1. Introduction
The World Health Organization (WHO) estimates that around 420,000 people die every year due to a foodborne illness. This has an economic impact of US$110 billionon the global economy. Furthermore, according to the WHO, approximately 18% of the infectious disease outbreaks are related to the water in Europe
[1], and this percentage may be higher in other continents. On the other hand, multidrug resistant (MDR) bacteria are a big concern not only in human health, but also in livestock industries. The global estimation of antimicrobial consumption (mg) per population correction unit (PCU) reported for cattle, chickens and pig is 45 mg/PCU, 148 mg/PCU and 172 mg/PCU, respectively, and a rise of antimicrobials in food animal production has been projected by 67% from 2010 to 2030, reaching 105,596 (±3605) tons of antimicrobials by 2030
[2]. In this sense, China consumes the largest quantity of antimicrobials, followed by the United States of America, Brazil, Germany and India
[2]. Moreover, transmission of MDR bacteria from animals to humans has been described
[3] and is subject of surveillance.
In the last decades, the use of bacteriophages has reappeared in Western countries as an alternative to chemicals treatments
[4]. Bacteriophages are recognized as the most abundant biological agents on Earth, due to their ubiquitous presence in the environment. Phages, for short, are able to lyse MDR bacteria and reduce the non-desirable effects produced by chemicals on food. According to their life cycle, they can be classified as virulent phages (lytic phages) or temperate phages (lysogenic phages)
[5]. Lytic phages use the genomic and biosynthetic machinery of the bacteria to produce their progeny, provoking the bacterial lysis and their consequent release. The phage-encoded endolysins are ultimately responsible to break down the bacterial peptidoglycan at the final stage of the cycle
[6]. Conversely, lysogenic phages are capable of incorporating their nucleic acid into the genome of the host cell or just remain like a plasmid into the host cell during multiple bacterial generations. Therefore, only lytic phages are usually used in bioremediation or phage therapy. Bacteria can become resistant to phages by modifying their receptors, turning them inaccessible or non-complementary to the phage receptor binding protein
[7]. Fortunately, these insensitive strains can be lysed using cocktails of phages instead of a single phage.
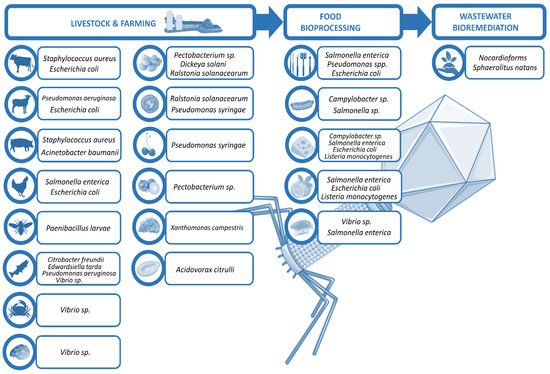
Phages in food industry can be applied at different stages
[8]: directly on animals or plants to eliminate the probability of bacterial infection and disease, in food production plants to prevent bacterial biofilm formation, or directly on food to preserve the product. This review excludes in vitro experiments and summarizes selected in vivo findings of phage use in non-human biocontrol, focusing on the treatment of live animals and plants that are relevant in the food industry, as well as the raw food products, and the biofilm control on surfaces during their processing and manufacturing, ending with the bioremediation of the wastewaters generated (
Figure 1).
Figure 1. Scheme of phage utilities in biocontrol.
3. Phage Therapy for Animals
Phage administration is an interesting alternative to antibiotics in animals. Many in vitro experiments against pathogenic bacteria infecting animals have been reported. Here we focus on recent in vivo studies to show the state of the art in this field (Table 2).
In farms, phage therapy has been studied to treat and prevent infections caused by
Salmonella enterica serovar Kentucky and
Escherichia coli in chickens. Two
S. enterica serovar Kentucky and three
E. coli O119 phages were able to reduce mortality from 30% in positive control groups up to 0% in treated chickens. Notably, the higher reduction of bacteria counts in cecum, heart and liver was obtained at day 23
[41].
