1000/1000
Hot
Most Recent

Infection with HTLV-1 has been associated with various clinical syndromes including co-infection with Strongyloides stercoralis, which is an intestinal parasitic nematode and the leading cause of strongyloidiasis in humans. Interestingly, HTLV-1 endemic areas coincide with regions citing high prevalence of S. stercoralis infection, making these communities optimal for elucidating the pathogenesis of co-infection and its clinical significance. HTLV-1 co-infection with S. stercoralis has been observed for decades in a number of published patient cases and case series; however, the implications of this co-infection remain elusive. Thus far, data suggest that S. stercoralis increases proviral load in patients co-infected with HTLV-1 compared to HTLV-1 infection alone. Furthermore, co-infection with HTLV-1 has been associated with shifting the immune response from Th2 to Th1, affecting the ability of the immune system to address the helminth infection. Thus, further research is required to fully elucidate the impact of each pathogen in co-infected patients. This review provides an analytical view of studies that have evaluated the variation within HTLV-1 patients in susceptibility to S. stercoralis infection, as well as the effects of strongyloidiasis on HTLV-1 pathogenesis. Further, it provides a compilation of available clinical reports on the epidemiology and pathology of HTLV-1 with S. stercoralis co-infection, as well as data from mechanistic studies, to facilitate moving this field forward.
A well-characterized disease manifestation associated with human T-cell leukemia/lymphoma virus type 1 (HTLV-1) is the co-infection with Strongyloides stercoralis. Although the mechanism of the co-infection is not fully understood, it is possible that the microorganisms interact in a complicated manner to influence the course of the combined disease. The epidemiology of HTLV-1 infection is intimately intertwined with its biology. Although worldwide prevalence of HTLV-1 is not at the levels seen for the epidemic of human immunodeficiency virus type 1 (HIV-1), HTLV-1 infection is nonetheless a significant problem affecting millions of individuals across the globe. While the exact numbers of people infected with HTLV-1 remain uncertain, estimates vary from a minimum of 5–10 million, based on data from epidemiologic studies covering areas with approximately 1.5 billion people [1], to as many as 20 million infected worldwide [2][3].
The prevalence of infection is heterogeneous across different populations, often achieving high levels (as high as 5–40%) in small population niches, even within an individual country (reviewed in [1][3]). Compared to HIV-1, HTLV-1 is relatively poorly infectious, relying on direct cell-to-cell transmission to spread the virus [4]. Given this feature, infected lymphocytes must be directly transmitted for efficient infection. Therefore, the most common modes are breast milk transmission of infected cells from mother to child, sexual transmission primarily male to female, and transmission from blood and blood products such as intravenous drug abuse [5]. Breast milk transmission is preventable in societies in which formula feeding is a practical and healthy alternative, such as Japan, which has seen a marked decrease in perinatal transmission in endemic areas [6]. These modes of transmission have also likely impacted the geographic distribution of this virus. Geographical clustering of HTLV-1 infection is well-known, dating back to its initial discovery. In fact, the clustering of adult T-cell leukemia/lymphoma (ATLL) cases in southwestern Japan was a sentinel observation suggesting the existence of a potential new human oncogenic virus [7]. In 1980, Poiesz et al. identified HTLV-1 from a U.S. patient with a similar CD4+ T-cell lymphoma [8]. Similarly, Yoshida and colleagues isolated HTLV-1 from ATLL patients, clearly identifying the virus as the causative agent of this malignancy [9]. Subsequently, the high levels of HTLV-1 both in Japan and in the Caribbean islands led to its identification as the cause of the demyelinating disease referred to as HTLV-associated myelopathy (HAM, originally described in Japan) or tropical spastic paraparesis (TSP, identified in patients of Caribbean origin) [10][11]. Other areas of endemic infection include regions of Sub-Saharan Africa, much of South America, and focal infection in the Middle East and in Australia and Melanesia.
