Adrenomedullin (AM) is a bioactive peptide with various physiological functions, including vasodilation, angiogenesis, anti-inflammation, organ protection, and tissue repair.
1. Introduction
Ulcerative colitis (UC) and Crohn’s disease (CD) are the most common types of chronic inflammatory bowel disease (IBD). The number of people with IBD worldwide reached over 6.8 million in 2017
[1]. The cause of IBD is unknown but is thought to be caused by abnormalities in intestinal immunity, a genetic predisposition, and environmental factors, such as diet and intestinal microflora.
Although there is no cure for IBD, there have been remarkable therapeutic developments that contribute to the induction and maintenance of remission over the past 20 years, including anti-TNF-alpha antibodies, tacrolimus, anti-IL-12/23p40 antibodies, Janus kinase (JAK) inhibitors, and integrin inhibitors. While these drugs have shown excellent efficacy, they also negatively affect patients’ immune systems, leading some patients to develop infections and malignant lymphomas.
Severe infections such as tuberculosis and Pneumocystis pneumonia require serious attention, particularly in elderly patients and patients with underlying diseases, such as diabetes mellitus. Further, the above treatments demonstrate a diminishing therapeutic response, as they are immunogenic and targets of anti-drug antibodies. Therefore, the development of new therapeutic agents for IBD requires efficacy, safety, and immunogenicity.
Adrenomedullin (AM), an endogenous vasodilatory peptide, was isolated from human pheochromocytoma by Kitamura et al. in 1993
[2]. AM was initially studied as a circulatory agonist but was later found to promote angiogenesis, organ protection, and anti-inflammatory immune activity. AM is also widely expressed in the gastrointestinal tract epithelia and effectively treats gastric ulcers and enteritis in animal models. Therefore, we began translational research on the clinical application of AM in IBD
[3].
2. Structure and Biosynthesis of Adrenomedullin
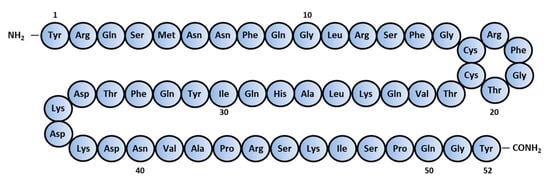
AM is composed of 52 amino acids
[2] and has a ring structure consisting of six amino acids and a C-terminal amide structure (
Figure 1). These two structural features are essential for their biological activity. It also shares homology with calcitonin gene-related peptide (CGRP), amylin, and adrenomedullin 2/intermedin (AM2/IMD) to form the calcitonin peptide superfamily.
Figure 1. Amino-acid sequence of human Adrenomedullin.
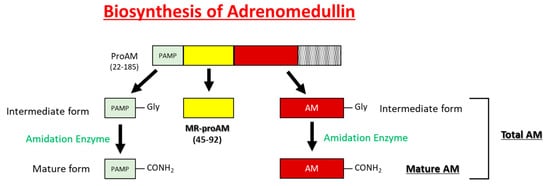
AM is widely expressed throughout the blood vessels, heart, lungs, kidneys, and gastrointestinal tract and is highly concentrated in the adrenal medulla. The pro-adrenomedullin N-terminal 20 peptide (PAMP) has a shorter duration of antihypertensive activity than AM and cooperatively regulates blood circulation with AM (
Figure 2)
[4]. Although mid regional pro-adrenomedullin (MR-pro ADM) has no biological activity, it has been attracting attention as a biomarker for the prognosis of heart failure
[5], myocardial infarction
[5], community-acquired pneumonia
[6], septic shock
[7], and COVID-19
[8][9].
Figure 2. Schematic representations of the processing of AM, MR-proAM, and PAMP from proAM.