Recently, a bacteriophage cocktail was used against
Pseudomonas aeruginosa that produces rhinosinusitis in sheep. A mix of four phages was able to reduce biofilm biomass on frontal sinus mucosa at concentrations of 10
8–10
10 PFU/mL with no safety concerns
[42].
Several murine mastitis models have showed that phage therapy could be also used against
Staphylococcus aureus in bovine mastitis caused by microbial infection
[43][44][45]. A previous study published in 2006 by Gill et al. analyzed the efficacy of a 5-day treatment consisting of phage K administered intramammary in lactating Holstein cows with subclinical mastitis caused by
S. aureus. Three out of 18 animals were cured (16.7%) compared to none out of 20 cows of the negative control group (0%)
[46]. Despite some success, the low efficacy could be explained by the data of Gill et al. showing that incubation of
S. aureus with whey and bovine serum resulted in inhibition of phage K lysis. Accordingly, they concluded that proteins could block sterically the phage K attachment to the bacteria, suggesting that
S. aureus could be more resistant to phages in vivo in mastitis infections than in vitro experiments
[47].
Infections caused by
E. coli O157:H7 and treatment with phage therapy in ruminants have been already reviewed
[48], revealing that further understanding of phage administration, effective multiplicity of infection (MOI) and correct analysis of results are necessary in cattle phage therapy
[49][50][51]. In sheep, no significant reductions of
E. coli O157:H7 were found compared to controls when a single phage was administered after oral
E. coli inoculation
[52][53]. However, a mix of two phages reduced more than 99% the presence of
E. coli in the lower intestinal tracts of treated animals
[54]. In addition, a cocktail of eight phages reduced significantly fecal
E. coli O157:H7, although not in the rumen, after 24 h post phage administration
[55].
In piglet studies, phages were able to kill methicillin-resistant
S. aureus (MRSA) in vitro but no reduction was observed in the nasal mucosa in vivo or ex vivo
[56]. This fact emphasizes the importance of considering other factors that may counteract phage efficacy in vivo, such as reduced adherence or increased clearance by the animal fluids. However, experiments conducted in growing pigs showed that dietary supplementation with a commercial cocktail of phages against
Salmonella enterica,
S. aureus,
E. coli and
Clostridium prefringens was more efficient than probiotics as growth promoters
[57], improving food digestibility, daily weight gain and gain per feed, among other parameters.
The presence of wounds is relatively common in swine. An hydrogel containing phages against
Acinetobacter baumanii was used to reduce wound infections in an ex vivo model of pig skin, and achieved a 90% reduction in bacterial counts after only 4 h of treatment
[58].
Another study showed that seven phages isolated from pig farms in the United Kingdom were able to lyse all 68
Salmonella strains tested, including MDR ones, offering a valuable alternative to antimicrobials to reduce infections and food poisoning
[59].
Another recent review
[60] summarized the known phages infecting
Paenibacillus larvae. This spore-forming bacterium attacks honeybee larvae causing the American foulbrood, which is the most widespread and destructive of the honeybee brood diseases, being able to destroy an entire colony in just three weeks. Importantly, all known bacteriophages against
P. larvae to date are lysogenic. Despite that, studies of phage therapy in vitro and in hives have shown higher survival rates of treated groups including prophylactic benefits. Lack of success in some cases was attributed to the lysogenic nature of the phages or their inability to access the gut.
