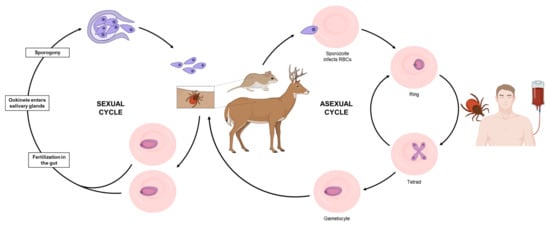
2. Current Treatments against Human Babesiosis
The current arsenal for the treatment of human babesiosis relies principally on four drugs: atovaquone, azithromycin, clindamycin, and quinine. Atovaquone is used to treat several human diseases, including
Pneumocystis jirovecii pneumonia
[7], toxoplasmosis
[8], and malaria (in combination with proguanil (Malarone)
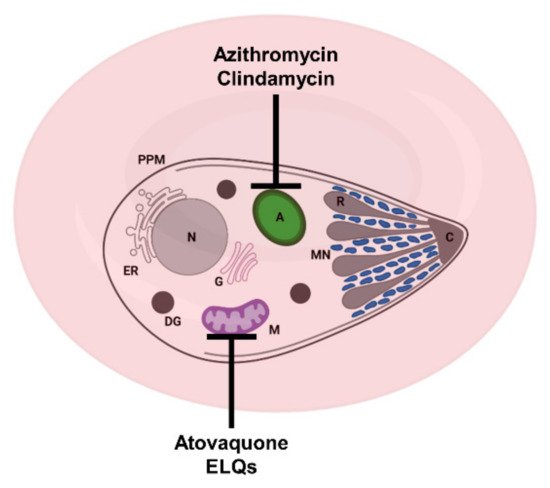
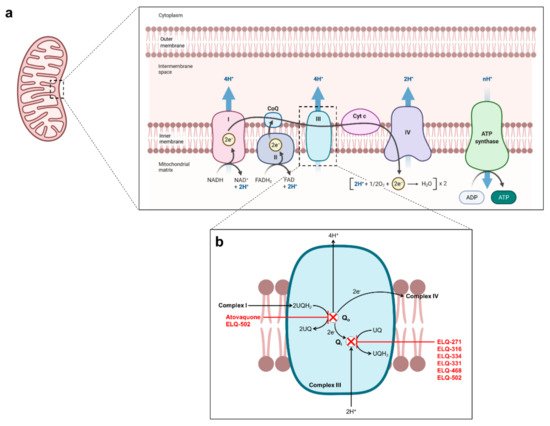
[9]. In apicomplexan parasites, atovaquone targets the cytochrome
bc1 complex of the mitochondrial electron transport chain (
Figure 2 and
Figure 3)
[10][11][12][13]. Azithromycin is a relatively broad-spectrum antibiotic indicated for the treatment of numerous bacterial infections, such as those caused by
Staphylococcus spp.
[14][15][16] and
Legionella spp.
[17]. The antibiotic is also used for the treatment of toxoplasmosis
[12] and, in combination with other drugs, for the treatment of malaria
[18]. Azithromycin is a well-characterized protein synthesis inhibitor, which in apicomplexan parasites targets the translation machinery in the apicoplast (
Figure 2)
[19][20][21]. It is worth noting that azithromycin was found to have a “delayed death” effect, in which parasite division produces viable daughter cells that are subsequently unable to divide in the following cycle
[19][21][22]. Clindamycin is another antibiotic commonly used for the treatment of various bacterial infections
[23] and repurposed for the treatment of parasitic infections. In combination with quinine, clindamycin is used for the treatment of both malaria and babesiosis
[24][25][26]. Several reports have suggested that clindamycin acts in a similar way as azithromycin and targets protein synthesis in the apicoplast (
Figure 2)
[19][21][22]. Furthermore, selection of clindamycin-resistant
T. gondii parasites showed cross-resistance to azithromycin, further suggesting a common target
[27]. Quinine is a widely used antimalarial agent, typically administered in combination with an antibiotic such as clindamycin or doxycycline
[28]. However, the drug is poorly tolerated and, as such, tends to be replaced by alternative drugs with fewer side-effects
[28][29]. In malaria parasites, several modes of action for quinine have been proposed. The most commonly reported mechanism of action involves the disruption of hemozoin formation, resulting in accumulation of free ferriprotoporphyrin IX, a by-product of hemoglobin degradation, which is deleterious to parasite growth
[30][31][32]. Confocal imaging using fluorescent derivatives of quinine and its structural analogues, quinidine and chloroquine, have shown accumulation of the probes in the digestive vacuole, consistent with the activity of this compound in this organelle
[32][33]. Unlike
Plasmodium parasites,
Babesia species lack a digestive vacuole, do not degrade hemoglobin, and do not produce hemozoin. Therefore, the mode of action of quinine against
Babesia parasites is likely to be different from that in
Plasmodium. Interestingly, fluorescent probes were found to bind to phospholipids and to accumulate in membranous structures, including the parasite plasma membrane, the endoplasmic reticulum, and the mitochondrion, suggesting that quinine may inactivate specific biological functions in these organelles
[32][33]. Another proposed hypothesis is that quinine acts as a DNA intercalator
[34][35][36]. However, the lack of fluorescence in the nucleus reported by Woodland et al. seem to refute interactions with DNA as a potential mode of action
[32][33]. More recently, a study in
P. falciparum using thermal shift assays suggested that the purine nucleoside phosphorylase (PfPNP) might also be a target of quinine
[37].
