Video Upload Options
High-energy and high-atom-number (HZE) space radiation poses an inevitable potential threat to astronauts on deep space exploration missions. In addition to inducing damage at the DNA level, space radiation also causes epigenetic changes, such as microRNAs, which is important to understanding of the machanisms underlying space radiobiological effects.
1. MicroRNAs
MicroRNAs (miRNAs) are a class of small single-stranded endogenous noncoding RNAs of 18-24 nucleotides at length, which are identified in various organisms, including mammals, plants, and many microorganisms. They are transcribed from the genomes by RNA polymerase II or RNA polymerase III, capable of suppressing the expression of a large number of genes by binding to the 3′-untranslated region (3′-UTR) of their transcripts [1].
Early studies have found that most of miRNA genes are located in the intergenic region 1 kb away from the annotated genes, while some of miRNA genes are located in the introns of known genes and share the transcriptional elements with their host genes. It is reported that about 50% of miRNA gene is located closely to other miRNAs as a cluster [2][3]. Most miRNA genes have their own promoters and the clustered miRNAs come from the polycistronic transcripts[4][5]. Most of miRNAs are transcribed from the genomes by RNA polymerase II or RNA polymerase III under strict regulation [4]. It was found by Bradley that 70% of the mammalian miRNA genes were located in a specific transcription unit, and 117 of 232 miRNAs were located in the introns, of which 90 were located in the introns of protein- encoding genes, and 27 were located in non-coding RNAs in the introns. Thus, miRNAs can be divided into three types according to their location: the exonic miRNAs located in the non-coding transcription units, the intronic miRNAs located in the non-coding transcription units, and the intronic miRNAs in the protein encoding units [6].
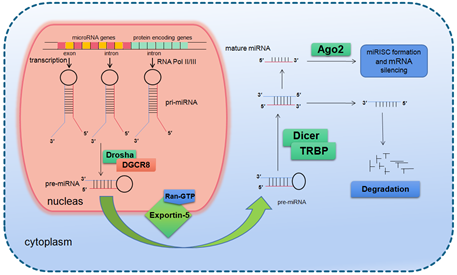
The primary transcription product of a miRNA gene is primary miRNA (pri-miRNA), which is thousands of nucleotides long and contains a hairpin structure. Once the primary miRNA is produced, its 5′ end was added with a methylation hat while the 3′ end was polyadenylated [7]. The modified primary miRNA is first cut by the nuclear ribonuclease III (RNase III) Drosha in the nucleus to produce about 70 nucleotides long precursor miRNA molecules (precursor miRNA, pre-miRNA) [8]. It is generally believed that the residual flanking sequence will be degraded in the nucleus, but its specific function remains to be clarified. Drosha is a conserved protein of 160 kDa size containing two series of RNase III domains (RIIIDs) and a double stranded RNA binding domain (dsRBD) [9], which in the human body associates with DiGeorge syndrome critical region gene 8 (DGCR8) to form a microprocessor complex of 650 kDa [10]. At present, it is believed that DGCR8 can assist Drosha to identify the substrate, and the determinant of the specificity for substrate recognition is the structure of pri-miRNA [11][12][13].
In mammals, the two-step splicing of pri-miRNA occurs in the nucleus and cytoplasm (Figure 1). The nuclear export of a pre-miRNA is mediated by nuclear transport receptor exportin-5. It combines pre-miRNA and co-factor Ran-GTP in the nucleus. Once exported, GTP is hydrolyzed to GDP, resulting in the release of pre-miRNA from the transport complex. When exportin-5 is knocked-down, the amounts of both pre-miRNA and mature miRNA in the cytoplasm decrease, but the pre-miRNA in the nucleus does not accumulate, which indicates that pre-miRNA is extremely unstable in the nucleus or gets stabilized by binding with exportin-5 [14][15]. The homologues of Drosha and DGCR8/Pasha were not found in plants and yeast. The study in Arabidopsis found the nucleoprotein Dcl1 was one of the Dicer-like proteins and was involved in processing miRNA [16][17].
Figure 1. Basic miRNA processing pathway.
After being exported to the cytoplasm, the pre-miRNA is further spliced by RNase III nuclease Dicer to generate ~22 nt double stranded RNA molecules. It is reported that the knockout of Dicer in Caenorhabditis elegans (C. elegans) causes the accumulation of pre-miRNA and the decrease of mature miRNA [18][19], providing direct evidence for Dicer as a RNA enzyme in the maturation process of miRNA. Dicer is about 200 kDa of the multi-domain protein, which contains two RIIIDs domains and a dsRBD domain like Drosha, and the difference is that Dicer contains a long N short sequence, including a DEAD-BOX RNA helicase domain, a DUF283 domain and a PAZ domain. The PAZ domain is also found in the Argonaute family of proteins, which specifically bind to the 3′ terminal of small RNA molecules [20][20,21]. There is only one kind of Dicer in mammals and nematodes, which plays a key role in the biogenesis of siRNA and miRNA [21][22][23]. The miRNA dimer was subsequently integrated into the complex called miRNP (ribonucleoprotein complex containing miRNA) or miRISC (miRNA-induced silencing complex). The life span of miRNA dimer is short. Once combined with Ago protein, the double strands are quickly unraveled, a chain is stabilized and the other is lost. The retention or degradation of the chain depends on its thermodynamic stability [24][25][26]. The mammalian Dicer/Ago/miRNA complex also interacts with a number of other proteins, such as Gemin3, Gemin4, Mov10, and GW182 [27][28][29][30].
Since the first miRNA Lin-4 was identified in nematode in 1993 as a development regulation gene, the number of miRNAs found in all species has reached more than 38,000 and the number of human miRNAs also exceeded 2600 (http://www.mirbase.org/). It is presumed that more than 60% of the human genes are regulated by miRNAs [31][32][33]. Precious studies suggest that miRNAs regulate gene expression through translational inhibition and/or degradation of target mRNAs [34][35]. There is no final conclusion on which mechanism will play a major role. It is speculated that the number, type and location of mismatch in miRNA/mRNA dimers as well as the cell type determines the triggering of degradation or translation inhibition [36][33]. A miRNA interacts with its target genes to form a regulatory network, which acts in almost all life processes. It has been shown by many studies that miRNAs are a key group of important modulators in biological processes and play important roles in development [37][38], organogenesis [39][40], cell proliferation and differentiation [41], cell cycle regulation [41][42], cell apoptosis [43][44][45], aging [46][47][48], pathogenesis [49][50], cellular response to stresses [51][52][53][54], etc.
