
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Marco Santin | + 2527 word(s) | 2527 | 2021-07-29 11:46:06 | | | |
| 2 | Beatrix Zheng | + 694 word(s) | 3221 | 2021-08-08 04:39:56 | | |
Video Upload Options
Plants continuously rely on light as an energy source and as the driver of many processes in their lifetimes. The ability to perceive different light radiations involves several photoreceptors, which in turn activate complex signalling cascades that ultimately lead to a rearrangement in plant metabolism as an adaptation strategy towards specific light conditions. This entry introduces the main classes of secondary metabolites and specifically focuses on the influence played by the different wavelengths on the content of these compounds in agricultural plants, because of their recognised roles as nutraceuticals.
1. Introduction
Plants rely on an uncountable number of secondary metabolites during their lifespans in order to perform several fundamental functions, such as attracting pollinators, mechanical support, protection from solar UV radiation, deterrents against pests, pathogens, and herbivores, interaction with other plants, and response to environmental stimuli/stresses [1]. Thanks to a network of photoreceptors and the following complex signalling routes, the different light wavelengths may impact the content of these metabolites by up- or downregulating specific sets of biosynthetic and regulatory genes.
2. Signal Transduction Pathways
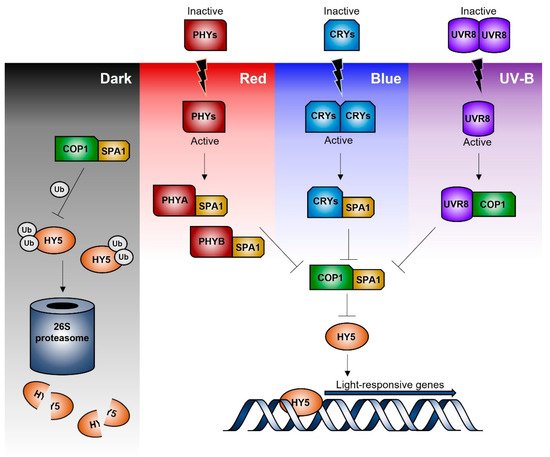
Independently from the light quality and kind of photoreceptor involved in light perception, the downstream event proceeds via a complex network of early signalling factors, central integrators, and final effectors. Please refer to some recent reviews [2][3][4][5] for a detailed summary of the current knowledge of the transcriptional network and mechanisms regulating the response to the different light spectral composition. Interestingly, CONSTITUTIVE PHOTOMORPHOGENIC 1 (COP1), which promotes the proteasome-mediates degradation of key factors involved in light signalling, is involved in the response to any light radiation, from UV to far-red wavelengths [6]. Similarly, the transcription factor ELONGATED HYOCOTYL 5 (HY5) has a central role as a final effector of all the light-dependent signalling routes, being able to bind to the promoters of about 4000 genes in Arabidopsis [6].
Figure 1 represents a simplified scheme of the signal transduction pathways leading to gene regulation in response to blue, red/far-red, and UV-B radiation. Briefly, under dark conditions, COP1/SPA (suppressor of Phytochrome A) ubiquitin ligase complex promotes the ubiquitination and degradation of HY5 via the 26S-proteasome pathway [4]. Upon light perception, the active blue- and red/far-red-photoreceptors (cryptochromes and phytochromes) interact with the COP1/SPA complex binding to SPA; thus, leading to COP1 disassembly and migration outside the nucleus. This prevents HY5 ubiquitination and subsequent degradation, so that HY5 may bind the promoter sequence of the light inducible target genes. Similarly, UVR8, after UVB-induced monomerization, can bind to COP1, leading to a functional disruption of the COP1/SPA complex and a consequent HY5 stabilization and functioning [7][8].
3. Plant Metabolism and Light
This review specifically focuses on the influence of the different light radiations, from red–far-red to UV-B, on the main classes of secondary metabolites, such as phenolic compounds, terpenoids, tocopherols, glucosinolates, and ascorbic acid in agricultural plant species, because of the recognised role that these compounds generally play as promoters of human wellness [9][10][11][12]. UV-C radiation was reported to modulate accumulation of health-promoting compounds in different plants and fruits of food interest, such as tomato fruit [13], bean seedlings [14] and peanut sprouts [15], this review exclusively discusses the effects of those wavelengths that reach the Earth’s surface, and to which plants have adapted fine-tuning perception mechanisms and consequent molecular and biochemical responses through evolution.
According to their chemical structures, terpenoids fulfil essential functions during plant life as, e.g., direct/indirect defensive compounds against biotic stressors, deterrent towards herbivores, photosynthetic pigments, signalling molecules mediating plant-plant, and plant–environment interaction [16][17][18][19].
Vitamin E has been widely studied due to its high antioxidant activity, especially preventing the oxidation of mono- and poly-unsaturated lipids. In addition, vitamin E compounds were shown to have hypolipidemic, antiatherogenic, antihypertensive, neuroprotective, anti-inflammatory, and many other beneficial effects for human health [20][21][22][23][24]. The main plant sources of tocopherols and tocotrienols are seeds (especially oilseeds) and nuts. In addition, they can be found in many plants and fruits, although their concentrations are limited due to their low lipid content [25].
Vitamin C, like the majority of the hydrosoluble vitamins, participates as a cofactor for many enzymes, e.g., members of the mono- and dioxygenases family [26], essentially contributing to the maintenance of the cell redox state, together with several other antioxidant molecules and enzymes. In plants, vitamin C is involved in many pathways and processes, e.g., the xanthophyll cycle, the flavonoids, and the glucosinolates pathways, and in the biosynthesis of plant hormones, such as ethylene, gibberellins, and abscisic acid [27][28][29][30][31]. Studies on the role of vitamin C role and its benefits in humans started when it was first noticed that vitamin C deficiency determined a potentially lethal disease called scurvy [32], negatively affecting the immune system, the collagenous architecture, and the regeneration process from wounds. Moreover, pharmacological effects of ascorbic acid against cancer and cardiovascular diseases were also observed [33][34]. The main dietary sources of vitamin C are fresh fruits and vegetables; therefore, their consumption has been widely encouraged by the main food and health organisations (e.g., the Food and Nutrition Board of the National Academy of Sciences, the European Food Safety Authority (EFSA), and the Food and Drug Administration (FDA)) throughout the years, and vitamin C deficiency symptoms have progressively reduced worldwide.