Since infected individuals are concentrated in discrete geographical locations, and the testing for HTLV-1 across much of the world is often unreliable, there is significant uncertainty in the global extent of HTLV-1 infection. Furthermore, many of the serological prevalence studies have used specific subpopulations, including blood donors or pregnant women seen at specific health care facilities. Data from such groups may not be representative of the general population as a whole. HTLV-1 co-infections with other pathogens, such as nematode S. stercoralis, have been appreciated for over three decades [12][13][14][15]. Studies of areas of high prevalence of both infections including case reports of co-infected patients, as represented in Figure 1, provided insights on the interactions between these two pathogens [16][17][18][19][20][21][22][23][24][25][26][27][28][29][30][31][32][33][34]. The geographic overlap of many areas of endemic infection for each pathogen have raised the question of whether HTLV-1 infection increases the incidence of S. stercoralis infection, or whether conversely, infection with this nematode might increase the incidence of HTLV-1 infection. Alternatively, it is possible that neither infection specifically alters the incidence of the other, but that co-infection may change the clinical picture of either infection. Overall, data support the later conclusion, including the possibilities that S. stercoralis infection might accelerate the onset of HTLV-1 disease, and that HTLV-1 infection is associated with a higher likelihood of higher parasite burden, more clinically apparent infection, and more severe and life-threatening disease [12][13][35].
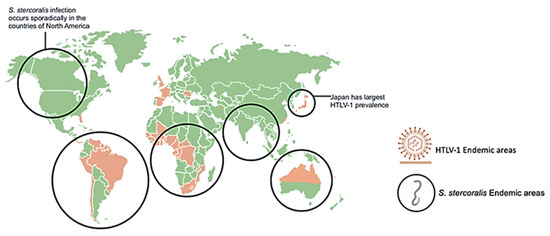
Figure 1. Geographical distribution of human T-cell leukemia/lymphoma virus type 1 (HTLV-1) and Strongyloides stercoralis with overlapping areas of prevalence. Areas highly prevalent with HLTV-1 infection include Japan, Northern Australia, Africa, and South America. Areas highly prevalent with S. stercoralis infection include tropical and sub-tropical areas across the globe including sporadic infection in North America. S. stercoralis image retrieved from https://www.cdc.gov/dpdx/strongyloidiasis/index.html.
Strongyloides is an intestinal nematode that may cause parasitic infection of humans. Estimates of global prevalence range between 100–370 million people currently infected, mostly in tropical and subtropical regions of the world [36][37][38]. The wide discrepancy in these estimates is explained in part by the difficulty in diagnosing these infections, the chronic nature of the disease, and the absence of pathognomonic signs and symptoms. As a soil transmitted helminth (STH), the infection is initiated by the invasion of the skin by the infective third stage larvae (L3). The parasites migrate extensively through the body, pass through the lungs, and develop into parthenogenic parasitic female worms in the small intestine. The adult worms release eggs, which eventually form first stage larvae (L1) in the feces. The L1 have three possible developmental pathways. The first is direct development on the ground through the transition of L1 into the infectious form L3, which can then invade the next host. In the second pathway, the L1 released on the ground develop into free living male and female adult worms that mate and produce offspring that eventually develop into infective L3. This developmental pathway ensures an abundance of larvae in the environment and thus enhances transmission of the infection to the next host. In the third pathway of development, parasites develop from L1 into autoinfective third stage larvae (L3a) without leaving the host, with the L3a reinfecting the same host. The transition from L1 to L3a and ultimately parthenogenetic female worms occurs at a slow rate, replenishing worms in the intestine as they die off. The result is not an increase in the number of adult worms in the intestine, but rather extremely long-term infections in the host, caused by the replenishing of the adult forms in the intestine for decades after the initial infection. Chronic infection with S. stercoralis typically presents as urticaria, diarrhea and abdominal pain, although these associations are not universal [39][40][41]. Hyperinfection may result if the infected individual is treated with steroids or is infected with HTLV-1 due to a disruption in the homeostatic relationship between the parasite and the host. Instead of parasites leaving the host in the feces as L1, they develop into L3a, exit the intestine in large numbers and disseminate to virtually every organ system with accompanying intestinal bacteria. Hyperinfection due to S. stercoralis results in multiple potential clinical manifestations with life-threatening consequences resulting in mortality rates of up to 85% to 100% [39][41][42][43]. Despite having separate clinical manifestations, the two pathogens appear to be closely associated, given the prevalence of the co-infection and the apparent clinical association of HTLV-1 and S. stercoralis hyperinfection. Characterizing the co-infection will require exploring the epidemiology of HTLV and S. stercoralis individually and considering each pathogen's biology.
