Your browser does not fully support modern features. Please upgrade for a smoother experience.

Submitted Successfully!
Thank you for your contribution! You can also upload a video entry or images related to this topic.
For video creation, please contact our Academic Video Service.
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Dylan Deska-Gauthier | + 2326 word(s) | 2326 | 2021-08-06 08:57:44 | | | |
| 2 | Camila Xu | Meta information modification | 2326 | 2021-08-17 04:08:55 | | |
Video Upload Options
We provide professional Academic Video Service to translate complex research into visually appealing presentations. Would you like to try it?
Cite
If you have any further questions, please contact Encyclopedia Editorial Office.
Deska-Gauthier, D. Spinal Cord Interneuron Differentiation. Encyclopedia. Available online: https://encyclopedia.pub/entry/13229 (accessed on 08 February 2026).
Deska-Gauthier D. Spinal Cord Interneuron Differentiation. Encyclopedia. Available at: https://encyclopedia.pub/entry/13229. Accessed February 08, 2026.
Deska-Gauthier, Dylan. "Spinal Cord Interneuron Differentiation" Encyclopedia, https://encyclopedia.pub/entry/13229 (accessed February 08, 2026).
Deska-Gauthier, D. (2021, August 16). Spinal Cord Interneuron Differentiation. In Encyclopedia. https://encyclopedia.pub/entry/13229
Deska-Gauthier, Dylan. "Spinal Cord Interneuron Differentiation." Encyclopedia. Web. 16 August, 2021.
Copy Citation
The spinal cord is comprised of vastly heterogeneous interneuron populations defined by unique molecular identities, intrinsic properties, connectivity and functional outputs.
spinal cord
interneuron
subpopulations
neurogenesis
mouse
zebra fish
temporal control
postmitotic differentiation
locomotion
1. Introduction
Interneuron (IN) circuits in the spinal cord are essential for patterned, rhythmic and flexible motor control. From basic to complex sensorimotor tasks, combinatorial IN recruitments in the spinal cord are required for successful execution of movement. The spinal cord is comprised of vastly heterogeneous IN populations defined by unique molecular identities, intrinsic properties, connectivity and functional outputs. This IN diversity enables the spinal cord to coordinate varied movement schemes through dynamic environments. Thus, understanding spinal IN diversity and the developmental mechanisms that give rise to it, is fundamental to understanding movement.
Early work in the mammalian spinal cord revealed a remarkable spatial organization of progenitor domains along the dorsoventral axis during early embryogenesis [1][2]. These 11 progenitor domains give rise to distinct post-mitotic interneuron (IN) and motor neuron (MN) cardinal classes (dI1–dI6 INs, dILA–B, V0–V3 INs, MNs) defined by respective transcription factor (TF) expression profiles. Physiological and anatomical studies have revealed general connectivity, electrophysiological properties and functional outputs of these cardinal IN classes across various model systems [3][4][5]. However, extensive subpopulation heterogeneity has become evident within each cardinal class [6][7][8]. Furthermore, the developmental mechanisms underlying such subpopulation diversities are beginning to be understood.
Neurogenesis timing is an essential developmental mechanism for neuronal diversity and organization throughout the central nervous system [9][10]. Likewise, it plays an instructive role in the development of IN circuits within the spinal cord [11]. Notably, spinal INs form the final circuits controlling the coordination and rhythmicity of movement. This enables behavioural quantifications of their circuit outputs. Thus, spinal IN circuits are ideal model systems for understanding how differential neurogenesis timing contributes to molecular, cellular and behavioural development in the central nervous system.
2. Lessons from the Mouse Spinal Cord
2.1. Early Temporal Mechanisms Guide Molecular Diversity in the Mouse Spinal Cord
One of most revolutionary breakthroughs in understanding the development of spinal neurons is the discovery of spatially organized progenitors during early embryogenesis. Graded morphogens sonic hedgehog (Shh), released from the floor plate, and bone morphogenic protein (BMP)/Wnt protein, released from the roof plate, pattern the positions and cross-inhibitory boundaries of 11 discrete progenitor domains along the dorsoventral spinal axis [1]. These progenitor domains, in turn, give rise to 13 distinct post-mitotic cardinal IN populations and MNs. However, accumulating evidence suggests vast subpopulation diversity within each cardinal population and differential neurogenesis timing as a potentially key developmental mechanism for such diversity.