4. Red and Far-Red Light
Similarly, when bilberries ( Vaccinium myrtillus L.), plants were exposed to monochromatic red light (7.8 μmol m −2 s −1 ) during the berry ripening period, a significant increment of total anthocyanins occurred, due to the positive effect of this radiation on petudinins and delphinidins, while peonidins decreased, and cyanidins and malvidins were unaffected [35]. This finding underlines an interesting aspect of the light–phenolic interaction, i.e., the diversity of response to the same stimulus shown by different subclasses of molecules belonging to the same metabolic class. A similar phytochemical specificity of response was also observed in wheat ( Triticum aestivum L.) sprouts grown under a 16-h light/8-h dark photoperiod under white, red, or blue light, for up to 12 days. Specifically, red light, at the end of the growing period, did not lead to a significant increase in the content of total phenylpropanoids in comparison to white light, but modified their composition, inducing an increase in quercetin and a decrease in 4-hydroxybenzoic acid [36].
A 7-day exposure of red clover ( Trifolium pratense L.) sprouts to red-light (630 nm, 150 μmol m −2 s −1 , LEDs as lighting sources) induced a significant decrease in zeaxanthin concentration, while β-carotene and lutein were unaffected by the treatment [37]. A negative impact of this radiation on β-carotene concentration (−42.5%) was instead observed in Romaine green baby leaf lettuce (cv. Thumper) treated with supplemental red light (638 nm, 150 μmol m −2 s −1 , LEDs lighting sources) for 3 days [38]. These results differed from the ones by Li and Kubota (2009) [39], who found that 12 days of supplemental far-red light, but not red-light , irradiation determined a decrease in xanthophylls and β-carotene concentration in “Red Cross” baby leaf lettuce. Moreover, red light (380 μmol m −2 s −1 , LED lighting sources) was ineffective in modifying the carotenoid content of another lettuce cultivar (“Red Fire”) when compared to white light [40].
Tocopherols are reported to be influenced by red light. Exposure to supplemental red light (638 nm, 150 μmol m −2 s −1 , LEDs lighting sources, 3 days) was effective in significantly increasing α- and γ-tocopherols of Romaine green baby leaf lettuce (cv. Thumper) [38]. The same authors [41] also detected significant accumulation of α-tocopherol in basil microgreens grown under increased or sole red radiation (638 nm). However, increased red radiation lowered the α-tocopherol content of parsley microgreens, which was instead incremented when cultivation occurred with sole red lightning. It is therefore evident that, as observed for phenolic compounds, tocopherols are also influenced by red radiation in a species-depending way.
The limited data available on light quality influence on glucosinolates show that the effect is highly dependent on the wavebands and the plant species. Under red irradiation (730 and 640 nm) sinigrin content of kale was higher as compared to plants grown under blue light [42]. Similarly, when three Chinese cabbage varieties were exposed for 24 h to fluorescent light supplemented with red LEDs (625 nm), the content of total glucosinolates increased in the variety characterised by a low content of these metabolites, while the variety with high glucosinolates positively reacted to supplemental blue radiation [43]. These authors also reported that different set of genes involved in glucosinolates biosynthesis were upregulated by red or blue radiations in Chinese cabbage. To confirm the genotype dependence of the light influence on glucosinolates biosynthesis, Qian et al. [44] did not observe any variation in the content of these compounds in Chinese kale sprouts exposed to red LED light.
5. Green Light
| Species | Cultivar | Phenolics | AC | T | AA | TP | GSL | Ref. | ||
|---|---|---|---|---|---|---|---|---|---|---|
| Tot | Flav | Ant | ||||||||
| Lettuce (Lactuca sativa. L.) | Youmaicai | ↑ | [50] | |||||||
| Butterhead | ↑ | [51] | ||||||||
| Tea leaves (Camellia sinensis L.O. Kuntze) | Zhonghuang 3 | ↑ | ↑ | [52] | ||||||
| Tomato plants (Solanum lycopersicum L.) | Komeett’ | =/↑ | [53] | |||||||
| Basil (Ocimum basilicum L.) | Improved Genovese Compact | =/↓ | =/↓ | =/↓ | =/↓ | [54] | ||||
| Red Rubin | =/↓ | =/↓ | =/↓ | =/↓ | ||||||
6. Blue Light
