Your browser does not fully support modern features. Please upgrade for a smoother experience.

Submitted Successfully!
Thank you for your contribution! You can also upload a video entry or images related to this topic.
For video creation, please contact our Academic Video Service.
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Mohamad Goldust | + 2546 word(s) | 2546 | 2021-08-04 03:37:05 | | | |
| 2 | Peter Tang | Meta information modification | 2546 | 2021-08-05 03:54:45 | | |
Video Upload Options
We provide professional Academic Video Service to translate complex research into visually appealing presentations. Would you like to try it?
Cite
If you have any further questions, please contact Encyclopedia Editorial Office.
Goldust, M. Chronic Kidney Disease-Associated Pruritus. Encyclopedia. Available online: https://encyclopedia.pub/entry/12791 (accessed on 07 February 2026).
Goldust M. Chronic Kidney Disease-Associated Pruritus. Encyclopedia. Available at: https://encyclopedia.pub/entry/12791. Accessed February 07, 2026.
Goldust, Mohamad. "Chronic Kidney Disease-Associated Pruritus" Encyclopedia, https://encyclopedia.pub/entry/12791 (accessed February 07, 2026).
Goldust, M. (2021, August 04). Chronic Kidney Disease-Associated Pruritus. In Encyclopedia. https://encyclopedia.pub/entry/12791
Goldust, Mohamad. "Chronic Kidney Disease-Associated Pruritus." Encyclopedia. Web. 04 August, 2021.
Copy Citation
Pruritus is a distressing condition associated with end-stage renal disease (ESRD), advanced chronic kidney disease (CKD), as well as maintenance dialysis and adversely affects the quality of life (QOL) of these patients.
chronic kidney disease
pruritus
end-stage renal disease
1. Introduction
Pruritus is a distressing comorbidity found commonly in patients with end-stage renal disease (ESRD), chronic kidney disease (CKD), and those on dialysis.It adversely affects the quality of life (QOL) of these patients. The pruritus associated with CKD has been termed “uremic pruritus”; however, since there is no direct relationship between pruritus and uremia, the term “CKD-associated pruritus” (CKD-aP) is preferred [1]. The lack of specific clinical features defining the disease and consensus guidelines on diagnosis and management has lead to under-diagnosis and poor patient management. In a large international dialysis outcome and practice pattern study (DOPPS), conducted from 1996 to 2015, comprising of 51,062 patients from 21 countries, medical directors in 69% of the facilities under-diagnosed pruritus. Almost 17% of patients having itchy skin did not mention this symptom [2].
2. Epidemiology
The prevalence of CKD-aP has been very variable in different studies. It has been reported to range from 20% to as high as 90% [2][3][4]. The prevalence has been reported to vary significantly in different countries and also within a country at different centers [5]. However, the prevalence has been observed to decline due to the availability of better dialysis facilities, different dialysis modalities, access, and adequacy. The prevalence also depends on the mode of dialysis, i.e., hemodialysis (HD) or peritoneal dialysis (PD). A large international dialysis outcome and practice pattern study (DOPPS) was conducted on hemodialysis (HD) patients from 12 countries [4]. It was completed in two phases, DOPSS I (1996–2001) with 17,034 patients and DOPPS II (2002–2004) with 12,839 patients. 