Rare earth elements (REEs) as defined by the International Union of Pure and Applied Chemistry (IUPAC) include metals characterized by similar properties, namely scandium (Sc), yttrium (Y) and all the lanthanides. The latter correspond to the chemical elements listed in the periodic table of Mendeleev that have an atomic number ranging from 57 for lanthanum (La) to 71 for lutetium (Lu). REEs are often subdivided into “light rare earths elements” (LREEs) and “heavy rare earths elements” (HREEs) according to their atomic numbers. Yttrium is oftentimes associated with HREEs due to chemical similarities, including ionic radii. In some cases, the elements from samarium to terbium are considered as the “middle rare earth elements” (MREEs).
1. Overview
Rare earth elements (REEs) are becoming more and more significant as they play crucial roles in many advanced technologies. Therefore, the development of optimized processes for their recovery, whether from primary resources or from secondary sources, has become necessary, including recovery from mine tailings, recycling of end-of-life products and urban and industrial waste. Ionic solvents, including ionic liquids (ILs) and deep-eutectic solvents (DESs), have attracted much attention since they represent an alternative to conventional processes for metal recovery. These systems are used as reactive agents in leaching and extraction processes.
2. Rare Earth Elements
Rare earth elements (REEs) as defined by the International Union of Pure and Applied Chemistry (IUPAC) include metals characterized by similar properties, namely scandium (Sc), yttrium (Y) and all the lanthanides. The latter correspond to the chemical elements listed in the periodic table of Mendeleev that have an atomic number ranging from 57 for lanthanum (La) to 71 for lutetium (Lu)
[1]. REEs are often subdivided into “light rare earths elements” (LREEs) and “heavy rare earths elements” (HREEs) according to their atomic numbers. Yttrium is oftentimes associated with HREEs due to chemical similarities, including ionic radii. In some cases, the elements from samarium to terbium are considered as the “middle rare earth elements” (MREEs)
[2].
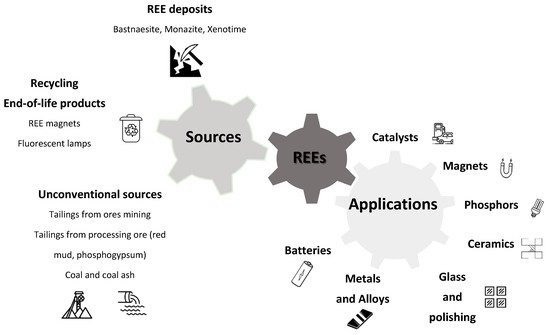
With the increasing uses of these elements, global demand is growing and their access is becoming crucial, leading to diversified sources of supply (Figure 1).
Figure 1. Use and sourcing of rare earth elements.
The properties of REEs are linked to their electronic configuration and in particular to the specificity of their electronic 4f sublayer, which allows numerous optical transitions and gives them particular magnetic and catalytic properties
[3]. REEs are mainly trivalent; however, different oxidation states can occur naturally for cerium and europium.
Due to these remarkable properties, REEs are used in many high-tech applications
[4][5], such as lasers
[6][7], permanent magnets
[8][9] energy storage
[10][11] and so on. REEs are therefore part of the so-called “technological” metals, the supply of which has become strategic, as it is threatened by the growth in world demand for these particular metals
[12][13]. REEs are also considered as “critical” elements by a number of institutions, such as the EU, which has identified these elements as being of high economic importance with a high risk associated with their supply. The latter includes geopolitical considerations, such as the quasi-monopoly over the production of these elements maintained by China.
REEs are commonly mined as co-products or as by-products of other materials. Primary sources of REEs include hard rock deposits of bastnaesite as well as alluvial deposits of monazite and xenotime
[14]. In addition to these ores, which contain the highest concentrations of REEs, other mineral resources can be considered, such as phosphate or apatites, which are admittedly poorer in REEs, but whose exploitation and processing in the future may be cost-effective
[15][16].
Due to the critical need for these materials, alternative sources need to be investigated, particularly in the context of the circular economy
[17][18]. Valorisation of secondary or unconventional sources becomes more and more relevant, both from economic and environmental perspectives
[19].
REE recovery from industrial landfilled stocks containing lower REE concentrations requires larger volumes. Among industrial wastes, bauxite residue (red mud) generated during the production of alumina from bauxite (the Bayer process) represents a promising source for REEs
[20][21]. It is mainly composed of the metallic oxides aluminium, iron and titanium, as well as up to 1000 ppm of REEs
[22][23].
Like bauxite residue, phosphogypsum, which is a waste generated by the fertilizer industry, has been identified as a significant source of REEs due to presence of these elements and the large volumes generated worldwide
[24][25].
