
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Attilio Converti | + 2325 word(s) | 2325 | 2020-10-07 05:04:15 | | | |
| 2 | Catherine Yang | Meta information modification | 2325 | 2020-10-22 05:09:57 | | |
Video Upload Options
Solid dispersions (SDs) are a technological strategy to improve the pharmacological potential of natural or synthetic bioactive molecules, due to the increase in its solubility and bioavailability, leading to a possible improvement of its biological activities. In this sense, the review sought to synthesize and critically examine the studies that address SDs with therapeutic applications, evaluated through in vitro and/or in vivo tests. This bibliographic survey shows the significant therapeutic potential of SDs in the context of the most diverse biological activities. Among these, including in vitro and/or in vivo antitumor, antiparasitic, antimicrobial, antioxidant, anti-inflammatory or cytoprotective activities, while additional activities, such as gastroprotective, hepatoprotective, antidiabetic or antinociceptive, were highlighted by in vivo studies.
Although SDs have already been studied and cited in the literature, the number of studies published with a focus on in vitro and in vivo trials is still relatively small, considering the great potential of these formulations in pharmaceutical technology and with the most diverse applications. The results of biological activity studies showed that SDs, as a drug release tool, is not a limiting factor for the execution of in vitro and in vivo tests. Additionally, it stands out as a promising system in which the active principle and the carrier interact, allowing, in most cases, an increase in the pharmacological potential due to changes in the physicochemical properties of the constituents. Thus, SDs can represent a safe and effective alternative for the development and improvement of drugs directed to a wide range of pharmacological treatments.
1. Introduction
Many drug candidates have low aqueous solubility, which can make their oral absorption inadequate. According to the literature, approximately 40% of marketed drugs are poorly soluble in water, as are, according to the Biopharmaceutical Classification System, about 40–90% of new drug candidates [1][2][3][4].
The slow dissolution rate and low solubility of some drugs lead to unpredictable bioavailability, non-reproducible clinical response or treatment inefficiency due to low therapeutic plasma levels; therefore, their use must be optimized using formulation strategies capable of improving their administration [5].
Several technological strategies have been developed and employed to circumvent this situation such as complexation with cyclodextrins, particle size reduction by micronization, crystal development, nanotechnology [5], salt formation and solid dispersions (SDs) [6][7]. However, some of these strategies have shown disadvantages including the development of active forms in vivo, high execution cost and considerable levels of toxicity. Some of these limits can be overcome by employing solid dispersions, a viable, well-established and widely-used strategy to increase the dissolution rate and solubility of poorly water-soluble drugs [4][6][7].
SDs can be defined as molecular mixtures of drugs that are not soluble in hydrophilic carriers, which exhibit a drug release profile driven by the polymer properties [8]. The so-called first-generation crystalline SDs, which use crystalline carriers such as urea and sugars including sucrose, dextrose and galactose, have the disadvantage of high thermodynamic stability that prevents a quick release of drugs [9]. In second-generation SDs drugs are dispersed in usually polymeric amorphous carriers [8], while the carriers used in third-generation SDs are surfactants or a mixture of amorphous polymers and surfactants [10].
The use of hydrophilic polymeric matrices to develop SDs for the release of commercial drugs or new drugs candidates has been shown to be a promising alternative able to improve their pharmacokinetic properties [7].
This review deals with SDs, mainly those belonging to the second-generation class that use polymers as carriers. It describes the main in vitro and in vivo activities (Figure 1) of SDs having different compositions and various preparation methods, intending to innovate and enhance the release of poorly water-soluble drugs, as well as improving the biological activities of the loaded bioactive compounds.
Figure 1. Representation of the application of solid dispersions in biological assays.
2. In Vitro Study of Solid Dispersions in Polymeric Matrices
This section deals with solid dispersions (SDs) prepared to improve the properties and release of poorly soluble drugs and drug candidates of natural or synthetic origin. The preparation and use of SDs have been reported in several in vitro studies, which have been numerically quantified and classified in Figure 2, based on the biological activities of their active compounds, while the main information on these studies is summarized in Table 1.
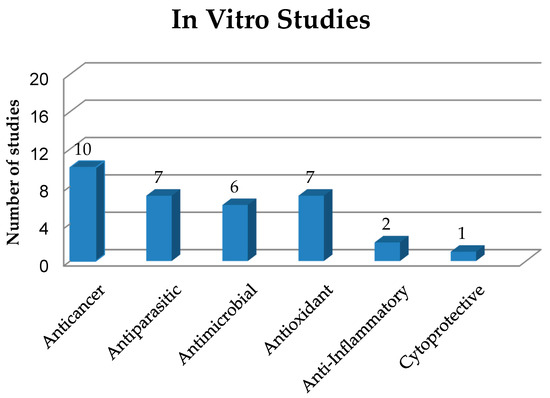
Figure 2. Quantification and classification of in vitro studies on solid dispersions published in the period from 2009 to 2020.
