
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Venu Babu Borugadda | + 4330 word(s) | 4330 | 2021-08-09 08:38:01 | | | |
| 2 | Vivi Li | Meta information modification | 4330 | 2021-08-23 05:51:10 | | |
Video Upload Options
Recently, due to the escalating usage of non-renewable fossil fuels such as coal, natural gas and petroleum coke in electricity and power generation, and associated issues with pollution and global warming, more attention is being paid to finding alternative renewable fuel sources. Thermochemical and hydrothermal conversion processes have been used to produce biochar and hydrochar, respectively, from waste renewable biomass. Char produced from the thermochemical and hydrothermal decomposition of biomass is considered an environmentally friendly replacement for solid hydrocarbon materials such as coal and petroleum coke. Unlike thermochemically derived biochar, hydrochar has received little attention due to the lack of literature on its production technologies, physicochemical characterization, and applications.
1. Introduction
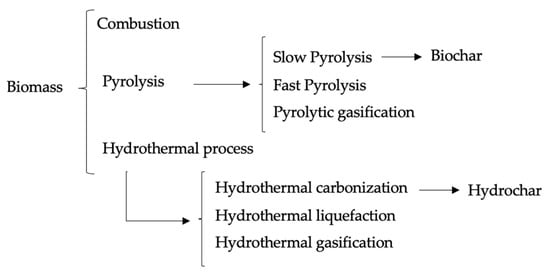
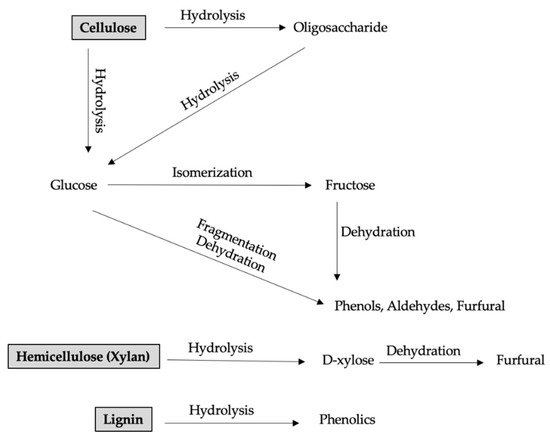
2. Hydrochar Production Technologies
| Feedstock | Temperature (℃) | Time (h) | Hydrochar Yield (wt.%) | Carbon Content (wt.%) | Atomic H/C Ratio | Higher Heating Value (MJ/kg) |
References |
|---|---|---|---|---|---|---|---|
| Chinese fan palm | 180 210 240 |
1 1 1 |
61 57 50 |
55.9 58.1 64.1 |
1.64 1.6 1.5 |
24.2 25.3 28.1 |
[35] |
| Chinese fan palm | 210 210 210 |
0.5 1 1.6 |
60 57 57 |
57.3 58.1 59.6 |
1.62 1.60 1.62 |
24.9 25.3 26.4 |
[35] |
| Apple chip pomace Grape pomace Rotten apple Apple juice pomace |
190 190 190 190 |
- | - | 55.9 55.7 62.5 53.9 |
0.13 0.1 0.09 0.11 |
- | [36] |
| Bamboo shoot shell | 210 270 |
0.5 0.5 |
56.4 31.9 |
51.3 >52 |
- | - | [37] |
| Corn cob residue | 250 250 |
0.55 | 46.6 45.7 |
61.7 63.6 |
0.08 0.07 |
24.3 24.9 |
[38] |
| Cotton stalk | 180 200 220 240 260 |
4 | 60 55 40 35 32 |
51.2 53.2 59 69 70.4 |
1.2 1.3 1.1 0.91 0.94 |
- | [39] |
| Cotton stalk | 240 | 1 2 4 6 8 |
240 | 61.9 64.8 69 69.6 70.6 |
0.92 1.03 0.91 0.94 0.92 |
- | [39] |
| Green waste | 190 | 1 | 80 | 48.8 | 1.2 | 19.2 | [40] |
| Moso bamboo | 220 260 300 |
1 | 51.8 40.5 35.6 |
- | - | 19.8 28.3 29.3 |
[41] |
| Rice husk | 200 300 |
6 6 |
66 43 |
40.8 45.6 |
1.27 0.84 |
15.7 17.8 |
[42] |
