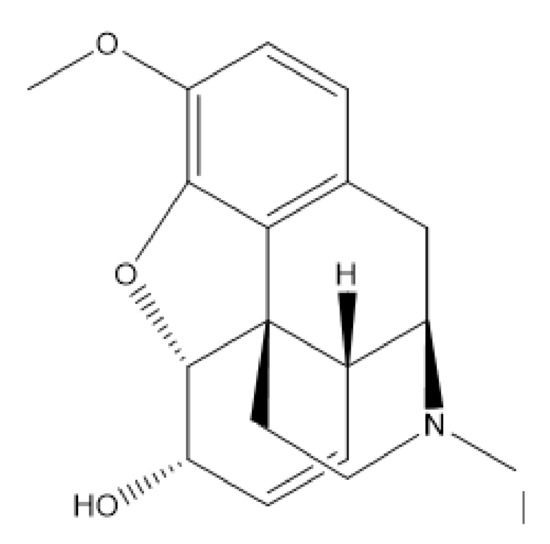
The chemical structure of codeine is given in Figure 1. It has a benzene ring with heteroatoms bound to the ring which is capable of absorbing light energy in the ultraviolet (UV) range, allowing this compound to be analyzed by UV spectrophotometry [5]. There are various forms of codeine salts and hydrates including codeine phosphate, codeine hydrobromide, codeine N-oxide, codeine monohydrate, codeine phosphate sesquihydrate, codeine hydrochloride, and codeine phosphate hemihydrate [6]. Codeine monohydrate is a codeine base that has colorless crystals and is odorless, slightly soluble in water, and freely soluble in ethanol [7]. Codeine phosphate is often used for pharmaceutical formulations. It is a crystalline powder that is odorless, soluble in water, and slightly soluble in ethanol [7].
As codeine (especially its combination with non-opiate analgesics) can be easily obtained over the counter and has a psychoactive effect, it is often misused. Misuse of opioid analgesics including codeine has become an emerging public health concern due to its associated adverse effects on human health [8]. Reliable methods for detecting codeine in various samples are critical for both quality assurance of the drug within pharmaceutical formulations and identification of potential misuse in the community. Various methods have been reported for qualitative and quantitative analysis of codeine [1][9]. Qualitative or simple yes/no analysis often serves as a rapid test to detect the drug presence. This can be achieved via colorimetric assays which provide easy-to-interpret visual results [10]. Codeine can be quantified optically using spectrophotometric methods due to the presence of the chromophore group [11]. This drug is also electro-active, enabling electrochemical measurements for drug quantification [12]. However, the complexity of the matrices often poses a challenge to codeine detection, and thus rigorous sample preparation and/or coupling with separation techniques may be required to achieve accurate determination of the drug. This review aims to assess various analytical methods for codeine detection in the literature and provide critical outlooks on their advantages/disadvantages for analyzing codeine in different sample types. We selected seminal papers on the topic (regardless of the year of publication) to provide a broad overview on applicable methods, with examples (if applicable) provided by more recent publications (last 10 years). We start by evaluating simple colorimetric approaches for qualitative detection and then present two detection methods often used for codeine quantification: optical/spectrophotometric and electrochemical methods. Hyphenated detection methods with separation techniques including chromatography and electromigration-based separation are reviewed next before we finish with conclusions.
Colorimetric assays offer simple visual readouts and do not require complicated instruments for the analysis [10]. The United Nations International Drug Control Programme recommends several colorimetric assays for codeine detection which include the Marquis, Mecke, nitric acid, and ferric sulfate tests [13]. Froehde’s reagent can also be used to identify morphine and codeine. The Marquis and Mecke reagents give a violet color in the presence of opiates including morphine, codeine, and heroin [14]. The detailed color responses from various colorimetric reagents for opiates are shown in Table 1. Codeine reacts with Lieberman’s reagent, producing a black color; with Mandelin’s reagent, producing a green color; and it produces a violet color in the Marquis test [15]. Codeine is also identified by nitric acid reagent by producing an initial orange color that slowly changes to yellow [13]. Nitric acid and Mandelin’s reagents can be used to differentiate codeine from other opiates due to the noticeably different colors they produce for different opiates. Although these colorimetric methods are simple and rapid, most of the chromogenic reagents give similar color responses in the presence of different opioid drugs, making these techniques lack selectivity [16].
