
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Narendar Dudhipala | + 7247 word(s) | 7247 | 2021-06-07 09:55:38 | | | |
| 2 | Vivi Li | Meta information modification | 7247 | 2021-06-08 03:04:59 | | |
Video Upload Options
Exosomes as nanosized vesicles are emerging as drug delivery systems for therapeutics owing to their natural origin, their ability to mediate intercellular communication, and their potential to encapsulate various biological molecules such as proteins and nucleic acids within the lipid bilayer membrane or in the lumen. Exosomes contain endogenous components (proteins, lipids, RNA) that could be used to deliver cargoes to target cells, offering an opportunity to diagnose and treat various diseases. Owing to their ability to travel safely in extracellular fluid and to transport cargoes to target cells with high efficacy, exosomes offer enhanced delivery of cargoes in vivo. However, several challenges related to the stabilization of the exosomes, the production of sufficient amounts of exosomes with safety and efficacy, the efficient loading of drugs into exosomes, the clearance of exosomes from circulation, and the transition from the bench scale to clinical production may limit their development and clinical use. For the clinical use of exosomes, it is important to understand the molecular mechanisms behind the transport and function of exosome vesicles.
1. Introduction
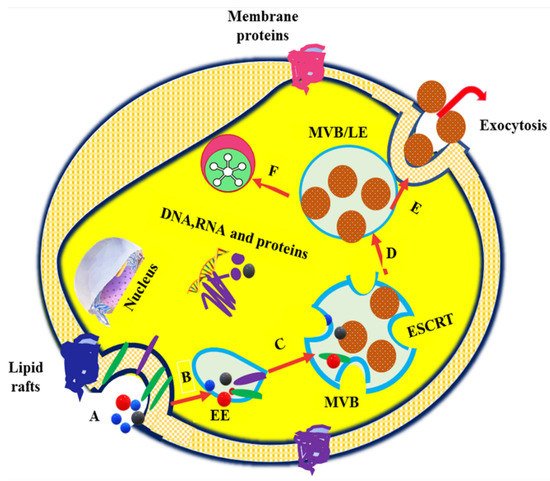
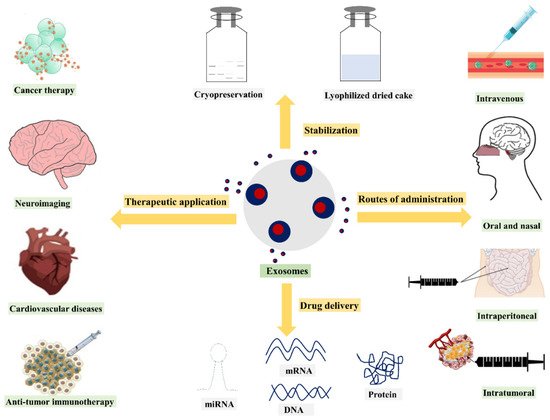
2. Exosomes in Drug Delivery
2.1. Protein and Peptide Delivery:
2.2. Exosomes in Gene Delivery
2.3. Exosomes in Delivery of Other Therapeutic Compounds
| Cargo Type | Origin of Exosomes | Disease Type | Isolation or Purification Method | Drug Loading Method | Outcome | Reference | |
|---|---|---|---|---|---|---|---|
| Proteins | Signal regulatory protein α | Human embryonic kidney293T cells | Cancer | Centrifugation | Transfection | Enhanced phagocytosis of tumor cells | [45] |
| Survivin-T34A | Melanoma cell lines | Pancreatic cancer | Centrifugation | NA | Apoptotic death of cells | [91] | |
| Antiepidermal growth factor receptor | Mouse neuroblastoma | Epidermoid carcinoma | Ultrafiltration/size exclusion liquid chromatography | NA | Target specificity | [48] | |
| 20S proteasome | Mesenchymal stem cells | Mouse myocardium | Tangential flow filtration | NA | Reduction in myocardial infraction | [92] | |
| Genetic substances | miRNA | Glioblastoma cells | Glioblastoma tumor | Differential centrifugation | Transfection | Providing diagnostic information | [69] |
| miRNA | Human cord blood endothelial colony-forming cells | Ischemic kidney injury | Centrifugation | Transfection | Protected kidney function and reduced kidney injury | [93] | |
| Spherical nucleic acids | PC-3 cells | Prostate cancer | Centrifugation | Naturally encased |
3000-fold-enhanced knockdown of miR-21 | [94] | |
| siRNA | Human embryonic kidneycells (HEK293) | Breast cancer | Sequential centrifugation | Electroporation | TPD52 gene expression was downregulated up to 70% compared with non-targeted exosomes | [76] | |
| Small molecules | Paclitaxel | Prostate cancer cell lines (PC-3 and LNCaP) | Autologous prostate cancer | Differential centrifugation | Co-incubation | Enhanced drug cytotoxicity to cancer cells | [95] |