As a key modulator in radiation response of human and other organisms, miRNAs has been found to respond to various radiation (both ionizing radiation (IR) and non-ionizing radiation) as well as to regulate a lot of genes involved in the cellular radiation response [55][56][57]. As a key radiation type confronted by the astronauts in the manned spaceflight missions, high-energy and high-Z (HZE) particles-related biological effects and the underlying mechanisms have attracted much attention from space radiobiologists. In particular research on HZE particle radiation-induced miRNAs and their roles in the regulation of the corresponding biological effects has made great progress.
2. Space Radiation and the Health Effects
With the deepening of space exploration, the duration of astronauts’ permanence in space will become longer and longer. The safety evaluation of space explorations beyond the low Earth orbit (LEO) inevitably is one of the most important topics in space science since the radiation levels exceed those routinely received by terrestrial radiation workers, or astronauts in near-Earth orbits such as the International Space Station (ISS). Because of the lack of atmosphere and magnetic field and thereby important shielding properties the only shielding measure is the spacecraft bulkhead and high energy space radiation is an unavoidable danger for astronauts in manned space missions.
Basically, there are three main sources of space radiation: (1) Trapped low Earth orbit (LEO) radiation, in which the main components are protons and electrons with relatively low energy. (2) Incidental and high dose radiation from solar particle events (SPEs). Solar flares emit a large number of high-energy protons, and a small amount of α particles and heavy ions. (3) Low dose rate galactic cosmic rays (GCR). The protons account for about 85%, followed by 14% α particles and 1% of heavy ions. Although low in dose and dose rate, GCR is highly energetic and powerfully penetrating. There are no shielding materials capable of blocking it completely. Exposure of astronauts to GCR causes clustered DNA damages that are difficult to repair, which are strongly lethal, mutagenic and carcinogenic [58][59][60]. Heavy ions are high in ionization density (linear energy transfer, LET), although their abundance is low [61][62][63]. In addition to the abovementioned radiation, secondary particles produced by these primary particles through the shielding materials form a secondary radiation environment, including photons, electrons, protons and neutrons, which can also lead to biological effects such as DNA damage, gene mutation, cell transformation, and other biological effects [64].
The human experience in space flights is only about six decades old, inaugurated by the first complete Earth orbit flight by Yuri Gagarin, and currently continuing on the International Space Station. To date, manned missions have been limited to near-Earth orbits, with the moon as our farthest destination from Earth. Historical space radiation career exposures for astronauts from all NASA missions through December 1999 (including early Mercury, Gemini, STS, and Apollo Missions) involved total exposures of less than about 20 mSv [65]. With the advent of Skylab and MIR, total career exposure levels increased to a maximum of nearly 200 mSv. It is estimated that astronauts will be exposed to 1.84 ± 0.30 mSv/day of GCR in interplanetary space and 0.64 ± 0.12 mSv/day of GCR on the Mars surface, amounting to a total mission dose equivalent of ~1.01 Sv for a round trip to Mars with 180 days (each way) cruise, and 500 days stay on the Mars surface for a particular solar cycle [66]. Besides, radiation tissue equivalent doses deposited in blood-forming organs when encountering a large SPE may reach 1.93 Sv even behind 5 g/cm2 Al shielding [67]. Missions into deep space, due to the requisite longer duration of the planned missions, may pose greater risks due to the increased potential for exposure to complex radiation fields comprised of a broad range of radiation types and energies from cosmic and unpredictable solar sources. Ionizing radiation prevalent in space such as protons, carbon, argon and iron ions covers both a broad range of energies and a highly diverse range of radiation quality. LETs of these particles range from <10 to >200 keV/µm. Protons are more prevalent with relatively low LET values, whereas the iron ions are relatively rare, but with high LET values. Dose rate measured in LEO is of the order of 1 mSv/day, but will be higher on missions to Mars as mentioned above. It is an enormous and complex task to assess the biological and clinical effects of all possible space radiation scenarios. Reminders of the presence of low-flux particle radiation fields have graphically been visualized in light-flash phenomena experienced by many space travelers [68]. Evidence exists from the accelerator-based human exposures with muons [69], pions [70], helium ions [71], carbon ions [72], and nitrogen ions [73]. Visual phenomena have also been noted by human subjects on exposure to neutrons of various energies. Several human subjects saw a multitude of bright colorless flashes on exposure to neutrons, which were described as “a bunch of stars moving or blinking” and are similar to light flashes and streaks seen by astronauts on translunar flight. These phenomena are caused by interaction with retinal rods by proton recoils and by α particles released from neutron reactions with carbon and oxygen [74][75][76]. However, space radiation exposure influences multiple organs and physiological systems in complicated ways. NASA has classified the biomedical consequences into four risk areas [77]: (1) Degenerative tissue effects from radiation exposure, e.g., cardiovascular disease, cataract formation, and premature aging; (2) Radiation-induced carcinogenesis; (3) CNS (central nervous system) injury caused by radiation exposure, leading to deficits in cognitive and executive function, inducing fatigue, and degrading crew performance; (4) Radiation syndromes caused by SPE. The high doses of radiations from large SPEs induce acute radiation syndrome effects, such as nausea, emesis, haemorrhaging, or, possibly, even death. However, the underlying molecular mechanisms are still under considerations.
3. MiRNAs Involved in the Biological Responses to Space Radiation
Up to now, a number of studies have found that the biological responses to space radiation are related to miRNAs, as demonstrated in Table 1. Khan et al. examined the miRNA expression in selected mouse organs (testis, brain, and liver) exposed to whole-body proton irradiation (2 Gy). By bioinformatics analysis they revealed dysregulation of 14 miRNAs in mouse testis, 8 in liver, and 8 in brain and a possible mechanism of proton particle involvement in the onset of tumorigenesis [78]. Templin et al. studied the expression changes of miRNA derived from mouse blood using quantitative real-time polymerase chain reaction (qRT-PCR) in mice exposed to 600 MeV protons at doses of 0.5 or 1.0 Gy. They found 26 miRNAs were differentially expressed and mouse blood miRNA signatures are radiation type- and dose-dependent [79].