Other important secondary sources that are challenging for REE recovery are acid mine drainages (AMDs)
[26] and wastewater sludges
[27]. AMD and mine water from operating and closed mining sites represent a potential secondary source for many elements of economic interest, including REEs
[28]. There is also significant potential for the recovery of these elements from coal and coal combustion by-products (coal fly ash) and the feasibility and implementation of industrial processes are under study
[29][30].
As REEs are also present in a large amount of technological equipment, recovery from recycling of specific wastes, and in particular from waste electrical and electronic equipment (WEEE), also called urban mines, represents an unconventional and alternative source with high potential
[18][31][32]. In this context, much research has recently been performed on end-of-life products containing high concentrations of REEs, such as magnets
[33][34] or fluorescent lamps
[35][36].
The processes currently used to recover rare earths from these natural or industrial ores (conventional and unconventional sources) consist in subjecting the ores, which have been previously crushed, to chemical treatments (with acidic or basic reagents) in order to obtain mineral concentrates. The chemical attack or leaching process is conventionally carried out by means of one or more acid reagents, including nitric acid, sulphuric acid, phosphoric acid, hydrochloric acid or a mixture of them (aqua regia). The so-called “pregnant leach solutions” are then subjected to an extraction/separation step using solvent extraction, ion exchange and/or precipitation
[37].
Among such separation techniques, solvent extraction or liquid–liquid extraction is the most commonly used process for the recovery of REEs from the different sources (primary and secondary mining, waste streams, end-of-life materials and so on). The solvent extraction process consists in bringing the aqueous phase constituted by the pregnant leach solution into contact with an organic phase comprising one or more extractants, in order to achieve an efficient and selective extraction of REEs. Although widely applied on an industrial scale, liquid–liquid extraction suffers from certain limitations, such as the use of large amounts of toxic solvents and/or flammable solvents, which are a potential threat to human health and the environment
[38]. For example, aliphatic diluents, such as isooctane, are classified as highly flammable liquids and vapours with safety concerns (they may be fatal if they are swallowed and enter the airways, very toxic to aquatic life, etc.), while
n-dodecane is classified as a combustible liquid and can be used as an alternative; however, it also presents health risks. Another major issue associated with the use of organic solvents in hydrometallurgy is their loss by evaporation. Besides the economic aspect, this leads to ecological concerns. Elimination or replacement of hazardous solvents in terms of toxicity and flammability, which has a clear impact on worker safety and environmental issues, is therefore one of the main challenges for metallurgical processes.
3. Description and Properties of Ionic Solvents
Due to their quasi non-flammability
[39] and non-volatility
[40][41], ionic solvents, including ionic liquids (ILs) and deep-eutectic solvents (DESs), are increasingly being used as alternative diluents or extractants to conventional volatile organic compounds in extraction processes
[42][43]. Recently, the term ‘‘ionometallurgy’’ was introduced by Abbott et al. for metallurgical processes using ionic solvents
[44]. One of the interesting features of using ionic solvents is that an electrochemical process for the dissolution and the purification of metallic species can be implemented thanks to their large electrochemical window
[45].
Ionic solvents can be considered as salts with melting points lower than 100 °C
[46][47] and modular physical and chemical properties. Two categories can be considered: the “conventional” ILs composed of a discrete cation and anion, and DES systems formed from an eutectic mixture of Lewis or Brønsted acids and bases, which can contain a diversity of ionic species (anionic and/or cationic)
[48][49][50].
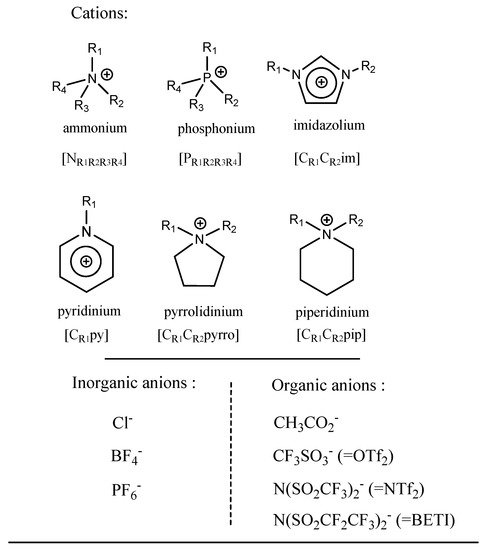
ILs exhibit a wide variety of chemical structures, but share some common characteristics, such as bulky organic cations (ammonium, phosphonium, imidazolium, pyridinium, pyrrolidinium, piperidinium, triazolium) combined with smaller inorganic (chloride, hexafluorophosphate, tetrafluoroborate, etc.) or organic (acetate, triflate, bis(trifluoromethyl)sulfonyl imide, etc.) anions (Figure 2).
Figure 2. Structures of common cations and anions used in ILs.