Recent work by Delile and colleagues utilized single cell RNA sequencing to systematically profile post-mitotic neurons across early embryonic stages (E9.5–E13.5) in the mouse spinal cord. They revealed a temporal emergence of shared transcription factor networks endowing subpopulation cluster identities across cardinal IN classes. This work was the first to systematically reveal previously underappreciated temporal mechanisms—acting in tandem with spatial controls—delineating IN subpopulation identities in the spinal cord. Indeed, several of these temporally regulated postmitotic TFs have been independently shown to be necessary for the specification and differentiation of subpopulation identities. Onecut TFs expressed across early-born spinal IN classes [12] are necessary for the differentiation of Renshaw cells (RCs) [13] and other spinal INs [14][15]; Pou2f2 and Zfhx TFs expressed across intermediate-born spinal IN classes [12] are necessary for proper migration [14][16] and molecular specification of laterally positioned V2a INs [17]; lastly, Nfib TFs expressed across late-born spinal IN classes [12] serve as molecular markers for a medial V2a IN subpopulation [17]. Together, this work has illuminated those neurons across the spinal cord may follow shared developmental temporal logic in their molecular diversification from spatially confined progenitor domains. However, understanding how and whether temporal mechanisms translate into distinct IN phenotypes, circuit integrations and functional outputs remains an ongoing question. Over the last decade, various studies have begun to investigate how differential neurogenesis timing orders the divergence of IN properties and functions.
2.2. Interneuron Subpopulations Emerge from Temporally Separated Progenitors
V1 INs, defined by engrailed-1 TF expression, arise from the p1 progenitor domain between embryonic days (E) 9.5 and E12.5 in the mouse spinal cord [5][13]. They project ipsilaterally and form inhibitory contacts onto both MNs and other IN classes in the ventral spinal cord [2]. In the mouse, V1 INs have been shown to be necessary for increased locomotor speed [7][18] and flexor–extensor alternation during walking [19][20]. Several classically characterized spinal IN types, such as RCs and inhibitory Ia-INs, were shown to be part of the V1 IN lineage [21]. They were among the first groups of subpopulations recognized within the cardinal populations. However, the vast heterogeneity of V1 INs was not fully revealed until the combinatorial expression of 19 distinct TFs was shown to delineate approximately 50 distinct V1 subsets throughout the lumbar and thoracic spinal cord [22][23][24].
More interestingly, in addition to revealing RC and Ia-IN V1 lineage, the same research groups showed that RCs and Ia-INs emerge from the p1 progenitor domain at different embryonic timepoints. They revealed that V1 INs could be organized into two general waves of neurogenesis: early (E9.5–E10.5) and late (E11.5–E12.5). The first wave of neurogenesis from the p1 progenitor domain gives rise to RCs [13][18], while the second wave gives rise to inhibitory Ia-INs, FoxP2+ V1 INs and other V1 IN subpopulations [18]. Early-born RCs are marked by the expression of a distinct TF profile (Foxd3, MafB, Onecut1 and Onecut2), as well as the calcium binding protein, calbindin [25][26]. Upon exiting from the p1 progenitor domain, RCs display a distinct ventrolateral migratory stream settling amongst lateral motor column MNs [25][27]. This early differentiation pathway allows RCs to form unique recurrent inhibitory circuits with MNs [26].
The early V1 IN birthdate determines a temporally ordered TF cascade necessary for the specification and maintenance of a RC-type specific phenotype [26]. Transcription factors Onecut1, Onecut2 and Foxd3 are responsible for the immediate postmitotic differentiation of RCs, including their calbindin expression, migration and circuit formation with MNs. Subsequently, downstream MafB expression is necessary for the maintenance of RC identity during late embryonic stages [26]. Thus, the specific early neurogenesis timing results in the postmitotic acquirement of a distinct TF expression cascade that facilitates the differentiation, maturation and circuit integration of RCs, separating them from other V1 INs.
While V1 IN subpopulation identity is correlated with neurogenesis time, to what extent their temporal expression profiles are endowed by intrinsic transcription programs or extrinsic signaling pathways remains an ongoing question. To begin to answer this question, Hoang and colleagues [28] established an in vitro model system of V1 IN diversification utilizing mouse embryonic stem cell (ESC) cultures. ESC V1 IN clades displayed transcription factor expressions, electrophysiological properties and connectivities that recapitulated those observed in the spinal cord [6][28]. Interestingly, ESC V1 IN subpopulations also displayed distinct neurogenesis birth orders in culture. Calbindin+ V1 INs were born first, followed by Foxp2+ V1 INs. These results, from a system in the absence of many surrounding extrinsic signaling sources, are similar to those observed in vivo [25][26].