| Feedstock | Hydrochar Yield | Temperature (℃) |
Time (h) |
Carbon (wt.%) |
Atomic H/C Ratio | Higher Heating Value (MJ/kg) |
References |
|---|---|---|---|---|---|---|---|
| Apple pomace | 17 16 |
250 | 0.08 60 |
63.2 69.8 |
0.1 0.02 |
26 28 |
[43] |
| Apple pomace | 17 12 |
250 300 |
0.08 | 63.2 73 |
0.1 0.06 |
26 32 |
[43] |
| Microalgae | 19.5 12.1 |
222 272 |
0.16 | 52.1 54.3 |
0.14 0.12 |
- | [28] |
| Microalgae | 19.5 15.4 |
222 | 0.16 0.58 |
52.1 55.6 |
0.14 0.12 |
- | [44] |
| Microalgae | 28.6 22.7 |
210 250 |
1 | 54.89 27.33 |
0.11 0.12 |
- | [45] |
| Sewage sludge | 56 | 220 | 0.5 | 7.9 | 0.16 | 3.6 | [29] |
| Wastewater-grown algae | - | 350 | 0.5 | 27 | 0.06 | 20.3 | [46] |
3. Applications of Activated Hydrochar
3.1. Agriculture and Crop Improvement
3.2. Pollutant Adsorption (from Wastewater and Flue Gases)
3.3. Catalyst Support
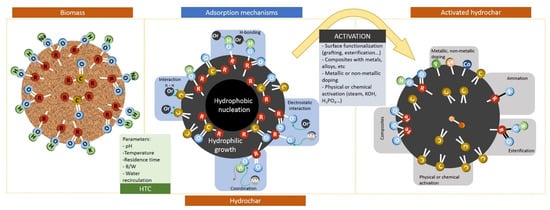
3.4. Energy Production
3.5. Carbon Sequestration
3.6. Electrochemical Devices
References
- Gollakota, A.R.K.; Kishore, N.; Gu, S. A review on hydrothermal liquefaction of biomass. Renew. Sustain. Energy Rev. 2018, 81, 1378–1392.
- Xu, D.; Lin, G.; Guo, S.; Wang, S.; Guo, Y.; Jing, Z. Catalytic hydrothermal liquefaction of algae and upgrading of biocrude: A critical review. Renew. Sustain. Energy Rev. 2018, 97, 103–118.
- Yen, H.W.; Hu, I.C.; Chen, C.Y.; Ho, S.H.; Lee, D.J.; Chang, J.S. Microalgae-based biorefinery–from biofuels to natural products. Bioresour. Technol. 2013, 135, 166–174.
- Vassilev, S.V.; Baxter, D.; Andersen, L.K.; Vassileva, C.G. An overview of the chemical composition of biomass. Fuel 2010, 89, 913–933.
- Masoumi, S.; Borugadda, V.B.; Dalai, A.K. Biocrude Oil Production via Hydrothermal Liquefaction of Algae and Upgradation Techniques to Liquid Transportation Fuels. In Biorefinery of Alternative Resources: Targeting Green Fuels and Platform Chemicals; Springer: Singapore, 2020; pp. 249–270.
- Qian, K.; Kumar, A.; Zhang, H.; Bellmer, D.; Huhnke, R. Recent advances in utilization of biochar. Renew. Sustain. Energy Rev. 2015, 42, 1055–1064.
- Cheng, F.; Li, X. Preparation and application of biochar-based catalysts for biofuel production. Catalysts 2018, 8, 346.
- Hoekman, S.K.; Broch, A.; Robbins, C.; Zielinska, B.; Felix, L. Hydrothermal carbonization (HTC) of selected woody and herbaceous biomass feedstocks. Biomass Convers. Biorefinery 2013, 3, 113–126.