Table 1. In vitro studies on solid dispersions.
| Carrier Type | Substance | Cell Type | Activity | Improved Characteristics | Reference |
|---|---|---|---|---|---|
| OHPP | Niclosamide | PC-3, HeLa, A549 | Anticancer | SDs showed higher cytotoxicity to target cells (lower IC50) than the niclosamide solution. | [11] |
| OHPP | Paclitaxel | PC-3, HeLa, A549 | Anticancer | SDs showed significantly higher cytotoxicity to target cells (lower IC50) than the paclitaxel solution. |
[12] |
| PVP/VA TPGS |
Paclitaxel | BT-474, MCF-7, SK-BR-3 | Anticancer | SDs showed higher cytotoxicity against cancer cells compared to the pure drug. | [13] |
| Brij®L4 | Chrysin | HT29 | Anticancer | The higher solubility of chrysin in SDs compared to water solution increased cytotoxicity. |
[14] |
| Poloxamer 407 | Curcumin (CM) | NCIH460, HeLa, HepG2, MCF-7 and PLP2 |
Anticancer | Anticancer SDs showed cytotoxicity against all tumor cell lines tested, but no toxic effects on non-tumor cells. | [15] |
| AChE, BChE, GST, MAO A-B |
Enzyme inhibitory /Antioxidant |
SD was able to inhibit the activities of AChE, BChE and GST in aqueous medium. |
|||
| LPS-stimulated murine macrophages (RAW 264.7) |
Anti-inflammatory | IC50 (inhibitory concentration of 50% NO production by macrophages) > 400 µg/mL. | |||
| PVP K30 | Zn(II)-curcumin complex |
HepG2, SK-HEP1 | Anticancer | SD of Zn(II)-curcumin complex had a potent anticancer effect. | [16] |
| HPMC, PVP K30, PEG 6000 |
Telaprevir | HepG2 | Anticancer | The antitumor activity was dose dependent and even with the addition of the polymer the drug maintained its efficacy. | [17] |
| Soluplus ® | Angelica gigas Nakai | HeLa, HEK 293 | Anticancer | SD at the concentration of 200 µg/mL showed a significant decrease (to only 17.37%) in cell viability. There was no toxicity to normal cells. | [18] |
| Eudragit S-100 | Berberine hydrochloride (HB) |
SW480, HCT116, Caco-2 | Anticancer | The release of HB from SDs was effective and cell viability was reduced in a dose and time dependent manner. | [19] |
| PVP K30 | IIIM-290 | Ehrlich ascites carcinoma cells |
Cytotoxic | Despite the reduced amount of IIIM-290 in SD, the IC50 value of SD was lower than that of IIIM-290 alone. |
[20] |
| Poloxamer 407 | Benznidazole | Epimastigotes of Trypanosoma cruzi |
Antichagasic | SDs enhanced drug solubility, release kinetics and parasitic activity. | [21] |
| Low-substituted HPC | Benznidazole | Epimastigotes and intracellular amastigotes of T. cruzi (CL-B5) |
Antichagasic | SDs had higher antiparasitic activity against amastigotes than epimastigotes. | [22] |
| Gelucire 50/13 | Ursolic acid | Trypomastigotes of T. cruzi Y |
Antichagasic | Increased antiparasitic activity. | [23] |
| PVP K30,PVP/VA, Kollidon-CL-M, sodium starch glycolate |
Praziquantel | Adult schistosomes of Schistosoma mansoni |
Antischistosomal | Increased solubility, better bioavailability and stronger antiparasitic activity. |
[24] |
| PVP K30 | Praziquantel | Newly transformed schistosomula of S. mansoni and adults |
Antischistosomal | Increased solubility, reduced dosage especially for children and increased antiparasitic activity |
[25] |
| Soluplus, PEG 400, Lutrol F127 and Lutrol F68 |
Artemether | Schizonts of Plasmodium falciparum 3D7 |
Antimalarial | Increased dissolution rate, amorphous form, increased solubility and, mainly, increased antimalarial activity. |
[26] |
| Soluplus, Kollidon VA64, Plasdone S630 |
Lumefantrine | ITG cells | Antimalarial | Increased antiparasitic activity. | [27] |
| Chitosan | Abietic acid | Staphylococcus epidermidis | Antimicrobial | SD exhibited better MIC values against S. epidermidis than chitosan and abietic acid alone. |