In aquaculture, the common carp has been used as a model to demonstrate the effectiveness of phage therapy against
Citrobacter freundii, using a single phage, IME-JL8. This bacterium belongs to the normal flora of fishes; however, it has been associated to systemic infection in common carp and other diseases in diverse fishes. Administration of phages into the carp decreased pro-inflammatory cytokines and protected the fish from infection when phages were administered one hour after bacteria inoculation, but not after 24 h, indicating that timing is relevant in phage therapy
[61]. Similarly, no adverse inflammatory response was induced by the ETP-1 phage in zebrafish (
Danio rerio), and twelve days of exposition to ETP-1 was able to increase survival from 18% in the control group up to 68% after infection with
Edwardsiella tarda bacteria
[62]. Another example can be found in the North African catfish (
Clarias gariepinus). Ulcerative lesions caused by
P. aeruginosa in North African catfish were reduced seven fold compared with untreated control after 8–10 days of treatment with a single phage
[63]. In addition, treatment with two different phages at MOI of 100 reached 100% of survival in Vietnamese striped catfish (
Pangasianodon hypophthalmus) infected with
Aeromonas hydrophila, which produces hemorrhagic septicemia, compared to 13% of survival in the control group
[64].
Vibrio sp. produce mortality in bivalve larvae and bacteriophages could be used as biocontrol agents in oyster hatcheries. Two different approaches have been described to solve this problem. The first consists on direct phage treatment comprising two phages, which diminished mortality rates from 77.9% in the control group to 28.2% after just 24 h of incubation
[65]. However, the second approach focuses on decontaminating microalgae as vectors for
Vibrio sp. infection of larval cultures. Phage administration in microalgae resulted in significant reduction of
Vibrio sp. within 2 h, suggesting that feeding larvae with decontaminated microalgae could be a promising preventive method to avoid infection of bivalve larvae
[66]. Curiously, in 2019, a study using a heterologous expression vector was performed against
Vibrio parahaemolyticus. The yeast
Pichia pastoris X-33 expressed the phage endolysin Vplys60 from bacteriophage qdv001 and the enzyme was shown to inhibit biofilm formation and to reduce mortality rates for the crustacean
Artemia franciscana [67]. In other studies, a phage treatment with two phages against
Vibrio anguillarum infection was effective at 72 h in zebrafish larvae
[68], and a cocktail of three phages isolated from sewage showed host specificity against eight
Vibrio coralliilyticus strains and a
Vibrio tubiashii strain, obtaining a decrease of over 90% in
V. coralliilyticus compared to the untreated control
[69].
These studies reveal that current results are more promising in aquaculture than in farms. More studies are needed to clarify the real sanitary and economic potential of phage-based therapies in the food industry. It is possible that, as it happens in humans, better results could be obtained by mixing phages and antibiotics due to the synergistic effect.
Table 2. Summary of reviewed studies using phage therapy in animals.
| Animal |
Infection/Colonization |
Bacteria |
Phage Therapy |
Outcome |
References |
| Chicken |
Salmonellosis and colibacillosis |
S. enterica serovar Kentucky and Escherichia coli O119 |
Siphoviridae (107 PFU) against serovar
Kentucky and Podoviridae (10 PFU) against Escherichia coli orally |
Reduction of mortality from 30% to 0% in treated group |
[41] |
| Sheep |
Rhinosinusitis |
Pseudomonas aeruginosa |