45% of patients had moderate to severe pruritus in DOPPS I and 42% in DOPPS II. In DOPPS phase V (2012–2015), it was observed that the number of patients with pruritus declined between 1996 and 2015 from 28% to 18% [2]. Another large dialysis outcome and practice pattern study (DOPPS) was conducted in Japan. It was completed in three phases, JDOPSS I (1999–2001; 65 facilities, 2757 patients), JDOPPS II (2002–2004; 60 facilities, 2286 patients), and JDOPPS III (2005–2008; 62 facilities, 2326 patients). The prevalence of moderate to extreme pruritus was 44%, while those bothered by itching to some extent was 35% [5]. In a large American study, the prevalence of CKD-aP was 60% [6]. In a study on 382 patients in China, 51.7% of patients were reported to have CKD-aP, with a higher incidence in patients on HD than PD [7]. In a meta-analysis of cross-sectional studies, including 42 studies, the prevalence of CKD-aP in each study ranged between 18% and 97.8%, and the overall prevalence of CKD-aP was 55%. The pooled prevalence in males and females was similar (55%). The prevalence of pruritus in patients undergoing HD was similar to those undergoing PD (55% vs. 56%) [8]. The prevalence was found to depend on the method of dialysis and, thus, is different in HD and PD patients. In a cross-sectional study from Korea, the prevalence of mild pruritus was higher in PD patients (62.5%) than in HD patients (48.3%). There were 223 PD and 425 HD patients enrolled in the study. The VAS score in PD patients was found to be significantly higher than in HD patients [9]. However, in another study, the pruritus prevalence and VAS score was higher in HD (38.2%) than PD (28.6%). This significantly lower intensity of pruritus in PD patients was attributed to better preservation of renal function and less inflammation in patients on PD than HD [10]. Thus there is no consensus as to which dialysis method is better to prevent CKD-aP. In a few studies, the prevalence was determined in CKD patients who had not yet started dialysis. In a cross-sectional study on patients with stage 2–5 CKD, 18.9% had pruritus [11]. They found no relationship between pruritus and the stage of CKD. However, in another cross-sectional study of 200 CKD and ESRD patients, pruritus was seen in 36% of cases, and the authors found raising skin manifestations with the worsening of kidney disease [12]. In another study, 56% of 55 patients with stage 4 and 5 CKD reported pruritus [13]. A study on 5658 patients with CKD stage 3–5 (nondialysis) reported moderate to extreme pruritus in 24% of patients, across all stages. In these patients, other features observed were moderate-to-extreme dry skin (73%) and restless sleep for at least 3 days out of the week (48%), which was 19% and 26%, respectively, for patients without pruritus [14]. Pediatric patients on dialysis seldom encounter severe pruritus. In a systematic review involving 199 children at various German pediatric dialysis centers, only 9.1% children had pruritus, and even if present, was not severe [15]. In another multicenter study in Poland, 20.8% of the pediatric population with CKD experienced pruritus. It was noted that the children on dialysis had higher rates of pruritus (23.5%) compared to those on conservative supportive care without dialysis (18.4%). Children with pruritus were more commonly associated with xerosis (66.7%) than those without (50.9%) [16]. Some literature reviews on CKD-aP report that kidney transplant seems to relieve pruritus [17]. In a study on 49 patients with successful kidney transplantation, it was observed that when sufficient renal function was restored after transplantation, the uremic skin alterations disappeared [18].
3. Etiopathogenesis
The pathogenesis of CKD-aPhas not been completely understood. Many theories have been proposed in various studies to explain it. The most common of them are highlighted in Figure 1:
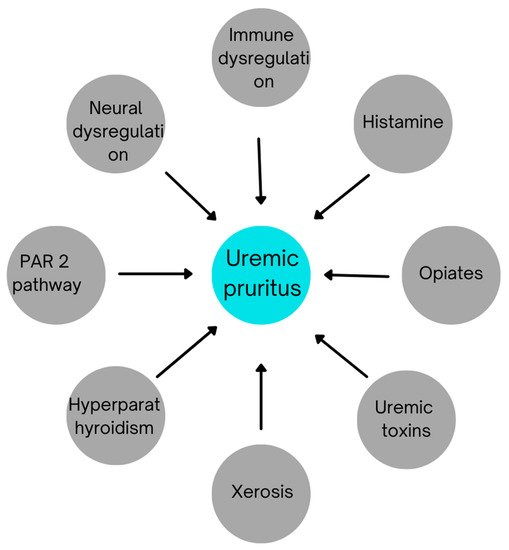
Figure 1. Etiological factors contributing to uremic pruritus.