Figure 2. Schematic representation of a Babesia-infected red blood cell and sites of action of some approved and experimental drugs. Azithromycin and clindamycin target the apicoplast; atovaquone and ELQs target the mitochondrion. A: apicoplast, C: conoid + polar rings, DG: dense granule, ER: endoplasmic reticulum, G: Golgi apparatus, M: mitochondrion, MN: microneme, PPM: parasite plasma membrane and R: rhoptry.
Figure 3. Proposed mechanism of action of atovaquone and endochin-like quinolones in Babesia mitochondrion. (a) Schematic representation the mitochondrial electron transfer chain. (b) Schematic representation of the parasite bc1 complex with proposed mode of action of atovaquone and ELQs.
The severity of babesiosis depends mainly on the host’s immune status, the presence of risk factors and the
Babesia species responsible for the infection. In symptomatic patients, babesiosis usually manifests with flu-like symptoms such as fever, fatigue, chills, sweats, and headache
[38]. For this moderate form of the disease, typically associated with a low parasitemia level (<4%)
[26], no hospital admission is required and a 7–10-day treatment course of oral atovaquone + azithromycin (500 mg azithromycin on day 1, followed by 250 mg on subsequent days + 750 mg b.i.d. atovaquone) is recommended
[26][38]. Babesiosis typically resolves within seven days from the start of the treatment, but asymptomatic, low level parasitemia may persist for up to one year
[26]. Monitoring of persistent parasitemia in immunocompetent individuals following treatment is usually not necessary. However, given the risk of transmission of
Babesia parasites through blood transfusion, these patients are excluded as blood donors
[3]. Immunocompromised individuals are more at risk of developing a severe form of babesiosis, resulting in complications such as acute respiratory distress syndrome, disseminated intravascular coagulation, severe hemolytic anemia, organ failure, splenic rupture, relapse, and death
[2][26]. A combination of oral clindamycin + quinine (600 mg + 650 mg, every 8 h) is the standard of care for the treatment of severe babesiosis
[26][38]. However, this treatment regimen is frequently associated with serious side effects, such as hearing loss, vertigo, and tinnitus. In some cases, these side effects can be so severe that dose reduction or discontinuation of treatment is required
[38]. Recently, it has been demonstrated that a combination of atovaquone + azithromycin is also suitable for the treatment of severe babesiosis, displaying comparable efficacy to clindamycin + quinine with fewer side effects
[39]. Although atovaquone + azithromycin is now the preferred course of treatment for severe babesiosis, the standard 7–10-day treatment regimen of oral atovaquone + azithromycin is usually not enough to eliminate
Babesia infection. Higher doses, longer treatment duration, and in some cases intravenous administration is required to clear the infection
[26]. It is also worth noting that the use of immunosuppressive agents such as Rituximab to treat prior illnesses (B cell lymphoid malignancies, rheumatoid arthritis, etc.) may lead to babesiosis relapse and extended persistence of
Babesia parasites
[40][41][42].