| Doxorubicin | Immature mouse dendritic cells transfected with the vector-expressing iRGD-Lamp2b fusion proteins | Breast cancer | Centrifugation and ultrafiltration | Electroporation | Specific drug delivery to the tumor site andinhibited tumor growth | [79] | |
| Curcumin | Tumor cells (GL26-Luc, BV2, 3T3L1, 4T1, CT26, A20, and EL-4) | Brain tumor and autoimmune encephalitis | Sucrose gradient centrifugation | Direct mixing | Inhibited brain inflammation and delayed brain tumor growth | [84] | |
| Dopamine | Kunming mouse blood | Parkinson’s disease | Ultracentrifugation | Co-incubation | Enhanced therapeutic effect due to brain specific drug delivery | [96] | |
3. Exosomes Drug Loading Techniques
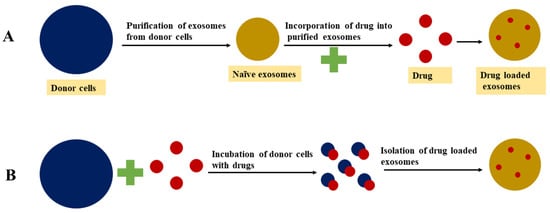
3.1. Passive Loading Approach
3.1.1. Incubation of Drugs with Exosomes
3.1.2. Incubation of Drugs with Donor Cells
3.2. Active Drug Loading Approaches
3.2.1. Sonication
3.2.2. Extrusion
3.2.3. Freeze–Thaw Cycles
3.2.4. Electroporation
3.2.5. Incubation with Membrane Permeabilizers
| Drug Loading Approach | Mechanism | Advantages | Disadvantages | |
|---|---|---|---|---|
| Passive loading | Incubation of exosomes and free drugs. | Diffusion of cargo into a cell or exosomal membrane. | Simple operation. Does not compromise the membrane integrity. |
Loading efficiency. Drugs may cause cytotoxicity to the donor cells. |
| Incubation of the donor cells with free drugs. | ||||
| Active loading | Sonication | Creation of micropores for diffusion by mechanical shear force. | Higher loading capacity than the simple incubation method. | Sonication-induced membrane damage is a roadblock for large scale application. Influence on exosome integrity and cargo aggregation. |
| Extrusion | Membrane recombination. | High cargo loading efficiency. Repeated extrusion provides a homogeneous blend of exosomes with cargoes. |
Recombination of exosomal surface structure may compromise the immune-privileged status of exosomes, making exosomes visible to immune cells such as mononuclear phagocytes. | |
| Freeze–thaw cycles | Membrane fusion. | Simple and effective strategy to load various cargoes (drugs, proteins, and peptides) into exosomes directly. | Repeated freeze–thaw may cause protein degeneration and exosome aggregation. Drug loading efficiency is lower than sonication and extrusion methods. |
|
| Electroporation | Creation of micropores for diffusion by the electric field. | High loading efficiency | The loading efficiency and aggregation of cargoes are major limitations. | |
| Incubation with membrane permeabilizers | Dissolves membrane molecules (cholesterol), create pores on the exosomal surface. | Higher loading capacity as compared with the simple incubation method | Saponin is hemolytically active in vivo, limiting the concentration (toxicity) of saponin used for drug loading. Extra purification process may be required to remove saponin. |
|
References
- Elsharkasy, O.M.; Nordin, J.Z.; Hagey, D.W.; de Jong, O.G.; Schiffelers, R.M.; Andaloussi, S.E.; Vader, P. Extracellular Vesicles as Drug Delivery Systems: Why and How? Adv. Drug Deliv. Rev. 2020, 159, 332–343.