The clinical presentations of HTLV-1 infection are broad and varied and have been extensively and elegantly reviewed, as have the key principles developed in the extensive literature on HTLV-1 pathogenesis, so only a limited number of salient features will be addressed here [44][45][46][47]. While less than 10% of infected individuals develop known HTLV-1-associated diseases, even those who do not exhibit overt HTLV-1 disease do exhibit shortened lifespans. While the mechanisms responsible for this increased overall mortality are not clear, HTLV-1 infection leads to a chronic inflammatory state with a significant on-going immune response to the virus. These increased death rates are potentially associated with chronic inflammation, resulting in a well-studied increase in the morbidity of cardiovascular disease and non-lymphoid malignancies [44][45].
The most dramatic presentation of HTLV-1 infection is ATLL, an aggressive malignancy of CD4+ T-cells. ATLL occurs in approximately 3–5% of HTLV-1 infected patients, often through breast milk transmission [46]. ATLL can present as a fulminant CD4+ T-cell leukemia, an aggressive lymphoma, or as a more indolent smoldering or chronic ATLL [47][48]. Prognosis of aggressive disease remains dismal, since the average survival times of the acute leukemias and lymphomas remain in the range of 8–10 months [49]. Due to the severity of this disease, therapy for HTLV-1 malignant disease is a rapidly evolving field [50][51]. Classical chemotherapy approaches have relatively little success, and the only potential cures to date follow allogeneic hematopoietic stem cell transplantation, resulting in reconstitution of the lymphoid population [52]. Over the past two decades, protocols using the anti-viral drugs, interferon-α and azidothymidine, have shown evidence of efficacy in inducing longer remissions in acute and chronic subtypes of ATLL, including up to 100% 5-year survival in the chronic subtypes [53]. Anti-proliferative and pro-apoptotic effects of the combination of these two agents appear to be the major contributors to their real, though limited therapeutic effects [54][55].
Another promising approach for ATLL therapy has been the use of antibodies directed against receptors present in high levels on the surface of HTLV-1 malignant T-cells. Early studies targeted the interleukin-2 (IL-2) receptor alpha subunit, anti-CD25, with some success, particularly using antibodies coupled to cytotoxic agents such as cytotoxic toxin subunits [56][57]. More recently, the C-C chemokine receptor type 4 (CCR4) has been shown to be a useful target for antibody directed therapy against ATLL. CCR4 is overexpressed and/or mutated in a significant proportion of ATLL, resulting in constitutive activation of downstream signaling [58][59]. CCR4 is also expressed on other, non-malignant T-cells infected by HTLV-1 [60]. A variety of studies assessing the effects of histone deacetylase inhibitors, arsenate derivatives, the immunomodulator lenalidomide, and other antibodies and small molecules are also in progress [47][50][51].
In addition to malignant disease, HTLTV-1 infection can result in several different inflammatory diseases that may resemble autoimmune disorders. The most prevalent of these inflammatory disorders is HAM/TSP, a demyelinating disorder involving primarily the spinal cord, found in up to 2–4% of HTLV-1-infected patients [60][61]. HAM/TSP occurrence is generally associated with infection in adulthood through sexual transmission or blood transfusion. It is important to note that patients with juvenile HAM/TSP have been seen [62][63] and that ATLL is a major cause of death in HAM/TSP patients [64], suggesting that the transmission and pathogenesis of these two classes of HTLV-1 disease may not be entirely unrelated. Prior to onset of symptoms, clinical latency is typically anywhere from a couple of years to decades following infection (reviewed in [65][66]). Subsequently, HAM/TSP initially presents in the lower extremities with chronic progression of spasticity and weakness (paraparesis), contributing to increasing gait disturbance and loss of mobility. Over time, patients can experience bladder dysfunction and increasing sensory deficits. Histologically, the disease is characterized by lymphoid inflammatory infiltrates throughout the central nervous system with accompanying demyelination, axon loss, and formation of gliotic scars.