[28] |
| DPPH radical scavenging | Antioxidant | SD had higher antioxidant power (IC50 of 0.61 mg/mL) than abietic acid alone (IC50 of 11 mg/mL). |
|||
| PVP K30 and HPMCAS | Griseofulvin | Dermatophytes of Trichophyton rubrum NCPF 935 |
Antimicrobial | SDs significantly reduced biofilm formation when compared to the control. |
[29] |
| Pluronic F127 | Gatifloxacin | Staphylococcus aureus | Antimicrobial | The gatifloxacin/Pluronic F127 system exhibited antimicrobial efficacy when compared to commercialized eye drops. |
[30] |
| PVP K30 | Curcumin | Salmonella enteritidis | Antimicrobial | SD had a strong antimicrobial effect on S. enteritidis, while CM alone did not show antimicrobial activity in vitro | [31] |
| HPMC | Curcumin | Escherichia coli | Antimicrobial | SD used to prepare phototoxic supersaturated solutions showed significant bactericidal activity against E. coli. | [32] |
| Polaxamer 407 | Curcumin | E. coli, Pseudomonas aeruginosa and S. aureus |
Antimicrobial | The association between SD and silver nanoparticles increased CM antimicrobial and antioxidant activities. |
[33] |
| DPPH radical scavenging | Antioxidant | ||||
| PVP K25 | Quercetin | DPPH radical scavenging | Antioxidant | Increased quercetin antioxidant activity in SD (0.61 ± 0.03 ≤ IC50 ≤ 1.00 ± 0.02 µg/mL). | [34] |
| Mannitol | Coenzyme Q10 | Intracellular ROS level | Antioxidant | The SD with the smallest particle size showed the greatest absorption of UVB radiation as well as the highest antioxidant activity in vitro. |
[35] |
| PVP K30 | Usnic acid | DPPH radical scavenging | Antioxidant | Increased usnic acid solubility and antioxidant activity. | [36] |
| PEG 4000 | Luteolin | DPPH | Antioxidant | Polymers increased luteolin solubility and antioxidant activity. | [37] |
| PVP K30, PEG 6000 and HPMC |
α,β-Amyrin | LPS-stimulated macrophages J774 |
Anti-inflammatory | SDs enhanced the anti-inflammatory activity of α,β-amyrin. | [7] |
| HPMC | Curcumin | HepG2 | Cytoprotective | SDs showed better cytoprotective activity than pure CM and inhibited cell death induced by t-BHP. | [38] |
OHPP, Octenylsuccinate hydroxypropyl phytoglycogen; IC50, Half inhibitory concentration; PVP/VA, Polyvinylpyrrolidone/vinyl acetate; TPGS, D-α-toco-pheryl polyethylene glycol-1000-succinate; PVP, Polyvinylpyrrolidone; SD, Solid dispersion; AChE, Acetylcholinesterase; BChE, Butyrylcholinesterase; CM, Curcumin; GST, Glutathione S-transferase; MAO, Monoamine oxidase; LPS, Lipopolysaccharide; NO, Nitric oxide; PEG, Polyethylene glycol; HPMC, Hydroxypropyl methylcellulose; ITG (chloroquine-resistant cell line); DPPH, 2,2-diphenyl-1-picryl-hydrazyl-hydrate; HPMCAS, Hydroxypropyl methylcellulose acetate succinate; MIC, Minimum inhibitory concentration; ROS, Reactive oxygen species; t-BHP, tert-Butylhydroperoxide.
3. In Vivo Studies of Solid Dispersions in Polymeric Matrices
As already mentioned, solid dispersions (SDs) have been used as a strategy in pharmaceutical technology to circumvent some limitations presented by drugs and new bioactive compounds such as low solubility and bioavailability. In this sense, this section addresses in vivo studies on SDs with different biological activities, as shown in Figura 3 and quantitatively expressed in Figure 4 . The main information about these studies is summarized in Table 2.
Figure 3. In vivo activities of solid dispersions.
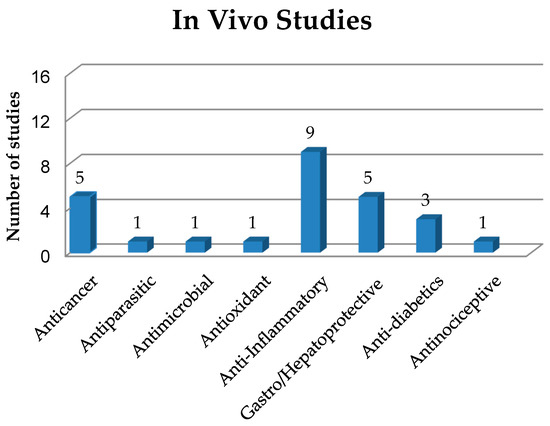
Figure 4. Quantification and classification of in vivo studies on solid dispersions published in the period from 2009 to 2020.
Table 2. In vivo studies of solid dispersions.