Hoang and colleagues [28] further assessed a potential causative link between neurogenesis timing and subpopulation generation. Through inhibition of Notch signaling, they were able to increase the rate of cell cycle exit and neurogenesis timing. When Notch was inhibited at early stages, there was a significant increase in the proportion of early-born Calbindin+ V1 INs, as well as other TFs belonging to the MafA+ V1 clade. This early-born subpopulation increase was accompanied by an almost complete loss of late-born Foxp2+ V1 INs. These experiments indicated that, when late-born V1 INs were prematurely pushed out of the p1 progenitor domain, they switched to an early-born subpopulation fate.
Taken together, both these in vivo and in vitro studies suggest that differential neurogenesis timing enables specific temporal transcription pathways in p1 progenitor cells to be translated into distinct V1 IN subpopulation fates. That is, the transcriptional identity of a V1 IN (or any spinal IN) at the time it becomes post-mitotic may instruct its subpopulation fate choice.
Beyond V1 INs, we have begun to investigate how neurogenesis timing underlies subpopulation divergence within the most ventral-originating V3 IN cardinal class. V3 INs, marked by TF Sim1 expression, also exit from the p3 progenitor domain between E9.5 and E12.5 in the mouse spinal cord. V3 INs are mostly commissural and excitatory INs [29][30]. They are functionally involved in coordinating excitation between left–right extensor centres [31] and robust locomotor pattern output [30]. As V3 INs become postmitotic, they form distinct dorsolateral and ventrolateral migratory streams. Early-born (E9.5–E10.5) V3 INs follow both dorsolateral and ventrolateral migratory streams and cluster across deep dorsal, intermediate and ventral laminae by postnatal day (P) 0. In contrast, late-born (E11.5–E12.5) V3 INs almost exclusively follow the ventrolateral migratory stream and cluster mostly within ventral laminae by P0 [32]. Furthermore, early-born V3 INs display both ascending and descending axonal projections, while late born V3 INs display significantly more descending axonal projections than ascending ones [32]. Thus, successive neurogenesis timing fate-restricts late-born V3 INs to anatomically confined subpopulations.
Whether distinct transcriptional pathways are restricted to different temporal waves of V3 neurogenesis, as seen in V1 INs, remains largely unknown. Though, we have shown some evidence that the Sim1 TF, while expressed in all V3 INs, exclusively affects the laminar clustering and electrophysiological properties of early-born dorsal and intermediate V3 IN clusters, but not late-born ventral V3 IN clusters. Much more work is required to reveal the role temporal mechanisms play in regulating the molecular pathways underlying V3 IN subpopulation diversity and how that diversity is then translated into functionally distinct circuit integrations.
2.3. Select Dorsal IN Populations Emerge from Temporally Separated Progenitors
Select sensory-related dorsal IN populations have also been shown to emerge during specific neurogenesis windows. Dorsal horn progenitors separate along both spatial and temporal axes of control. Two dorsal progenitor lineages, dILA and dILB INs, emerge from Lbx1+ dorsal progenitors during specifically late neurogenesis stages. dILA coexpress Gbx1 with Pax2 and Lbx1 and are inhibitory, while dILB coexpress Lmx1b with Lbx1 and are excitatory [33][34][35]. Of particular note, Gbx1 is exclusively expressed and is necessary for the specification and differentiation of late-born dILA INs [26][36][37].
Loss of Gbx1 resulted in abnormal hindlimb gaits during locomotion, as well as sensory processing [36][37]. Gbx1 knockout (KO) mice displayed a duck-type gait characterized by hyper flexion during the swing phase and a decrease in average locomotor speed in open field tests. They also displayed reduced thermal pain sensitivity and increased slips during beam walking [37]. However, whether this was due to Gbx1 early expression in the floor plate, ISL1+ motor neurons, or dILA INs is not clear. However, Gbx1 KO mice displayed intact motor strength and no changes in the number of ISL1+ MNs, nor sensory innervations patterns [37]. Taken together, this suggests that temporally regulated expression of Gbx1 in late-born dorsal INs is necessary for the specification of dILA INs, which are involved in distinct aspects of sensorimotor control.