3. Adrenomedullin Receptors
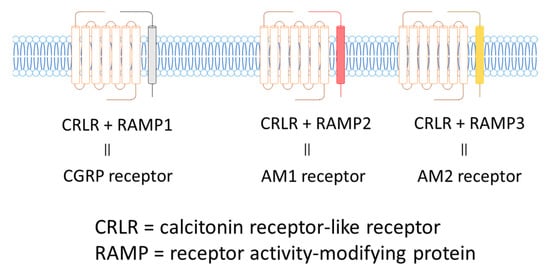
The AM receptor consists of a complex of calcitonin receptor-like receptors (CRLR; a seven-transmembrane G protein receptor) and a receptor activity-modifying protein (RAMP; a single transmembrane receptor). There are three subtypes of RAMPs, RAMP1, RAMP2, and RAMP3, and the CRLR/RAMP1 complex has a high affinity for CGRP, while the CRLR/RAMP2 and CRLR/RAMP3 complexes have high affinity for AM. The CRLR/RAMP2 complex comprises the AM1 receptor, and the CRLR/RAMP3 complex constitutes the AM2 receptor (Figure 3).
Figure 3. CGRP and AM Receptors.
AM contributes to the development and homeostasis of blood vessels and lymphatic vessels; AM knockout mice develop defective embryos due to defective blood and lymphatic vessel formation. Furthermore, studies using RAMP2 and RAMP3 gene knockout mice report that the AM1 receptor is involved in angiogenesis and vascular homeostasis, while the AM2 receptor regulates lymph vessel function
[10]. Thus, we speculate that AM influences vascular and lymphatic system ecological functions through two receptors.
4. General Physiological Effects of Adrenomedullin
AM circulates the blood constitutively and is detected in healthy individuals; its expression is enhanced by mechanical stimuli, such as:
-
Myocardial and vascular wall stretching
-
Organ ischemia and hypoxia
-
Inflammatory factors, such as inflammatory cytokines, angiotensin II, oxidative stress, and various tissue stress.
AM promotes various pathophysiological effects, such as (
Table 1)
[11]:
Table 1. Diverse physiological effects of adrenomedullin.
| Vasodilation |
| Angiogenesis |
| Cardioprotection |
| Nephroprotection |
| Anti-oxidation |
| Anti-apoptosis |
| Tissue repair and regeneration |
In a study of blood levels in patients with diseases, increases in blood (AM) were observed in cardiovascular diseases such as essential hypertension, heart failure, and renal failure
[12]. Elevated blood (AM) is associated with the development of inflammatory diseases such as pancreatitis and sepsis
[13] in patients with IBD
[14], the severity of which increases in a concentration-dependent manner.
5. Anti-Inflammatory Effects of Adrenomedullin
Blood (AM) is markedly elevated during severe inflammation, such as burns, pancreatitis, and systemic inflammatory response syndrome (SIRS) associated with sepsis
[12].
In vitro studies using cultured vascular smooth muscle cells (VSMCs) and endothelial cells have shown that pro-inflammatory cytokines such as IL-1, tumor necrosis factor (TNF)-α, and lipopolysaccharide (LPS) stimulate AM expression in smooth muscle and endothelial cells
[15]. In addition, the monocyte/macrophage cell line (RAW 264.7), murine celiac macrophages, and peripheral blood-derived monocytes have shown that monocyte-macrophage differentiation enhances AM production. Further, AM inhibits monocyte and macrophage TNF-α and IL-6 secretion following LPS-stimulation
[16]. In an in vivo mouse study of sepsis treated with LPS and D-galactosamine, AM-overexpressing mice had demonstrated milder liver failure than wild-type mice, demonstrating that endogenous AM protects against SIRS
[17]. Further, septic mice models treated with AM demonstrated improved hemodynamics and decreased pro-inflammatory cytokines such as TNF-α, IL-1β, and IL-6
[18] than control mice. Thus, AM ameliorates inflammatory conditions and is expected to be applied clinically to treat various inflammatory diseases.
6. Physiological Functions of Adrenomedullin in the Gastrointestinal Tract
AM is widely expressed throughout the mucosal epithelium, glandular duct cells, neuroendocrine cells, and smooth muscle cells of the gastrointestinal tract, between the oral cavity and the large intestine
[19]. The physiological effects of AM include suppressed gastric acid secretion via somatostatin in the stomach, enhanced electrolyte secretion in the colon, suppressed gastrointestinal motility, and changes to microcirculation flux. Additionally, AM has similar physiological and antibacterial effects as defensins and may contribute to the mucosal defense system by regulating the oral and intestinal microbiome
[20].