4. Risk Factors Associated with CKD-aP
There are various risk factors associated with CKD-aP. Many studies have evaluated these factors and have reported variable results. In an international prospective cohort study on HD patients, the patients with pruritus were older, had higher median C-reactive protein, and had hepatitis B or C antibodies and low serum albumin. However, no association was observed with parathyroid hormone (PTH), Kt/V, serum calcium, phosphorus, calcium phosphorus product, and hemodiafiltration [2]. DOPPS data suggested that the likelihood of moderate to extreme pruritus was more in male patients or patients with a history of systemic diseases involving the heart, lungs, central nervous system, or liver. Higher serum phosphorus (>5.5 md/dL) or serum calcium (>10.2 mg/dL) levels, higher calcium phosphate (>80 mg2/dL2), lower serum or white blood cells >6700 /mL were also independent risk factors. The following factors were found to be negatively associated with pruritus: early ESRD (3 months or less), or prolonged ESRD >10 years, or serum ferritin concentration ≥400 ng/mL. It was also observed that an increase in dialysis dose improved pruritus [4]. In a Japanese DOPPS (JDOPPS) study, some additional risk factors were identified for CKD-aP, such as: hypertension, high PTH levels, smoked within the past year, and arteriovenous graft as the primary vascular access. They did not find any relation of CKD-aP with hepatitis B, serum creatinine, body mass index, dialyzer membrane type or flux of the dialyzer, percentage transferrin saturation, neutrophil count, and residual renal function [5]. In a study on 5658 patients with CKD stage 3–5 managed without dialysis, the following patient characteristics were associated with a higher prevalence of moderate-to-extreme pruritus: older age, female sex, history of lung disease and diabetes, higher serum phosphate, and lower hemoglobin levels. The prevalence was higher in patients with advanced stages of CKD [14]. Ozenet al. from Turkey found inflammatory mediators to be elevated in patients with CKD-aP. They found that WBC counts ≥ 6.7 × 103/μL increased the risk of pruritus by 1.73 times. Serum proinflammatory cytokines (IL-6, CRP, etc.) were found to be higher in CKD-aP patients [19]. In a prospective cohort study of 85 PD patients in Taiwan, 28.2% of patients had pruritus. Technical faults in PD lead to a higher intensity of pruritus and worse patient survival in patients undergoing dialysis. They also found that higher blood levels of iPTH, higher dietary protein intake, long duration of dialysis, weekly total Kt/V ≤ 1.88, and high-sensitivity CRP were independent determinants of higher Visual Analog Scale scores of pruritus intensity [20]. A cross-sectional study was conducted in Korea amongst 223 PD and 425 HD patients. They observed that peritoneal dialysis and BMI were independently responsible for pruritus. The severity of pruritus was negatively correlated to total weekly Kt/V in PD patients and serum albumin levels in HD patients. In PD patients, it was positively correlated with duration of dialysis, total cholesterol levels, and systolic BP. Amongst all these factors, serum albumin levels in HD and total weekly Kt/V in PD were independently found significant. However, clinically, there was no significant difference between PD and HD patients in the distribution of pruritus, frequency of pruritus-related sleep disturbances, pruritus intensity, and scratching activity [9]. Ko et al. followed up 111 patients of maintenance hemodialysis with CKD-aP for 4 years. They compared the parameters in patients with improved and unimproved pruritus at the end of follow-up. Lower levels of calcium and phosphorus products, low serum phosphorus, and ferritin were seen in patients with improved pruritus. In the study, the factors responsible for increased severity of pruritus over time were: female gender, low uric acid at baseline, use of low-flux dialyzer, high urea nitrogen, and β2- microglobulin, lowKt/V at baseline, low baseline VAS score, total bilirubin at baseline, high creatinine, preprandial glucose, and high calcium and phosphate product at baseline. It was also observed that a baseline Kt/V˂ 1.5 had severe pruritus, while the use of a high-flux dialyzer led to improvement in pruritus [21]. A study by Tinghai Hu et al. did not find serum calcium levels to be associated with pruritus. In a meta-analysis of cross-sectional studies, dialysis duration was negatively associated with the prevalence of CKD-associated pruritus [7]. To summarize all these findings, low levels of serum albumin and high levels of serum C-reactive protein, phosphorus, PTH, and calcium phosphorous products are positively correlated with higher intensity of pruritus. Coinfection with Hepatitis C and white blood cells >6700 /mL also increases the intensity of pruritus. Dialysis efficiency also affects pruritus, and low Kt/V increases pruritus. Older age, female sex, and presence of comorbidities such as lung disease and diabetes mellitus are also poor prognostic factors.