One downside of a prolonged treatment regimen and dose escalation is the risk of developing drug resistance. Previous reports have established the emergence of mutations in the cytochrome b (Cytb) of
Babesia parasites in humans and animal models following treatment with atovaquone
[11][42][43]. In 2016, Lemieux et al. examined clinical isolates of relapsing babesiosis and identified a methionine to isoleucine mutation (M134I) in the Q
o site (atovaquone-binding site) of the BmCytb
[43]. This same mutation was observed in a murine model of
B. microti infection
[11], as well as in other apicomplexan parasites, such as
P. falciparum and
T. gondii [43]. Later, Simon et al. reported a Y272C mutation in the BmCytb Q
o site in a patient presenting with relapsed
B. microti infection following an atovaquone + azithromycin treatment course
[42]. In both cases, these mutations have been shown to impact the atovaquone-binding domain
[44] and appear to be associated with decreased sensitivity to the drug
[42][43]. With regard to azithromycin resistance, sequencing of clinical isolates obtained from patients with relapsing babesiosis identified mutations in the ribosomal protein subunit L4 (RPL4) encoded by the apicoplast genome
[42][43]. Lemieux et al. identified three substitutions in the RPL4: R86H, R86C and S73L
[43]. Simon et al. observed the same R86C mutation in a patient presenting with relapsing babesiosis following atovaquone + azithromycin treatment
[42]. Similar mutations associated with azithromycin resistance have been reported in
P. falciparum [20] and
S. pneumoniae [45] RPL4. Alternative management strategies for human babesiosis in the case of persistent relapse include the use of different drug combinations such as atovaquone + azithromycin + clindamycin, atovaquone + clindamycin, atovaquone + proguanil, or atovaquone + azithromycin + clindamycin + quinine
[26][41][46][47]. The introduction of other drugs such as doxycycline, moxifloxacin, pentamidine, trimethoprim-sulfamethoxazole or artemisinin to treatment regimens with the standard therapies was also reported
[40][48]. A recent study in a small cohort of patients suffering from Lyme disease and babesiosis co-infection suggested improvement, and in some cases remission, following one course of disulfiram monotherapy
[49]. In patients with high parasitemia (>10%), exchange transfusion is recommended and often results in a rapid reduction of the parasite load
[26][50].
Despite clinical evidence that atovaquone, azithromycin, clindamycin and quinine can be used to manage human babesiosis, preclinical evaluation of these drugs in different models of
Babesia infection has not demonstrated unanimous results with regards to their efficacy. Clindamycin showed only limited activity at a dose of 300 mg/kg (p.o.) in
B. microti-infected Mongolian jirds
[51]. When evaluated in
B. microti-infected hamster, a course of 150 mg/kg (i.m. or p.o.) of clindamycin resulted in a two-fold decrease in peak parasitemia. Similar results were obtained when clindamycin was administered in combination with quinine
[52]. AbouLaila et al. reported a ~three-fold decrease in peak parasitemia following i.p. injection of 500 mg/kg of clindamycin in
B. microti-infected Balb/c mice
[53]. Another study using the same Balb/c model of
B. microti infection showed that oral administration of clindamycin at 25, 50, and 100 mg/kg did not lead to reduction of parasite burden
[54]. Similar results were obtained by Lawres et al. following oral administration of 10 or 50 mg/kg of clindamycin to immunocomprimized mice infected with
B. microti [11]. The consensus seems to be more apparent in the case of quinine, where most studies report no effect on parasitemia following administration of quinine as a single drug
[11][52][54]. Interestingly, a combination of clindamycin + quinine was reported to achieve up to 70% suppression of parasitemia
[55] and result in a faster resolution of parasitemia compared to clindamycin alone
[52], suggesting a potential synergy between the two drugs. Preclinical investigation of azithromycin efficiency against
Babesia parasites also yielded inconsistent results. In
B. microti-infected Balb/c mice, a four-day treatment course with azithromycin at 25, 50, and 100 mg/kg was found to be potent, resulting in 75–96% suppression of parasitemia
[54]. In contrast, the evaluation of azithromycin in
B. microti-infected SCID mice showed no effect on parasitemia at 10 and 50 mg/kg after a seven-day treatment course
[11]. Similar results were obtained in
B. microti-infected hamsters, where 150 mg/kg azithromycin treatment regimen, administered daily for almost two weeks, showed no apparent effect on parasitemia
[56]. Out of the four clinically used drugs in the treatment of babesiosis, only atovaquone seems to consistently show high potency against
Babesia parasites
[11][56][57][58][59]. Studies carried out in
B. microti-infected hamsters and SCID mice reported fast clearance of parasitemia following treatment with atovaquone
[11][56]. However, recrudescence due to atovaquone-resistant parasites was observed
[11][56]. In
B. microti-infected hamsters, a combination therapy of atovaquone + azithromycin resulted in rapid clearance of parasitemia without recrudescence
[56]. In a lethal model of
B. microti infection in hamsters, atovaquone monotherapy was found to be superior to a combination of clindamycin + quinine, resulting in low to undetectable parasitemia and extended survival
[58]. Potency of atovaquone was also demonstrated in
B. divergens [59] and
B. duncani [57] models, with IC
50 values in the low nanomolar range. In gerbils, although prophylaxis experiments were not successful, a dose of atovaquone as low as 0.5 mg/kg was found to efficiently prevent
B. divergens infection, so long as daily treatment was maintained several days post-infection
[59]. In the case of
B. duncani, a treatment course of 10 mg/kg atovaquone resulted in a clear reduction of parasitemia and 80% survival using a mouse model of lethal infection
[57]. The results derived from the evaluation of atovaquone, azithromycin, clindamycin, and quinine in preclinical models of babesiosis are summarized in
Table 1.