Appropriate combinations of the cation and anion allow adjustment of the physicochemical properties of the ILs (viscosity, density, hydrophilicity and solubility). The multiple possibilities make it possible to consider them as “designer solvents” that are attractive potential alternatives for organic molecular solvents
[51].
In conventional approaches applied for metal extraction, ILs are usually hydrophobic to avoid their miscibility in the aqueous phases. Their hydrophobicity is governed by the length of the alkyl chain carried by the cation, as well as by the shape, symmetry and the nature of the anion associated. Typically the applied organic cations present long carbon chains, and the anions are usually hydrophobic anions such as hexafluorophosphate [PF
6−] or bis(trifluoromethyl)sulfonyl imide [NTf
2−]. IL density and viscosity are essential parameters to take into account in the implementation of an extraction process, as they affect the transport properties, such as diffusion
[52]. Longer alkyl chain lengths result in lower density and higher viscosity while, for different anions, these parameters may evolve independently from their hydrophobicity
[53]. Viscosity behaviours have also been assigned to the molecular structure of ionic liquids and to the presence of hydrogen bonding and van der Waals interactions
[54]. In addition, for an identical cation, ILs based on a weak or non-coordinating anion have a lower viscosity than those composed of a complexing anion.
Eutectic mixtures are usually considered a new class of ILs and they gained attention because some of them can be prepared from natural sources, which tends to make them more biodegradable, biocompatible and sustainable
[55][56]. In this case, they are defined as natural DESs (NADESs)
[57][58]. DESs usually result from a mixture of compounds that interact through hydrogen bonding, which leads to charge delocalization resulting in a depression of the melting point of the mixture
[49][59][60][61][62].
A four-group classification has been proposed by Abbott et al. based on their constituents
[48][49].
Types I to III DESs can be defined by the general formula Cat+X−·Y−, where:
- -
-
Cat+ is usually a quaternary ammonium or phosphonium salt;
- -
-
X is the anionic moiety (generally a halide anion);
- -
-
Y is a metal chloride for type I, a metal chloride hydrate for type II and a hydrogen bond donor for type III.
Type IV DESs involve a metal chloride hydrate and hydrogen bond donor.
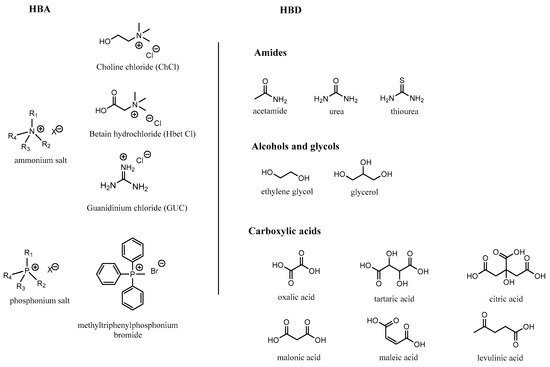
Most common DESs imply a hydrogen bond donor (HBD) and an ionic hydrogen bond acceptor (HBA), leading to type III DESs as defined by Abbott et al.
[48]. DESs are mainly hydrophilic mixtures as they are based on at least one ionic species and strong hydrogen bonding interactions. Most popular DESs are based on quaternary ammonium salts and choline chloride
[63] for their HBA part, and on urea, carboxylic acids, alcohols and glycols for their HBD. The most common structures of the HBA and HBD are illustrated in
Figure 3. As for the ILs, judicious choices for each component allow the control of parameters such as density, viscosity, acidity or basicity, hydrophobicity, polarity, volatility and the extractability properties of the DES.
Figure 3. Structures of some hydrogen bond acceptors (halide salts) and hydrogen bond donors (amines, alcohols, carboxylic acids) used in the formation of deep-eutectic solvents.
Among these properties, the acidic character of DESs, also called acidic deep-eutectic solvents (ADESs), has been highlighted for applications in dissolution, extraction and metal electrodeposition
[60]. The acidity of ADESs can be controlled with the acidity of the HBD and these DESs are often divided into two classes: Brønsted and Lewis DESs.
Also, since 2015, with the concept of hydrophobic deep-eutectic solvents (HDESs) proposed by van Osch et al.
[64], interest in the synthesis and application of HDESs is strongly increasing
[65][66][67]. It is assumed that the hydrophobic character depends on the hydrophobicity of the components forming the DES. HDESs are mainly type III, involving bulky quaternary ammonium or phosphonium halide ILs as HBAs and hydrophobic carboxylic acids as HBDs. This trend for hydrophobic eutectic mixtures was recently investigated for mixtures solely composed of non-ionic species
[68][69]. A fifth class of DES has therefore been proposed to complete the Abbott classification so that it includes such fully organic hydrophobic eutectic solvents involving strong interaction between a hydrogen bond donor and a hydrogen bond acceptor
[70]. These type V non-ionic DESs represent a relatively new class of eutectics which have strong potential for the recovery of REEs through solvent extraction.