| Species | Cultivar | Phenolics | AC | T | AA | TP | GSL | Ref. | ||
|---|---|---|---|---|---|---|---|---|---|---|
| Tot | Flav | Ant. | ||||||||
| Green leafy lettuce (Lactuca sativa. L.) | Thumper | ↓ | = | ↓/↑ | ↑ | [133] | ||||
| ↑ | [167] | |||||||||
| Grizzly | ↑ | [168] | ||||||||
| Red leafy lettuce (Lactuca sativa. L.) | Red Cross | = | ↑ | [131] | ||||||
| Red clover (Trifolium pratense L.) | ↑ | [144] | ||||||||
| Chinese cabbage (Brassica campestris L.) | ↑ | [169] | ||||||||
| Mustard (Brassica juncea L.) | Red Lion | ↑ | ↑ | [170] | ||||||
| Beet (Beta vulgaris L.) | Bulls Blood | ↑ | ↑ | |||||||
| Parsley (Petroselinum crispum Mill.) | Plain Leaved or French | ↑ | ↑ | |||||||
| Buckwheat (Fagopyrum esculentum) | Möench | ↑ | ↑ | [128] | ||||||
| Wheat (Triticum aestivum L.) | ↑/↓ | [145] | ||||||||
| Soybean (Glycine max L.) | Dongnong 690 | ↑ | ↓ | [166] | ||||||
| Bilberry fruit (Vaccinium myrtillus L.) | ↑ | [148] | ||||||||
| Apple fruit (Malus domestica Borkh.) | Mishima Fuji | ↑ | [171] | |||||||
| Jonathan | ↑ | |||||||||
| Strawberry (Fragaria × Ananassa) | ↑ | [172,173] | ||||||||
| Fengguang | ↑ | [174] | ||||||||
| Cowpea (Vigna unguiculata L. Walp.) | ↓/↑/= | [147] | ||||||||
| Tartary buckwheat (Fagopyrum tataricum Gaertn.) |
↓ | [146] | ||||||||
| Pak choi (Brassica rapa ssp. chinensis) | ↓/= | [150] | ||||||||
| Tomato fruit (Solanum lycopersicum L.) | Micro-Tom | ↑ | [175] | |||||||
| Satsuma mandarin fruit (Citrus unshiu Marc.) | ↓/↑/= | [152] | ||||||||
| Tea leaves (Camellia sinensis) | Jinxuan | ↑ | [149] | |||||||
| Basil (Ocimum basilicum L.) | Genovese | ↑ | [176] | |||||||
| Satsuma mandarin fruit (Citrus unshiu Marc.) | ↑ | [177] | ||||||||
| Valencia orange fruit (Citrus sinensis Osbeck) | ↑ | |||||||||
| Lisbon lemon fruit (Citrus limon Burm.f.) | ↑ | |||||||||
| Canola (Brassica napus L.) | ↑/= | [178] | ||||||||
| Mustard (Brassica juncea L.) | ↓ | [179] | ||||||||
7. UV-A Radiation
| Species | Cultivar | Phenolics | AC | T | AA | TP | GSL | Ref. | ||
|---|---|---|---|---|---|---|---|---|---|---|
| Tot | Flav | Ant | ||||||||
| Pak-choi (Brassica rapa ssp. chinensis var. communis) | Red leaf cv. | ↑/= | ↑ | ↑ | ↑ | ↑/= | ↑/↓ | ↑ | ↑ | [55][56] |
| Green leaf cv. | ↑/= | ↑/= | ↑ | ↑ | = | ↓ | ↑ | |||
| Turnip (Brassica rapa subsp. rapa) | Tsuda | ↑ | [57] | |||||||
| Broccoli (Brassica oleracea L., var. italica) | Waltham 29 | =/↓ | =/↓ | [58] | ||||||
| Monopoly | =/↓ | = | [59] | |||||||
| Broccoli (Brassica oleracea L., var. gemmifera DC) | ↑/↓ | ↑/= | [60] | |||||||
| Lettuce (Lactuca sativa. L.) | Yanzhi | = | ↑ | ↑ | ↑ | ↓ | ↑ | [61] | ||
| Red butter | ↑ | ↑ | ↑ | = | ↓ | ↑ | ||||
| Klee | ↑/= | ↑/= | ↑ | ↑ | [62] | |||||
| Red leaf cvs. | = | [63] | ||||||||
| Green leaf cvs. | = | |||||||||
| Hongyeom | ↑/= | ↑/= | ↑/= | [64] | ||||||
| Tomato plant (Solanum lycopersicum L.) | Oxheart | ↓ | = | = | = | [65] | ||||
| Cherry | = | ↓ | ↓ | ↓/= | ||||||
| Roma | = | = | ↓ | ↑/= | ||||||
| MicroTom | ↑ | [66] | ||||||||
| Tomato fruit (Solanum lycopersicum L.) | Budenovka | ↑ | ↑ | ↑/= | [67] | |||||
| Bull Heart | ↑ | ↑ | ↑/= | |||||||
| Gina | ↑ | ↑ | ↑/= | |||||||
| Micro-Tom | ↑ | [66] | ||||||||
| Sowthistle (Ixeris dentata Nakai) | ↑/= | ↑/= | ↑/= | [68] | ||||||
| Grape berry (Vitis vinifera L.) | Cabernet Sauvignon | ↑ | [69] | |||||||
| Blueberry (Vaccinium corymbosum L.) | Duke | ↓ | = | [70] | ||||||
| Peach fruit (Prunus persica L. Batsch) | Hujingmilu | ↑ | [71] | |||||||
| Yulu | = | |||||||||
| Basil (Ocimum basilicum L.) | Genovese | ↑/= | ↑ | ↓ | ↑/↓ | [56][72][73][74][75] | ||||
| Beet (Beta vulgaris L.) | Bulls Blood | ↑/↓ | ↑ | [56] | ||||||
| Rice (Oryza sativa L.) | Kanchana | ↑ | ↑ | [76] | ||||||
| Mattatriveni | ↓/= | |||||||||
| Harsha | ↑/= | |||||||||
| Broccoli (Brassica oleracea L. var. italica) | Waltham 29 | = | ↑ | [77] | ||||||
| Wheat (Triticum aestivum L.) | Sumai188 | ↑ | [78] | |||||||
| Mung bean (Vigna radiata) | ↑/↓ | ↑ | ↑ | [79] | ||||||
| Peppermint (Mentha piperita L.) | Rubescens | ↑ | ↑/↓ | [80] | ||||||
8. UV-B Radiation
| Species | Cultivar | Phenolics | AC | T | AA | TP | GSL | Ref. | ||
|---|---|---|---|---|---|---|---|---|---|---|
| Tot | Flav | Ant | ||||||||
| Basil (Ocimum basilicum L.) | Genovese | ↑/= | ↑/= | [72][73][74][75] | ||||||
| Cinnamon | ↑/↓ | ↑/=/↓ | [81] | |||||||