| Carrier Type | Substance | Animal | Dose | Activity | Improved Characteristics | References |
|---|---|---|---|---|---|---|
| PVP K30 | IIIM-290 | Swiss male mice (18–23 g) |
25, 50 and 75 mg/kg | Anticancer | SD was able to reduce the IIIM-290 dose by at least 1.5-fold thanks to its efficiency in Ehrlich solid tumor model. | [20] |
| Soluplus® | 9-Nitrocamptothecin | Male Sprague-Dawley rats (20 ± 2 g) |
4 mg/kg | Anticancer | SD showed higher tumor growth inhibitory rate than the pure compound due to improved oral bioavailability. | [39] |
| (+)-Xylitol | (−)-Oleocanthal | Athymic nude mice | 10 mg/kg | Anticancer | Treatment with SD showed prevention, lower growth rate and less recurrence of tumor. | [40] |
| PVP K30 | Zinc(II)-curcumin complex |
B-NDG, BALB/c mice | 100 mg/kg | Anticancer | SD reduced tumor size and weight in animals. | [17] |
| PVP K30 | Selaginella doederleinii Hieron |
BALB/c mice | 200 mg/kg | Anticancer | SD reduced tumor size as well as the level of tumor angiogenesis. | [41] |
| Low substituted HPC | Benznidazole | Female NMRI mice (25 ± 2 g) |
25 mg/kg/day | Antichagasic | The best SD showed a 96.65% trypanocidal activity, expressed as percentage reduction in the area under the parasitic curve. | [22] |
| PVP K30 | Curcumin (CM) | Male Cobb-Vantress broiler chickens |
1 g/kg of feed | Antimicrobial | The synergistic effect of 0.05% CM/PVP SD with 0.05% boric acid reduced colonization of Salmonella enteritidis in crop and ceca-cecal tonsils. | [31] |
| PVP K30 | Taurine-zinc complex | Female Sprague-Dawley rats (240–260 g) |
100 and 200 mg/kg/day |
Antioxidant | SDs protected rat gastric mucosa from ethanol-induced injury and increased SOD activity and glutathione level. |
[42]] |
| Gastroprotective | ||||||
| Kollidon (VA64) | Triacetylated andrographolide (TA) |
Male Kunming mice |
50, 100 and 200 mg/kg/day |
Anti-inflammatory | TA-SD prepared with VA64 significantly improved the drug activity against ulcerative colitis. | [43] |
| PVP K30, Poloxamer 188 | Curcumin | Female CD-1 mice | 100 mg/kg oral doses | Anti-inflammatory | CM-SD prepared with PVP decreased matrix metallo-peptidase 9 expression and levels of IL-1β and IL-6 cytokines. | [44] |
| Gelucire®50/13-Aerosil® | Curcumin | Rat | 10 to 100 mg/kg | Anti-inflammatory | A CM-SD dose of 100 mg/kg was more effective than 5 mg/kg indomethacin in reducing edema | [45] |
| HPMC, lecithin and isomalt |
Curcumin | Male Sprague-Dawley rats |
5 mg/kg | Anti-inflammatory | A CM-SD dose of 5 mg/kg had greater anti-inflammatory activity than 50 mg/kg curcumin alone. | [46] |
| Crospovidone | Aceclofenac | Male Sprague-Dawley rats |
1 g/cm2 (topical) |
Anti-inflammatory | The enhanced drug permeation increased the intensity of the anti-inflammatory response. | [47] |
| PEG 8000 | Ibuprofen | Wistar rats | 20 mg/kg | Anti-inflammatory | All SDs showed better anti-inflammatory activity than the pure drug, allowing up to 90% edema inhibition after 6 h. | [48] |
| Urea and mannitol | Flurbiprofen | Rat | 11.69 mg/kg | Anti-inflammatory | SD showed better inhibition of rat paw edema up to 16 h. | [49] |
| Paracetamol | Meloxicam | Rat | - | Anti-inflammatory | SDs reduced by more than 50% the volume of carrageenan-induced tail edema compared to the physical mixture. | [50] |
| PVP K30 | Chelerythrine (CHE) | Mice | 10 mg/kg | Anti-inflammatory | SD enhanced CHE anti-inflammatory effect by reducing the levels of TNF-α, IL-6 and NO in mice serum. | [51] |
| HPMC | Curcumin | Male BABL/c mice | 200 and 400 mg/kg | Hepatoprotective | The best SD increased the hepatoprotective efficacy of CM. |
[38] |
| HPC | Nobiletin | Male Sprague-Dawley rats (220 g) | 2 mg of drug/kg | Hepatoprotective | SD was more effective than the crystalline drug in rats with acute liver injury | [52] |
| PVP K30 | Silymarin | Adult male albino rats (150–200 g) | 25 mg/kg | Hepatoprotective | The best SD improved biomarker rates and had a significantly better hepatoprotective effect than the commercial extract | [53] |
| PVP K30 | Silymarin | Male Sprague-Dawley rats (190–210 g) | 50 mg/kg | Hepatoprotective | SD improved drug solubility and hepatoprotective activity, reducing the AST levels. | [54] |
| Poloxamer 188 | Repaglinide | Wistar rats (150–250 g) ) |
(1 mg of drug) | Antihyperglycemic | SD obtained by the microwave method improved the drug anti-hyperglycemic activity. | [55] |
| Soluplus1 and PEG 4000 | Glimepiride | Albino Wistar rats (200–250 g) |
0.0285 mg of drug/kg | Anti-diabetic | SD reduced the glucose level in rats more than the pure drug and a commercial product | [5] |
| PVP K17 | Pioglitazone | Male Swiss albino mice (25–30 g) | 30 mg/kg SD | Antihyperglycemic | SD reduced the mean glucose level in mice more than the pure drug and a commercial product. | [56] |
| HPMC | Hecogenin acetate | Male Swiss mice (28–35 g) |
40 mg/kg | Antinociceptive | Both the drug alone and its SD with HPMC-reduced mechanical and thermal hyperalgesia induced by crushing of the sciatic nerve in mice. | [57] |
PVP, Polyvinylpyrrolidone; SD, Solid dispersion; HPC, Hydroxypropyl cellulose; CM, Curcumin; CM/PVP SD, curcumin/polyvinylpyrrolidone solid dispersion; SOD, Superoxide dismutase; PEG, Polyethylene glycol; TNF-α, tumor necrosis factor alpha; IL, interleukin; NO, Nitric oxide; HPMC, Hydroxypropyl methylcellulose; AST, Aspartate aminotransferase.