Table 1. Studies relating to miRNAs responding to space radiation.[80][81][82][83]
|
Studied Materials |
Radiation |
MiRNA Expression |
Target Proteins/Pathways |
MiRNA Detection Methods |
Predicted Biological Consequences |
Refs |
|
Testis, brain and liver tissues of Balb/C male mice, 4 h after exposure |
2 Gy whole-body proton irradiation |
Dysregulation of 14 mouse testis, 8 liver, and 8 brain miRNAs, with 20 up- and 10 down-regulated |
- |
NGS for profiling, no validation |
Regulation of DNA damage response and tumorigenesis |
[78] |
|
Blood of C57BL/6 male mice, 6 h/24 h after exposure |
0.5 or 1.0 Gy whole-body proton irradiation |
Dysregulation of 26 miRNAs, with 5 up- and 21 down-regulated |
- |
qRT-PCR |
Regulation of nucleic acid metabolic process and system development |
[79] |
|
Bystander 3-D artificial human tissues, 8 h, 1, 2, 3, 4, 5 and 7 days post-IR |
5.4 Gy α particle irradiation for the first layer of cells |
Dysregulation of 12 miRNAs, with 7 up- and 5 down-regulated |
DNMT3a, MCL1, BCL2, RB1, E2F1/ apoptosis pathway, TNF signaling pathway, cellular senescence pathway |
miRNA microarray for profiling, no validation |
Apoptosis, cell cycle deregulation and DNA hypomethylation |
[84] |
|
Three human cell lines (A549, THP-1 and HFL), 24 h/72 h after exposure |
0.5, 1.0 or 1.5 Gy α particle irradiation |
Dysregulation of 13 miRNAs, with 8 up- and 5 down-regulated |
- |
miRNA microarray for profiling, qRT-PCR for validation |
Regulation of ribosomal assembly, lung carcinoma development, TGF-β signaling, cell communication, etc |
[85] |
|
Immortalized human bronchial epithelial cells (BEAS-2B), 40 passages after exposure |
0.1, 0.25, 0.5 or 1 Gy α particle irradiation |
Dysregulation of 68 miRNAs, with 20 up- and 48 down-regulated |
PEG10, ARHGAP26, IRS1/ β-catenin pathway, PI3K-Akt signaling pathway, TGF-β signaling pathway |
miRNA microarray for profiling, qRT-PCR for validation |
Promotion of malignant transformation |
[86] |
|
Blood of C57BL/6 male mice, 6 h/24 h after exposure |
Whole-body irradiation by 0.1 or 0.5 Gy iron ions |
Dysregulation of 14 miRNAs, with 6 up- and 8 down-regulated |
- |
qRT-PCR |
Regulation of transcription, nucleic-acid metabolism, and development |
[87] |
|
Testis of Swiss-Webster male mice, 4 weeks after exposure |
Whole-body irradiation by 2 Gy carbon ions |
Dysregulation of 70 miRNAs, with 56 up- and 14 down-regulated |
- |
NGS for profiling, qRT-PCR for validation |
Apoptosis of spermatogenic cells |
[88] |
|
Blood of Kunming male mice, 24 h after exposure |
Whole-body irradiation by 0.5 or 2 Gy carbon ions |
Dysregulation of 12 miRNAs, with 6 up- and 6 down-regulated |
- |
miRNA microarray for profiling, qRT-PCR for validation |
Regulation of cell cycle transition, immune system and carcinogenesis |
[89] |
|
Blood of Kunming male mice, 24 h after exposure |
Whole-body irradiation by 0.25 or 0.5 Gy carbon ions |
Upregulation of let-7a-5p and miR-200b-5p |
- |
miRNA microarray for profiling, qRT-PCR for validation |
- |
[90] |
Besides the involvement of miRNAs in response to proton radiation, they were also found to participate in the biological responses to particles of higher LET, such as α particle and heavy ions. Kovalchuk et al. analyzed microRNAome changes in bystander tissues after 5.4 Gy α-particle microbeam irradiation of 3-D artificial human tissues using miRNA microarrays and found that miRNAs play a profound role in the manifestation of late radiation-induced bystander effect (RIBE) end points . Chauhan et al. studied the miRNA expression patterns in three human cell lines (A549, THP-1 and HFL) exposed to 0.5 Gy, 1.0 Gy and 1.5 Gy of α-particles at a low dose-rate (0.98 ± 0.01 Gy/h), and found cell-specific responses of 13 miRNAs. Besides, bioinformatics analysis suggested the α-particle induced miRNA mapped to target genes related to ribosomal assembly, lung carcinoma development, TGF-β signaling, cell communication and keratin sulfate . Nie et al. studied the malignant transformation of immortalized human bronchial epithelial cells (BEAS-2B) irradiated by 0.25 Gy α-particles using miRNA-mRNA networks. Sixty-eight miRNAs were found to be dysregulated, among which miR-107 and miR-494 were predicted to play a role in α-particles-mediated cellular malignant transformation processes by bioinformatics analysis .
Templin et al. also compared the miRNA expression signatures in peripheral blood of mice exposed to either γ-rays or 56Fe ions and found that miRNA expression signatures were radiation type-specific and dose- and time-dependent . He et al. investigated the toxicity in testis of mice following enterocoelia irradiation with 2 Gy carbon ions by miRNA sequencing and bioinformatics analyses. Differentially expressed miRNAs were found to be involved in the regulation of metabolism, development, and reproduction. Wei et al. analyzed miRNA expression profiles with miRNA PCR arrays at 24 h post heavy ion irradiation, and developed a universal model to predict the degree of exposure to different radiation types with high sensitivity and specificity based on five miRNAs (miR-183-5p, miR-9-3p, miR-200b-5p, miR-342-3p and miR-574-5p) that showed a significant response to 0.1–2 Gy of carbon-ion, iron-ion or X-rays. In another study, this team exposed Kunming mice to different doses of carbon ions and X-rays and found two miRNAs (let-7a-5p, miR-200b-5p) in the serum of irradiated mice were up-regulated significantly and exhibited dose- (0.1~2 Gy) and time-dependence (6~72 h), which may serve as potential noninvasive indicators for space radiation.