- Shen, Y. A review on hydrothermal carbonization of biomass and plastic wastes to energy products. Biomass Bioenergy 2020, 134, 105479.
- Mumme, J.; Eckervogt, L.; Pielert, J.; Diakité, M.; Rupp, F.; Kern, J. Hydrothermal carbonization of anaerobically digested maize silage. Bioresour. Technol. 2011, 102, 9255–9260.
- Leng, L.; Huang, H. An overview of the effect of pyrolysis process parameters on biochar stability. Bioresour. Technol. 2018, 270, 627–642.
- Papari, S.; Hawboldt, K. A review on condensing system for biomass pyrolysis process. Fuel Process. Technol. 2018, 180, 1–13.
- Ayinla, R.T.; Dennis, J.O.; Zaid, H.M.; Sanusi, Y.K.; Usman, F.; Adebayo, L.L. A review of technical advances of recent palm bio-waste conversion to activated carbon for energy storage. J. Clean. Prod. 2019, 229, 1427–1442.
- Namazi, A.B.; Allen, D.G.; Jia, C.Q. Benefits of microwave heating method in production of activated carbon. Can. J. Chem. Eng. 2016, 94, 1262–1268.
- Hu, B.; Wang, K.; Wu, L.; Yu, S.H.; Antonietti, M.; Titirici, M.M. Engineering carbon materials from the hydrothermal carbonization process of biomass. Adv. Mater. 2010, 22, 813–828.
- Martinot, E.; Dienst, C.; Weiliang, L.; Qimin, C. Renewable energy futures: Targets, scenarios, and pathways. Annu. Rev. Environ. Resour. 2007, 32, 205–239.
- Lee, J.; Kim, K.H.; Kwon, E.E. Biochar as a catalyst. Renew. Sustain. Energy Rev. 2017, 77, 70–79.
- Mohan, D.; Sarswat, A.; Ok, Y.S.; Pittman, C.U., Jr. Organic and inorganic contaminants removal from water with biochar, a renewable, low cost and sustainable adsorbent–a critical review. Bioresour. Technol. 2014, 160, 191–202.
- Uchimiya, M.; Wartelle, L.H.; Klasson, K.T.; Fortier, C.A.; Lima, I.M. Influence of pyrolysis temperature on biochar property and function as a heavy metal sorbent in soil. J. Agric. Food Chem. 2011, 59, 2501–2510.
- Elliott, D.C. Review of recent reports on process technology for thermochemical conversion of whole algae to liquid fuels. Algal Res. 2016, 13, 255–263.
- Kumar, M.; Oyedun, A.O.; Kumar, A. A review on the current status of various hydrothermal technologies on biomass feedstock. Renew. Sustain. Energy Rev. 2018, 81, 1742–1770.
- Sharma, R.; Jasrotia, K.; Singh, N.; Ghosh, P.; Sharma, N.R.; Singh, J.; Kumar, A. A comprehensive review on hydrothermal carbonization of biomass and its applications. Chem. Afr. 2020, 3, 1–19.
- Tekin, K.; Karagöz, S.; Bektaş, S. A review of hydrothermal biomass processing. Renew. Sustain. Energy Rev. 2014, 40, 673–687.
- Zhang, Z.; Zhu, Z.; Shen, B.; Liu, L. Insights into biochar and hydrochar production and applications: A review. Energy 2019, 171, 581–598.
- Broch, A.; Jena, U.; Hoekman, S.K.; Langford, J. Analysis of solid and aqueous phase products from hydrothermal carbonization of whole and lipid-extracted algae. Energies 2014, 7, 62–79.
- Toor, S.S.; Rosendahl, L.; Rudolf, A. Hydrothermal liquefaction of biomass: A review of subcritical water technologies. Energy 2011, 36, 2328–2342.
- Elliott, D.C.; Hart, T.R.; Schmidt, A.J.; Neuenschwander, G.G.; Rotness, L.J.; Olarte, M.V.; Holladay, J.E. Process development for hydrothermal liquefaction of algae feedstocks in a continuous-flow reactor. Algal. Res. 2013, 2, 445–454.
