
Video Upload Options
Next-generation renewable energy sources and perovskite solar cells have revolutionised photovoltaics research and the photovoltaic industry. However, the presence of toxic lead in perovskite solar cells hampers their commercialisation. Lead-free tin-based perovskite solar cells are a potential alternative solution to this problem; however, numerous technological issues must be addressed before the efficiency and stability of tin-based perovskite solar cells can match those of lead-based perovskite solar cells.
1. Introduction
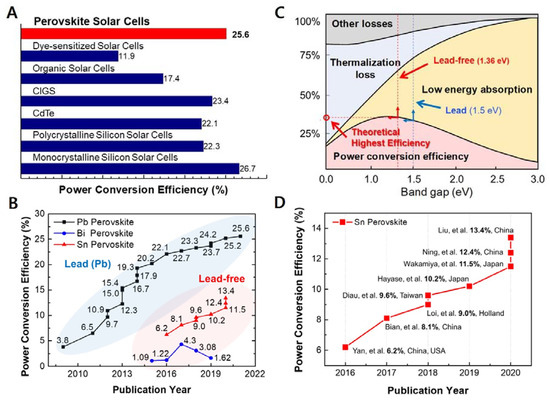
Since a pioneering report by Kojima et al., in 2009 [1] and a subsequent breakthrough by Kim et al. and Lee et al. [2][3] metal halide perovskite solar cells (PSCs) have substantially transformed the field of photovoltaics. Such next-generation light harvesters exhibit an exceptionally high-power conversion efficiency (PCE) owing to the long exciton diffusion length [4], high absorption coefficient [5], and defect tolerance [6] of perovskite materials. Presently, the highest PCE certified by the National Renewable Energy Laboratory (NREL) is 25.6% [7][8][9][10]. This value is higher than that of other types of photovoltaics and extremely close to that of monocrystalline silicon solar cells ( Figure 1 A). With rapid and substantial improvements in PSC technology, the intrinsic device instability of PSCs has also been greatly improved to the level of commercialization [11][12]. However, the presence of lead (Pb) in perovskite materials lowers the market value of PSCs, as Pb is perceived to be toxic and detrimental to the environment. The use of Pb in electronic products is banned by the Restriction of Hazardous Substances (RoHS) directive in Europe [13][14][15]. According to the World Health Organization (WHO), the human body cannot purge Pb through metabolism, and children are at a particularly high risk of Pb poisoning [16][17]. Moreover, Pb can easily spread via airborne particles into the air, water, and soil [18][19]. Thus, the scientific community has been searching for Pb alternatives. Metals such as tin (Sn), bismuth (Bi), antimony (Sb) have been reported to form metal halide perovskite crystals, and their application in PSCs has been proven to be effective ( Figure 1 B) [20][21]. Sn-based PSCs (Sn-PSCs) have thus far shown the best prospects. Although there are fewer reports on Sn-PSCs compared with those on Pb-PSCs, the PCE and stability of Sn-based PSCs have been improved rapidly. In addition, the ideal bandgap of Sn-based perovskite materials plays a crucial role ( Figure 1 C). Sn-based perovskites such as methylammonium tin iodide (MASnI 3), formamidinium tin iodide (FASnI 3), and caesium tin iodide (CsSnI 3) have direct bandgaps of approximately 1.20, 1.41, and 1.3 eV, respectively, which are narrower than those of Pb-based perovskites [22]. However, Sn 2+ in Sn-based perovskite is prone to form Sn 4+ . Although the product of this oxidation reaction is SnO 2, which is environment friendly, it dramatically lowers the device stability. This review focuses on Sn-PSCs that have significantly contributed to the development of Sn-PSC technology thus far ( Figure 1 D). Chronological accounts of Sn-PSCs from their initial development to the latest certified PCE can shed light on future prospects and research directions. Furthermore, this work reviews papers on solar cell–battery integration technologies to assess the potential of lead-free Sn-based PSCs as light harvesters in energy storage systems (ESSs).