In addition to the characteristic of being rich in high LET radiation, the effects of space radiation on astronauts are affected by a variety of other space environmental factors, such as microgravity, weak magnetic field, and short circadian rhythm. As shown in Table 2, several studies have been carried out to identify the miRNAs participating in the regulation of compound effect of space radiation and other space environment factors. Researchers from Padua University analyzed miRNA expression profile in human peripheral blood lymphocytes (PBL) incubated for 4 and 24 h in normal gravity (1 g) and in modeled microgravity (MMG) during the repair time after irradiation with 0.2 or 2 Gy of γ-rays. They found that MMG altered miRNA expression signature of irradiated PBL by decreasing the number of radio-responsive miRNAs. Integrated analyses of transcriptome and microRNAome suggested that modeled microgravity affected the DNA-damage response to irradiation. Fu et al. analyzed RNA expression profiles in human lymphoblastoid TK6 cells incubated for 24 h under static or stimulated microgravity after 2 Gy γ-ray irradiation. Although no differentially-expressed miRNAs were identified under either simulated microgravity or irradiation conditions, both miR-15b and miR-221 were found to be differentially expressed under compound conditions with an interactive effect.
Table 2. Studies relating to miRNAs responding to space compound environment.
|
Studied Materials |
Experimental Treatments |
MiRNA Expression |
Target Proteins/Pathways |
MiRNA Detection Methods |
Predicted Biological Consequences |
Refs |
|
Human peripheral blood lymphocytes |
4 or 24 h incubation in modeled microgravity after irradiation with 0.2 or 2 Gy γ-rays |
Downregulation of let-7i*, miR-7, miR-7-1*, miR-144, miR-200a, miR-598, miR-650, upregulation of miR-27a, miR-99b by combined action |
ATM, FANCF, STAT5A, BAX/DNA damage response pathway, p53 pathway |
miRNA microarray for profiling, qRT-PCR for validation |
Modeled microgravity inhibits the DNA-damage response to IR |
[80] |
|
Human lymphoblastoid TK6 cells |
24 h incubation under simulated microgravity after irradiation with 2 Gy γ-rays |
Hsa-miR-15b was downregulated while hsa-miR-221 was upregulated by combined action |
Apoptosis pathway, NF-κB pathway, TNF signaling pathway |
miRNA microarray for profiling, qRT-PCR for validation |
Regulation of apoptosis process and immune response by combined action |
[81] |
|
C. elegans (Bristol N2) |
7 h after a 16.5-day shuttle spaceflight on Shenzhou-8, with 1.92 mGy space radiation in static slot and 2.27 mGy in centrifuge slot |
The expression of 23 miRNAs changed when exposed to a space synthetic environment and a space radiation environment |
Capg-2, deb-1, ZK180.5, egl-5, C07H4.1, wrk-1, nep-11, odr-2, eff-1, air-2, bath-41, etc/DNA damage response pathway, apoptosis pathway |
miRNA microarray for profiling, qRT-PCR for validation |
Regulation of embryonic development, growth and body morphogenesis, biological behavioural responses, DNA damage response, etc |
|
|
C. elegans (wild-type strain vs. ced-1 mutant) |
7 h after a 16.5-day shuttle spaceflight on Shenzhou-8, with 1.92 mGy space radiation in static slot and 2.27 mGy in centrifuge slot |
Differential miRNA expression increased from 43 (ground control condition) to 57 and 91 in spaceflight and spaceflight control conditions |
Ced-10, drp-1, hsp-1/ DNA damage response pathway, p53 pathway |
miRNA microarray for profiling, qRT-PCR for validation |
Regulation of apoptosis, neurogenesis larval development, ATP metabolism and GTPase-mediated signal transduction |
[91] |
The asterisk (*) indicates a miRNA expressed at low levels relative to the miRNA from the opposite arm of the same pre-miRNA hairpin.
Furthermore, some studies were also carried out to gain insight into the effects of spaceflight on miRNA expression profile. Some researchers from Dr. Yeqing Sun’s group explored the miRNA expression profile changes in space-flown C. elegans larvae that experienced the 16.5-day shuttle spaceflight on Shenzhou-8 spacecraft. 23 miRNAs were found to express differentially in spaceflight groups compared with the ground control group and were predicted to be involved in the regulation of developmental processes, growth and body morphogenesis, DNA damage response as well as biological behavioural responses [89,90]. Gao et al. from the same team also investigated miRNAome and mRNA expression in the ced-1 C. elegans mutant vs. the wild-type strain, both of which underwent spaceflight, spaceflight 1g-centrifuge control and ground control conditions during the Shenzhou-8 mission, and found differential miRNA expression increased from 43 (ground control condition) to 57 and 91 in spaceflight and spaceflight control conditions, respectively, suggesting several miRNAs responding to space radiation were deregulated by microgravity, which is consistent with the finding of Girardi’s group. Some of differentially expressed miRNAs were predicted to regulate apoptosis, neurogenesis larval development and ATP metabolism [91].
Considering the above studies, we can see that for the same cell or tissue type, the miRNA expression profile is radiation type-, dose- and time-dependent. The miRNA expression profiles are also significantly different in varying cell or tissue types even though exposed to the same radiation of the same dose. This suggests that we should consider radiation type, dose, biological samples, as well as sample collection time comprehensively when designing space radiation-related miRNA experiments. In addition, we note that both miR-150-5p and miR-342-3p are down-regulated in blood samples of different mouse models (C57BL/6 or Kunming mice) at 24 h after exposed to different radiation (0.5 Gy iron or carbon ions), implying their important regulatory roles in biological effects of heavy ion radiation. Although a large number of miRNAs have been identified to be involved in the responses to space radiation, few functional experiments were carried out. Zhu et al. found that high LET IR promoted miR-21 expression through the EGFR/STAT3 pathway and miR-21 played an important role in high LET IR-induced carcinogenesis [92][93]. Zhang et al. found miR-21 promoted formation of high LET (Fe ions) irradiation-induced reactive oxygen species (ROS) by targeting both SOD3 and TNFα and contributed to an elevation of IR-induced cell transformation [94]. Wang et al. found that radiation didn’t induce any lung tumorigenesis in miR-21–/– mice while miR-21 knock-in mice showed high spontaneous lung tumor incidences. By further analysis, miR-21 was found to be upregulated by oxygen, silicon and iron ion irradiation and contribute to radiation-induced lung tumorigenesis, whereas its level in serum could be used as a biomarker for predicting high-LET radiation-induced lung tumorigenesis [95]. Kim et al. found high energy proton irradiation induced down-regulation of miR-31-5p in mouse serum while its inhibition protected human colonic epithelial cells against ionizing radiation in an hMLH1-dependent manner [96]. However, most studies involving space radiation-related miRNAs are limited to description of radiation-induced changes of miRNA profiles, the functions of the differentially expressed miRNAs and the underlying mechanisms in space radiation-induced biological effects are still not revealed.