4. Conclusions
Solid dispersions are a technological strategy to improve the pharmacological potential of natural or synthetic bioactive molecules, thanks to an increase in their solubility and bioavailability, thus leading to possible enhancement of their biological activities. In this context, this review sought to summarize and critically examine studies conducted with this approach both in vitro and in vivo, which have provided evidence of a significant therapeutic potential that solid dispersions could offer in the context of the most diverse biological activities.
As poorly water-soluble active ingredients incorporated in solid dispersions exhibited improved physicochemical and pharmacological properties, these systems can be targeted in the pharmaceutical industry as possible therapeutic alternatives for certain diseases, with greater selectivity, safety and efficacy than the existing treatments.
References
- Kambayashi, A.; Kiyota, T.; Fujiwara, M.; Dressman, J.B. PBPK modeling coupled with biorelevant dissolution to forecast the oral performance of amorphous solid dispersion formulations. Eur. J. Pharm. Sci. 2019, 135, 83–90.
- Jermain, S.V.; Brough, C.; Williams, R.O. Amorphous solid dispersions and nanocrystal technologies for poorly water-soluble drug delivery—An update. Int. J. Pharm. 2018, 535, 379–392.
- Loftsson, T.; Brewster, M.E. Pharmaceutical applications of cyclodextrins: Basic science and product development. J. Pharm. Pharmacol. 2010, 62, 1607–1621.
- Worku, Z.A.; Aarts, J.; Singh, A.; Mooter, G.V.D. Drug–Polymer Miscibility across a Spray Dryer: A Case Study of Naproxen and Miconazole Solid Dispersions. Mol. Pharm. 2014, 11, 1094–1101.
- Reginald-Opara, J.N.; Attama, A.; Ofokansi, K.; Umeyor, C.; Kenechukwu, F. Molecular interaction between glimepiride and Soluplus®-PEG 4000 hybrid based solid dispersions: Characterisation and anti-diabetic studies. Int. J. Pharm. 2015, 496, 741–750.
- Danda, L.J.D.A.; Batista, L.D.M.; Melo, V.C.S.; Sobrinho, J.L.S.; Soares, M.F.D.L.R. Combining amorphous solid dispersions for improved kinetic solubility of posaconazole simultaneously released from soluble PVP/VA64 and an insoluble ammonio methacrylate copolymer. Eur. J. Pharm. Sci. 2019, 133, 79–85.
- Júnior, W.F.D.S.; Pinheiro, J.G.D.O.; De Menezes, D.L.B.; Silva, N.E.D.S.E.; De Almeida, P.D.O.; Lima, E.S.; Veiga-Junior, V.F.; De Azevedo, E.P.; Lima, E.S. Development, Physicochemical Characterization and In Vitro Anti-Inflammatory Activity of Solid Dispersions of α,β Amyrin Isolated from Protium Oilresin. Molecules 2017, 22, 1512.
- Vasconcelos, T.; Sarmento, B.; Costa, P.C. Solid dispersions as strategy to improve oral bioavailability of poor water soluble drugs. Drug Discov. Today 2007, 12, 1068–1075.
- Leuner, C. Improving drug solubility for oral delivery using solid dispersions. Eur. J. Pharm. Biopharm. 2000, 50, 47–60.
- Zhang, Z.; Chen, Y.; Deng, J.; Jia, X.-B.; Zhou, J.; Lv, H. Solid dispersion of berberine–phospholipid complex/TPGS 1000/SiO2: Preparation, characterization and in vivo studies. Int. J. Pharm. 2014, 465, 306–316.
- Xie, Y.; Yao, Y. Octenylsuccinate hydroxypropyl phytoglycogen enhances the solubility and in-vitro antitumor efficacy of niclosamide. Int. J. Pharm. 2018, 535, 157–163.
- Xie, Y.; Yao, Y. Incorporation with Dendrimer-Like Biopolymer Leads to Improved Soluble Amount and In Vitro Anticancer Efficacy of Paclitaxel. J. Pharm. Sci. 2019, 108, 1984–1990.
- Choi, J.-S.; Cho, N.H.; Kim, D.-H.; Park, J.-S. Comparison of paclitaxel solid dispersion and polymeric micelles for improved oral bioavailability and in vitro anti-cancer effects. Mater. Sci. Eng. C 2019, 100, 247–259.