Sensory mediating cerebrospinal fluid contacting neurons were also shown to have a characteristically late neurogenesis timing in the mouse spinal cord. These neurons are born as late as E14–E16 from the oligodendrocite and p2 progenitor domains [38]. This neurogenesis window is well beyond the common neurogenesis window observed in the mouse spinal cord. Cerebrospinal fluid contacting neurons settle around the central canal and display a unique morphology with the extension of a dendrite into the central canal, unique mechanosensitive channel expression and distinct electrophysiological properties [38]. While their function has not been shown in mice, they are likely functionally distinct from other p2 originating IN types, V2a and V2b INs. Indeed, in the zebrafish cerebrospinal fluid contacting neurons have been shown responsible for sensing spinal bending and mediating postural control during locomotion [39][40][41].
2.4. Neurogenesis Timing Can Restrict in Specific Circuit Wirings
Timing of neuronal differentiation may play a role beyond defining molecular identity of spinal subpopulations, to guiding the formation of distinct circuit connectivity. While limited studies have been performed to date, there is evidence for IN neurogenesis timing and motor pool specific wiring.
Trans-synaptic viral tracing by Tripodi and colleges [42] revealed that ipsilateral dI4-6 INs respectively innervating flexor or extensor MNs were spatially, synaptically and temporally separated. Last-order extensor INs were positioned more medially and received high levels of proprioceptive innervation, while flexor INs were positioned more laterally and received less proprioceptive innervations. Interestingly, last-order flexor INs were early-born cells (around E10.5), while last-order extensor INs were born later (around E12.5) (Figure 1A,B [42]). The late-born Lbx1+ last-order extensor INs were most likely from late dILA,B progenitors [24][30][38][43]. Thus, the time within which an IN becomes post-mitotic may position it in a specific functional pathway.
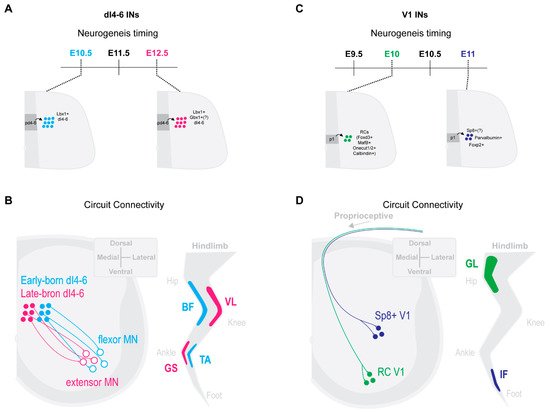
Figure 1. IN neurogenesis timing and circuit connectivity in the mouse spinal cord. (A) dI4-6 last-order INs emerge during either early-born (E10.5) or late-born (E12.5) neurogenesis waves [42]. Late-born dI4-6 INs are likely dILA/B IN populations and some likely express Gbx1+ [26][36][37]. (B) By postnatal stages, early-born dI4-6 INs are positioned preferentially laterally and innervate flexor MNs, while late-born dI4-6 INs are positioned preferentially medially and innervate extensor MNs [42] (extensor muscles, vastus lateralis (VL), gastrocnemius (GS); flexor muscles, biceps femoris (BF), Tibialis anterior (TA)). (C) Early-born V1 INs (E10) differentiate into Renshaw cells (RCs) [13][25], while late-born V1 INs (E11) (differentiate into Foxp2+ V1 INs [25], Parvalbumin+ V1 INs [25] and, likely, Sp8+ V1 INs [27]. (D) By postnatal stages, early-born RC V1 INs settle within more ventral clusters and receive more proprioceptive innervations from proximal hip muscles [6]. Presumptive later-born Sp8+ V1 INs [28] settle within more dorsal clusters and receive more proprioceptive innervations from distal foot muscles [6] (proximal muscle, Gluteus (GL); distal muscle, intrinsic foot (IF)).
Likewise, neurogenetically separated V1 INs display distinct microcircuit connectivity. Early-born V1 RCs [13][25] settled within more ventral clusters and received more proprioceptive innervations from proximal hip muscles [6]. In contrast, presumptive later-born Sp8+ V1 INs [28] settled within more dorsal clusters and received more proprioceptive innervations from distal foot muscles [6] (Figure 1C,D). While the functional relevance of these distinct sensory innervation patterns remains to be determined, it suggests an intriguing link between timing of an INs differentiation and microcircuit specific integration.