- Masoumi, S.; Boahene, P.E.; Dalai, A.K. Biocrude oil and hydrochar production and characterization obtained from hydrothermal liquefaction of microalgae in methanol-water system. Energy 2021, 217, 119344.
- Lai, F.Y.; Chang, Y.C.; Huang, H.J.; Wu, G.Q.; Xiong, J.B.; Pan, Z.Q.; Zhou, C.F. Liquefaction of sewage sludge in ethanol-water mixed solvents for bio-oil and biochar products. Energy 2018, 148, 629–641.
- Feng, H.; Zhang, B.; He, Z.; Wang, S.; Salih, O.; Wang, Q. Study on co-liquefaction of Spirulina and Spartina alterniflora in ethanol-water co-solvent for bio-oil. Energy 2018, 155, 1093–1101.
- Zhang, J.; Zhang, Y.; Luo, Z. Hydrothermal liquefaction of chlorella pyrenoidosa in ethanol-water for bio-crude production. Energy Procedia 2014, 61, 1961–1964.
- Azadi, P.; Farnood, R. Review of heterogeneous catalysts for sub-and supercritical water gasification of biomass and wastes. Int. J. Hydrog. Energy 2011, 36, 9529–9541.
- Okolie, J.A.; Nanda, S.; Dalai, A.K.; Kozinski, J.A. Optimization and modeling of process parameters during hydrothermal gasification of biomass model compounds to generate hydrogen-rich gas products. Int. J. Hydrog. Energy 2020, 45, 18275–18288.
- Yang, C.; Wang, S.; Yang, J.; Xu, D.; Li, Y.; Li, J.; Zhang, Y. Hydrothermal Liquefaction and Gasification of Biomass and Model Compounds: A Review; Green Chemistry: Lodon, UK, 2020.
- Yao, Z.; Ma, X. Hydrothermal carbonization of Chinese fan palm. Bioresour. Technol. 2019, 282, 28–36.
- Salaudeen, S.A.; Acharya, B.; Dutta, A. Steam gasification of hydrochar derived from hydrothermal carbonization of fruit wastes. Renew. Energy 2021, 171, 582–591.
- Guo, S.; Dong, X.; Zhu, C.; Han, Y.; Ma, F.; Wu, T. Pyrolysis behaviors and thermodynamics properties of hydrochar from bamboo (Phyllostachys heterocycla cv. pubescens) shoot shell. Bioresour. Technol. 2017, 233, 92–98.
- Zhang, L.; Liu, S.; Wang, B.; Wang, Q.; Yang, G.; Chen, J. Effect of residence time on hydrothermal carbonization of corn cob residual. BioResources 2015, 10, 3979–3986.
- Zhu, G.; Yang, L.; Gao, Y.; Xu, J.; Chen, H.; Zhu, Y.; Zhu, C. Characterization and pelletization of cotton stalk hydrochar from HTC and combustion kinetics of hydrochar pellets by TGA. Fuel 2019, 244, 479–491.
- Shao, Y.; Tan, H.; Shen, D.; Zhou, Y.; Jin, Z.; Zhou, D.; Long, Y. Synthesis of improved hydrochar by microwave hydrothermal carbonization of green waste. Fuel 2020, 266, 117146.
- Yan, W.; Perez, S.; Sheng, K. Upgrading fuel quality of moso bamboo via low temperature thermochemical treatments: Dry torrefaction and hydrothermal carbonization. Fuel 2017, 196, 473–480.
- Kalderis, D.; Kotti, M.S.; Méndez, A.; Gascó, G. Characterization of hydrochars produced by hydrothermal carbonization of rice husk. Solid Earth 2014, 5, 477–483.
- Li, C.; Yang, X.; Zhang, Z.; Zhou, D.; Zhang, L.; Zhang, S.; Chen, J. Hydrothermal liquefaction of desert shrub Salix psammophila to high value-added chemicals and hydrochar with recycled processing water. BioResources 2013, 8, 2981–2997.