| Rice (Oryza sativa L.) | Kanchana | ↑ | ↑ | [76] | ||||||
| Mattatriveni | ↓/= | |||||||||
| Harsha | ↑/= | |||||||||
| Broccoli (Brassica oleracea L. var. italica) | Waltham 29 | = | ↑/= | ↑ | [58][77][82] | |||||
| Broccoli (Brassica oleracea var. gemmifera DC) | = | [60] | ||||||||
| Wheat (Triticum aestivum L.) | Sumai188 | ↑ | [78] | |||||||
| Mung bean (Vigna radiata) | ↑/↓ | ↑ | ↑ | [79] | ||||||
| Peppermint (Mentha piperita L.) | Rubescens | ↑ | ↑/↓ | [80] | ||||||
| Lettuce (Lactuca sativa. L.) | Red leaf cvs. | ↑ | ↑ | ↑ | ↓ | [63][83] | ||||
| Green leaf cvs. | ↑ | ↑ | ↑ | |||||||
| Peach fruit (Prunus persica L.) | Suncrest | ↑/=/↓ | = | ↓/↑ | =/↓ | [84][85] | ||||
| Big Top | ↑/=/↓ | ↓/↑ | ↑ | |||||||
| Babygold 7 | ↓ | ↓/↑ | = | |||||||
| Fairtime | ↓/↑ | ↓/↑ | ↓/↑ | ↓ | [86][87] | |||||
| Yulu | ↑ | [71] | ||||||||
| Hujingmilu | ↑ | |||||||||
| Tomato fruit (Solanum lycopersicum L.) | Money Maker | ↑ | ↑ | [88][89] | ||||||
| Zhenfen 202 | ↓ | [90] | ||||||||
| Bell pepper fruit (Capsicum annum L.) | Angus | ↑ | [91] | |||||||
| Green lime fruit (Citrus latifolia Tan.) | ↑ | [92] | ||||||||
| Spinach (Spinacia oleracea L.) | Meridian | ↑ | [93] | |||||||
| Maize (Zea mays L.) | ↓ | [94] | ||||||||
| Cucumber (Cucumis sativus L.) | Long green | ↓ | [95] | |||||||
| Apple fruit (Malus domestica Borkh.) | Aroma | ↑ | [96] | |||||||
9. Conclusions
References
- Ashraf, M.A.; Iqbal, M.; Rasheed, R.; Hussain, I.; Riaz, M.; Arif, M.S. Environmental Stress and Secondary Metabolites in Plants: An Overview. In Plant Metabolites and Regulation under Environmental Stress; Elsevier: Amsterdam, The Netherlands, 2018; pp. 153–167. ISBN 9780128126905.
- Teixeira, R.T. Distinct Responses to Light in Plants. Plants 2020, 9, 894.
- Pierik, R.; Ballaré, C.L. Control of Plant Growth and Defense by Photoreceptors: From Mechanisms to Opportunities in Agriculture. Mol. Plant 2021, 14, 61–76.
- Han, X.; Huang, X.; Deng, X.W. The Photomorphogenic Central Repressor COP1: Conservation and Functional Diversification during Evolution. Plant Commun. 2020, 1, 1–13.
- Jing, Y.; Lin, R. Transcriptional Regulatory Network of the Light Signaling Pathways. New Phytol. 2020, 227, 683–697.
- Bhatnagar, A.; Singh, S.; Khurana, J.P.; Burman, N. HY5-COP1: The Central Module of Light Signaling Pathway. J. Plant Biochem. Biotechnol. 2020, 29, 590–610.
- Huang, X.; Yang, P.; Ouyang, X.; Chen, L.; Deng, X.W. Photoactivated UVR8-COP1 Module Determines Photomorphogenic UV-B Signaling Output in Arabidopsis. PLoS Genet. 2014, 10.
- Ulm, R.; Jenkins, G.I. Q&A: How Do Plants Sense and Respond to UV-B Radiation? BMC Biol. 2015, 13, 45.
- Amengual, J. Bioactive Properties of Carotenoids in Human Health. Nutrients 2019, 11, 2388.
- Fraga, C.G.; Croft, K.D.; Kennedy, D.O.; Tomás-Barberán, F.A. The Effects of Polyphenols and Other Bioactives on Human Health. Food Funct. 2019, 10, 514–528.
- Miekus, N.; Marszałek, K.; Podlacha, M.; Iqbal, A.; Puchalski, C.; Swiergiel, A.H. Health Benefits of Plant-Derived Sulfur Compounds, Glucosinolates, and Organosulfur Compounds. Molecules 2020, 25, 3804.
- Szewczyk, K.; Chojnacka, A.; Górnicka, M. Tocopherols and Tocotrienols—Bioactive Dietary Compounds; What Is Certain, What Is Doubt? Int. J. Mol. Sci. 2021, 22, 6222.
- Liu, L.H.; Zabaras, D.; Bennett, L.E.; Aguas, P.; Woonton, B.W. Effects of UV-C, Red Light and Sun Light on the Carotenoid Content and Physical Qualities of Tomatoes during Post-Harvest Storage. Food Chem. 2009, 115, 495–500.
- Hernandez-Aguilar, C.; Dominguez-Pacheco, A.; Tenango, M.P.; Valderrama-Bravo, C.; Hernández, M.S.; Cruz-Orea, A.; Ordonez-Miranda, J. Characterization of Bean Seeds, Germination, and Phenolic Compounds of Seedlings by UV-C Radiation. J. Plant Growth Regul. 2021, 40, 642–655.
- Zhu, T.; Yang, J.; Zhang, D.; Cai, Q.; Zhou, D.; Tu, S.; Liu, Q.; Tu, K. Effects of White LED Light and UV-C Radiation on Stilbene Biosynthesis and Phytochemicals Accumulation Identified by UHPLC-MS/MS during Peanut (Arachis Hypogaea L.) Germination. J. Agric. Food Chem. 2020, 68, 5900–5909.
- Abdallah, I.I.; Quax, W.J. A Glimpse into the Biosynthesis of Terpenoids. Kne Life Sci. 2017, 3, 81.
- Abbas, F.; Ke, Y.; Yu, R.; Yue, Y.; Amanullah, S.; Jahangir, M.M.; Fan, Y. Volatile Terpenoids: Multiple Functions, Biosynthesis, Modulation and Manipulation by Genetic Engineering. Planta 2017, 246, 803–816.