The major treatment of HAM/TSP is corticosteroids, which despite years of use, has only recently demonstrated responses in enough patients to be deemed clinically effective [67]. A little over half of patients have short-term responses, while two-thirds have longer term stable or improved disease. Nonetheless, significant numbers of patients have little or no long-term improvement or become refractory to steroid therapy. One therapy of note, mogamulizumab (anti-CCR4 antibody), has been recently reported to reduce the symptoms of spasticity and motor deficits, the presence of cerebrospinal fluid inflammatory markers and the levels of HTLV-1 proviral load. This antibody may work by targeting both CD4+CCR4+ and CD8+CCR4+ T-cell populations, both of which were shown to be reservoirs containing HTLV-1 proviruses in HAM/TSP patients [60], suggesting a promising new therapeutic approach for HAM/TSP [68] as well as ATLL.
HTLV-1 can also be associated with a generalized cytokine dysregulation and immunodeficiency, resulting in accentuation of other infectious processes in some HTLV-1 infected individuals. One notable example is HTLV-1-associated infective dermatitis, an eczematous rash, predominantly found in HTLV-1 infected children. This disease manifestation is characterized by pronounced lymphoid infiltration of the skin and associated with overgrowth of common pathogenic skin bacteria, particularly Staphylococcus aureus and β-hemolytic streptococci (reviewed in [69]). While this infective dermatitis is generally quite responsive to antibiotic treatment, it may be an early indicator of an underlying HTLV-1 infection and has been suspected to be a precursor to other HTLV-1-associated diseases [70]. The best characterized of these interactions between HTLV-1 and other infectious processes is the well-documented association between HTLV-1 and S. stercoralis.
Given early epidemiologic studies and the various cases reported in the literature, there appears to be a strong association of S. stercoralis infection with HTLV-1 infection [13][71]. Interactions between the pathogens may therefore change the course of each infection. Shortly after the discovery of HTLV-1 and S. stercoralis co-infection, Yamaguchi et al hypothesized that this interaction may be responsible for the earlier onset of ATLL seen in Caribbean and African patients (approximately 43 years [72]) as compared with ATLL onset in Japan (approximately 56 years [73]). Further data through the 1990s provided some support for the hypothesis that S. stercoralis co-infection is a co-factor for ATLL. Plumelle et al. reported a dramatic difference in age of onset of ATLL in S. stercoralis co-infected patients (median age 39 years) as compared with non-co-infected patients (median age 70 years, [74]). In this early study which was prior to more recent treatment modalities, the overall survival for co-infected individuals was independent of age and was more favorable for co-infected individuals (167 days) compared to HTLV-1 infection alone (30 days). There have been some interesting published findings that begin to explain possible mechanisms for S. stercoralis in altering the course of HTLV-1 infection. Specifically, S. stercoralis may have a role in changing both viral load and expression of immune-related genes, potentially worsening the clinical course of HTLV-1 infection.
The effect S. stercoralis has on HTLV-1 has often been assessed through comparison of HTLV-1 carriers with and without helminths. Gabet et al. used quantitative PCR methods to study HTLV-1 DNA proviral load in 18 patients with S. stercoralis+/HTLV-1+ along with 15 patients with HTLV-1 alone from French Guyana and Martinique [75]. Their main conclusion was that S. stercoralis infection stimulates the oligoclonal proliferation of HTLV-1-infected cells. This was hypothesized to account for the possibly shorter latency period in those that develop ATLL; expanding oligoclonal populations of HTLV-1 infected cells undergoing increased rounds of replication were hypothesized to have acquired additional cellular mutations enhancing the likelihood of malignant transformation. A mechanism of how S. stercoralis led to clonal expansion was not elucidated. As noted above, enhanced viral load has been associated with more severe infection and potential clinical manifestations. Gabet et al. looked only at HTLV-1 carriers without clinical manifestations of ATLL or HAM/TSP leaving open the possibility that the interactions between viral dynamics and nematode infection may differ in those already experiencing an HTLV-1-associated disease manifestation.