5. Clinical Features
The pruritus in patients with CKD presents with diverse clinical features. However, the pruritus is generally persistent and recurrent, bilaterally symmetrical, and worse at night. It is almost present daily and mostly presents over the trunk and limbs, with the back more commonly affected. Heat and dryness seem to aggravate pruritus. There are generally no primary skin lesions associated with pruritus, but secondary lesions due to scratching may be seen, such as: excoriation, linear crusts, ulcerations, impetigo, papules and prurigonodularis [6]. Amongst 103 HD patients in the United States, almost 84% of patients had daily or nearly daily pruritus. The itching was present over a wide area which was discontinuous but bilaterally symmetric and may migrate over time. The itching was persistent, it was severe at night as compared to daytime, and was aggravated by showering, dialysis, heat, stress, cold, and physical activity [22]. In DOPPS, it was observed that almost 50% of patients experienced itching throughout the day with no specific time; however, one-third of patients were most bothered at night. The presence of itching was not associated with the timing of dialysis; however, itching was reported to be worse during dialysis in 15% of patients, soon after dialysis in 9% of patients, or on nondialysis days in 14% of patients [2]. In a study by Tinghai Hu et al., the authors found that in most patients, pruritus was symmetric, while some had generalized pruritus. The pruritus was persistent and recurrent. The majority of patients suffered from daily itching, while the rest had itching occasionally in a week or a month. There were no typical skin lesions; however, xerosis was commonly present. Heat and dryness were found to aggravate itching [7]. Ozenet al. reported pruritus to be generalized in 35.3% of patients, with 50.4% experiencing moderate pruritus amongst the 249 HD patients studied. A total of 39.1% of patients had severe pruritus on the day after dialysis. The mean pruritus severity was 6.47 ± 1.56. If the patient had dry skin, they were 0.194 times more likely to have very severe pruritus [19]. In a cross-sectional study on 167 HD patients from Iran, pruritus was generalized in 70%of patients. The pruritus was limited to the trunk and limbs in 14.3% each and to the head and neck in 1.4%. In patients with pruritus, neuropathy was significant in 63.8% of patients [23]. Minato et al. studied 46 PD patients and found that pruritus affected almost all parts of the body, with the back being the most common site (70%). The other parts involved were lower limbs (67%), chest and abdomen (59%), upper limbs (28%), and head and neck (22%). The intensity was higher in the night times, as shown by the VAS score [24]. A study from Turkey on 181 HD patients reported itching before the hemodialysis session in 86.7% of patients, during hemodialysis in 72.9% of patients, and after hemodialysis in 49.7% of patients. The majority of patients had itching for 6–12 h, whereas only 1.1% had itching throughout the day. The itch was moderate in 40.3%, mild in 30.4%, severe in 28.2%, and unbearable in 2%. The course of itching was “a little bit better, but still present” in 38.7%, while in 5%, it was “getting worse”, as explained by the patients.In 65.2% of cases 6–10 anatomical regions were involved, most commonly over the back followed by upper arm, chest, and abdomen. Palms, soles and face/head had very minimalinvolvement [25].
6. Diagnosis
The clinical presentation of CKD-aP is highly variable; the onset of symptoms varies amongst the patients and the intensity of pruritus may range from mild to severe. The itching frequency and timings also differ amongst them. Pruritus in CKD-aP may not be associated with any primary skin lesions, while in some patients, it may be associated with secondary skin lesions such as crusts, papules, ulcerations, erosions, impetigo, and prurigonodularis. It is a diagnosis of exclusion in the absence of other systemic conditions causing itch, such as:liver disorders, hematologic abnormalities, comorbid skin conditions, infestations, etc. [26].
The criteria for diagnosis of uremic pruritus is [26]:
-
Pruritus appears shortly before the onset of dialysis, or at any time, without evidence of any other active disease that could explain the pruritus.
-
More than or equal to three episodes of itch during a period of 2 weeks, with the symptom appearing a few times a day, lasting at least few minutes, and troubling the patient.
-
The appearance of an itch in a regular pattern during a period of 6 months, but less frequently than listed above.
Since itching is a subjective phenomenon, it is typically accessed via patient-reported outcomes (PROs). Several PRO scales have been developed for the measurement of pruritus intensity, which can be either unidimensional (measures only severity of pruritus) or multidimensional (measures severity and other characteristics of pruritus, such as: duration, impact on activities of daily life, direction, degree, and location of pruritus). There are also few scales that focus predominantly on the quality of life [16]. A transformation equation has been suggested to correlate the data between the multidimensional (5-D itch scale) and unidimensional (numeric rating scale (NRS)) scales [27]. The commonly used scales have been listed in Table 1.
Table 1. Symptom scales for evaluation and treatment of uremic pruritus [16].
|
Measurement |
Tool |
|---|---|
|
Unidimensional |
|
|
Multidimensional |
|
7. Treatment
The patients with CKD-aP are mainly treated by nephrologists, primary care physicians, and dermatologists. In an international study, almost 50% of patients were treated by a nephrologist, 19% by a primary care physician, and only 24% were managed by a dermatologist [2]. Since there are many factors associated with CKD-aP, and these factors vary considerably amongst patients, no universal treatment can apply to all. The patients have to be properly evaluated, including proper history and examination, and the treatment planned accordingly.
