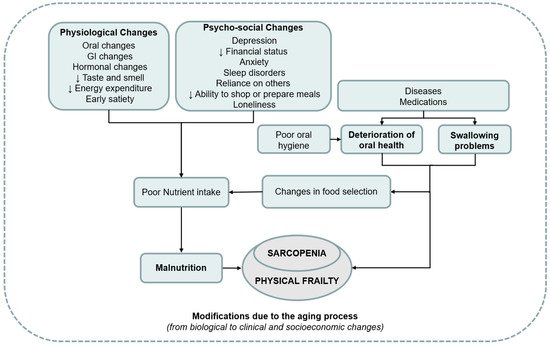
3. Sarcopenia and Oral Status
Sarcopenia, defined as the progressive and accelerated loss of muscle mass and function, is a major determinant of several adverse outcomes including frailty, disability, and mortality
[13][37]. Although sarcopenia is a condition commonly observed with the aging process, it can also occur earlier in life
[38]. Since 2016, sarcopenia has been recognized as an independent condition with an International Classification of Disease, 10th Revision, Clinical Modification (ICD-10-CM) Diagnosis Code
[39]. Recently, the European Working Group on Sarcopenia in Older People (EWGSOP)
[38] updated their consensus on definition and diagnosis (EWGSOP2). In this revised consensus, low muscle strength is considered a key characteristic of sarcopenia, and poor physical performance is identified as indicative of severe sarcopenia. Moreover, EWGSOP2 have recommended specific cut-off points to identify and characterize the sarcopenic condition, and provide an algorithm that can be used for case-finding.
Sarcopenia has a complex multifactorial pathogenesis, which involves lifestyle habits (i.e., malnutrition, physical inactivity), disease triggers, and age-dependent biological changes (i.e., chronic inflammation, mitochondrial abnormalities, loss of neuromuscular junctions, reduced satellite cell numbers, hormonal alterations)
[40][41]. Sarcopenia is a whole-body process, affecting not only lower extremities, but also muscles dedicated to breathing, mastication, and swallowing
[14][18][19][20][21][22]. In particular, swallowing is a complex mechanism involving several head and neck muscles simultaneously and in conjunction to coordinate the entire process
[42]. Several age-related changes, such as as reduction of tissue elasticity, changes of the head and neck anatomy, reduced oral and pharyngeal sensitivity, and impaired dental status, may contribute to different degrees to a subtle swallowing impairment, the so called “presbyphagia”. It is usually an asymptomatic condition in which swallowing function is preserved, but tends to slowly worsen as the aging process advances
[16][43]. Presbyphagia may increase the risk of dysphagia and aspiration in older people, especially during acute illnesses and other stressors
[44]. Moreover, reductions in muscle mass of the geniohyoid, pterygoid, masseter, tongue, and pharyngeal muscles have been documented in older individuals
[20][45][46][47]. Several authors also reported a decline in the strength of the swallowing muscles with aging or sarcopenia
[48]. Maximal tongue strength decreases with aging
[48][49][50][51], and there is some evidence that aging leads to a decreased jaw-opening force in older men. Several authors also reported an association between tongue strength and handgrip strength
[52][53]. A decrease in tongue strength has been associated with a decline of activities of daily living
[54], and a reduced tongue thickness has been noted in people with low body weight
[20].
Lip function is also important for feeding. In fact, poor lip muscle closure may cause leakage through the corners of the mouth
[55]. Additionally, decreased lip strength has been suggested to occur due to sarcopenia and to be related to difficulties in eating and drinking (i.e., dysphagia)
[49]. Lip force has been associated with hand grip strength and lip pendency has been associated with aging
[49][56].
Indeed, since it has been shown that skeletal muscle mass and strength decline may affect both swallowing and general muscle groups, a new condition, called “sarcopenic dysphagia” has been coined
[22][56][57]. Swallowing muscles are characterized by a high percentage of type II fibers, which are more easily affected by malnutrition and sarcopenia than type I muscle fibers
[22]. However, some cranial muscles, including the jaw-closers, are very different in fiber-type composition than other skeletal muscle groups (i.e., limbs or abdomen). For instance, the masseter muscle, which originates from the zygomatic arch, contains both type I and type II fibers, but shows a predominance of type I muscle fibers, which are more strongly affected by inactivity rather than aging
[58][59]. Given that the meal texture of older people frequently becomes softer, less power of tongue movement and of masseter muscle is required, which may result in decreased activity of these muscles.
Interestingly, poor oral health may predispose one to a chronic low-grade inflammatory state through periodontal disease, which is a well-known risk factor for frailty and sarcopenia
[25][60][61]. In fact, the detrimental effects of periodontitis are not confined solely to the oral cavity, but extend systemically, leading to metabolic alterations
[62], including insulin resistance
[63], diabetes
[63][64], arthritis
[65], and heart disease
[66]. Furthermore, alterations in mitochondrial function leading to oxidative stress through the production of reactive oxygen species (ROS) have also been reported to mediate both oral and systemic pathologies (i.e., sarcopenia)
[40][67][68][69]. Given their regulatory role as signaling molecules in autophagy, it has been speculated that elevated ROS production in periodontal disease could lead to autophagic alterations
[70]. Bullon et al.
[71] found high levels of mitochondrial-derived ROS, accompanied by mitochondrial dysfunction in peripheral blood mononuclear cells from patients with periodontitis. Moreover, oral gingiva seems to be highly responsive to the lipopolysaccharides (LPS), which are bacterial endotoxins prevalent in periodontal disease. In fact, gingival fibroblasts, which play an important role in remodeling periodontal soft tissues, may directly interact with LPS. In particular, LPS from
Porphyromonas gingivalis enhances the production of inflammatory cytokines
[72].
Porphyromonas gingivalis has been found to be responsible for high mitochondrial ROS and coenzyme Q10 levels, and for mitochondrial dysfunction, given its influence on the amount of respiratory chain complex I and III
[70][71]. Indeed, LPS-mediated mitochondrial dysfunction could explain the oxidative stress onset in patients with periodontitis. Furthermore, Hamalainen et al.
[29] reported an association between periodontitis and quicker declines in handgrip strength.
On the other hand, as discussed in the previous section, the variety of dental problems experienced by older people can lead to a decline in general health through poor nutrient intake, pain, and low quality of life
[25]. Poor oral status has been reported to affect 71% of patients in rehabilitation settings
[73] and 91% of people in acute-care hospitals
[74], and has been associated with malnutrition, dysphagia, and reduced activities of daily living
[17]. Hence, poor oral status may lead to sarcopenia through poor nutrient intake. Moreover, inflammation further contributes to malnutrition through various mechanisms, such as anorexia, decreased nutrient intake, altered metabolism (i.e., elevation of resting energy expenditure), and increased muscle catabolism
[75]. Chronic inflammation is a common underlying factor, not only in the etiology of sarcopenia, but also for frailty. In fact, sarcopenia and frailty are closely related and show a remarkable overlap especially in the physical function domain
[76][77][78]. The presence of oral problems, alone or in combination with sarcopenia, may thus represent the biological substratum of the disabling cascade experienced by many frail individuals.