- Dudareva, N.; Klempien, A.; Muhlemann, J.K.; Kaplan, I. Biosynthesis, Function and Metabolic Engineering of Plant Volatile Organic Compounds. New Phytol. 2013, 198, 16–32.
- Yu, F.; Utsumi, R. Diversity, Regulation, and Genetic Manipulation of Plant Mono- and Sesquiterpenoid Biosynthesis. Cell. Mol. Life Sci. 2009, 66, 3043–3052.
- Mocchegiani, E.; Costarelli, L.; Giacconi, R.; Malavolta, M.; Basso, A.; Piacenza, F.; Ostan, R.; Cevenini, E.; Gonos, E.S.; Franceschi, C.; et al. Vitamin E-Gene Interactions in Aging and Inflammatory Age-Related Diseases: Implications for Treatment. A Systematic Review. Ageing Res. Rev. 2014, 14, 81–101.
- Rashid Khan, M.; Ahsan, H.; Siddiqui, S.; Siddiqui, W.A. Tocotrienols Have a Nephroprotective Action against Lipid-Induced Chronic Renal Dysfunction in Rats. Ren. Fail. 2015, 37, 136–143.
- Kirmizis, D.; Chatzidimitriou, D. Vascular Health and Risk Management Antiatherogenic Effects of Vitamin e: The Search for the Holy Grail. Vasc. Health Risk Manag. 2009, 5, 767–774.
- Minhajuddin, M.; Beg, Z.H.; Iqbal, J. Hypolipidemic and Antioxidant Properties of Tocotrienol Rich Fraction Isolated from Rice Bran Oil in Experimentally Induced Hyperlipidemic Rats. Food Chem. Toxicol. 2005, 43, 747–753.
- Kizhakekuttu, T.J.; Widlansky, M.E. Natural Antioxidants and Hypertension: Promise and Challenges. Cardiovasc. Ther. 2010, 28, 20–32.
- García-Closas, R.; Berenguer, A.; Tormo, M.J.; Sánchez, M.J.; Quirós, J.R.; Navarro, C.; Arnaud, R.; Dorronsoro, M.; Chirlaque, M.D.; Barricarte, A.; et al. Dietary Sources of Vitamin C, Vitamin E and Specific Carotenoids in Spain. Br. J. Nutr. 2004, 91, 1005–1011.
- Fenech, M.; Amaya, I.; Valpuesta, V.; Botella, M.A. Vitamin C Content in Fruits: Biosynthesis and Regulation. Front. Plant Sci. 2019, 9.
- Bilska, K.; Wojciechowska, N.; Alipour, S.; Kalemba, E.M. Ascorbic Acid—the Little-Known Antioxidant in Woody Plants. Antioxidants 2019, 8, 645.
- Page, M.; Sultana, N.; Paszkiewicz, K.; Florance, H.; Smirnoff, N. The Influence of Ascorbate on Anthocyanin Accumulation during High Light Acclimation in Arabidopsis Thaliana: Further Evidence for Redox Control of Anthocyanin Synthesis. PlantCell Environ. 2012, 35, 388–404.
- Müller-Moulé, P.; Conklin, P.L.; Niyogi, K.K. Ascorbate Deficiency Can Limit Violaxanthin De-Epoxidase Activity in Vivo. Plant Physiol. 2002, 128, 970–977.
- Chen, S.-Y.; Chien, C.-T.; Chung, J.-D.; Yang, Y.-S.; Kuo, S.-R. Dormancy-Break and Germination in Seeds of Prunus Campanulata (Rosaceae): Role of Covering Layers and Changes in Concentration of Abscisic Acid and Gibberellins. Seed Sci. Res. 2007, 17, 21–32.
- De Tullio, M.C.; Arrigoni, O. The Ascorbic Acid System in Seeds: To Protect and to Serve. Seed Sci. Res. 2003, 13, 249–260.
- Sauberlich, H.E. A history of scurvy and vitamin C. In Vitamin C in Health and Disease; Marcel Dekker Inc.: New York, NY, USA, 1997; pp. 1–24. ISBN 0824793137.
- Ngo, B.; van Riper, J.M.; Cantley, L.C.; Yun, J. Targeting Cancer Vulnerabilities with High-Dose Vitamin C. Nat. Rev. Cancer 2019, 19, 271–282.
- Aune, D.; Keum, N.N.; Giovannucci, E.; Fadnes, L.T.; Boffetta, P.; Greenwood, D.C.; Tonstad, S.; Vatten, L.J.; Riboli, E.; Norat, T. Dietary Intake and Blood Concentrations of Antioxidants and the Risk of Cardiovascular Disease, Total Cancer, and All-Cause Mortality: A Systematic Review and Dose-Response Meta-Analysis of Prospective Studies. Am. J. Clin. Nutr. 2018, 108, 1069–1091.
- Zoratti, L.; Sarala, M.; Carvalho, E.; Karppinen, K.; Martens, S.; Giongo, L.; Häggman, H.; Jaakola, L. Monochromatic Light Increases Anthocyanin Content during Fruit Development in Bilberry. BMC Plant Biol. 2014, 14.
- Cuong, D.M.; Ha, T.W.; Park, C.H.; Kim, N.S.; Yeo, H.J.; Chun, S.W.; Kim, C.; Park, S.U. Effects of LED Lights on Expression of Genes Involved in Phenylpropanoid Biosynthesis and Accumulation of Phenylpropanoids in Wheat Sprout. Agronomy 2019, 9, 307.
- Gałazka-Czarnecka, I.; Korzeniewska, E.; Czarnecki, A.; Kiełbasa, P.; Drózdz, T. Modelling of Carotenoids Content in Red Clover Sprouts Using Light of Different Wavelength and Pulsed Electric Field. Appl. Sci. 2020, 10, 4143.
- Samuoliene, G.; Brazaityte, A.; Sirtautas, R.; Viršile, A.; Sakalauskaite, J.; Sakalauskiene, S.; Duchovskis, P. LED Illumination Affects Bioactive Compounds in Romaine Baby Leaf Lettuce. J. Sci. Food Agric. 2013, 93, 3286–3291.