While combinations of atovaquone + azithromycin and clindamycin + quinine have been used for more than 20 years for the treatment of human babesiosis
[60], the efficacy of these drugs and their primary modes of action in
Babesia parasites have only recently started to be elucidated.
Table 1. Reported efficacy of atovaquone, azithromycin, clindamycin and quinine in animal models of babesiosis.
| Drug |
Treatment Regimen |
Model |
Effect |
Ref. |
| Atovaquone |
20 mg/kg (p.o.), 5 d |
B. microti
Balb/c mice |
~5.7 × reduction in peak parasitemia. |
[61] |
| 25 mg/kg (p.o.), 4 d |
B. microti
Balb/c mice |
77% suppression of parasitemia at DPI 9. |
[54] |
| 50 mg/kg (p.o.), 4 d |
B. microti
Balb/c mice |
87% suppression of parasitemia at DPI 9. |
[54] |
| 100 mg/kg (p.o.), 4 d |
B. microti
Balb/c mice |
93% suppression of parasitemia at DPI 9. |
[54] |
| 10 mg/kg (p.o.), 7 d |
B. microti
SCID mice |
Parasitemia clearance followed by recrudescence by D5-9 post-treatment. |
[11] |
| 10 mg/kg (p.o.), 10 d |
B. microti
SCID mice |
Parasitemia clearance followed by recrudescence by D14 post-treatment. |
[57] |
| 10 mg/kg (p.o.), 10 d |
B. duncani
C3H/HeJ mice |
Parasitemia clearance followed by recrudescence by D10 post-treatment. 80% survival. |
[57] |
| Azithromycin |
25 mg/kg (p.o.), 4 d |
B. microti
Balb/c mice |
75% suppression of parasitemia at DPI 9. |
[54] |
| 50 mg/kg (p.o.), 4 d |
B. microti
Balb/c mice |
96% suppression of parasitemia at DPI 9. |
[54] |
| 100 mg/kg (p.o.), 4 d |
B. microti
Balb/c mice |
95% suppression of parasitemia at DPI 9. |
[54] |
| 10 mg/kg (p.o.), 7 d |
B. microti
SCID mice |
No effect. |
[11] |
| 50 mg/kg (p.o.), 7 d |
B. microti
SCID mice |
No effect. |
[11] |
| Clindamycin |
300 mg/kg (p.o.), 5d |
B. microti
Mongolian jirds |
9.4% suppression of parasitemia at DPI 9. |
[51] |
| 150 mg/kg (i.m.), 8d |
B. microti
Golden hamsters |
~2× reduction in peak parasitemia. |
[52] |
| 150 mg/kg (p.o.), 8d |
B. microti
Golden hamsters |
~2× reduction in peak parasitemia. |
[52] |
| 500 mg/kg (i.p.), 5d |
B. microti
Balb/c mice |
~3.2× reduction in peak parasitemia. |
[53] |
| 25 mg/kg (p.o.), 4 d |
B. microti
Balb/c mice |
No effect. |
[54] |
| 50 mg/kg (p.o.), 4 d |
B. microti
Balb/c mice |
No effect. |
[54] |
| 100 mg/kg (p.o.), 4 d |
B. microti Balb/c mice |
No effect. |
[54] |
| 10 mg/kg (p.o.), 7 d |
B. microti
SCID mice |
No effect. |
[11] |
| 50 mg/kg (p.o.), 7 d |
B. microti
SCID mice |
No effect. |
[11] |
| Quinine |
125 mg/kg (s.c.), 8d |
B. microti
Golden hamsters |
No effect. |
[52] |
| 250 mg/kg (p.o.), 8d |
B. microti
Golden hamsters |
No effect. |
[52] |
| 25 mg/kg (p.o.), 4 d |
B. microti
Balb/c mice |
No effect. |
[54] |
| 50 mg/kg (p.o.), 4 d |
B. microti
Balb/c mice |
No effect. |
[54] |
| 100 mg/kg (p.o.), 4 d |
B. microti
Balb/c mice |
No effect. |
[54] |
| 10 mg/kg (p.o.), 7 d |
B. microti
SCID mice |
No effect. |
[11] |
| 50 mg/kg (p.o.), 7 d |
B. microti
SCID mice |
No effect. |
[11] |
| 100 mg/kg (p.o.), 7 d |
B. microti
SCID mice |
No effect. |
[11] |