- Svenson, S. Clinical Translation of Nanomedicines. Curr. Opin. Solid State Mater. Sci. 2012, 16, 287–294.
- Van der Meel, R.; Fens, M.H.A.M.; Vader, P.; van Solinge, W.W.; Eniola-Adefeso, O.; Schiffelers, R.M. Extracellular Vesicles as Drug Delivery Systems: Lessons from the Liposome Field. J. Control. Release 2014, 195, 72–85.
- Bulbake, U.; Doppalapudi, S.; Kommineni, N.; Khan, W. Liposomal Formulations in Clinical Use: An Updated Review. Pharmaceutics 2017, 9, 12.
- Balachandran, B.; Yuana, Y. Extracellular Vesicles-Based Drug Delivery System for Cancer Treatment. Cogent Med. 2019, 6, 1635806.
- Sercombe, L.; Veerati, T.; Moheimani, F.; Wu, S.Y.; Sood, A.K.; Hua, S. Advances and Challenges of Liposome Assisted Drug Delivery. Front. Pharmacol. 2015, 6, 286.
- Shao, J.; Zaro, J.; Shen, Y. Advances in Exosome-Based Drug Delivery and Tumor Targeting: From Tissue Distribution to Intracellular Fate. Int. J. Nanomed. 2020, 15, 9355–9371.
- Kooijmans, S.A.A.; Vader, P.; van Dommelen, S.M.; van Solinge, W.W.; Schiffelers, R.M. Exosome Mimetics: A Novel Class of Drug Delivery Systems. Int. J. Nanomed. 2012, 7, 1525–1541.
- Schiller, M.; Bekeredjian-Ding, I.; Heyder, P.; Blank, N.; Ho, A.D.; Lorenz, H.-M. Autoantigens Are Translocated into Small Apoptotic Bodies during Early Stages of Apoptosis. Cell Death Differ. 2008, 15, 183–191.
- Gregory, C.D.; Pound, J.D. Microenvironmental Influences of Apoptosis In Vivo and In Vitro. Apoptosis 2010, 15, 1029–1049.
- Wang, J.; Chen, D.; Ho, E.A. Challenges in the Development and Establishment of Exosome-Based Drug Delivery Systems. J. Control. Release 2021, 329, 894–906.
- Ha, D.; Yang, N.; Nadithe, V. Exosomes as Therapeutic Drug Carriers and Delivery Vehicles across Biological Membranes: Current Perspectives and Future Challenges. Acta Pharm. Sin. B 2016, 6, 287–296.
- Bunggulawa, E.J.; Wang, W.; Yin, T.; Wang, N.; Durkan, C.; Wang, Y.; Wang, G. Recent Advancements in the Use of Exosomes as Drug Delivery Systems. J. Nanobiotechnology 2018, 16, 81.
- Gebeyehu, A.; Kommineni, N.; Meckes, D.; Sachdeva, M. Exosome Vehicles as Nano-Drug Delivery Materials for Chemotherapeutic Drugs. CRT 2021.
- Mittelbrunn, M.; Sánchez-Madrid, F. Intercellular Communication: Diverse Structures for Exchange of Genetic Information. Nat. Rev. Mol. Cell Biol. 2012, 13, 328–335.