Cocktail of 4 phages (Pa193, Pa204, Pa222, and Pa223) at 108–1010 PFU/mL |
Reduction of biofilm biomass on sinus mucosa |
[42] |
| Cow |
Subclinical mastitis |
Staphylococcus aureus |
Phage K (1011 PFU) intramammary infusions for 5 days |
3/18 cows were cured compared to 0/20 of control group |
[46] |
| Sheep |
Gut |
Escherichia coli O157:H7 |
Oral phage KH1 (1011 PFU) or DC22 (1013 PFU) |
No reduction of strain O157:H7 |
[52][53] |
| Sheep |
Gut |
Escherichia coli O157:H7 |
Cocktail of CEV1 (T4-like) and CEV2 (T5-like) orally |
Reduction >99% of Escherichia coli in the lower intestinal tract |
[54] |
| Sheep |
Gut |
Escherichia coli O157:H7 |
Cocktail of 8 phages orally |
Reduction of fecal Escherichia coli O157:H7, but not in the rumen, 24 h after phage administration |
[55] |
| Pig |
Nasal colonization |
MRSA V0608892/1 strain |
P68 (Podovirus) and K* 710 (Myovirus) in gel |
No reduction observed in the nasal mucosa |
[56] |
| Pig |
Prevention |
Salmonella enterica, Staphylococcus aureus, Escherichia coli and Clostridium prefringens |
Cocktail of phages orally |
Compared to probiotics, phages had better results as growth promoters, improving digestibility, daily weight gain and gain per feed |
[57] |
| Pig |
Ex vivo skin infection |
Acinetobacter baumannii |
IME-AB2 (Myoviridae) via gel |
Reduction of 90% of bacterial counts 4 h post-treatment |
[58] |
| Honeybee larvae |
American foulbrood |
Paenibacillus larvae |
Cocktail of phages 1, 5 and 9 |
Higher survival rates in hives of treated groups including prophylactic benefits |
[60][70] |
| Common carp |
Sepsis |
Citrobacter freundii |
IME-JL8 (Siphoviridae) |
Decreased pro-inflammatory cytokines and protection of fish from infection when phages were administered 1 h after bacteria, but not after 24 h |
[61] |
| Zebrafish |
Sepsis |
Edwardsiella tarda |
ETP-1 for 12 d |
Increment of survival from 18% to 68% |
[62] |
| North African catfish |
Ulcerative lesions |
Pseudomonas aeruginosa |
Single phage for 8–10 d |
7-fold reduction of ulcerative lesions |
[63] |
| Vietnamese striped catfish |
Hemorrhagic septicemia |
Aeromonas hydrophila |
Φ2 and Φ5 |
Increment of survival from 13% to 100% |
[64] |
| Bivalve larvae |
Infection |
Vibrio sp. |
Cocktail of Φ5, Φ6 and Φ7 |
Reduction of mortality from 77.9% to 28.2% |
[65] |
| Microalgae food of bivalve larvae |
Infection |
Vibrio harveyi |
Cocktail of Φ1, Φ2, Φ3 and Φ4 |
10 times reduction of bacteria after 2 h |
[66] |
| Zebrafish larvae |
Infection |
Vibrio anguillarum |
VA-1 phage |
Mortality rate after 72 post-infection was reduced from 17€ to 3%. |
[68] |
| Larval Pacific oysters |
Infection |
Vibrio coralliilyticus |
Cocktail of vB_VcorM-GR7B, vB_VcorM-GR11A, and vB_VcorM-GR28A |
Mortality reduction of >90% respect to the control group |
[69] |
4. Phage Therapy for Plants
Different pathogenic bacteria produce significant economic losses in plant production worldwide. This section focuses on recent advances in phage use against pathogens infecting economically relevant plants such as potatoes, tomatoes, cherries, onions, kohlrabies and melons.
Potatoes:
Pectobacterium atrosepticum is a pathogenic bacterium causing soft rot disease and blackleg disease. A cocktail of six phages infected 93% of tested strains and succeeded for biocontrol by decreasing disease incidence (61%) and severity (64%)
[71]. Another study treated a mixed infection caused by two different
P. atrosepticum strains with a cocktail of three bacteriophages and the results showed that the average weight of rotten tissue decreased significantly from 5.39 g in infected plants to 0.31 g in treated tubers
[72]. Semi-in planta potato bioassays showed that a cocktail of six phages were able to suppress the growth of a mix of