- Masoumi, S.; Dalai, A.K. Optimized production and characterization of highly porous activated carbon from algal-derived hydrochar. J. Clean. Prod. 2020, 263, 121427.
- Saber, M.; Takahashi, F.; Yoshikawa, K. Characterization and application of microalgae hydrochar as a low-cost adsorbent for Cu (II) ion removal from aqueous solutions. Environ. Sci. Pollut. Res. 2018, 25, 32721–32734.
- Yu, J.; Tang, T.; Cheng, F.; Huang, D.; Martin, J.L.; Brewer, C.E.; Luo, H. Waste-to-wealth application of wastewater treatment algae-derived hydrochar for Pb (II) adsorption. MethodsX 2021, 8, 101263.
- Sohi, S.; Krull, E.; Lopez-Capel, E.; Bol, R. A review of biochar and its use and function in soil. Adv. Agron. 2010, 105, 47–82.
- Warnock, D.D.; Lehmann, J.; Kuyper, T.W.; Rillig, M.C. Mycorrhizal responses to biochar in soil–concepts and mechanisms. Plant Soil 2017, 300, 9–20.
- Cheng, C.H.; Lehmann, J.; Engelhard, M.H. Natural oxidation of black carbon in soils: Changes in molecular form and surface charge along a climosequence. Geochim. Cosmochim. Acta 2008, 72, 1598–1610.
- Rillig, M.C.; Wagner, M.; Salem, M.; Antunes, P.M.; George, C.; Ramke, H.G.; Titirici, M.M.; Antonietti, M. Material derived from hydrothermal carbonization: Effects on plant growth and arbuscular mycorrhiza. Appl. Soil Ecol. 2010, 45, 238–242.
- Kambo, H.S.; Strength, D.A. Storage, and combustion characteristics of densified lignocellulosic biomass produced via torrefaction and hydrothermal carbonization. Appl. Energy 2014, 135, 182–191.
- Schimmelpfennig, S.; Muller, C.; Grunhage, L.; Koch, C.; Kammann, C. Biochar, hydrochar and uncarbonized feedstock application to permanent grassland—Effects on greenhouse gas emissions and plant growth. Agric. Ecosyst. Environ. 2014, 191, 39–52.
- Bargmann, I.; Rillig, M.C.; Kruse, A.; Greef, J.M.; Kucke, M. Effects of hydrochar application on the dynamics of soluble nitrogen in soils and on plant availability. J. Plant Nutr. Soil Sci. 2014, 177, 48–58.
- Qambrani, N.A.; Rahman, M.M.; Won, S.; Shim, S.; Ra, C. Biochar properties and eco-friendly applications for climate change mitigation, waste management, and wastewater treatment: A review. Renew. Sustain. Energy Rev. 2017, 79, 255–273.
- Xue, Y.; Gao, B.; Yao, Y.; Inyang, M.; Zhang, M.; Zimmerman, A.R.; Ro, K.S. Hydrogen peroxide modification enhances the ability of biochar (hydrochar) produced from hydrothermal carbonization of peanut hull to remove aqueous heavy metals: Batch and column tests. Chem. Eng. J. 2012, 200–202, 673–680.
- Fang, J.; Gao, B.; Chen, J.; Zimmerman, A.R. Hydrochars derived from plant biomass under various conditions: Characterization and potential applications and impacts. Chem. Eng. J. 2015, 267, 253–259.
- Ronix, A.; Pezoti, O.; Souza, L.S.; Souza, I.P.A.F.; Bedin, K.C.; Souza, P.S.C.; Silva, T.S.; Melo, S.A.R.; Cazetta, A.L.; Almeida, V.C. Hydrothermal carbonization of coffee husk: Optimization of experimental parameters and adsorption of methylene blue dye. J. Environ. Chem. Eng. 2017, 5, 4841–4849.
- Li, Y.; Meas, A.; Shan, S.; Yang, R.; Gai, X. Bioresource Technology Production and optimization of bamboo hydrochars for adsorption of Congo red and 2-naphthol. Bioresour. Technol. 2016, 207, 379–386.