References
- Bartel, D. P., MicroRNAs: Target Recognition and Regulatory Functions. Cell 2009, 136, 215–233.
- Lau, N. C.; Lim, L. P.; Weinstein, E. G.; Bartel, D. P. An abundant class of tiny RNAs with probable regulatory roles in Caenorhabditis elegans. Science 2001, 294, 858–862.
- Mourelatos, Z.; Dostie, J.; Paushkin, S.; Sharma, A.; Charroux, B.; Abel, L.; Rappsilber, J.; Mann, M.; Dreyfuss, G. miRNPs: a novel class of ribonucleoproteins containing numerous microRNAs. Genes Dev. 2002, 16, 720–728.
- Cai, X.; Hagedorn, C. H.; Cullen, B. R. Human microRNAs are processed from capped, polyadenylated transcripts that can also function as mRNAs. RNA 2004, 10, 1957–1966.
- Lee, Y.; Kim, M.; Han, J.; Yeom, K. H.; Lee, S.; Baek, S. H.; Kim, V. N. MicroRNA genes are transcribed by RNA polymerase II. EMBO J. 2004, 23, 4051–4060.
- Rodriguez, R. E.; Mecchia, M. A.; Debernardi, J. M.; Schommer, C.; Weigel, D.; Palatnik, J. F. Control of cell proliferation in Arabidopsis thaliana by microRNA miR396. Development 2010, 137, 103–112.
- Kim, V. N., MicroRNA biogenesis: coordinated cropping and dicing. Nat. Rev. Mol. Cell. Biol. 2005, 6, 376–385.
- Kim, S.; Song, M. L.; Min, H.; Hwang, I.; Baek, S. K.; Kwon, T. K.; Park, J. W. miRNA biogenesis-associated RNase III nucleases Drosha and Dicer are upregulated in colorectal adenocarcinoma. Oncol. Lett. 2017, 14, 4379–4383.
- Pong, S. K.; Gullerova, M. Noncanonical functions of microRNA pathway enzymes—Drosha, DGCR8, Dicer and Ago proteins. FEBS Lett. 2018, 592, 2973–2986.
- Nguyen, T. A.; Park, J.; Dang, T. L.; Choi, Y. G.; Kim, V. N. Microprocessor depends on hemin to recognize the apical loop of primary microRNA. Nucleic Acids Res. 2018, 46, 5726–5736.
- Spadotto, V.; Giambruno, R.; Massignani, E.; Mihailovich, M.; Maniaci, M.; Patuzzo, F.; Ghini, F.; Nicassio, F.; Bonaldi, T., PRMT1-mediated methylation of the microprocessor-associated proteins regulates microRNA biogenesis. Nucleic Acids Res. 2020, 48, 96–115.
- Fletcher, C. E.; Godfrey, J. D.; Shibakawa, A.; Bushell, M.; Bevan, C. L. A novel role for GSK3beta as a modulator of Drosha microprocessor activity and MicroRNA biogenesis. Nucleic Acids Res. 2017, 45, 2809–2828.
- Nguyen, T. L.; Nguyen, T. D.; Bao, S.; Li, S.; Nguyen, T. A. The internal loops in the lower stem of primary microRNA transcripts facilitate single cleavage of human Microprocessor. Nucleic Acids Res. 2020, 48, 2579–2593.
- Wu, K.; He, J.; Pu, W.; Peng, Y. The Role of Exportin-5 in MicroRNA Biogenesis and Cancer. Genom. Proteom. Bioinform. 2018, 16, 120–126.
- Hessam, S.; Sand, M.; Skrygan, M.; Gambichler, T.; Bechara, F. G. Inflammation induced changes in the expression levels of components of the microRNA maturation machinery Drosha, Dicer, Drosha co-factor DGRC8 and Exportin-5 in inflammatory lesions of hidradenitis suppurativa patients. J. Dermatol. Sci. 2016, 82, 166–174.
- Reis, R. S.; Eamens, A. L.; Roberts, T. H.; Waterhouse, P. M. Chimeric DCL1-Partnering Proteins Provide Insights into the MicroRNA Pathway. Front. Plant Sci. 2015, 6, 1201.
- Song, L.; Han, M. H.; Lesicka, J.; Fedoroff, N. Arabidopsis primary microRNA processing proteins HYL1 and DCL1 define a nuclear body distinct from the Cajal body. Proc. Natl. Acad. Sci. USA 2007, 104, 5437–5442.
- Nelson, C.; Ambros, V., Trans-splicing of the C. elegans let-7 primary transcript developmentally regulates let-7 microRNA biogenesis and let-7 family microRNA activity. Development 2019, 146, dev172031.
- Zisoulis, D. G.; Kai, Z. S.; Chang, R. K.; Pasquinelli, A. E., Autoregulation of microRNA biogenesis by let-7 and Argonaute. Nature 2012, 486, 541–544.
- Elkarhat, Z.; Elkhattabi, L.; Charoute, H.; Morjane, I.; Errouagui, A.; Carey, F.; Nasser, B.; Barakat, A.; Rouba, H. Identification of deleterious missense variants of human Piwi like RNA-mediated gene silencing 1 gene and their impact on PAZ domain structure, stability, flexibility and dimension: in silico analysis. J. Biomol. Struct. Dyn. 2019, 1–7, doi:10.1080/07391102.2019.1678522.
- Kandeel, M.; Kitade, Y. In silico molecular docking analysis of the human Argonaute 2 PAZ domain reveals insights into RNA interference. J. Comput.-Aided Mol. Des. 2013, 27, 605–614.
- Fukunaga, R.; Han, B. W.; Hung, J. H.; Xu, J.; Weng, Z.; Zamore, P. D. Dicer Partner Proteins Tune the Length of Mature miRNAs in Flies and Mammals. Cell 2012, 151, 912.
- Betancur, J. G.; Tomari, Y., Dicer is dispensable for asymmetric RISC loading in mammals. RNA 2012, 18, 24–30.
- Garner, A. L.; Lorenz, D. A.; Sandoval, J.; Gallagher, E. E.; Kerk, S. A.; Kaur, T.; Menon, A. Tetracyclines as Inhibitors of Pre-microRNA Maturation: A Disconnection between RNA Binding and Inhibition. ACS Med. Chem. Lett. 2019, 10, 816–821.
- Dallaire, P.; Tan, H.; Szulwach, K.; Ma, C.; Jin, P.; Major, F. Structural dynamics control the MicroRNA maturation pathway. Nucleic Acids Res. 2016, 44, 9956–9964.