2. Development of Pb-Free Sn-Based Perovskite Solar Cells
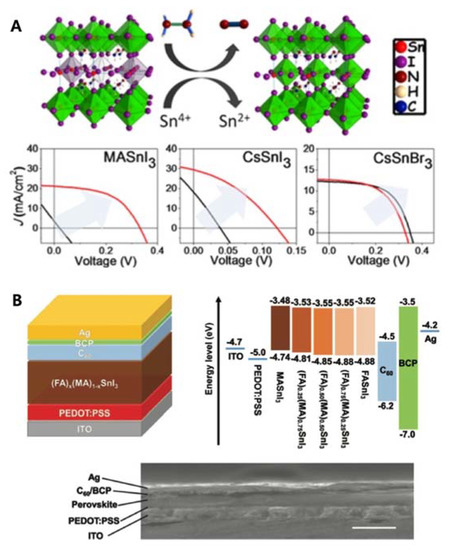
The first Sn-PSCs were reported by Kanatzidis and co-workers in 2014 [23]. The same year, Snaith and co-workers reported a higher PCE of 6.40% using the same device configuration [24]. However, they used MASnI 3 as the active layer in an n-i-p structure, of which the device performance was difficult to reproduce due to the poor stability nature of this device configuration [25]. The first inverted-type (p-i-n) Sn-PSCs were demonstrated by Yan and co-workers. They used poly(3,4-ethylenedioxythiophene) polystyrene sulfonate (PEDOT:PSS) as the hole-transporting layer, and C 60 and bathocuproine (BCP) as the electron-transporting layers [26]. Thus, a PCE of 6.22% was obtained ( Table 1 A). The key improvement was the addition of SnF 2 during diethyl ether antisolvent dripping, which resulted in highly uniform and pinhole-free compact FASnI 3 perovskite films. Kanatzidis and colleagues deepened the understanding of Sn-PSCs by including Cs in Sn-based perovskite fabrication [27]. Through the reduction of a vapour atmosphere during the preparation of Sn-based perovskites, PCEs of 3.89%, 1.83%, and 3.04% for MASnI 3, CsSnI 3, and CsSnBr 3, respectively, were obtained. Furthermore, the device stability and performance were considerably improved when the Sn 4+ /Sn 2+ ratio was reduced by 20% ( Table 1 B, Figure 2 A). Seok and co-workers reported a PCE of 4.8%, which remained stable for more than 100 d ( Table 1 C) [28]. The key to obtaining such a high device stability was the use of a SnF 2-pyrazine complex, which could disperse into the perovskite film better than SnF 2 alone. Kanatzidis and co-workers also reported a new type of hollow Sn-based perovskite [29], in which ethylenediammonium (en) served as the A-site cation in a FASnI 3 perovskite structure, forming {en}FASnI 3, a new hybrid perovskite structure. They showed that changing the A-site cation significantly increased the bandgap compared to X-site anion tuning, owing to the unique ability of en to create Schottky defects in perovskites. A relatively high PCE of 7.14% was attained with a device stability time of over 1000 h ( Table 1 D). The Sn-PSCs in the initial stage of development report PCEs under 8% despite the gradual improment over time. They rely on single A-site cation system until the work by Ke et al. [29], which demonstate en-added Sn perovskite. However, the amount of en was too small to regard the the perovskite composition as a mixed cation. Rather, en served more as an additive than a cation.
| Report | Year [Ref.] | Device Structure | JSC [mA m−2] | VOC [V] | FF | PCE [%] |
|---|---|---|---|---|---|---|
| A | 2016 [26] | Glass/ITO/PEDOT:PSS/FASnI3/C60/BCP/Ag | 22.07 | 0.47 | 0.60 | 6.22 |
| B | 2016 [27] | Glass/FTO/c-TiO2/m-TiO2/MASnI3/PTAA/Au | 19.92 | 0.38 | 0.51 | 3.86 |
| C | 2016 [28] | Glass/FTO/c-TiO2/m-TiO2/FASnI3/spiro-MeOTAD/Au | 23.70 | 0.32 | 0.63 | 4.80 |
| D | 2017 [29] | Glass/FTO/c-TiO2/m-TiO2/{en}FASnI3/spiro-MeOTAD/Au | 22.54 | 0.48 | 0.66 | 7.14 |
Note: ITO, indium tin oxide; FTO, fluorine-doped tin oxide.