P. atrosepticum and
P. carotovorum subsp
. carotovorum against soft rot development
[73]. Curiously, another study described that phage Pc1 infects
P. carotovorum subsp.
carotovorum more efficiently when zinc is not present in the medium, suggesting that inorganic composition of soil is relevant when phage therapy is considered for biocontrol
[74]. On the contrary, a Tasmanian potato farm study showed the protective effect of beneficial streptomycetes in soil and pointed that in case of treating the pathogenic strains of
Streptomyces with phage therapy, a preliminary host range analysis should be performed since a deleterious effect against beneficial streptomycetes might produce opportunistic fungal infections
[75]. In a different studio, a cocktail of six phages was used to combat the potato pathogen
Dickeya solani in soft rots. The cocktail was able to reduce the disease incidence in infected tubers from 93.3% to 48.9% and decrease the diseased tissue by 75.3%
[76]. Similar results were found previously with T4-related phages. The treatment of rotting of potato tubers with one phage decreased weight of rot from 4 g to 0.5 g at MOI of 100
[77]. Interestingly, the injection of six phages prior infection protected 80% of potato plants from the
Ralstonia solanacearum wilt. Phage treatment of contaminated soil also reduced more than 5-fold the presence of this pathogenic bacteria compared to the control soil one week after phage spraying. Efficiency was shown to depend on timing of phage administration, suggesting that phage administration should be performed just after the first sign of bacterial wilt
[78].
Tomatoes: Several studios with tomato plants are available in the literature. The application of phage PE204 to the root system of tomato plants completely inhibited bacterial wilt caused by
R. solanacearum [79]. Phages isolated from river water also reduced significantly bacterial wilt and cocktails were the most effective candidates
[80]. A greenhouse experiment with combinations of phages against
R. solanacearum suggested that cocktails of phages select slow-growing resistant bacteria which reduces the severity of the disease
[81]. Importantly, a seedling-based method has been recently developed by mixing phages and tomato seedlings in sterile conical tubes before applying
Pseudomonas syringae to screen phage effectiveness. The authors propose this method before choosing phage candidates in phage biocontrol
[82].
Cherries: A treatment with thirteen individual phages or two cocktails produced a reduction in the disease progression and a decrease of 15–40% of
P. syringae in cherry leaves
[83].
Onions: Recently, a phage-biocontrol study was performed against soft rot caused by
Pectobacterium sp. in onions using field trials. The results showed significant higher number of plants in the treatments compared to the positive controls, with concomitant increased bulb and foliage mass and also reduced soft rot disease symptoms
[84].
Kohlrabies: The administration of a single phage at a MOI of 10 was able to reduce black rot disease symptoms due to
Xanthomonas campestris pv.
campestris up to 45%
[85].
Melons:
Acidovorax citrulli causing fruit blotch was treated with a single phage and 27% of disease severity was shown compared with 80% of disease of the control group, moreover, phage was detected by PCR in foliar tissues 8 h after phage addition to the soil
[86].
Summarizing, the use of phage-biocontrol shows a certain effect in vegetables, mainly when cocktails are administered in a short period time after infection. If this strategy results beneficial, it could be administered in the irrigation water to help decrease losses caused by pathogenic bacteria in cultures of economic relevance.
5. Phages on Surfaces
Bacteria are able to attach different surfaces as glass, metals, polymers, foods, as well as to other organisms
[87]. The greatest risk of food contamination resides on food-contact surfaces. For this reason, biofilms are a big deal in food industry, since they can spoil the equipment and contaminate food, increasing production costs
[88]. The interactions between bacteria and food-processing surfaces begins with a non-specific adhesion and ends with specific adhesions and the biofilm formation
[89].