- Regmi, P.; Moscoso, J.L.G.; Kumar, S.; Cao, X.; Mao, J.; Schafran, G. Removal of copper and cadmium from aqueous solution using switchgrass biochar produced via hydrothermal carbonization process. J. Environ. Manag. 2012, 109, 61–69.
- Liu, H.; Zhang, F.S. Removal of lead from water using biochars prepared from hydrothermal liquefaction of biomass. J. Hazard. Mater. 2009, 167, 933–939.
- Fagnani, H.M.C.; da Silva, C.T.P.; Pereira, M.M.; Rinaldi, A.W.; Arroyo, P.A.; de Barros, M.A.S.D. CO2 adsorption in hydrochar produced from waste biomass. SN Appl. Sci. 2019, 1, 1031.
- Gallucci, K.; Taglieri, L.; Papa, A.A.; Lauro, F.D.; Ahmad, Z.; Gallifuoco, A. Non-Energy Valorization of Residual Biomasses via HTC: CO2 Capture onto Activated Hydrochars. Appl. Sci. 2020, 10, 1879.
- Spataru, A.; Jain, R.; Chung, J.W.; Gerner, G.; Krebs, R.; Lens, P.N.L. Enhanced adsorption of orthophosphate and copper onto hydrochar derived from sewage sludge by KOH activation. RSC Adv. 2016, 6, 101827–101834.
- Fernandez-Sanroman, A.; Lama, G.; Pazos, M.; Rosales, E.; Sanroman, M.A. Bridging the gap to hydrochar production and its application into frameworks of bioenergy, environmental and biocatalysis areas. Bioresour. Technol. 2021, 320, 124399.
- Castro, M.O.; de Santiago, M.Q.; Nascimiento, K.S.; Cavada, B.S.; de Castro Miguel, E.; de Paula, A.J.; Ferreira, O.P. Hydrochar as protein support: Preservation of biomolecule properties with non-covalent immobilization. J. Material. Sci. 2017, 52, 13378–13389.
- Primozic, M.; Podrepsek, G.H.; Pavlovic, I.; Skerget, M.; Knez, Z.; Leitgeb, M. Enzyme immobilization onto biochar produced by the hydrothermal carbonization of biomass. Acta Chim. Slov. 2019, 66, 732–739.
- Kang, S.; Ye, J.; Zhang, Y.; Chang, J. Preparation of biomass hydrochar derived sulfonated catalysts and their catalytic effects for 5-hydroxymethylfurfural production. RSC Adv. 2013, 3, 7360–7366.
- Norouzi, O.; Heidari, M.; Martinez, M.M.; Dutta, A. New Insights for the Future Design of Composites Composed of Hydrochar and Zeolite for Developing Advanced Biofuels from Cranberry Pomace. Energies 2020, 13, 6600.
- Norouzi, O.; Heidari, M.; Maria, F.D.; Dutta, A. Design of a ternary 3D composite from hydrochar, zeolite and magnetite powder for direct conversion of biomass to gasoline. Chem. Eng. J. 2021, 410, 128323.
- Gai, C.; Zhang, F.; Guo, Y.; Peng, N.; Liu, T.; Lang, Q.; Xia, Y.; Liu, Z. Hydrochar-Supported, in Situ-Generated Nickel Nanoparticles for Sorption-Enhanced Catalytic Gasification of Sewage Sludge. ACS Sustain. Chem. Eng. 2017, 5, 7613–7622.
- Falco, C.; Marco-Lozar, J.P.; Salinas-Torres, D.; Morallon, E.; Cazorla-Amoros, D.; Titirici, M.M. Tailoring the porosity of chemically activated hydrothermal carbons: Influence of the precursor and hydrothermal carbonization temperature. Carbon 2013, 62, 346–355.
- Reza, M.T.; Andert, J.; Wirth, B.; Busch, D.; Pielert, J.; Lynam, J.G.; Mumme, J. Hydrothermal carbonization of biomass for energy and crop production. Appl. Bioenergy 2014, 1, 11–29.