- Lee, S.H.; Lee, Y.-S.; Song, J.G.; Han, H.-K. Improved In vivo Effect of Chrysin as an Absorption Enhancer Via the Preparation of Ternary Solid Dispersion with Brij®L4 and Aminoclay. Curr. Drug Deliv. 2018, 16, 86–92.
- De Sá, I.S.; Peron, A.P.; Leimann, F.V.; Bressan, G.N.; Krum, B.N.; Fachinetto, R.; Pinela, J.; Calhelha, R.C.; Barreiro, M.F.; Ferreira, I.C.; et al. In vitro and in vivo evaluation of enzymatic and antioxidant activity, cytotoxicity and genotoxicity of curcumin-loaded solid dispersions. Food Chem. Toxicol. 2019, 125, 29–37.
- Wu, R.; Mei, X.; Ye, Y.; Xue, T.; Wang, J.; Sun, W.; Lin, C.; Xue, R.; Zhang, J.; Xu, D. Zn(II)-curcumin solid dispersion impairs hepatocellular carcinoma growth and enhances chemotherapy by modulating gut microbiota-mediated zinc homeostasis. Pharmacol. Res. 2019, 150, 104454.
- Xiong, X.; Zhang, M.; Hou, Q.; Tang, P.; Suo, Z.; Zhu, Y.; Li, H. Solid dispersions of telaprevir with improved solubility prepared by co-milling: Formulation, physicochemical characterization and cytotoxicity evaluation. Mater. Sci. Eng. C 2019, 105, 110012.
- Jiang, Y.; Piao, J.; Cho, H.-J.; Kang, W.-S.; Kim, H. Improvement in antiproliferative activity of Angelica gigas Nakai by solid dispersion formation via hot-melt extrusion and induction of cell cycle arrest and apoptosis in HeLa cells. Biosci. Biotechnol. Biochem. 2015, 79, 1635–1643.
- Guo, S.; Wang, G.; Wu, T.; Bai, F.; Xu, J.; Zhang, X. Solid dispersion of berberine hydrochloride and Eudragit® S100: Formulation, physicochemical characterization and cytotoxicity evaluation. J. Drug Deliv. Sci. Technol. 2017, 40, 21–27.
- Kumar, V.; Mintoo, M.J.; Mondhe, D.M.; Bharate, S.B.; Vishwakarma, R.A.; Bharate, S.S. Binary and ternary solid dispersions of an anticancer preclinical lead, IIIM-290: In vitro and in vivo studies. Int. J. Pharm. 2019, 570, 118683.
- Simonazzi, A.; Davies, C.; Cid, A.G.; Gonzo, E.; Parada, L.; Bermúdez, J.M. Preparation and Characterization of Poloxamer 407 Solid Dispersions as an Alternative Strategy to Improve Benznidazole Bioperformance. J. Pharm. Sci. 2018, 107, 2829–2836.
- Fonseca-Berzal, C.; Palmeiro-Roldán, R.; Escario, J.A.; Torrado, S.; Arán, V.J.; Torrado-Santiago, S.; Gómez-Barrio, A. Novel solid dispersions of benznidazole: Preparation, dissolution profile and biological evaluation as alternative antichagasic drug delivery system. Exp. Parasitol. 2015, 149, 84–91.
- Eloy, J.O.; Saraiva, J.; De Albuquerque, S.; Marchetti, J.M. Solid Dispersion of Ursolic Acid in Gelucire 50/13: A Strategy to Enhance Drug Release and Trypanocidal Activity. AAPS PharmSciTech 2012, 13, 1436–1445.
- Perissutti, B.; Passerini, N.; Trastullo, R.; Keiser, J.; Zanolla, D.; Zingone, G.; Voinovich, D.; Albertini, B. An explorative analysis of process and formulation variables affecting comilling in a vibrational mill: The case of praziquantel. Int. J. Pharm. 2017, 533, 402–412.
- Albertini, B.; Perissutti, B.; Bertoni, S.; Zanolla, D.; Franceschinis, E.; Voinovich, D.; Lombardo, F.; Keiser, J.; Passerini, N. Combining Mechanochemistry and Spray Congealing for New Praziquantel Pediatric Formulations in Schistosomiasis Treatment. Int. J. Mol. Sci. 2019, 20, 1233.
- Fule, R.A.; Meer, T.A.; Sav, A.R.; Amin, P. Artemether-Soluplus Hot-Melt Extrudate Solid Dispersion Systems for Solubility and Dissolution Rate Enhancement with Amorphous State Characteristics. J. Pharm. 2013, 2013, 1–15.
- Fule, R.; Meer, T.; Sav, A.; Amin, P. Solubility and dissolution rate enhancement of lumefantrine using hot melt extrusion technology with physicochemical characterisation. J. Pharm. Investig. 2013, 43, 305–321.
- Crucitti, V.C.; Migneco, L.M.; Piozzi, A.; Taresco, V.; Garnett, M.; Argent, R.H.; Francolini, I. Intermolecular interaction and solid state characterization of abietic acid/chitosan solid dispersions possessing antimicrobial and antioxidant properties. Eur. J. Pharm. Biopharm. 2018, 125, 114–123.