- Li, Q.; Kubota, C. Effects of Supplemental Light Quality on Growth and Phytochemicals of Baby Leaf Lettuce. Environ. Exp. Bot. 2009, 67, 59–64.
- Ohashi-Kaneko, K.; Takase, M.; Kon, N.; Fujiwara, K.; Kurata, K. Effect of Light Quality on Growth and Vegetable Quality in Leaf Lettuce, Spinach and Komatsuna. Environ. Control Biol. 2007, 45, 189–198.
- Samuoliene, G.; Brazaityte, A.; Viršile, A.; Jankauskiene, J.; Sakalauskiene, S.; Duchovskis, P. Red Light-Dose or Wavelength-Dependent Photoresponse of Antioxidants in Herb Microgreens. PLoS ONE 2016, 11.
- Frisch, T.; Motawia, M.S.; Olsen, C.E.; Agerbirk, N.; Møller, B.L.; Bjarnholt, N. Diversified Glucosinolate Metabolism: Biosynthesis of Hydrogen Cyanide and of the Hydroxynitrile Glucoside Alliarinoside in Relation to Sinigrin Metabolism in Alliaria Petiolata. Front. Plant Sci. 2015, 6.
- Moon, J.; Jeong, M.J.; Lee, S.I.; Lee, J.G.; Hwang, H.; Yu, J.; Kim, Y.R.; Park, S.W.; Kim, J.A. Effect of LED Mixed Light Conditions on the Glucosinolate Pathway in Brassica Rapa. J. Plant Biotechnol. 2015, 42, 245–256.
- Qian, H.; Liu, T.; Deng, M.; Miao, H.; Cai, C.; Shen, W.; Wang, Q. Effects of Light Quality on Main Health-Promoting Compounds and Antioxidant Capacity of Chinese Kale Sprouts. Food Chem. 2016, 196, 1232–1238.
- Terashima, I.; Fujita, T.; Inoue, T.; Chow, W.S.; Oguchi, R. Green Light Drives Leaf Photosynthesis More Efficiently than Red Light in Strong White Light: Revisiting the Enigmatic Question of Why Leaves Are Green. Plant Cell Physiol. 2009, 50, 684–697.
- Bouly, J.P.; Schleicher, E.; Dionisio-Sese, M.; Vandenbussche, F.; van der Straeten, D.; Bakrim, N.; Meier, S.; Batschauer, A.; Galland, P.; Bittl, R.; et al. Cryptochrome Blue Light Photoreceptors Are Activated through Interconversion of Flavin Redox States. J. Biol. Chem. 2007, 282, 9383–9391.
- Smith, H.L.; Mcausland, L.; Murchie, E.H. Don’t Ignore the Green Light: Exploring Diverse Roles in Plant Processes. J. Exp. Bot. 2017, 68, 2099–2110.
- Battle, M.W.; Jones, M.A. Cryptochromes Integrate Green Light Signals into the Circadian System. Plant Cell Environ. 2020, 43, 16–27.
- Schenkels, L.; Saeys, W.; Lauwers, A.; de Proft, M.P. Green Light Induces Shade Avoidance to Alter Plant Morphology and Increases Biomass Production in Ocimum Basilicum L. Sci. Hortic. 2020, 261.
- Liu, H.; Fu, Y.; Wang, M.; Liu, H. Green Light Enhances Growth, Photosynthetic Pigments and CO2 Assimilation Efficiency of Lettuce as Revealed by ‘Knock out’ of the 480–560 Nm Spectral Waveband. Photosynthetica 2017, 55, 144–152.
- Bian, Z.; Yang, Q.; Li, T.; Cheng, R.; Barnett, Y.; Lu, C. Study of the Beneficial Effects of Green Light on Lettuce Grown under Short-Term Continuous Red and Blue Light-Emitting Diodes. Physiol. Plant. 2018, 164, 226–240.
- Zheng, C.; Ma, J.Q.; Ma, C.L.; Shen, S.Y.; Liu, Y.F.; Chen, L. Regulation of Growth and Flavonoid Formation of Tea Plants (Camellia Sinensis) by Blue and Green Light. J. Agric. Food Chem. 2019, 67, 2408–2419.
- Kaiser, E.; Weerheim, K.; Schipper, R.; Dieleman, J.A. Partial Replacement of Red and Blue by Green Light Increases Biomass and Yield in Tomato. Sci. Hortic. 2019, 249, 271–279.
- Dou, H.; Niu, G.; Gu, M. Photosynthesis, Morphology, Yield, and Phytochemical Accumulation in Basil Plants Influenced by Substituting Green Light for Partial Red and/or Blue Light. HortScience 2019, 54, 1769–1776.
- Mao, P.; Duan, F.; Zheng, Y.; Yang, Q. Blue and UV-A Light Wavelengths Positively Affected Accumulation Profiles of Healthy Compounds in Pak-Choi. J. Sci. Food Agric. 2021, 101, 1676–1684.
- Brazaityte, A.; Viršile, A.; Jankauskiene, J.; Sakalauskiene, S.; Samuoliene, G.; Sirtautas, R.; Novičkovas, A.; Dabašinskas, L.; Miliauskiene, J.; Vaštakaite, V.; et al. Effect of Supplemental UV-A Irradiation in Solid-State Lighting on the Growth and Phytochemical Content of Microgreens. Int. Agrophysics 2015, 29, 13–22.
- Wang, Y.; Zhou, B.; Sun, M.; Li, Y.; Kawabata, S. UV-A Light Induces Anthocyanin Biosynthesis in a Manner Distinct from Synergistic Blue + UV-B Light and UV-A/Blue Light Responses in Different Parts of the Hypocotyls in Turnip Seedlings. Plant Cell Physiol. 2012, 53, 1470–1480.
- Moreira-Rodríguez, M.; Nair, V.; Benavides, J.; Cisneros-Zevallos, L.; Jacobo-Velázquez, D.A. UVA, UVB Light Doses and Harvesting Time Differentially Tailor Glucosinolate and Phenolic Profiles in Broccoli Sprouts. Molecules 2017, 22, 1065.