Sn-based perovskites have excellent prospects to replace Pb, yet the easy oxidation and the low formation energy of Sn vacancies of Sn 2+ serve as a great hindrance to high PCE. One of the strategies to overcome this is engineering the A-site cation of ASnI 3. Mixing the A-site cations not only improve the film quality, such as morphology, but it can also change the optoelectrical properties of the Sn perovskites. This is because the perovskite crystal structure of ABX 3 is determined chiefly by the size of the A-site cation. Therefore, A-site cation engineering was used initially to fine tune the bandgap and to change the chemical composition of the Sn perovskite precursors. Initially, researchers mixed MA + , FA + , and Cs + A-site cations. The candidates extended to HA, EA, GA, EDA, TN, BEA, PN, BA, HEA, 4AMP, PEA, PPA, and 5-AVA in the later stage of the Sn-PSC evolution.
Loi and co-workers reported a method for lowering the background carrier density arising from the intrinsic defects in Sn perovskites. Sn vacancies and Sn 4 + were reduced by more than one order of magnitude via the deposition of near-single-crystalline FASnI 3, which possesses the orthorhombic a-axis in the out-of-plane direction [31]. This was achieved by mixing a small amount of a layered two-dimensional Sn perovskite (0.08 M) with 0.92 M of three-dimensional FASnI 3. The fabricated devices had a PCE of 9.0%, with negligible hysteresis and light soaking owing to a low trap density and efficient charge collection. SnF 2 was used as a reducing agent, as recommended in previous reports ( Table 3 F).
| Report | Year [Ref.] | Device Structure | JSC [mA m−2] | VOC [V] | FF | PCE [%] |
|---|---|---|---|---|---|---|
| F | 2018 [31] | Glass/ITO/PEDOT:PSS/FASnI3 (PEAI)/C60/ BCP/Ag | 24.1 | 0.53 | 0.71 | 9.00 |
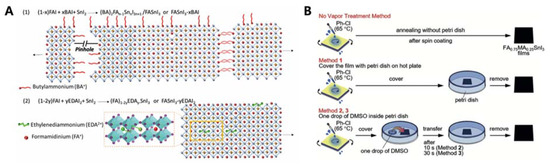
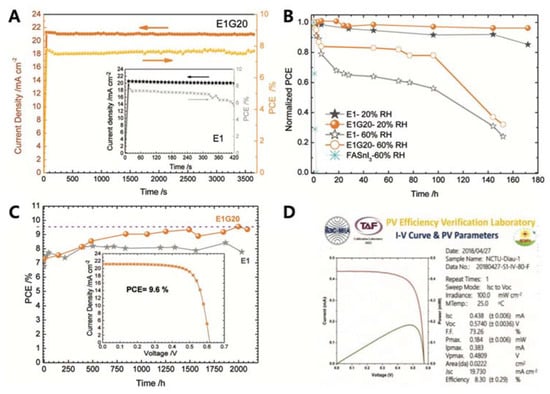
Diau and co-workers studied the doping effect of ethylenediammonium diiodide (EDAI 2) and butylammonium iodide (BAI) on organic cations, which passivated the defect sites and improved the film morphology and perovskite crystallinity [32]. The addition of BAI changed the orientation of the perovskite crystal growth and enhanced the connectivity of the crystal grains. The existing pinholes were passivated by the addition of EDAI 2. This prevented the oxidation of Sn 2+ to Sn 4+ and promoted slow relaxation of the crystal structure, as evidenced by X-ray diffraction, X-ray photoelectron spectroscopy, and photoluminescence decay measurements. With the addition of EDAI 2 (1%), the initial PCE of 7.4%, obtained for the FASnI 3-based PSC, increased to 8.9% after ageing. The resulting devices retained their PCE for more than 2000 h ( Table 4 H and Figure 3 A). Hayase and co-workers achieved highly stable and efficient Sn-PSCs by adding Ge to a Sn-based perovskite precursor. The Ge–Sn mixed perovskite film exhibited a bandgap between 1.4 to 1.5 eV, based on photoacoustic spectroscopy results [33]. The amount of added Ge was optimised at 5%, and thus, the perovskite structure was FA 0.75 MA 0.25 Sn 1−x GexI 3, as confirmed by X-ray diffraction and X-ray photoelectron spectroscopy measurements. The initial PCE of 4.48% increased to 6.90% after 72 h of ageing. The devices underwent measurements outside a glove box, without encapsulation ( Table 4 H). Subsequently, two simple methods for improving Sn-PSCs were proposed in a report by Liu et al. [34], The first method involved the use of a high-temperature antisolvent for full coverage of the Sn-based perovskites on PEDOT:PSS. The second method entailed annealing under a low partial pressure of dimethyl sulfoxide vapour, which increased the perovskite crystallite domains. A PCE of 7.20% was obtained owing to topographical and electrical improvements ( Table 5 J and Figure 3 B). Diau and co-workers achieved a PCE close to 10% in an inverted-type Sn-PSC, which used a mixture of nonpolar organic cation, guanidinium (GA + ), and formamidinium (FA + ) [35]. FASnI 3 was fabricated in the presence of 1% EDAI 2 as an additive, with an optimised ratio of GAI to FAI of 20:80. This composition resulted in a PCE of 8.5%, which then increased to 9.6% after storage inside a glove box ( Table 4 K). The devices were stable under continuous 1 sun illumination for 1 h without encapsulation. A certified PCE of 8.3% was obtained, which was the highest to the best of our knowledge ( Figure 4 ). Han and colleagues reduced the crystallisation speed of Sn-based perovskites by adding poly (vinyl alcohol) (PVA) which induced hydrogen bonding interactions during the growth of FASnI 3 [36]. This reduced the number of defect sites, which was reflected by the increased V OC . The recorded V OC of 0.63 V is ascribed to the suppressed migration of iodide ions in the presence of the PVA additive. A PCE of 8.92% was attained, and the device exhibited stable operation for 400 h ( Table 4 L).
| Report | Year [Ref.] | Device Structure | JSC [mA m−2] | VOC [V] | FF | PCE [%] |
|---|---|---|---|---|---|---|
| H | 2018 [32] | Glass/ITO/PEDOT:PSS/FASnI3 (1%EDAI2)/C60/BCP/Ag | 21.30 | 0.58 | 0.72 | 8.9 |
| I | 2018 [33] | Glass/ITO/PEDOT:PSS/FA0.75MA0.25Sn1-xGexI3/C60/BCP/Ag | 21.90 | 0.45 | 0.70 | 6.90 |
| J | 2018 [34] | Glass/ITO/PEDOT:PSS/FA0.75MA0.25SnI3/ C60/BCP/Ag | 19.40 | 0.55 | 0.61 | 7.20 |
| K | 2019 [35] | Glass/ITO/PEDOT:PSS/GAxFA0.98-xSnI3 (1%EDAI2)/C60/BCP/Ag | 21.20 | 0.62 | 0.73 | 9.60 |
| L | 2019 [36] | Glass/ITO/PEDOT:PSS/FASnI3-PVA/C60/BCP/Ag | 20.37 | 0.63 | 0.69 | 8.92 |
3. Conclusions
We have reviewed the development of Pb-free Sn-PSCs. By studying the progress of the Sn-PSC technology, we noticed that the enhancement of V OC played an important role in obtaining a high PCE. This was generally achieved by tuning the energy levels of Sn-based perovskites and charge-transporting layers. The Fermi level and bandgap of Sn-based perovskites were controlled by using different chemical compositions. Different electron-transporting layers, such as fullerene derivatives, were explored to find a material that matched the energy levels of the Sn-based perovskite materials. The absorption range could be widened by tuning the bandgap of the Sn-based perovskite layer; however, this could result in reductions in V OC and FF. In addition, the increase in J SC did not match the decrease in V OC and FF. Furthermore, passivation and protection of Sn 2+ from oxidation are important for increasing the FF and device stability. Various chemical compounds were used as additives. Further, higher PCEs for Sn-PSCs can potentially be achieved by investigating unexplored hole-transporting materials. High-performance Sn-PSCs are limited to inverted architectures and PEDOT: PSS hole-transporting layers. However, the highest PCEs have been reported for the normal architecture of Pb-based PSCs; thus, investigating methods for improving the performance of the normal architecture of Sn-PSCs is crucial. In this context, it is important to inhibit the damage caused by spiro-MeOTAD and its components. To this end, carbon nanotube electrodes which are laminated before the application of spiro-MeOTAD can be used [11][37][38][39][40]. Another method is to apply poly(triarylamine) (PTAA), which has yielded relatively high PCEs. The challenge in utilising PTAA lies in its wettability. A Sn-based perovskite precursor that contains DMSO cannot sufficiently cover the surface of PTAA as the latter is non-protic polar and highly dielectric. Modifying the PTAA surface could be a good solution for enhancing its applicability in Sn-PSCs.