- Al-Obaidi, H.; Kowalczyk, R.M.; Kalgudi, R.; Zariwala, M.G. Griseofulvin solvate solid dispersions with synergistic effect against fungal biofilms. Colloids Surfaces B Biointerfaces 2019, 184, 110540.
- Kanoujia, J.; Kushwaha, P.S.; Saraf, S.A. Evaluation of gatifloxacin pluronic micelles and development of its formulation for ocular delivery. Drug Deliv. Transl. Res. 2014, 4, 334–343.
- Hernandez-Patlan, D.; Solis-Cruz, B.; Pontin, K.P.; Latorre, J.D.; Baxter, M.F.A.; Hernandez-Velasco, X.; Merino-Guzman, R.; Méndez-Albores, A.; Hargis, B.M.; Lopez-Arellano, R.; et al. Evaluation of a Solid Dispersion of Curcumin With Polyvinylpyrrolidone and Boric Acid Against Salmonella Enteritidis Infection and Intestinal Permeability in Broiler Chickens: A Pilot Study. Front. Microbiol. 2018, 9, 1289.
- Hegge, A.B.; Vukićević, M.; Bruzell, E.; Kristensen, S.; Tønnesen, H. Solid dispersions for preparation of phototoxic supersaturated solutions for antimicrobial photodynamic therapy (aPDT). Eur. J. Pharm. Biopharm. 2013, 83, 95–105.
- Alves, T.; Chaud, M.; Grotto, D.; Jozala, A.F.; Pandit, R.; Rai, M.; Dos Santos, C.A. Association of Silver Nanoparticles and Curcumin Solid Dispersion: Antimicrobial and Antioxidant Properties. AAPS PharmSciTech 2017, 19, 225–231.
- Costa, A.R.D.M.; Marquiafável, F.S.; Vaz, M.M.D.O.L.L.; Rocha, B.A.; Bueno, P.C.P.; Amaral, P.L.M.; Barud, H.D.S.; Berreta-Silva, A.A. Quercetin-PVP K25 solid dispersions. J. Therm. Anal. Calorim. 2010, 104, 273–278.
- Ge, Y.; Zhao, X.; Wang, S.; Zu, Y.; Zhao, T.; Sang, M.; Sui, X.; Wang, K. 10 (CoQ10) Prepared by Heat Melt and High-pressure Homogenization Method. Curr. Nanosci. 2014, 10, 292–296.
- Fitriani, L.; Rismawati, E.; Umar, S.; Zaini, E. Solid dispersion of usnic acid-PVP K30 and evaluation of antioxidant activity. Rasayan J. Chem. 2018, 11, 1643–1648.
- Alshehri, S.; Imam, S.S.; Altamimi, M.A.; Hussain, A.; Shakeel, F.; Elzayat, E.M.; Mohsin, K.; Ibrahim, M.; Alanazi, F. Enhanced Dissolution of Luteolin by Solid Dispersion Prepared by Different Methods: Physicochemical Characterization and Antioxidant Activity. ACS Omega 2020, 5, 6461–6471.
- Shin, M.-S.; Yu, J.S.; Lee, J.; Ji, Y.S.; Joung, H.J.; Han, Y.-M.; Yoo, H.H.; Kang, K.; Shin, Y. A Hydroxypropyl Methylcellulose-Based Solid Dispersion of Curcumin with Enhanced Bioavailability and its Hepatoprotective Activity. Biomolecules 2019, 9, 281.
- Lian, X.; Dong, J.; Zhang, J.; Teng, Y.; Lin, Q.; Fu, Y.; Gong, T. Soluplus® based 9-nitrocamptothecin solid dispersion for peroral administration: Preparation, characterization, in vitro and in vivo evaluation. Int. J. Pharm. 2014, 477, 399–407.
- Qusa, M.H.; Siddique, A.B.; Nazzal, S.; El Sayed, K. Novel olive oil phenolic (−)-oleocanthal (+)-xylitol-based solid dispersion formulations with potent oral anti-breast cancer activities. Int. J. Pharm. 2019, 569, 118596.
- Chen, B.; Wang, X.; Zhang, Y.; Huang, K.; Liu, H.; Xu, D.; Li, S.; Liu, Q.; Huang, J.; Yao, H.; et al. Improved solubility, dissolution rate and oral bioavailability of main biflavonoids from Selaginella doederleinii extract by amorphous solid dispersion. Drug Deliv. 2020, 27, 309–322.
- Yu, C.; Mei, X.-T.; Zheng, Y.-P.; Xu, D. Gastroprotective effect of taurine zinc solid dispersions against absolute ethanol-induced gastric lesions is mediated by enhancement of antioxidant activity and endogenous PGE2 production and attenuation of NO production. Eur. J. Pharmacol. 2014, 740, 329–336.