- Heijnen, H.F.; Schiel, A.E.; Fijnheer, R.; Geuze, H.J.; Sixma, J.J. Activated Platelets Release Two Types of Membrane Vesicles: Microvesicles by Surface Shedding and Exosomes Derived from Exocytosis of Multivesicular Bodies and Alpha-Granules. Blood 1999, 94, 3791–3799.
- Savina, A.; Furlán, M.; Vidal, M.; Colombo, M.I. Exosome Release Is Regulated by a Calcium-Dependent Mechanism in K562 Cells. J. Biol. Chem. 2003, 278, 20083–20090.
- Hung, W.-T.; Hong, X.; Christenson, L.K.; McGinnis, L.K. Extracellular Vesicles from Bovine Follicular Fluid Support Cumulus Expansion. Biol. Reprod. 2015, 93, 117.
- Machtinger, R.; Laurent, L.C.; Baccarelli, A.A. Extracellular Vesicles: Roles in Gamete Maturation, Fertilization and Embryo Implantation. Hum. Reprod. Update 2016, 22, 182–193.
- Patton, A.L.; McCallie, B.; Parks, J.C.; Schoolcraft, W.B.; Katz-Jaffe, M. Exosome Bound MicroRNAs Transcriptionally Regulate Embryo-Endometrial Dialogue Impacting Implantation Potential for AMA Patients. Fertil. Steril. 2015, 104, e308.
- Matsuno, Y.; Onuma, A.; Fujioka, Y.A.; Yasuhara, K.; Fujii, W.; Naito, K.; Sugiura, K. Effects of Exosome-like Vesicles on Cumulus Expansion in Pigs In Vitro. J. Reprod. Dev. 2017, 63, 51–58.
- Qu, P.; Qing, S.; Liu, R.; Qin, H.; Wang, W.; Qiao, F.; Ge, H.; Liu, J.; Zhang, Y.; Cui, W.; et al. Effects of Embryo-Derived Exosomes on the Development of Bovine Cloned Embryos. PLoS ONE 2017, 12, e0174535.
- Mobarak, H.; Heidarpour, M.; Lolicato, F.; Nouri, M.; Rahbarghazi, R.; Mahdipour, M. Physiological Impact of Extracellular Vesicles on Female Reproductive System; Highlights to Possible Restorative Effects on Female Age-Related Fertility. Biofactors 2019, 45, 293–303.
- Tsochandaridis, M.; Nasca, L.; Toga, C.; Levy-Mozziconacci, A. Circulating MicroRNAs as Clinical Biomarkers in the Predictions of Pregnancy Complications. Biomed. Res. Int. 2015, 2015, 294954.
- 25 Koh, Y.Q.; Peiris, H.N.; Vaswani, K.; Reed, S.; Rice, G.E.; Salomon, C.; Mitchell, M.D. Characterization of Exosomal Release in Bovine Endometrial Intercaruncular Stromal Cells. Reprod. Biol. Endocrinol. 2016, 14, 78.
- Sullivan, R.; Frenette, G.; Girouard, J. Epididymosomes Are Involved in the Acquisition of New Sperm Proteins during Epididymal Transit. Asian J. Androl. 2007, 9, 483–491.
- Andrews, R.E.; Galileo, D.S.; Martin-DeLeon, P.A. Plasma Membrane Ca2+-ATPase 4: Interaction with Constitutive Nitric Oxide Synthases in Human Sperm and Prostasomes Which Carry Ca2+/CaM-Dependent Serine Kinase. Mol. Hum. Reprod. 2015, 21, 832–843.
- Choy, K.H.K.; Chan, S.Y.; Lam, W.; Jin, J.; Zheng, T.; Yu, S.S.; Wang, W.; Li, L.; Xie, G.; Yim, H.C.H.; et al. The Repertoire of Testicular Extracellular Vesicles Cargoes and Their Involvement in Inter-Compartmental Communication Required for Spermatogenesis. bioRxiv 2021.