References
- Jessell, T.M. Neuronal Specification in the Spinal Cord: Inductive Signals and Transcriptional Codes. Nat. Rev. Genet. 2000, 1, 20–29.
- Lu, D.C.; Eniu, T.; Alaynick, W.A. Molecular and Cellular Development of Spinal Cord Locomotor Circuitry. Front. Mol. Neurosci. 2015, 8, 25.
- Deska-Gauthier, D.; Zhang, Y. The Functional Diversity of Spinal Interneurons and Locomotor Control. Curr. Opin. Physiol. 2019, 8, 99–108.
- Goulding, M. Circuits Controlling Vertebrate Locomotion: Moving in a New Direction. Nat. Rev. Neurosci. 2009, 10, 507–518.
- Kiehn, O. Decoding the Organization of Spinal Circuits That Control Locomotion. Nat. Rev. Neurosci. 2016, 17, 224–238.
- Bikoff, J.B.; Gabitto, M.I.; Rivard, A.F.; Drobac, E.; Machado, T.; Miri, A.; Brenner-Morton, S.; Famojure, E.; Diaz, C.; Alvarez, F.J.; et al. Spinal Inhibitory Interneuron Diversity Delineates Variant Motor Microcircuits. Cell 2016, 165, 207–219.
- Gosgnach, S.; Bikoff, J.B.; Dougherty, K.J.; El Manira, A.; Lanuza, G.M.; Zhang, Y. Delineating the Diversity of Spinal In-ter-neurons in Locomotor Circuits. J. Neurosci. 2017, 37, 10835–10841.
- Ziskind-Conhaim, L.; Hochman, S. Diversity of Molecularly Defined Spinal Interneurons Engaged in Mammalian Locomotor Pattern Generation. J. Neurophysiol. 2017, 118, 2956–2974.
- Holguera, I.; Desplan, C. Neuronal Specification in Space and Time. Sci. 2018, 362, 176–180.
- Oberst, P.; Agirman, G.; Jabaudon, D. Principles of Progenitor Temporal Patterning in the Developing Invertebrate and Ver-tebrate Nervous System. Curr. Opin. Neurobiol. 2019, 56, 185–193.
- Sagner, A.; Briscoe, J. Establishing Neuronal Diversity in the Spinal Cord: A Time and a Place. Development 2019, 146.
- Delile, J.; Rayon, T.; Melchionda, M.; Edwards, A.; Briscoe, J.; Sagner, A. Single Cell Transcriptomics Reveals Spatial and Temporal Dynamics of Gene Expression in the Developing Mouse Spinal Cord. Development 2019, 146.
- Stam, F.J.; Hendricks, T.J.; Zhang, J.; Geiman, E.J.; Francius, C.; Labosky, P.; Clotman, F.; Goulding, M. Renshaw Cell In-ter-neuron Specialization Is Controlled by a Temporally Restricted Transcription Factor Program. Development 2012, 139, 179–190.
- Harris, A.; Masgutova, G.; Collin, A.; Toch, M.; Hidalgo-Figueroa, M.; Jacob, B.; Corcoran, L.M.; Francius, C.; Clotman, F. Onecut Factors and Pou2f2 Regulate the Distribution of V2 Interneurons in the Mouse Developing Spinal Cord. Front. Cell. Neurosci. 2019, 13, 184.
- Kabayiza, K.U.; Masgutova, G.; Harris, A.; Rucchin, V.; Jacob, B.; Clotman, F. The Onecut Transcription Factors Regulate Differentiation and Distribution of Dorsal Interneurons During Spinal Cord Development. Front. Mol. Neurosci. 2017, 10, 157.
- Masgutova, G.; Harris, A.; Jacob, B.; Corcoran, L.M.; Clotman, F. Pou2f2 Regulates the Distribution of Dorsal Interneurons in the Mouse Developing Spinal Cord. Front. Mol. Neurosci. 2019, 12, 263.
- Hayashi, M.; Hinckley, C.A.; Driscoll, S.P.; Moore, N.J.; Levine, A.; Hilde, K.L.; Sharma, K.; Pfaff, S.L. Graded Arrays of Spinal and Supraspinal V2a Interneuron Subtypes Underlie Forelimb and Hindlimb Motor Control. Neuron 2018, 97, 869–884.e5.
- Falgairolle, M.; O’Donovan, M.J. V1 interneurons regulate the pattern and frequency of locomotor-like activity in the neonatal mouse spinal cord. PLoS Biol. 2019, 17, e3000447.