Table 1. Color responses of different chromogenic reagents for detecting codeine, heroin, and morphine.
| Compound | Codeine | Heroin | Morphine | Ref. |
|---|---|---|---|---|
| Marquis | violet | violet | violet | [15] |
| Mecke | blue to green | blue to green | blue to green | [15][17] |
| Froehde | light green | n/a | violet to grey | [15][18] |
| Mandelin | green to blue | blue-grey | light grey | [15] |
| Lieberman | Black | n/a | black | [15] |
| Ferric sulfate | n/a | n/a | red | [13] |
| Nitric acid | orange slowly changing to yellow | yellow slowly changing to light green | orange rapidly changing to red then slowly to yellow | [13] |
| AuNPs | green | - | - | [19] |
A rapid and selective colorimetric method for the detection of codeine sulfate has been developed using unmodified gold nanoprobes by Lodha et al. [19]. Citrate-stabilized gold nanoparticles (AuNPs) were synthesized and could react with codeine sulfate to produce a green color. The kinetics of AuNPs aggregation in the presence of codeine sulfate were obtained by measuring the absorbance and color intensity at the red, green, and blue channels as well as the total RGB. The results showed that there is a significant change in absorbance intensity from 520 to 582 nm upon increasing the concentration of codeine sulfate [19].
While limitations exist within the discussed colorimetric methods, they are still useful techniques for screening samples in large quantities, especially for those in resource-limited settings. In addition, it is possible to improve selectivity or even embedding specificity into the colorimetric approach by using specific recognition elements such as enzymes, antibodies, aptamers, or molecularly imprinted polymers [20][21][22][23]. However, as the assay complexity may increase with these specific recognition elements integrated, one should consider the cost-to-benefit ratio for the intended assay application.
A rapid and selective colorimetric method for the detection of codeine sulfate has been developed using unmodified gold nanoprobes by Lodha et al. [19]. Citrate-stabilized gold nanoparticles (AuNPs) were synthesized and could react with codeine sulfate to produce a green color. The kinetics of AuNPs aggregation in the presence of codeine sulfate were obtained by measuring the absorbance and color intensity at the red, green, and blue channels as well as the total RGB. The results showed that there is a significant change in absorbance intensity from 520 to 582 nm upon increasing the concentration of codeine sulfate [19].
While limitations exist within the discussed colorimetric methods, they are still useful techniques for screening samples in large quantities, especially for those in resource-limited settings. In addition, it is possible to improve selectivity or even embedding specificity into the colorimetric approach by using specific recognition elements such as enzymes, antibodies, aptamers, or molecularly imprinted polymers [20][21][22][23]. However, as the assay complexity may increase with these specific recognition elements integrated, one should consider the cost-to-benefit ratio for the intended assay application.
A rapid and selective colorimetric method for the detection of codeine sulfate has been developed using unmodified gold nanoprobes by Lodha et al. [19]. Citrate-stabilized gold nanoparticles (AuNPs) were synthesized and could react with codeine sulfate to produce a green color. The kinetics of AuNPs aggregation in the presence of codeine sulfate were obtained by measuring the absorbance and color intensity at the red, green, and blue channels as well as the total RGB. The results showed that there is a significant change in absorbance intensity from 520 to 582 nm upon increasing the concentration of codeine sulfate [19].
While limitations exist within the discussed colorimetric methods, they are still useful techniques for screening samples in large quantities, especially for those in resource-limited settings. In addition, it is possible to improve selectivity or even embedding specificity into the colorimetric approach by using specific recognition elements such as enzymes, antibodies, aptamers, or molecularly imprinted polymers [20][21][22][23]. However, as the assay complexity may increase with these specific recognition elements integrated, one should consider the cost-to-benefit ratio for the intended assay application.
A rapid and selective colorimetric method for the detection of codeine sulfate has been developed using unmodified gold nanoprobes by Lodha et al. [19]. Citrate-stabilized gold nanoparticles (AuNPs) were synthesized and could react with codeine sulfate to produce a green color. The kinetics of AuNPs aggregation in the presence of codeine sulfate were obtained by measuring the absorbance and color intensity at the red, green, and blue channels as well as the total RGB. The results showed that there is a significant change in absorbance intensity from 520 to 582 nm upon increasing the concentration of codeine sulfate [19].
While limitations exist within the discussed colorimetric methods, they are still useful techniques for screening samples in large quantities, especially for those in resource-limited settings. In addition, it is possible to improve selectivity or even embedding specificity into the colorimetric approach by using specific recognition elements such as enzymes, antibodies, aptamers, or molecularly imprinted polymers [20][21][22][23]. However, as the assay complexity may increase with these specific recognition elements integrated, one should consider the cost-to-benefit ratio for the intended assay application.