Subsequent studies have also correlated viral load and clonal proliferation of HTLV-1 in co-infected patients using a larger sample size, including individuals with HTLV-1-specific disease presentations. Applying the pioneering deep sequencing of HTLV-1 integrations sites, 61 individuals with HTLV-1 alone (i.e., no co-infection), were studied [76]. This included 14 asymptomatic HTLV-1 carriers, 26 HAM/TSP patients, 20 ATLL patients, and 1 with uveitis. In addition, 14 patients co-infected with S. stercoralis and HTLV-1 were included. This study had the advantage of samples representing of several clinical presentations of HTLV-1 patients, including uveitis and neurological diseases. An increased number of HTLV-1-infected clones was observed in S. stercoralis patients. In addition, there was higher proviral load in co-infected patients due to increased mean clone abundance (i.e., a greater abundance of individual HTLV-1-infected cell clones within the overall population of lymphocytes). This indicates that in the presence of S. stercoralis infection, either endogenous restrictions on clonal proliferation are over-ridden or there is some spur to increased proliferation in already proliferating clones. A surprising observation from this study was that the previously identified association between HTLV-1 infection and the genomic context of proviral integration, was not observed in proliferating clones in the setting of S. stercoralis co-infection. Both of these observations suggest that in the presence of Strongyloides co-infection there is an additional drive for proliferation of subsets of HTLV-1 infected cells, which in turn may provide a relevant contribution to the early observations of earlier onset of ATLL in co-infected patients.
In addition to the impact of S. stercoralis on HTLV-1 viral load, the immune response in these patients is of great interest. The shift to a Th1 response is well-documented in HTLV-1 carriers, and this is likely involved in the mechanism of co-infection with helminths because the body cannot induce an effective Th2 response against S. stercoralis. It is worth mentioning, however, that some studies have shown increased Th2 responses in HTLV-1 patients with the parasites compared to those without them. Porto et al. studied 310 HTLV-1 carriers and 32 HAM/TSP patients in Brazil [77]. Specifically, 35 HTLV-1 carriers co-infected with helminths were compared with a control group of 35 without helminths. Those co-infected had lower interferon gamma (IFN-γ), and increased IL-5 and IL-10; indicating an overall decreased Th1 response compared to HTLV-1 carriers, associated with increased Th2 response. This study also contrasts to other recent studies because the patients with the S. stercoralis showed a lower HTLV-1 proviral load. The lower viral burden may have been the reason for the predominant Th2 response rather than Th1. These observations raise questions as to whether Strongyloides always induces proliferation of HTLV-1-infected cells, and which T-cell response predominates.
Other work has sought to better understand the role of IL-2 in the mechanism of the co-infection. Satoh et al. studied 123 patients in Okinawa, showing that HTLV-1/S. stercoralis co-infected patients had higher proviral load and increased CD4+25+ T-cells, compared to patients with HTLV-1 alone [78]. This corroborates other work described above. Additionally, it was determined that S. stercoralis antigen activated the IL-2 promoter, supporting growth of cell lines with HTLV-1 or transduced with Tax. Overall, this was a new finding and showed that S. stercoralis can induce polyclonal expansion of HTLV-1 cells, through activation of the IL-2/IL-2R receptor system. Later work in Brazil came to similar findings through the study of 30 patients with Strongyloides and HTLV-1 and 60 patients with HTLV-1 only [79]. Co-infected patients showed significantly higher levels of secretory IL-2 receptor (sIL-2R) than patients with HTLV-1 alone, and also of interest, higher levels of sIL-2R were noted before treatment of S. stercoralis. Treatment of helminth infection decreased these levels, again suggesting a role of the parasites in modulating HTLV-1 infection. This further suggests a role of helminthic infection in enhancing T-cell proliferation through IL-2 induction and induction of IL-2 receptor levels when co-infected with HTLV-1. Further studies that might integrate this work on the effects of S. stercoralis on IL-2/IL-2R function with the apparent increased drive for HTLV-1 clonal abundance and proliferation in the setting of co-infection [80] will be of great interest and may help establish key mechanistic links between S. stercoralis infection and HTLV-1-associated disease. It also would have implications for both T-cell functions and T-cell oncogenesis. In addition to potential effects on oncogenesis, other studies are exploring the impact of HTLV-1 and S. stercoralis infections on the incidence of other malignancies. Co-infection has been independently associated with a number of different cancers other than ATLL; for example, HTLV-1 infection has been associated with increased non-ATLL lymphomas and hepatocellular carcinoma in a long-term follow-up of a cohort in Okinawa [81]. However, despite relatively common co-infection, it is unclear if co-infection increases the risk for non-ATLL cancers as well.