- Zhang, J.; Lanuza, G.M.; Britz, O.; Wang, Z.; Siembab, V.C.; Zhang, Y.; Velasquez, T.; Alvarez, F.J.; Frank, E.; Goulding, M. V1 and v2b interneurons secure the alternating flexor-extensor motor activity mice require for limbed locomotion. Neuron 2014, 82, 138–150.
- Britz, O.; Zhang, J.; Grossmann, K.S.; Dyck, J.; Kim, J.C.; Dymecki, S.; Gosgnach, S.; Goulding, M. A genetically defined asymmetry underlies the inhibitory control of flexor-extensor locomotor movements. Elife 2015, 4, e04718.
- Alvarez, F.J.; Jonas, P.C.; Sapir, T.; Hartley, R.; Berrocal, M.C.; Geiman, E.J.; Todd, A.J.; Goulding, M. Postnatal Phenotype and Localization of Spinal Cord V1 Derived Interneurons. J. Comp. Neurol. 2005, 493, 177–192.
- Bikoff, J.B. Interneuron Diversity and Function in the Spinal Motor System. Curr. Opin. Physiol. 2019, 8, 36–43.
- Gabitto, M.I.; Pakman, A.; Bikoff, J.B.; Abbott, L.; Jessell, T.M.; Paninski, L. Bayesian Sparse Regression Analysis Documents the Diversity of Spinal Inhibitory Interneurons. Cell 2016, 165, 220–233.
- Sweeney, L.B.; Bikoff, J.B.; Gabitto, M.I.; Brenner-Morton, S.; Baek, M.; Yang, J.H.; Tabak, E.G.; Dasen, J.S.; Kintner, C.R.; Jessell, T.M. Origin and Segmental Diversity of Spinal Inhibitory Interneurons. Neuron 2018, 97, 341–355.e3.
- Benito-Gonzalez, A.; Alvarez, F.J. Renshaw Cells and Ia Inhibitory Interneurons Are Generated at Different Times from P1 Progenitors and Differentiate Shortly After Exiting the Cell Cycle. J. Neurosci. 2012, 32, 1156–1170.
- Doe, C.Q. Temporal Patterning in the Drosophila CNS. Annu. Rev. Cell Dev. Biol. 2017, 33, 219–240.
- Buckley, D.M.; Burroughs-Garcia, J.; Kriks, S.; Lewandoski, M.; Waters, S.T. Gbx1 and Gbx2 Are Essential for Normal Pat-terning and Development of Interneurons and Motor Neurons in the Embryonic Spinal Cord. J. Dev. Biol. 2020, 8, 9.
- Boeri, J.; Le Corronc, H.; Lejeune, F.-X.; Le Bras, B.; Mouffle, C.; Angelim, M.K.S.; Mangin, J.-M.; Branchereau, P.; Legendre, P.; Czarnecki, A. Persistent Sodium Current Drives Excitability of Immature Renshaw Cells in Early Embryonic Spinal Networks. J. Neurosci. 2018, 38, 7667–7682.
- Hoang, P.; Chalif, J.I.; Bikoff, J.B.; Jessell, T.M.; Mentis, G.Z.; Wichterle, H. Subtype Diversification and Synaptic Specificity of Stem Cell-Derived Spinal Interneurons. Neuron 2018, 100, 135–149.e7.
- Allan, D.W.; Thor, S. Transcriptional Selectors, Masters, and Combinatorial Codes: Regulatory Principles of Neural SubtypeSpecification. Wiley Interdiscip. Rev. Dev. Biol. 2015, 4, 505–528.
- Blacklaws, J.; Deska-Gauthier, D.; Jones, C.T.; Petracca, Y.L.; Liu, M.; Zhang, H.; Fawcett, J.P.; Glover, J.C.; Lanuza, G.M.; Zhang, Y. Sim1is Required for the Migration and Axonal Projections of V3 Interneurons in the Developing Mouse Spinal Cord. Dev. Neurobiol. 2015, 75, 1003–1017.
- Zhang, Y.; Narayan, S.; Geiman, E.; Lanuza, G.; Velasquez, T.; Shanks, B.; Akay, T.; Dyck, J.; Pearson, K.; Gosgnach, S.; et al. V3 Spinal Neurons Establish a Robust and Balanced Locomotor Rhythm During Walking. Neuron 2008, 60, 84–96.