References
- Kojima, A.; Teshima, K.; Shirai, Y.; Miyasaka, T. Organometal Halide Perovskites as Visible-Light Sensitizers for Photovoltaic Cells. J. Am. Chem. Soc. 2009, 131, 6050–6051.
- Kim, H.-S.; Lee, C.-R.; Im, J.-H.; Lee, K.-B.; Moehl, T.; Marchioro, A.; Moon, S.-J.; Humphry-Baker, R.; Yum, J.-H.; Moser, J.E.; et al. Lead Iodide Perovskite Sensitized All-Solid-State Submicron Thin Film Mesoscopic Solar Cell with Efficiency Exceeding 9%. Sci. Rep. 2012, 2, 591.
- Lee, M.M.; Teuscher, J.; Miyasaka, T.; Murakami, T.N.; Snaith, H.J. Efficient Hybrid Solar Cells Based on Meso-Superstructured Organometal Halide Perovskites. Science 2012, 338, 643–647.
- Xing, G.; Mathews, N.; Sun, S.; Lim, S.S.; Lam, Y.M.; Gratzel, M.; Mhaisalkar, S.; Sum, T.C. Long-Range Balanced Electron- and Hole-Transport Lengths in Organic-Inorganic CH3NH3PbI3. Science 2013, 342, 344–347.
- Lee, J.-W.; Seol, D.-J.; Cho, A.-N.; Park, N.-G. High-Efficiency Perovskite Solar Cells Based on the Black Polymorph of HC(NH2) 2PbI3. Adv. Mater. 2014, 26, 4991–4998.
- Yin, W.-J.; Shi, T.; Yan, Y. Unusual defect physics in CH3NH3PbI3 perovskite solar cell absorber. Appl. Phys. Lett. 2014, 104, 063903.
- Jeong, M.; Choi, I.W.; Go, E.M.; Cho, Y.; Kim, M.; Lee, B.; Jeong, S.; Jo, Y.; Choi, H.W.; Lee, J.; et al. Stable perovskite solar cells with efficiency exceeding 24.8% and 0.3-V voltage loss. Science 2020, 369, 1615–1620.
- Jeong, J.; Kim, M.; Seo, J.; Lu, H.; Ahlawat, P.; Mishra, A.; Yang, Y.; Hope, M.A.; Eickemeyer, F.T.; Kim, M.; et al. Pseudo-halide anion engineering for α-FAPbI3 perovskite solar cells. Nature 2021, 592, 381–385.
- Min, H.; Kim, M.; Lee, S.-U.; Kim, H.; Kim, G.; Choi, K.; Lee, J.H.; Seok, S., II. Efficient, stable solar cells by using inherent bandgap of α-phase formamidinium lead iodide. Science 2019, 366, 749–753.
- Kim, M.; Kim, G.-H.; Lee, T.K.; Choi, I.W.; Choi, H.W.; Jo, Y.; Yoon, Y.J.; Kim, J.W.; Lee, J.; Huh, D.; et al. Methylammonium Chloride Induces Intermediate Phase Stabilization for Efficient Perovskite Solar Cells. Joule 2019, 3, 2179–2192.
- Jeon, I.; Shawky, A.; Lin, H.-S.H.-S.; Seo, S.; Okada, H.; Lee, J.-W.J.-W.; Pal, A.; Tan, S.; Anisimov, A.; Kauppinen, E.I.E.I.; et al. Controlled Redox of Lithium-Ion Endohedral Fullerene for Efficient and Stable Metal Electrode-Free Perovskite Solar Cells. J. Am. Chem. Soc. 2019, 141, 16553–16558.
- Jeon, I.; Ueno, H.; Seo, S.; Aitola, K.; Nishikubo, R.; Saeki, A.; Okada, H.; Boschloo, G.; Maruyama, S.; Matsuo, Y. Lithium-Ion Endohedral Fullerene (Li + @C60) Dopants in Stable Perovskite Solar Cells Induce Instant Doping and Anti-Oxidation. Angew. Chem. Int. Ed. 2018, 57, 4607–4611.