HTLV-1 appears to have specific effects on how the body reacts to S. stercoralis in patients with the co-infection. Clinically, this appears to be manifest as a significant association between HTLV-1 infection and the occurrence of the aggressive and potentially life-threatening clinical syndrome of S. stercoralis hyperinfection [80][82]. In particular, Gotuzzo et al. in a study from Lima, Peru demonstrated a startlingly high rate of HTLV-1 co-infection in patients with hyperinfection (almost 86%), but not in those with simple intestinal infestation, which did not differ in frequency between HTLV-1+ and HTLV-1 uninfected patients [82]. Similar results in a Brazilian population were reviewed by Carvalho [80]. The increased susceptibility of HTLV-1 and S. stercoralis co-infected patients to aggressive parasite infection, strongly suggests that HTLV-1 infection may compromise an effective host response limiting nematode infection. A series of studies have measured various cytokine and IgE levels to determine possible associations between cytokine production and parasite expulsion in co-infection. In typical immune responses to the presence of S. stercoralis, a Th2 response includes an increase in the production of IL-4, which promotes isotype switching in B cells and the production of IgE [83]. Subsequently, IgE primes eosinophils and increases the anti-parasitic response. In co-infection with HTLV-1, there is increased production of IFN-γ, resulting in a shift from a Th2 response to a Th1 response. Given that a Th2 response is necessary to expel the parasite, the presence of a Th1 response would exacerbate the phenotype of disease and symptoms related to the S. stercoralis infection.
Several studies observed that with increased production of IFN-γ characteristic of a Th1 response, HTLV-1 infection results in decreases in IL-4, IL-5, and IgE [83][84]. These changes in cytokine production, specifically the apparent decrease in IL-4 and IL-5, then decreases the immune response to the parasite. A 2002 study included 32 HTLV-1 positive and 47 HTLV-1 negative, S. stercoralis-infected individuals from Japan [78]. Further, 21 S. stercoralis negative individuals, of whom, 9 were HTLV-1 positive, were also included [85]. Patients were diagnosed with S. stercoralis, and the effect of treatment of strongyloidiasis in HTLV-1 carriers was observed. This study observed serum IgG4 levels were higher in patients with co-infection, supporting the hypothesis that IgG4 may block production of IgE. Increased IFN-γ and tumor growth factor beta (TGF-β) were also seen in co-infected patients, followed by a decrease in IL-4. As noted above IL-4 is required for B cells to undergo isotype switching to produce IgE. Another study examined the effect of HTLV-1 infection on serologic and skin tests for strongyloidiasis to assess the impact on IgE and eosinophils [84]. Data was collected from three groups, including HTLV-1 seropositive individuals identified at blood banks; residents of an S. stercoralis endemic area in Salvador, Brazil who tested positive for helminth infection via fecal examination; and patients hospitalized for severe infection. This study concluded that HTLV-1 reduced the sensitivity of parasite specific IgE testing as well as immediate hypersensitivity skin test. The decrease in IL-4 also apparently prevented an increase in intestinal fluid content associated with parasite rejection. These findings were in accordance with prior knowledge of the role of IL-4. Observed effects were attributed to the impaired isotype switching from IgG to IgE as illustrated in Figure 2. This overall shift away from a typical anti-parasitic response, results in less elimination of the S. stercoralis infection through eosinophil activation and specific immunoglobulin production. Subsequently, the patient may develop disseminated strongyloidiasis and further complications. The same data set was also used to measure various cytokine and IgE levels to determine other possible association between cytokine production and parasite expulsion. The results included a negative correlation between IFN-γ and total IgE, which was in accordance with predicted levels in co-infection. Other results included an increase in IL-10 and a decrease in IL-13, which corresponded with the expected Th2 to Th1 shift.