- Rechner, O.; Neugart, S.; Schreiner, M.; Wu, S.; Poehling, H.M. Different Narrow-Band Light Ranges Alter Plant Secondary Metabolism and Plant Defense Response to Aphids. J. Chem. Ecol. 2016, 42, 989–1003.
- Acharya, J.; Rechner, O.; Neugart, S.; Schreiner, M.; Poehling, H.M. Effects of Light-Emitting Diode Treatments on Brevicoryne Brassicae Performance Mediated by Secondary Metabolites in Brussels Sprouts. J. Plant Dis. Prot. 2016, 123, 321–330.
- He, R.; Zhang, Y.; Song, S.; Su, W.; Hao, Y.; Liu, H. UV-A and FR Irradiation Improves Growth and Nutritional Properties of Lettuce Grown in an Artificial Light Plant Factory. Food Chem. 2021, 345.
- Chen, Y.; Li, T.; Yang, Q.; Zhang, Y.; Zou, J.; Bian, Z.; Wen, X. UVA Radiation Is Beneficial for Yield and Quality of Indoor Cultivated Lettuce. Front. Plant Sci. 2019, 10.
- Caldwell, C.R.; Britz, S.J. Effect of Supplemental Ultraviolet Radiation on the Carotenoid and Chlorophyll Composition of Green House-Grown Leaf Lettuce (Lactuca Sativa L.) Cultivars. J. Food Compos. Anal. 2006, 19, 637–644.
- Lee, M.J.; Son, J.E.; Oh, M.M. Growth and Phenolic Compounds of Lactuca Sativa L. Grown in a Closed-Type Plant Production System with UV-A, -B, or -C Lamp. J. Sci. Food Agric. 2014, 94, 197–204.
- Mariz-Ponte, N.; Mendes, R.J.; Sario, S.; Melo, P.; Santos, C. Moderate UV-A Supplementation Benefits Tomato Seed and Seedling Invigoration: A Contribution to the Use of UV in Seed Technology. Sci. Hortic. 2018, 235, 357–366.
- Guo, J.; Wang, M.H. Ultraviolet A-Specific Induction of Anthocyanin Biosynthesis and PAL Expression in Tomato (Solanum Lycopersicum L.). Plant Growth Regul. 2010, 62, 1–8.
- Dyshlyuk, L.; Babich, O.; Prosekov, A.; Ivanova, S.; Pavsky, V.; Chaplygina, T. The Effect of Postharvest Ultraviolet Irradiation on the Content of Antioxidant Compounds and the Activity of Antioxidant Enzymes in Tomato. Heliyon 2020, 6.
- Lee, M.J.; Son, J.E.; Oh, M.M. Growth and Phenolic Content of Sowthistle Grown in a Closed-Type Plant Production System with a UV-A or UV-B Lamp. Hortic. Environ. Biotechnol. 2013, 54, 492–500.
- Zhang, Z.Z.; Che, X.N.; Pan, Q.H.; Li, X.X.; Duan, C.Q. Transcriptional Activation of Flavan-3-Ols Biosynthesis in Grape Berries by UV Irradiation Depending on Developmental Stage. Plant Sci. 2013, 208, 64–74.
- Nguyen, C.T.T.; Kim, J.; Yoo, K.S.; Lim, S.; Lee, E.J. Effect of Prestorage UV-A, -B, and -C Radiation on Fruit Quality and Anthocyanin of “Duke” Blueberries during Cold Storage. J. Agric. Food Chem. 2014, 62, 12144–12151.
- Zhao, Y.; Dong, W.; Wang, K.; Zhang, B.; Allan, A.C.; Lin-Wang, K.; Chen, K.; Xu, C. Differential Sensitivity of Fruit Pigmentation to Ultraviolet Light between Two Peach Cultivars. Front. Plant Sci. 2017, 8.
- Mosadegh, H.; Trivellini, A.; Ferrante, A.; Lucchesini, M.; Vernieri, P.; Mensuali, A. Applications of UV-B Lighting to Enhance Phenolic Accumulation of Sweet Basil. Sci. Hortic. 2018, 229, 107–116.
- Nascimento, L.B.D.S.; Brunetti, C.; Agati, G.; Iacono, C.L.; Detti, C.; Giordani, E.; Ferrini, F.; Gori, A. Short-Term Pre-Harvest Uv-b Supplement Enhances the Polyphenol Content and Antioxidant Capacity of Ocimum Basilicum Leaves during Storage. Plants 2020, 9, 797.
- Ioannidis, D.; Bonner, L.; Johnson, C.B. UV-B Is Required for Normal Development of Oil Glands in Ocimum Basilicum L. (Sweet Basil). Ann. Bot. 2002, 90, 453–460.
- Chang, X.; Alderson, P.G.; Wright, C.J. Enhanced UV-B Radiation Alters Basil (Ocimum Basilicum L.) Growth and Stimulates the Synthesis of Volatile Oils. J. Hortic. For. 2009, 1, 27–31.
- Faseela, P.; Puthur, J.T. The Imprints of the High Light and UV-B Stresses in Oryza Sativa L. ‘Kanchana’ Seedlings Are Differentially Modulated. J. Photochem. Photobiol. B Biol. 2018, 178, 551–559.
- Moreira-Rodríguez, M.; Nair, V.; Benavides, J.; Cisneros-Zevallos, L.; Jacobo-Velázquez, D.A. UVA, UVB Light, and Methyl Jasmonate, Alone or Combined, Redirect the Biosynthesis of Glucosinolates, Phenolics, Carotenoids, and Chlorophylls in Broccoli Sprouts. Int. J. Mol. Sci. 2017, 18, 2330.
- Chen, Z.; Ma, Y.; Weng, Y.; Yang, R.; Gu, Z.; Wang, P. Effects of UV-B Radiation on Phenolic Accumulation, Antioxidant Activity and Physiological Changes in Wheat (Triticum Aestivum L.)Seedlings. Food Biosci. 2019, 30.