References
- Patel, T.S.; Freedman, B.I.; Yosipovitch, G. An Update on Pruritus Associated With CKD. Am. J. Kidney Dis. 2007, 50, 11–20.
- Rayner, H.C.; Larkina, M.; Wang, M.; Graham-Brown, M.; Van Der Veer, S.N.; Ecder, T.; Hasegawa, T.; Kleophas, W.; Bieber, B.A.; Tentori, F.; et al. International Comparisons of Prevalence, Awareness, and Treatment of Pruritus in People on Hemodialysis. Clin. J. Am. Soc. Nephrol. 2017, 12, 2000–2007.
- Shirazian, S.; Aina, O.; Park, Y.; Chowdhury, N.; Leger, K.; Hou, L.; Miyawaki, N.; Mathur, V.S. Chronic kidney disease-associated pruritus: Impact on quality of life and current management challenges. Int. J. Nephrol. Renov. Dis. 2017, 10, 11–26.
- Pisoni, R.L.; Wikström, B.; Elder, S.J.; Akizawa, T.; Asano, Y.; Keen, M.L.; Saran, R.; Mendelssohn, D.C.; Young, E.W.; Port, F.K. Pruritus in haemodialysis patients: International results from the Dialysis Outcomes and Practice Patterns Study (DOPPS). Nephrol. Dial. Transplant. 2006, 21, 3495–3505.
- Kimata, N.; Fuller, D.S.; Saito, A.; Akizawa, T.; Fukuhara, S.; Pisoni, R.L.; Robinson, B.M.; Akiba, T. Pruritus in hemodialysis patients: Results from the Japanese Dialysis Outcomes and Practice Patterns Study (JDOPPS). Hemodial. Int. 2014, 18, 657–667.
- Sood, V.; Ramakrishnan, K.; Bond, T.C.; Claxton, A.; Kootsikas, M.; Agnese, W.; Sibbel, S. Clinical characteristics and outcomes of end-stage renal disease patients with self-reported pruritus symptoms. Int. J. Nephrol. Renov. Dis. 2013, 7, 1–12.
- Hu, T.; Wang, B.; Liao, X.; Wang, S. Clinical features and risk factors of pruritus in patients with chronic renal failure. Exp. Ther. Med. 2019, 18, 964–971.
- Hu, X.; Sang, Y.; Yang, M.; Chen, X.; Tang, W. Prevalence of chronic kidney disease-associated pruritus among adult dialysis patients. Medicine 2018, 97, e10633.
- Min, J.W.; Kim, S.H.; Kim, Y.O.; Jin, D.C.; Song, H.C.; Choi, E.J.; Kim, Y.L.; Kim, Y.S.; Kang, S.W.; Kim, N.H.; et al. Comparison of uremic pruritus between patients undergoing hemodialysis and peritoneal dialysis. Kidney Res. Clin. Pract. 2016, 35, 107–113.
- Wu, H.-Y.; Peng, Y.-S.; Chen, H.-Y.; Tsai, W.-C.; Yang, J.-Y.; Hsu, S.-P.; Pai, M.-F.; Lu, H.-M.; Chiang, J.-F.; Ko, M.-J.; et al. A Comparison of Uremic Pruritus in Patients Receiving Peritoneal Dialysis and Hemodialysis. Medicine 2016, 95, e2935.
- Stolic, R.; Trajković, G.; Perić, V.; Jovanovic, A.; Lazarević, T.; Subarić-Gorgieva, G. Parametres of clinical and biohumoral status of pruritus in patients on chronic haemodialysis. Med. Pregl. 2007, 60 (Suppl. S2), 101–103.
- Patel, T.; Ishiuji, Y.; Yosipovitch, G. Nocturnal itch: Why do we itch at night? Acta Derm. Venereol. 2007, 87, 295–298.
- Santoro, D.; Satta, E.; Messina, S.; Costantino, G.; Savica, V.; Bellinghieri, G. Pain in end-stage renal disease: A frequent and neglected clinical problem. Clin. Nephrol. 2013, 79 (Suppl. S1), S2–S11.
- Sukul, N.; Speyer, E.; Tu, C.; Bieber, B.A.; Li, Y.; Lopes, A.A.; Asahi, K.; Mariani, L.; Laville, M.; Rayner, H.C.; et al. Pruritus and patient reported outcomes in non-dialysis CKD. Clin. J. Am. Soc. Nephrol. 2019, 14, 673–681.