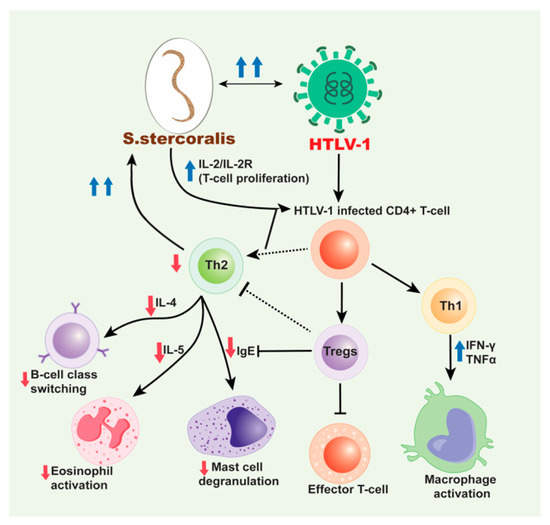
Figure 2. Mechanism for HTLV-1/S. stercoralis co-infection. HTLV-1 infected CD4+ T-cells induce a predominant Th1 response over a Th2 response and produce high production of interferon gamma (IFN-γ) and tumor necrosis factor alpha (TNF-α). This decreases the production of cytokines, IL-4 and IL-5 subsequently inhibiting B cell isotype switching to IgE and decreasing eosinophil activation, respectively and also decrease in the mast cell degranulation. The increased Th1 response also contributes to the decreased Th2 response with the release of IL-10. HTLV-1 has also been associated with an increase in Treg cells, which are CD4+CD25+FOXP3+ T-cells that lead to immunosuppression and downregulation. These cells inhibit effector T-cells and likewise help decrease the Th2 response seen in HTLV-1/S. stercoralis co-infection. With the decrease in IgE production and decreased eosinophil and mast cell activation and priming, the S. stercoralis parasite cannot be eliminated from the body. Therefore, with cases of co-infection, disseminated strongyloidiasis is likely. Moreover, the dissemination of S. stercoralis results in the IL-2/IL-2R which leads to increase in the polyclonal expansion of HTLV-1 infected T-cells which leads to these sequences of events.
Despite many studies that showed a change in IgE production, a recent study found unchanged serum IgE levels with co-infection. The study observed Japanese patients with S. stercoralis treated between 1991–2014 [86]. Patients received treatment with ivermectin and despite HTLV-1 co-infection their serum IgE was unchanged. The two cohorts were used to collect data on symptoms and serum IgE and eosinophil counts. This study also demonstrated that serum IgE levels were lower than expected in female patients under 70 years old, indicating a potential relationship between sex and serum IgE levels. There was also a proposed impact of age on serum IgE levels, potentially contributing to the normal IgE levels seen in patients included in this study. Patients under 70 years of age typically showed normal IgE levels even with co-infection compared to individuals over 70 years of age. This particular study unfortunately does not appear to have measured IL-4/IL-5 levels to correlate to IgE. Additionally, few of these studies correlate the response to Strongyloides to HTLV-1 proviral load; therefore, it is unclear if worsening HTLV-1 infection causes an incremental impairment of immune response to the helminths.
The Th2 to Th1 shift is one of the potential mechanisms for the impact of HTlV-1 on the S. stercoralis infection. However, other changes in the CD4+ T-cell population have also been noted. A current area of interest is the impact HTLV-1 has on Treg cells. Several studies show associations between Treg cells and IgE expression, which may contribute to the pathogenic mechanisms of HTLV-1/S. stercoralis co-infection [87][88]. Montes et al. compared a cohort of co-infected patients, and in addition to observing higher parasite burden and higher HTLV-1 proviral loads, the relative proportion of Tregs was significantly higher than in patients infected with either agent alone [89]. HTLV-1 co-infection was associated with increased Tregs and decreased ex vivo S. stercoralis larval antigen-driven production of IL-5. This decrease in IL-5 production is potentially also followed by decreased eosinophil differentiation, decreased helminth killing, and decreased mast cell degranulation. Malpica et al. [90] extended these observations to determine how increased Tregs impacted the local immune response to S. stercoralis in the infected intestine. The study demonstrated increased overall CD3, CD8 and Treg cell responses in duodenal biopsies of co-infected patients versus non-specific duodenitis or normal patients. However, there was also a relative decrease of the aforementioned cells and eosinophils adjacent to the parasites. Recently, intriguing follow-up studies showed that successful treatment of S. stercoralis infection in co-infected patients resulted in decreased Tregs, however the defect in IL-5 production in response to Strongyloides antigen persisted [91]. Thus, HTLV-1 infection induces long-term and persistent deficits in the host response to S. stercoralis even following parasite eradication. Many of these findings corroborate the proposed role of HTLV-1 in this co-infection. The presence of the retrovirus results in increased Treg cells and a downregulation of effector T-cells, likely contributing to increased parasite burden (as depicted in Figure 2), although it is unclear if this is specific to the S. stercoralis species. However, future studies could help determine other immune responses to co-infection beyond cytokine production and Treg promotion as well as the potential confounding role of corticosteroid treatment in the impact of HTLV-1 on S. stercoralis.