- Wang, H.; Gui, M.; Tian, X.; Xin, X.; Wang, T.; Li, J. Effects of UV-B on Vitamin C, Phenolics, Flavonoids and Their Related Enzyme Activities in Mung Bean Sprouts (Vigna Radiata). Int. J. Food Sci. Technol. 2017, 52, 827–833.
- Dolzhenko, Y.; Bertea, C.M.; Occhipinti, A.; Bossi, S.; Maffei, M.E. UV-B Modulates the Interplay between Terpenoids and Flavonoids in Peppermint (Mentha × piperita L.). J. Photochem. Photobiol. B Biol. 2010, 100, 67–75.
- Sakalauskaite, J.; Viskelis, P.; Dambrauskiene, E.; Sakalauskiene, S.; Samuoliene, G.; Brazaityte, A.; Duchovskis, P.; Urbonavičiene, D. The Effects of Different UV-B Radiation Intensities on Morphological and Biochemical Characteristics in Ocimum Basilicum L. J. Sci. Food Agric. 2013, 93, 1266–1271.
- Mewis, I.; Schreiner, M.; Nguyen, C.N.; Krumbein, A.; Ulrichs, C.; Lohse, M.; Zrenner, R. UV-B Irradiation Changes Specifically the Secondary Metabolite Profile in Broccoli Sprouts: Induced Signaling Overlaps with Defense Response to Biotic Stressors. Plant Cell Physiol. 2012, 53, 1546–1560.
- Assumpção, C.F.; Assis, R.Q.; Hermes Poletto, V.S.; Castagna, A.; Ranieri, A.; Neugart, S.; Flôres, S.H.; de Oliveira Rios, A. Application of Supplemental UV-B Radiation in Pre-Harvest to Enhance Health-Promoting Compounds Accumulation in Green and Red Lettuce. J. Food Process. Preserv. 2019, 43.
- Scattino, C.; Castagna, A.; Neugart, S.; Chan, H.M.; Schreiner, M.; Crisosto, C.H.; Tonutti, P.; Ranieri, A. Post-Harvest UV-B Irradiation Induces Changes of Phenol Contents and Corresponding Biosynthetic Gene Expression in Peaches and Nectarines. Food Chem. 2014, 163, 51–60.
- Sgherri, C.; Scattino, C.; Pinzino, C.; Tonutti, P.; Ranieri, A.M. Ultraviolet-B Radiation Applied to Detached Peach Fruit: A Study of Free Radical Generation by EPR Spin Trapping. Plant Physiol. Biochem. 2015, 96, 124–131.
- Santin, M.; Lucini, L.; Castagna, A.; Rocchetti, G.; Hauser, M.T.; Ranieri, A. Comparative “Phenol-Omics” and Gene Expression Analyses in Peach (Prunus Persica) Skin in Response to Different Postharvest UV-B Treatments. Plant Physiol. Biochem. 2019, 135, 511–519.
- Santin, M.; Lucini, L.; Castagna, A.; Chiodelli, G.; Hauser, M.-T.; Ranieri, A. Post-Harvest UV-B Radiation Modulates Metabolite Profile in Peach Fruit. Postharvest Biol. Technol. 2018, 139, 127–134.
- Castagna, A.; Dall’Asta, C.; Chiavaro, E.; Galaverna, G.; Ranieri, A. Effect of Post-Harvest UV-B Irradiation on Polyphenol Profile and Antioxidant Activity in Flesh and Peel of Tomato Fruits. Food Bioprocess Technol. 2014, 7, 2241–2250.
- Castagna, A.; Chiavaro, E.; Dall’Asta, C.; Rinaldi, M.; Galaverna, G.; Ranieri, A. Effect of Postharvest UV-B Irradiation on Nutraceutical Quality and Physical Properties of Tomato Fruits. Food Chem. 2013, 137, 151–158.
- Liu, C.; Han, X.; Cai, L.; Lu, X.; Ying, T.; Jiang, Z. Postharvest UV-B Irradiation Maintains Sensory Qualities and Enhances Antioxidant Capacity in Tomato Fruit during Storage. Postharvest Biol. Technol. 2011, 59, 232–237.
- Martínez-Zamora, L.; Castillejo, N.; Artés-Hernández, F. Postharvest UV-B and Photoperiod with Blue + Red LEDs as Strategies to Stimulate Carotenogenesis in Bell Peppers. Appl. Sci. 2021, 11, 3736.
- Srilaong, V.; Aiamla-or, S.; Soontornwat, A.; Shigyo, M.; Yamauchi, N. UV-B Irradiation Retards Chlorophyll Degradation in Lime (Citrus Latifolia Tan.) Fruit. Postharvest Biol. Technol. 2011, 59, 110–112.
- DeLong, J.M.; Steffen, K.L. Photosynthetic Function, Lipid Peroxidation, and-Tocopherol Content in Spinach Leaves during Exposure to UV-B Radiation. Can. J. Plant Sci. 1997, 77, 453–459.
- Carletti, P.; Masi, A.; Wonisch, A.; Grill, D.; Tausz, M.; Ferretti, M. Changes in Antioxidant and Pigment Pool Dimensions in UV-B Irradiated Maize Seedlings. Environ. Exp. Bot. 2003, 50, 149–157.
- Jain, K.; Kataria, S.; Guruprasad, K.N. Changes in Antioxidant Defenses of Cucumber Cotyledons in Response to UV-B and to the Free Radical Generating Compound AAPH. Plant Sci. 2003, 165, 551–557.
- Hagen, S.F.; Borge, G.I.A.; Bengtsson, G.B.; Bilger, W.; Berge, A.; Haffner, K.; Solhaug, K.A. Phenolic Contents and Other Health and Sensory Related Properties of Apple Fruit (Malus Domestica Borkh., Cv. Aroma): Effect of Postharvest UV-B Irradiation. Postharvest Biol. Technol. 2007, 45, 1–10.