Salmonella fimbriae facilitate attachment and the presence of cellulose enhances biofilm formation on certain abiotic surfaces
[90]. In 2019, Islam et al. isolated three broad-ranged lytic phages, LPSTLL, LPST94 and LPST153, from environmental water samples. The cocktail reduced
Salmonella biofilms by 44–63% on 96-well microplates. On food-processing surfaces such as stainless steel the cocktail was able to reduce biofilms cells up to 6.42 log CFU
[91]. Remarkably, Sadekuzzaman et al. showed that bacteriophages reduced
Salmonella in biofilms after only two hours of treatment by 3 and 2 log CFU/cm
2 on stainless steel and rubber, respectively, while adhered viable cells on lettuce were reduced around 1 log CFU per unit
[92]. Gong et al. exhibited that a cocktail of six phages was able to diminish by 84.2% the
Salmonella population on the boots of workers (which is relevant to prevent re-contamination of rendered meals) in a rendering-processing environment. This reduction increased in combination with sodium hypochlorite (92.9%) and scrubbing (93.2%) after a treatment three times for one week
[93]. Interestingly, it has been shown a synergistic effect in the combination of bacteriophages and chlorine with a reduction of biofilm growth by 94% and the ability to remove pre-existing biofilms by 88%, whereas chlorine alone could not eliminate them
[94].
Pseudomonas is the most frequently reported genus of the bacteria found after sanitation on food processing surfaces. This genus is able to resist in niches with nutrients, surface materials, temperatures and stress factors that are problematic for other bacteria, such as machines, floors, drains or stainless steel
[95]. Magin et al. tested 14.1 and LUZ7 phages isolated from drinking and thermal water against 24 h old biofilms produced by
P. aeruginosa PAO1 and D1 strains. Results showed that phage treatment produced a reduction of 1.7 log CFU/cm
2 of bacteria in biofilms formed on stainless-steel surface compared with untreated biofilms
[96].
On the other hand,
E. coli can attach to a variety of surfaces including stainless steel, teflon, glass, polystyrene, polypropylene, PVC and biotic surfaces, which are commonly employed in food industry. Wang et al. tested the AZO145A phage against the Shiga toxigenic
E. coli O145:H25 strain, known to be a strong biofilm former, on stainless steel coupons. Bacteriophage addition on biofilms grown during 24, 48 and 72 h was able to reduce cells 2.9, 1.9 and 1.9 log CFU/coupon, respectively, compared to the control
[97].
Overall, bacteriophages show great promise in decreasing the formation of new biofilms, but most importantly, in removing pre-existing ones in combination with other agents such as bleach.
7. Discussion
Phages are promising candidates in the fight against MDR bacteria. Recent studies report that phage treatment is able to reduce bacterial load and biofilm formation in biotic and abiotic media, indicating that this approach can be useful in biotechnology. However, one of the main concerns when considering this alternative is the narrow host range of most of the phages. This can be reverted almost completely with the use of phage cocktails. Moreover, cocktail use decreases the appearance of phage-resistant strains. High MOI and a rapid administration have been shown to increase successful rates of phage therapy in controlled experiments, however, in the real practice these two parameters cannot be determined.
Two different approaches of phage therapy have been proposed depending on the goal, humans and non-humans. In the first case, phage therapy is administered usually in combination with antibiotics due to their synergistic effect, while in the second case studies are performed typically only with phages to avoid antibiotics. Lessons learnt for human use could be helpful to succeed in non-human practice. Successful case reports in humans usually have administered cocktails of phages combined with antibiotics in multiple doses. Therefore, the combination of cocktails and low levels of antibiotics could improve the results of ineffective phage therapy in non-human use. On the other hand, there are cases in which neither antimicrobials nor phages can solve the injury, such as the case of toxigenic strains in which the harmful effect is due to the toxin. A less explored alternative is the induction of prophages that are latent in the bacterial genomes
[106] with compounds like EDTA, sodium citrate
[107], glycolic acid, N-acetyl cysteine, vinegar or plant extracts like stevia
[108], which would solve issues such as the host range restriction or bacterial resistance, and would improve the reaching to intracellular bacteria
[106].
In conclusion, further research is necessary to elaborate standard protocols in each specific field, including farms, aquaculture, surfaces or bioremediation in terms of timing, administration or cocktail composition, although the current phage products available in the market show that this alternative is already a real choice in biocontrol.