- Curtin, S. J.; Michno, J. M.; Campbell, B. W.; Gil-Humanes, J.; Mathioni, S. M.; Hammond, R.; Gutierrez-Gonzalez, J. J.; Donohue, R. C.; Kantar, M. B.; Eamens, A. L.; et al. MicroRNA Maturation and MicroRNA Target Gene Expression Regulation Are Severely Disrupted in Soybean dicer-like1 Double Mutants. G3 (Bethesda) 2015, 6, 423–433.
- Komiya, R.; Nonomura, K. Isolation and bioinformatic analyses of small RNAs interacting with germ cell-specific argonaute in rice. Methods Mol. Biol. 2014, 1093, 235–245.
- Till, S.; Lejeune, E.; Thermann, R.; Bortfeld, M.; Hothorn, M.; Enderle, D.; Heinrich, C.; Hentze, M. W.; Ladurner, A. G. A conserved motif in Argonaute-interacting proteins mediates functional interactions through the Argonaute PIWI domain. Nat. Struct. Mol. Biol. 2007, 14, 897–903.
- Bridge, K. S.; Shah, K. M.; Li, Y.; Foxler, D. E.; Wong, S. C. K.; Miller, D. C.; Davidson, K. M.; Foster, J. G.; Rose, R.; Hodgkinson, M. R.; et al. Argonaute Utilization for miRNA Silencing Is Determined by Phosphorylation-Dependent Recruitment of LIM-Domain-Containing Proteins. Cell Rep. 2017, 20, 173–187.
- Leung, A. K.; Sharp, P. A. Quantifying Argonaute proteins in and out of GW/P-bodies: implications in microRNA activities. Adv. Exp. Med. Biol. 2013, 768, 165–182.
- Friedman, R. C.; Farh, K. K.; Burge, C. B.; Bartel, D. P. Most mammalian mRNAs are conserved targets of microRNAs. Genome Res. 2009, 19, 92–105.
- Lim, L. P.; Lau, N. C.; Garrett-Engele, P.; Grimson, A.; Schelter, J. M.; Castle, J.; Bartel, D. P.; Linsley, P. S.; Johnson, J. M. Microarray analysis shows that some microRNAs downregulate large numbers of target mRNAs. Nature 2005, 433, 769–773.
- Aleman, L. M.; Doench, J.; Sharp, P. A. Comparison of siRNA-induced off-target RNA and protein effects. RNA 2007, 13, 385–395.
- Miao, L.; Yao, H.; Li, C.; Pu, M.; Yao, X.; Yang, H.; Qi, X.; Ren, J.; Wang, Y. A dual inhibition: microRNA-552 suppresses both transcription and translation of cytochrome P450 2E1. Biochim. Biophys. Acta 2016, 1859, 650–662.
- Subasic, D.; Brummer, A.; Wu, Y.; Pinto, S. M.; Imig, J.; Keller, M.; Jovanovic, M.; Lightfoot, H. L.; Nasso, S.; Goetze, S.; et al. Cooperative target mRNA destabilization and translation inhibition by miR-58 microRNA family in C. elegans. Genome Res. 2015, 25, 1680–1691.
- Kulkarni, V.; Naqvi, A. R.; Uttamani, J. R.; Nares, S. MiRNA-Target Interaction Reveals Cell-Specific Post-Transcriptional Regulation in Mammalian Cell Lines. Int. J. Mol. Sci. 2016, 17, 72.
- Mahmoodian Sani, M. R.; Hashemzadeh-Chaleshtori, M.; Saidijam, M.; Jami, M. S.; Ghasemi-Dehkordi, P. MicroRNA-183 Family in Inner Ear: Hair Cell Development and Deafness. J. Audiol. Otol. 2016, 20, 131–138.
- Kittelmann, S.; McGregor, A. P. Modulation and Evolution of Animal Development through microRNA Regulation of Gene Expression. Genes 2019, 10, 321.
- Hayashi, T.; Hoffman, M. P. Exosomal microRNA communication between tissues during organogenesis. RNA Biol. 2017, 14, 1683–1689.
- Shrestha, A.; Mukhametshina, R. T.; Taghizadeh, S.; Vasquez-Pacheco, E.; Cabrera-Fuentes, H.; Rizvanov, A.; Mari, B.; Carraro, G.; Bellusci, S. MicroRNA-142 is a multifaceted regulator in organogenesis, homeostasis, and disease. Dev. Dyn. An Off. Publ. Am. Assoc. Anat. 2017, 246, 285–290.
- Sengupta, D.; Govindaraj, V.; Kar, S. Alteration in microRNA-17-92 dynamics accounts for differential nature of cellular proliferation. FEBS Lett. 2018, 592, 446–458.
- Rissland, O. S.; Hong, S. J.; Bartel, D. P. MicroRNA destabilization enables dynamic regulation of the miR-16 family in response to cell-cycle changes. Mol. Cell 2011, 43, 993–1004.
- Ye, H.; Jin, Q.; Wang, X.; Li, Y. MicroRNA-802 Inhibits Cell Proliferation and Induces Apoptosis in Human Laryngeal Cancer by Targeting cAMP-Regulated Phosphoprotein 19. Cancer Manag. Res. 2020, 12, 419–430.
- Wei, Y.; Lu, S.; Hu, Y.; Guo, L.; Wu, X.; Liu, X.; Sun, Y. MicroRNA-135a Regulates VEGFC Expression and Promotes Luteinized Granulosa Cell Apoptosis in Polycystic Ovary Syndrome. Reprod. Sci. 2020, 27, 1436–1442.
- Li, S.; Xie, Y.; Yang, B.; Huang, S.; Zhang, Y.; Jia, Z.; Ding, G.; Zhang, A. MicroRNA-214 targets COX-2 to antagonize indoxyl sulfate (IS)-induced endothelial cell apoptosis. Apoptosis Int. J. Program. Cell Death 2020, 25, 92–104.
- Umansky, S. Aging and aging-associated diseases: a microRNA-based endocrine regulation hypothesis. Aging 2018, 10, 2557–2569.
- Margolis, L. M.; Rivas, D. A. Potential Role of MicroRNA in the Anabolic Capacity of Skeletal Muscle With Aging. Exerc. Sport Sci. Rev. 2018, 46, 86–91.