- Baskaran, S.; Panner Selvam, M.K.; Agarwal, A. Exosomes of Male Reproduction. Adv. Clin. Chem. 2020, 95, 149–163.
- Zhou, Y.; Tian, T.; Zhu, Y.; Jaffar Ali, D.; Hu, F.; Qi, Y.; Sun, B.; Xiao, Z. Exosomes Transfer Among Different Species Cells and Mediating MiRNAs Delivery. J. Cell. Biochem. 2017, 118, 4267–4274.
- Baj-Krzyworzeka, M.; Majka, M.; Pratico, D.; Ratajczak, J.; Vilaire, G.; Kijowski, J.; Reca, R.; Janowska-Wieczorek, A.; Ratajczak, M.Z. Platelet-Derived Microparticles Stimulate Proliferation, Survival, Adhesion, and Chemotaxis of Hematopoietic Cells. Exp. Hematol. 2002, 30, 450–459.
- Al-Nedawi, K.; Meehan, B.; Kerbel, R.S.; Allison, A.C.; Rak, J. Endothelial Expression of Autocrine VEGF upon the Uptake of Tumor-Derived Microvesicles Containing Oncogenic EGFR. Proc. Natl. Acad. Sci. USA 2009, 106, 3794–3799.
- Tian, T.; Wang, Y.; Wang, H.; Zhu, Z.; Xiao, Z. Visualizing of the Cellular Uptake and Intracellular Trafficking of Exosomes by Live-Cell Microscopy. J. Cell. Biochem. 2010, 111, 488–496.
- Mayer, A. Intracellular Membrane Fusion: SNAREs Only? Curr. Opin. Cell Biol. 1999, 11, 447–452.
- Simpson, R.J.; Jensen, S.S.; Lim, J.W.E. Proteomic Profiling of Exosomes: Current Perspectives. Proteomics 2008, 8, 4083–4099.
- Valadi, H.; Ekström, K.; Bossios, A.; Sjöstrand, M.; Lee, J.J.; Lötvall, J.O. Exosome-Mediated Transfer of MRNAs and MicroRNAs Is a Novel Mechanism of Genetic Exchange between Cells. Nat. Cell Biol. 2007, 9, 654–659.
- Yeo, R.W.Y.; Lai, R.C.; Zhang, B.; Tan, S.S.; Yin, Y.; Teh, B.J.; Lim, S.K. Mesenchymal Stem Cell: An Efficient Mass Producer of Exosomes for Drug Delivery. Adv. Drug Deliv. Rev. 2013, 65, 336–341.
- Cabeza, L.; Perazzoli, G.; Peña, M.; Cepero, A.; Luque, C.; Melguizo, C.; Prados, J. Cancer Therapy Based on Extracellular Vesicles as Drug Delivery Vehicles. J. Control. Release 2020, 327, 296–315.
- Armstrong, J.P.K.; Stevens, M.M. Strategic Design of Extracellular Vesicle Drug Delivery Systems. Adv. Drug Deliv. Rev. 2018, 130, 12–16.
- Kwon, S.; Shin, S.; Do, M.; Oh, B.H.; Song, Y.; Bui, V.D.; Lee, E.S.; Jo, D.-G.; Cho, Y.W.; Kim, D.-H.; et al. Engineering Approaches for Effective Therapeutic Applications Based on Extracellular Vesicles. J. Control. Release 2021, 330, 15–30.
- Milman, N.; Ginini, L.; Gil, Z. Exosomes and Their Role in Tumorigenesis and Anticancer Drug Resistance. Drug Resist. Updates 2019, 45, 1–12.
- Tan, A.; Rajadas, J.; Seifalian, A.M. Exosomes as Nano-Theranostic Delivery Platforms for Gene Therapy. Adv. Drug Deliv. Rev. 2013, 65, 357–367.