- Gou, J.; Fei, S.; Xue, B.; Zhang, J.; Zhang, Y.; Wang, X.; Zhang, Y.; Yin, T.; He, H.; Tang, X. Triacetylated andrographolide solid dispersions: Preparation, stability study and in vivo anti-inflammation in mice ulcerative colitis model. J. Drug Deliv. Sci. Technol. 2019, 51, 91–100.
- He, Y.; Liu, H.; Bian, W.; Liu, Y.; Liu, X.; Ma, S.; Zheng, X.; Du, Z.; Zhang, K.; Ouyang, D. Molecular Interactions for the Curcumin-Polymer Complex with Enhanced Anti-Inflammatory Effects. Pharmaceutics 2019, 11, 442.
- Teixeira, C.C.C.; Mendonça, L.M.; Bergamaschi, M.M.; Queiroz, R.H.C.; Souza, G.E.P.; Antunes, L.M.G.; Freitas, L. Microparticles Containing Curcumin Solid Dispersion: Stability, Bioavailability and Anti-Inflammatory Activity. AAPS PharmSciTech 2015, 17, 252–261.
- Chuah, A.M.; Jacob, B.; Jie, Z.; Ramesh, S.; Mandal, S.; Puthan, J.K.; Deshpande, P.; Vaidyanathan, V.V.; Gelling, R.W.; Patel, G.; et al. Enhanced bioavailability and bioefficacy of an amorphous solid dispersion of curcumin. Food Chem. 2014, 156, 227–233.
- Jana, S.; Ali, S.A.; Nayak, A.K.; Sen, K.K.; Basu, S.K. Development of topical gel containing aceclofenac-crospovidone solid dispersion by “Quality by Design (QbD)” approach. Chem. Eng. Res. Des. 2014, 92, 2095–2105.
- Kenechukwu, F.C.; Ofokansi, K.C.; Ezugwu, R.O.; Attama, A.A. Improved dissolution and anti-inflammatory activity of ibuprofen-polyethylene glycol 8000 solid dispersion systems. Int. J. Pharm. Investig. 2016, 6, 139–147.
- Malipeddi, V.R.; Dua, K.; Awasthi, R. Development and characterization of solid dispersion-microsphere controlled release system for poorly water-soluble drug. Drug Deliv. Transl. Res. 2016, 6, 540–550.
- Al-Remawi, M.; Ali, A.M.A.; Khames, A.; Hamaidi, M. Meloxicam-Paracetamol Binary Solid Dispersion Systems with Enhanced Solubility and Dissolution Rate:Preparation, Characterization and In Vivo Evaluation. J. Pharm. Innov. 2017, 12, 206–215.
- Li, W.; Qing, S.; Zhi, W.; Yao, H.; Fu, C.; Niu, X. The pharmacokinetics and anti-inflammatory effects of chelerythrine solid dispersions in vivo. J. Drug Deliv. Sci. Technol. 2017, 40, 51–58.
- Onoue, S.; Nakamura, T.; Uchida, A.; Ogawa, K.; Yuminoki, K.; Hashimoto, N.; Hiza, A.; Tsukaguchi, Y.; Asakawa, T.; Kan, T.; et al. Physicochemical and biopharmaceutical characterization of amorphous solid dispersion of nobiletin, a citrus polymethoxylated flavone, with improved hepatoprotective effects. Eur. J. Pharm. Sci. 2013, 49, 453–460.
- Balata, G.; Shamrool, H. Spherical agglomeration versus solid dispersion as different trials to optimize dissolution and bioactivity of silymarin. J. Drug Deliv. Sci. Technol. 2014, 24, 478–485.
- Hwang, D.H.; Kim, Y.-I.; Cho, K.H.; Poudel, B.K.; Choi, J.Y.; Kim, D.-W.; Shin, Y.-J.; Bae, O.-N.; Yousaf, A.M.; Yong, C.S.; et al. A novel solid dispersion system for natural product-loaded medicine: Silymarin-loaded solid dispersion with enhanced oral bioavailability and hepatoprotective activity. J. Microencapsul. 2014, 31, 619–626.
- Zawar, L.R.; Bari, S.B. Preparation, characterization and in vivo evaluation of antihyperglycemic activity of microwave generated repaglinide solid dispersion. Chem. Pharm. Bull. 2012, 60, 482–487.
- Pokharkar, V.; Kutwal, M.; Mandpe, L. Pioglitazone Solid Dispersion System Prepared by Spray Drying Method: In Vitro and In Vivo Evaluation. PDA J. Pharm. Sci. Technol. 2013, 67, 23–34.
- Moreira, C.D.L.D.F.A.; Pinheiro, J.G.D.O.; Da Silva-Júnior, W.F.; Barbosa, E.G.; Lavra, Z.M.M.; Pereira, E.W.M.; Resende, M.M.; De Azevedo, E.P.; Quintans-Júnior, L.J.; Araújo, A.A.D.S.; et al. Amorphous solid dispersions of hecogenin acetate using different polymers for enhancement of solubility and improvement of anti-hyperalgesic effect in neuropathic pain model in mice. Biomed. Pharmacother. 2018, 97, 870–879.