- Lyu, M.; Yun, J.-H.; Chen, P.; Hao, M.; Wang, L. Addressing Toxicity of Lead: Progress and Applications of Low-Toxic Metal Halide Perovskites and Their Derivatives. Adv. Energy Mater. 2017, 7, 1602512.
- Abate, A. Perovskite Solar Cells Go Lead Free. Joule 2017, 1, 659–664.
- Hu, H.; Dong, B.; Zhang, W. Low-toxic metal halide perovskites: Opportunities and future challenges. J. Mater. Chem. A 2017, 5, 11436–11449.
- Mahaffey, K.R. Environmental lead toxicity: Nutrition as a component of intervention. Environ. Health Perspect. 1990, 89, 75–78.
- Wani, A.L.; Ara, A.; Usmani, J.A. Lead toxicity: A review. Interdiscip. Toxicol. 2015, 8, 55–64.
- Li, X.; Zhang, F.; He, H.; Berry, J.J.; Zhu, K.; Xu, T. On-device lead sequestration for perovskite solar cells. Nature 2020, 578, 555–558.
- Serrano-Lujan, L.; Espinosa, N.; Larsen-Olsen, T.T.; Abad, J.; Urbina, A.; Krebs, F.C. Tin- and Lead-Based Perovskite Solar Cells under Scrutiny: An Environmental Perspective. Adv. Energy Mater. 2015, 5, 1501119.
- Ke, W.; Stoumpos, C.C.; Kanatzidis, M.G. “Unleaded” Perovskites: Status Quo and Future Prospects of Tin-Based Perovskite Solar Cells. Adv. Mater. 2019, 31, 1803230.
- Giustino, F.; Snaith, H.J. Toward Lead-Free Perovskite Solar Cells. ACS Energy Lett. 2016, 1, 1233–1240.
- Stoumpos, C.C.; Malliakas, C.D.; Kanatzidis, M.G. Semiconducting Tin and Lead Iodide Perovskites with Organic Cations: Phase Transitions, High Mobilities, and Near-Infrared Photoluminescent Properties. Inorg. Chem. 2013, 52, 9019–9038.
- Hao, F.; Stoumpos, C.C.; Cao, D.H.; Chang, R.P.H.; Kanatzidis, M.G. Lead-free solid-state organic–inorganic halide perovskite solar cells. Nat. Photonics 2014, 8, 489–494.
- Noel, N.K.; Stranks, S.D.; Abate, A.; Wehrenfennig, C.; Guarnera, S.; Haghighirad, A.-A.A.; Sadhanala, A.; Eperon, G.E.; Pathak, S.K.; Johnston, M.B.; et al. Lead-Free Organic-Inorganic Tin Halide Perovskites for Photovoltaic Applications. Energy Environ. Sci. 2014, 7, 3061–3068.
- Diau, E.W.-G.; Jokar, E.; Rameez, M. Strategies to Improve Performance and Stability for Tin-Based Perovskite Solar Cells. ACS Energy Lett. 2019, 4, 1930–1937.
- Liao, W.; Zhao, D.; Yu, Y.; Grice, C.R.; Wang, C.; Cimaroli, A.J.; Schulz, P.; Meng, W.; Zhu, K.; Xiong, R.; et al. Lead-Free Inverted Planar Formamidinium Tin Triiodide Perovskite Solar Cells Achieving Power Conversion Efficiencies up to 6.22%. Adv. Mater. 2016, 28, 9333–9340.
- Song, T.B.; Yokoyama, T.; Stoumpos, C.C.; Logsdon, J.; Cao, D.H.; Wasielewski, M.R.; Aramaki, S.; Kanatzidis, M.G. Importance of reducing vapor atmosphere in the fabrication of Tin-based perovskite solar cells. J. Am. Chem. Soc. 2017, 139, 836–842.
- Lee, S.J.; Shin, S.S.; Kim, Y.C.; Kim, D.; Ahn, T.K.; Noh, J.H.; Seo, J.; Seok, S., II. Fabrication of Efficient Formamidinium Tin Iodide Perovskite Solar Cells through SnF2–Pyrazine Complex. J. Am. Chem. Soc. 2016, 138, 3974–3977.