- McCormick, R.; Goljanek-Whysall, K. MicroRNA Dysregulation in Aging and Pathologies of the Skeletal Muscle. Int. Rev. cell Mol. Biol. 2017, 334, 265–308.
- Matsuyama, H.; Suzuki, H. I. Systems and Synthetic microRNA Biology: From Biogenesis to Disease Pathogenesis. Int. J. Mol. Sci. 2019, 21, 132.
- Aziz, N. B.; Mahmudunnabi, R. G.; Umer, M.; Sharma, S.; Rashid, M. A.; Alhamhoom, Y.; Shim, Y. B.; Salomon, C.; Shiddiky, M. J. A. MicroRNAs in ovarian cancer and recent advances in the development of microRNA-based biosensors. Analyst 2020, 145, 2038–2057.
- Ashrafizadeh, M.; Ahmadi, Z.; Samarghandian, S.; Mohammadinejad, R.; Yaribeygi, H.; Sathyapalan, T.; Sahebkar, A. MicroRNA-mediated regulation of Nrf2 signaling pathway: Implications in disease therapy and protection against oxidative stress. Life Sci. 2020, 244, 117329.
- Cosentino, G.; Plantamura, I.; Cataldo, A.; Iorio, M. V. MicroRNA and Oxidative Stress Interplay in the Context of Breast Cancer Pathogenesis. Int. J. Mol. Sci. 2019, 20, 5143.
- Lin, Y. H., MicroRNA Networks Modulate Oxidative Stress in Cancer. Int. J. Mol. Sci. 2019, 20, 4497.
- Biggar, K. K.; Storey, K. B. Functional impact of microRNA regulation in models of extreme stress adaptation. J. Mol. Cell. Biol. 2018, 10, 93–101.
- Sun, Y.; Hawkins, P. G.; Bi, N.; Dess, R. T.; Tewari, M.; Hearn, J. W. D.; Hayman, J. A.; Kalemkerian, G. P.; Lawrence, T. S.; Ten Haken, R. K.; et al. Serum MicroRNA Signature Predicts Response to High-Dose Radiation Therapy in Locally Advanced Non-Small Cell Lung Cancer. Int. J. Radiat. Oncol. Biol. Phys. 2018, 100, 107–114.
- Halimi, M.; Asghari, S. M.; Sariri, R.; Moslemi, D.; Parsian, H. Cellular Response to Ionizing Radiation: A MicroRNA Story. Int. J. Mol. Cell. Med. 2012, 1, 178–184.
- Kraemer, A.; Anastasov, N.; Angermeier, M.; Winkler, K.; Atkinson, M. J.; Moertl, S. MicroRNA-mediated processes are essential for the cellular radiation response. Radiat. Res. 2011, 176, 575–586.
- Tsao, D.; Kalogerinis, P.; Tabrizi, I.; Dingfelder, M.; Stewart, R. D.; Georgakilas, A. G. Induction and processing of oxidative clustered DNA lesions in Fe-56-Ion-irradiated human monocytes. Radiat. Res. 2007, 168, 87–97.
- Zhang, X. R.; Ye, C. Y.; Sun, F.; Wei, W. J.; Hu, B. R.; Wang, J. F. Both Complexity and Location of DNA Damage Contribute to Cellular Senescence Induced by Ionizing Radiation. PloS ONE 2016, 11, e0155725.
- Lorat, Y.; Brunner, C. U.; Schanz, S.; Jakob, B.; Taucher-Scholz, G.; Rube, C. E. Nanoscale analysis of clustered DNA damage after high-LET irradiation by quantitative electron microscopy—The heavy burden to repair. DNA Repair 2015, 28, 93–106.
- Nelson, G. A. Space Radiation and Human Exposures, A Primer. Radiat. Res. 2016, 185, 349–58.
- Takahashi, A.; Ikeda, H.; Yoshida, Y. Role of High-Linear Energy Transfer Radiobiology in Space Radiation Exposure Risks. Int. J. Part. Ther. 2018, 5, 151–159.
- Norbury, J. W.; Slaba, T. C.; Aghara, S.; Badavi, F. F.; Blattnig, S. R.; Clowdsley, M. S.; Heilbronn, L. H.; Lee, K.; Maung, K. M.; Mertens, C. J.; et al. Advances in space radiation physics and transport at NASA. Life Sci. Space Res. 2019, 22, 98–124.
- Inozemtsev, K. O.; Kushin, V. V.; Stradi, A.; Ambrozova, I.; Kodaira, S.; Szabo, J.; Tolochek, R. V.; Shurshakov, V. A. Measurement of Different Components of Secondary Radiation Onboard International Space Station by Means of Passive Detectors. Radiat. Prot. Dosim. 2018, 181, 412–417.
- Cucinotta, F. A.; Schimmerling, W.; Wilson, J. W.; Peterson, L. E.; Badhwar, G. D.; Saganti, P. B.; Dicello, J. F. Space radiation cancer risks and uncertainties for Mars missions. Radiat. Res. 2001, 156, 682–688.
- Hassler, D. M.; Zeitlin, C.; Wimmer-Schweingruber, R. F.; Ehresmann, B.; Rafkin, S.; Eigenbrode, J. L.; Brinza, D. E.; Weigle, G.; Bottcher, S.; Bohm, E.; et al. Mars' Surface Radiation Environment Measured with the Mars Science Laboratory's Curiosity Rover. Science 2014, 343, 1244797.
- Hellweg, C. E.; Baumstark-Khan, C. Getting ready for the manned mission to Mars: the astronauts' risk from space radiation. Die Nat. 2007, 94, 517–526.
- McNulty, P. J.; Pease, V. P.; Bond, V. P. Role of Cerenkov radiation in the eye-flashes observed by Apollo astronauts. Life Sci. Space Res. 1976, 14, 205–217.
- McNulty, P. J.; Pease, V. P.; Bond, V. P. Muon-induced visual sensations. J. Opt. Soc. Am. 1976, 66, 49–55.
- McNulty, P. J.; Pease, V. P.; Bond, V. P. Visual sensations induced by Cerenkov radiation. Science 1975, 189, 453–454.
- Tobias, S.; Abramson, T. Interaction among anxiety, stress, response mode, and familiarity of subject matter on achievement from programmed instruction. J. Educ. Psychol. 1971, 62, 357–364.
- McNulty, P. J.; Pease, V. P.; Bond, V. P. Visual phenomena induced by relativistic carbon ions with and without Cerenkov radiation. Science 1978, 201, 341–343.