- Danner, S.M.; Zhang, H.; Shevtsova, N.; Borowska-Fielding, J.; Deska-Gauthier, D.; Rybak, I.A.; Zhang, Y. Spinal V3 In-ter-neurons and Left–Right Coordination in Mammalian Locomotion. Front. Cell. Neurosci. 2019, 13, 516.
- Deska-Gauthier, D.; Borowska-Fielding, J.; Jones, C.T.; Zhang, Y. The Temporal Neurogenesis Patterning of Spinal p3–V3 Interneurons into Divergent Subpopulation Assemblies. J. Neurosci. 2020, 40, 1440–1452.
- Müller, T.; Brohmann, H.; Pierani, A.; Heppenstall, P.A.; Lewin, G.; Jessell, T.M.; Birchmeier, C. The Homeodomain Factor Lbx1 Distinguishes Two Major Programs of Neuronal Differentiation in the Dorsal Spinal Cord. Neuron 2002, 34, 551–562.
- John, A.; Wildner, H.; Britsch, S. The Homeodomain Transcription Factor Gbx1 Identifies a Subpopulation of Late-Born GABAergic Interneurons in the Developing Dorsal Spinal Cord. Dev. Dyn. 2005, 234, 767–771.
- Lai, H.C.; Seal, R.P.; Johnson, J.E. Making Sense Out of Spinal Cord Somatosensory Development. Development 2016, 143, 3434–3448.
- Buckley, D.M.; Burroughs-Garcia, J.; Lewandoski, M.; Waters, S.T. Characterization of the Gbx1−/− Mouse Mutant: A Re-quirement for Gbx1 in Normal Locomotion and Sensorimotor Circuit Development. PLoS ONE 2013, 8, e56214.
- Meziane, H.; Fraulob, V.; Riet, F.; Krężel, W.; Selloum, M.; Geffarth, M.; Acampora, D.; Herault, Y.; Simeone, A.; Brand, M.; et al. The Homeodomain factorGbx1is Required for Locomotion and Cell Specification in the Dorsal Spinal Cord. PeerJ 2013, 1, e142.
- Petracca, Y.L.; Sartoretti, M.M.; Di Bella, D.J.; Marin-Burgin, A.; Carcagno, A.L.; Schinder, A.F.; Lanuza, G.M. The Late and Dual Origin of Cerebrospinal Fluid-Contacting Neurons in the Mouse Spinal Cord. Development 2016, 143, 880–891.
- Böhm, U.L.; Prendergast, A.; Djenoune, L.; Figueiredo, S.N.; Gomez, J.; Stokes, C.; Kaiser, S.; Suster, M.; Kawakami, K.; Charpentier, M.; et al. CSF-Contacting Neurons Regulate Locomotion by Relaying Mechanical Stimuli to Spinal Circuits. Nat. Commun. 2016, 7, 10866.
- Fidelin, K.; Djenoune, L.; Stokes, C.; Prendergast, A.; Gomez, J.; Baradel, A.; del Bene, F.; Wyart, C. State-Dependent Modu-lation of Locomotion by GABAergic Spinal Sensory Neurons. Curr. Biol. 2015, 25, 3035–3047.
- Hubbard, J.; Böhm, U.L.; Prendergast, A.; Tseng, P.-E.B.; Newman, M.; Stokes, C.; Wyart, C. Intraspinal Sensory Neurons Provide Powerful Inhibition to Motor Circuits Ensuring Postural Control During Locomotion. Curr. Biol. 2016, 26, 2841–2853.
More
Information
Subjects:
Neurosciences
Contributor
MDPI registered users' name will be linked to their SciProfiles pages. To register with us, please refer to https://encyclopedia.pub/register
:
View Times:
806
Revisions:
2 times
(View History)
Update Date:
17 Aug 2021
Notice
You are not a member of the advisory board for this topic. If you want to update advisory board member profile, please contact office@encyclopedia.pub.
OK
Confirm
Only members of the Encyclopedia advisory board for this topic are allowed to note entries. Would you like to become an advisory board member of the Encyclopedia?
Yes
No
${ textCharacter }/${ maxCharacter }
Submit
Cancel
Back
Comments
${ item }
|
More
No more~
There is no comment~
${ textCharacter }/${ maxCharacter }
Submit
Cancel
${ selectedItem.replyTextCharacter }/${ selectedItem.replyMaxCharacter }
Submit
Cancel
Confirm
Are you sure to Delete?
Yes
No