- Ke, W.; Stoumpos, C.C.; Zhu, M.; Mao, L.; Spanopoulos, I.; Liu, J.; Kontsevoi, O.Y.; Chen, M.; Sarma, D.; Zhang, Y.; et al. Enhanced photovoltaic performance and stability with a new type of hollow 3D perovskite FASnI3. Sci. Adv. 2017, 3, e1701293.
- Zhao, Z.; Gu, F.; Li, Y.; Sun, W.; Ye, S.; Rao, H.; Liu, Z.; Bian, Z.; Huang, C. Mixed-Organic-Cation Tin Iodide for Lead-Free Perovskite Solar Cells with an Efficiency of 8.12%. Adv. Sci. 2017, 4.
- Shao, S.; Liu, J.; Portale, G.; Fang, H.H.; Blake, G.R.; Brink, G.H.t.; Koster, L.J.A.; Loi, M.A. Highly Reproducible Sn-Based Hybrid Perovskite Solar Cells with 9% Efficiency. Adv. Energy Mater. 2018, 8, 1702019.
- Jokar, E.; Chien, C.H.; Fathi, A.; Rameez, M.; Chang, Y.H.; Diau, E.W.G. Slow surface passivation and crystal relaxation with additives to improve device performance and durability for tin-based perovskite solar cells. Energy Environ. Sci. 2018, 11, 2353–2362.
- Ito, N.; Kamarudin, M.A.; Hirotani, D.; Zhang, Y.; Shen, Q.; Ogomi, Y.; Iikubo, S.; Minemoto, T.; Yoshino, K.; Hayase, S. Mixed Sn-Ge Perovskite for Enhanced Perovskite Solar Cell Performance in Air. J. Phys. Chem. Lett. 2018, 9, 1682–1688.
- Liu, J.; Ozaki, M.; Yakumaru, S.; Handa, T.; Nishikubo, R.; Kanemitsu, Y.; Saeki, A.; Murata, Y.; Murdey, R.; Wakamiya, A. Lead-Free Solar Cells based on Tin Halide Perovskite Films with High Coverage and Improved Aggregation. Angew. Chem. Int. Ed. 2018, 57, 13221–13225.
- Jokar, E.; Chien, C.H.; Tsai, C.M.; Fathi, A.; Diau, E.W.G. Robust Tin-Based Perovskite Solar Cells with Hybrid Organic Cations to Attain Efficiency Approaching 10%. Adv. Mater. 2019, 31, 1804835.
- Meng, X.; Lin, J.; Liu, X.; He, X.; Wang, Y.; Noda, T.; Wu, T.; Yang, X.; Han, L. Highly Stable and Efficient FASnI3-Based Perovskite Solar Cells by Introducing Hydrogen Bonding. Adv. Mater. 2019, 31, 1903721.
- Lee, J.-W.; Jeon, I.; Lin, H.-S.; Seo, S.; Han, T.-H.; Anisimov, A.; Kauppinen, E.I.; Matsuo, Y.; Maruyama, S.; Yang, Y. Vapor-Assisted Ex-Situ Doping of Carbon Nanotube toward Efficient and Stable Perovskite Solar Cells. Nano Lett. 2019, 19, 2223–2230.
- Jeon, I.; Xiang, R.; Shawky, A.; Matsuo, Y.; Maruyama, S. Single-Walled Carbon Nanotubes in Emerging Solar Cells: Synthesis and Electrode Applications. Adv. Energy Mater. 2019, 9, 1801312.
- Jeon, I.; Shawky, A.; Seo, S.; Qian, Y.; Anisimov, A.; Kauppinen, E.I.; Matsuo, Y.; Maruyama, S. Carbon nanotubes to outperform metal electrodes in perovskite solar cells via dopant engineering and hole-selectivity enhancement. J. Mater. Chem. A 2020, 8, 11141–11147.
- Jeon, I.; Matsuo, Y.; Maruyama, S. Single-Walled Carbon Nanotubes in Solar Cells. In Single-Walled Carbon Nanotubes. Topics in Current Chemistry Collections; Springer: Cham, Switzerland, 2018; Volume 376, pp. 271–298.