- Budinger, T. F.; Lyman, J. T.; Tobias, C. A. Visual perception of accelerated nitrogen nuclei interacting with the human retina. Nature 1972, 239, 209–211.
- Fremlin, J. H. Causality and tachyons. Nature 1970, 226, 194–195.
- Budinger, T. F.; Bichsel, H.; Tobias, C. A. Visual phenomena noted by human subjects on exposure to neutrons of energies less than 25 million electron volts. Science 1971, 172, 868–870.
- Charman, W. N.; Dennis, J. A.; Fazio, G. G.; Jelley, J. V. Visual sensations produced by single fast particles. Nature 1971, 230, 522–524.
- Chancellor, J. C.; Scott, G. B.; Sutton, J. P. Space Radiation: The Number One Risk to Astronaut Health beyond Low Earth Orbit. Life (Basel) 2014, 4, 491–510.
- Khan, S. Y.; Tariq, M. A.; Perrott, J. P.; Brumbaugh, C. D.; Kim, H. J.; Shabbir, M. I.; Ramesh, G. T.; Pourmand, N. Distinctive microRNA expression signatures in proton-irradiated mice. Mol. Cell. Biochem. 2013, 382, 225–235.
- Templin, T.; Young, E. F.; Smilenov, L. B. Proton radiation-induced miRNA signatures in mouse blood: characterization and comparison with 56Fe-ion and gamma radiation. Int. J. Radiat. Biol. 2012, 88, 531–539.
- Girardi, C.; De Pitta, C.; Casara, S.; Sales, G.; Lanfranchi, G.; Celotti, L.; Mognato, M. Analysis of miRNA and mRNA expression profiles highlights alterations in ionizing radiation response of human lymphocytes under modeled microgravity. PLoS ONE 2012, 7, e31293.
- Fu, H.; Su, F.; Zhu, J.; Zheng, X.; Ge, C. Effect of simulated microgravity and ionizing radiation on expression profiles of miRNA, lncRNA, and mRNA in human lymphoblastoid cells. Life Sci. Space Res. 2020, 24, 1–8.
- Gao, Y.; Xu, D.; Zhao, L.; Zhang, M.; Sun, Y. Q. Effects of microgravity on DNA damage response in Caenorhabditis elegans during Shenzhou-8 spaceflight. Int. J. Radiat. Biol. 2015, 91, 531–539.
- Xu, D.; Gao, Y.; Huang, L.; Sun, Y. Changes in miRNA expression profile of space-flown Caenorhabditis elegans during Shenzhou-8 mission. Life Sci. Space Res. 2014, 1, 44–52.
- Kovalchuk, O.; Zemp, F. J.; Filkowski, J. N.; Altamirano, A. M.; Dickey, J. S.; Jenkins-Baker, G.; Marino, S. A.; Brenner, D. J.; Bonner, W. M.; Sedelnikova, O. A. microRNAome changes in bystander three-dimensional human tissue models suggest priming of apoptotic pathways. Carcinogenesis 2010, 31, 1882–1888.
- Chauhan, V.; Howland, M.; Wilkins, R. Effects of alpha-Particle Radiation on MicroRNA Responses in Human Cell-Lines. Open Biochem. J. 2012, 6, 16–22.
- Nie, J. H.; Chen, Z. H.; Shao, C. L.; Pei, W. W.; Zhang, J.; Zhang, S. Y.; Jiao, Y.; Tong, J. Analysis of the miRNA-mRNA networks in malignant transformation BEAS-2B cells induced by alpha-particles. J. Toxicol. Environ. Health Part A 2016, 79, 427–435.
- Templin, T.; Amundson, S. A.; Brenner, D. J.; Smilenov, L. B. Whole mouse blood microRNA as biomarkers for exposure to gamma-rays and (56)Fe ion. Int. J. Radiat. Biol. 2011, 87, 653–662.
- He, Y.; Zhang, Y.; Li, H.; Zhang, H.; Li, Z.; Xiao, L.; Hu, J.; Ma, Y.; Zhang, Q.; Zhao, X. Comparative Profiling of MicroRNAs Reveals the Underlying Toxicological Mechanism in Mice Testis Following Carbon Ion Radiation. Dose-Response A Publ. Int. Hormesis Soc. 2018, 16, 1559325818778633.
- Wei, W.; He, J.; Wang, J.; Ding, N.; Wang, B.; Lin, S.; Zhang, X.; Hua, J.; Li, H.; Hu, B. Serum microRNAs as Early Indicators for Estimation of Exposure Degree in Response to Ionizing Irradiation. Radiat. Res. 2017, 188, 342–354.
- Wei, W. J.; Wang, J. F.; He, J. P.; Xie, X. D. Serum microRNA as noninvasive indicator for space radiation. Acta Astronaut. 2018, 152, 101–104.
- Gao, Y.; Li, S.; Xu, D.; Wang, J.; Sun, Y. Changes in apoptotic microRNA and mRNA expression profiling in Caenorhabditis elegans during the Shenzhou-8 mission. J. Radiat. Res. 2015, 56, 872–882.
- Zhu, Y.; Yu, X. Y.; Fu, H. J.; Wang, H. Y.; Wang, P.; Zheng, X. F.; Wang, Y. MicroRNA-21 is involved in ionizing radiation-promoted liver carcinogenesis. Int. J. Clin. Exp. Med. 2010, 3, 211–222.
- Shi, Y.; Zhang, X. M.; Tang, X. B.; Wang, P.; Wang, H. C.; Wang, Y. MiR-21 is Continually Elevated Long-Term in the Brain after Exposure to Ionizing Radiation. Radiat. Res. 2012, 177, 124–128.
- Zhang, X. M.; Ng, W. L.; Wang, P.; Tian, L. L.; Werner, E.; Wang, H. C.; Doetsch, P.; Wang, Y. MicroRNA-21 Modulates the Levels of Reactive Oxygen Species by Targeting SOD3 and TNF alpha. Cancer Res. 2012, 72, 4707–4713.
- Wang, J.; Zhang, X.; Wang, P.; Wang, X.; Farris, A. B., 3rd; Wang, Y. Lessons learned using different mouse models during space radiation-induced lung tumorigenesis experiments. Life Sci. Space Res. 2016, 9, 48–55.
- Kim, S. B.; Zhang, L.; Barron, S.; Shay, J. W. Inhibition of microRNA-31-5p protects human colonic epithelial cells against ionizing radiation. Life Sci. Space Res. 2014, 1, 67–73.