- Mettang, T.; Pauli-Magnus, C.; Alscher, D.M. Uraemic pruritus–new perspectives and insights from recent trials. Nephr. Dial. Transplant. 2002, 17, 1558–1563.
- Reszke, R.; Szepietowski, J.C. End-stage renal disease chronic itch and its management. Dermatol. Clin. 2018, 36, 277–292.
- Combs, S.A.; Teixeira, P.; Germain, M.J. Pruritus in kidney disease. Semin. Nephrol. 2015, 35, 383–391.
- Altmeyer, P.; Kachel, H.G.; Schäfer, G.; Fassbinder, W. Normalisierung der urämischen Hautveränderungen nach Nierentransplantation [Normalization of uremic skin changes following kidney transplantation]. Hautarzt 1986, 37, 217–221.
- Ozen, N.; Cinar, F.I.; Askin, D.; Mut, D. Uremic pruritus and associated factors in hemodialysis patients: A multi-center study. Kidney Res. Clin. Pr. 2018, 37, 138–147.
- Wu, H.Y.; Huang, J.W.; Tsai, W.C.; Peng, Y.S.; Chen, H.Y.; Yang, J.Y.; Hsu, S.P.; Pai, M.F.; Ko, M.J.; Hung, K.Y.; et al. Prognostic importance and determinants of uremic pruritus in patients receiving peritoneal dialysis: A prospective cohort study. PLoS ONE 2018, 13, e0203474.
- Ko, M.J.; Wu, H.Y.; Chen, H.Y.; Chiu, Y.L.; Hsu, J.P.; Pai, M.F.; YehYang, J.; Lai, C.F.; Lu, H.M.; Huang, S.C. Uremic pruritus, dialysis adequacy, and metabolic profiles in hemodialysis patients: A prospective 5-year cohort study. PLoS ONE. 2013, 8, e71404.
- Mathur, V.S.; Lindberg, J.; Germain, M.; Block, G.; Tumlin, J.; Smith, M.; Grewal, M.; McGuire, D. For the ITCH national registry investigators a longitudinal study of Uremic Pruritus in hemodialysis patients. Clin. J. Am. Soc. Nephrol. 2010, 5, 1410–1419.
- Akhyani, M.; Ganji, M.R.; Samadi, N.; Khamesan, B.; Daneshpazhooh, M. Pruritus in hemodialysis patients. BMC Dermatol. 2005, 5, 7.
- Minato, S.; Hirai, K.; Morino, J.; Kaneko, S.; Yanai, K.; Mutsuyoshi, Y.; Ishii, H.; Matsuyama, M.; Kitano, T.; Shindo, M.; et al. Factors associated with Uremic Pruritus in patients undergoing peritoneal dialysis. Int. J. Nephrol. Renov. Dis. 2020, 13, 1–9.
- Ersoy, N.A.; Akyar, I. Multidimensional pruritus assessment in hemodialysis patients. BMC Nephrol. 2019, 20, 42.
- Keithi-Reddy, S.R.; Patel, T.V.; Armstrong, A.W.; Singh, A.K. Uremic pruritus. Kidney Int. 2007, 72, 373–377.
- Lai, J.W.; Chen, H.C.; Chou, C.Y.; Yen, H.R.; Li, T.C.; Sun, M.F.; Chang, H.H.; Huang, C.C.; Tsai, F.J.; Tschen, J.; et al. Transformation of 5-D itch scale and numerical rating scale in chronic hemodialysis patients. BMC Nephrol. 2017, 18, 56.
More
Information
Subjects:
Dermatology
Contributor
MDPI registered users' name will be linked to their SciProfiles pages. To register with us, please refer to https://encyclopedia.pub/register
:
View Times:
807
Revisions:
2 times
(View History)
Update Date:
05 Aug 2021
Notice
You are not a member of the advisory board for this topic. If you want to update advisory board member profile, please contact office@encyclopedia.pub.
OK
Confirm
Only members of the Encyclopedia advisory board for this topic are allowed to note entries. Would you like to become an advisory board member of the Encyclopedia?
Yes
No
${ textCharacter }/${ maxCharacter }
Submit
Cancel
Back
Comments
${ item }
|
More
No more~
There is no comment~
${ textCharacter }/${ maxCharacter }
Submit
Cancel
${ selectedItem.replyTextCharacter }/${ selectedItem.replyMaxCharacter }
Submit
Cancel
Confirm
Are you sure to Delete?
Yes
No