- Reza, M.T.; Lynam, J.G.; Vasquez, V.R.; Coronella, C.J. Pelletization of biochar from hydrothermally carbonized wood. Environ. Prog. Sustain. Energy 2012, 31, 225–234.
- Liu, X.V.; Hoekman, S.K.; Farthing, W.; Felix, L. TC2015: Life cycle analysis of co-formed coal fines and hydrochar produced in twin-screw extruder (TSE). Environ. Prog. Sustain. Energy 2017, 36, 668–676.
- Sadish, O.; Sebastian, S.P.; Banu, K.S.P.; Mahendran, R. Hydrochar as an Energy Alternative to Coal: Effect of Temperature on Hydrothermal Carbonization of Paper Board Mill Sludge. Int. J. Curr. Microbiol. Appl. Sci. 2019, 8, 1668–1675.
- Oumabady, S.; Sebastian, P.S.; Kamaludeen, S.P.B.; Ramasamy, M.; Kalaiselvi, P.; Parameswari, E. Preparation and Characterization of Optimized Hydrochar from Paper Board Mill Sludge. Sci. Rep. 2020, 10, 773.
- Wang, T.; Zhai, Y.; Zhu, Y.; Gan, X.; Zheng, L.; Peng, C.; Wang, B.; Li, C.; Zeng, G. Evaluation of the clean characteristics and combustion behavior of hydrochar derived from food waste towards solid biofuel production. Bioresour. Technol. 2018, 266, 275–283.
- Gaunt, J.L.; Lehmann, J. Energy balance and emissions associated with biochar sequestration and pyrolysis bioenergy production. Environ. Sci. Technol. 2008, 42, 4152–4158.
- Lee, J.W.; Hawkins, B.; Day, D.M.; Reicosky, D.C. Sustainability: The capacity of smokeless biomass pyrolysis for energy production, global carbon capture and sequestration. Energy Environ. Sci. 2010, 3, 1695–1705.
- Liu, D.; Wang, Y.; Jia, B.; Wei, J.; Liu, C.; Zhu, J.; Tang, S.; Wu, S.; Chen, G. Microwave-Assisted Hydrothermal Preparation of Corn Straw Hydrochar as Supercapacitor Electrode Materials. ACS Omega 2020, 5, 26084–26093.
- Malghani, S.; Gleixner, G.; Trumbore, S.E. Chars produced by slow pyrolysis and hydrothermal carbonization vary in carbon sequestration potential and greenhouse gases emissions. Soil Biol. Biochem. 2013, 62, 137–146.
- Kammann, C.; Ratering, S.; Eckhard, C.; Müller, C. Biochar and hydrochar effects on greenhouse gas (carbon dioxide, nitrous oxide, and methane) fluxes from soils. J. Environ. Qual. 2012, 41, 1052–1066.
- Li, Y.; Liu, X. Activated carbon/ZnO composites prepared using hydrochars as intermediate and their electrochemical performance in supercapacitor. Mater. Chem. Phys. 2014, 148, 380–386.
- Ding, L.; Wang, Z.; Li, Y.; Du, Y.; Liu, H.; Guo, Y. A novel hydrochar and nickel composite for the electrochemical supercapacitor electrode material. Mater. Lett. 2012, 74, 111–114.
- Cheng, B.H.; Zeng, R.J.; Jiang, H. Recent developments of post-modification of biochar for electrochemical energy storage. Bioresour. Technol. 2017, 246, 224–233.
- Guo, J.; Zhang, J.; Jiang, F.; Zhao, S.; Su, Q.; Du, G. Microporous carbon nanosheets derived from corncobs for lithium–sulfur batteries. Electrochim. Acta 2015, 176, 853–860.
- Norouzi, O.; Pourhosseini, S.E.M.; Nader, H.R.; Maria, F.D.; Dutta, A. Integrated hybrid architecture of metal and biochar for high performance asymmetric supercapacitors. Sci. Rep. 2021, 11, 5387.