- Graner, M.W.; Alzate, O.; Dechkovskaia, A.M.; Keene, J.D.; Sampson, J.H.; Mitchell, D.A.; Bigner, D.D. Proteomic and Immunologic Analyses of Brain Tumor Exosomes. FASEB J. 2009, 23, 1541–1557.
- Van Dommelen, S.M.; Vader, P.; Lakhal, S.; Kooijmans, S.A.A.; van Solinge, W.W.; Wood, M.J.A.; Schiffelers, R.M. Microvesicles and Exosomes: Opportunities for Cell-Derived Membrane Vesicles in Drug Delivery. J. Control. Release 2012, 161, 635–644.
- Cho, E.; Nam, G.-H.; Hong, Y.; Kim, Y.K.; Kim, D.-H.; Yang, Y.; Kim, I.-S. Comparison of Exosomes and Ferritin Protein Nanocages for the Delivery of Membrane Protein Therapeutics. J. Control. Release 2018, 279, 326–335.
- Théry, C.; Zitvogel, L.; Amigorena, S. Exosomes: Composition, Biogenesis and Function. Nat. Rev. Immunol. 2002, 2, 569–579.
- Khan, S.; Bennit, H.F.; Wall, N.R. The Emerging Role of Exosomes in Survivin Secretion. Histol. Histopathol. 2015, 30, 43–50.
- Kooijmans, S.A.A.; Aleza, C.G.; Roffler, S.R.; van Solinge, W.W.; Vader, P.; Schiffelers, R.M. Display of GPI-Anchored Anti-EGFR Nanobodies on Extracellular Vesicles Promotes Tumour Cell Targeting. J. Extracell. Vesicles 2016, 5, 31053.
- Peng, H.; Ji, W.; Zhao, R.; Yang, J.; Lu, Z.; Li, Y.; Zhang, X. Exosome: A Significant Nano-Scale Drug Delivery Carrier. J. Mater. Chem. B 2020, 8, 7591–7608.
- Gehrmann, U.; Hiltbrunner, S.; Georgoudaki, A.-M.; Karlsson, M.C.; Näslund, T.I.; Gabrielsson, S. Synergistic Induction of Adaptive Antitumor Immunity by Codelivery of Antigen with α-Galactosylceramide on Exosomes. Cancer Res. 2013, 73, 3865–3876.
- Tian, T.; Zhang, H.-X.; He, C.-P.; Fan, S.; Zhu, Y.-L.; Qi, C.; Huang, N.-P.; Xiao, Z.-D.; Lu, Z.-H.; Tannous, B.A.; et al. Surface Functionalized Exosomes as Targeted Drug Delivery Vehicles for Cerebral Ischemia Therapy. Biomaterials 2018, 150, 137–149.
- De Toro, J.; Herschlik, L.; Waldner, C.; Mongini, C. Emerging Roles of Exosomes in Normal and Pathological Conditions: New Insights for Diagnosis and Therapeutic Applications. Front. Immunol. 2015, 6, 203.
- Anand, P.K. Exosomal Membrane Molecules Are Potent Immune Response Modulators. Commun. Integr. Biol. 2010, 3, 405–408.
- Gehrmann, U.; Näslund, T.I.; Hiltbrunner, S.; Larssen, P.; Gabrielsson, S. Harnessing the Exosome-Induced Immune Response for Cancer Immunotherapy. Semin. Cancer Biol. 2014, 28, 58–67.
- Jesus, S.; Soares, E.; Cruz, M.T.; Borges, O. Exosomes as Adjuvants for the Recombinant Hepatitis B Antigen: First Report. Eur. J. Pharm. Biopharm. 2018, 133, 1–11.
- Liu, Y.; Fu, N.; Su, J.; Wang, X.; Li, X. Rapid Enkephalin Delivery Using Exosomes to Promote Neurons Recovery in Ischemic Stroke by Inhibiting Neuronal P53/Caspase-3. BioMed Res. Int. 2019, 2019, e4273290.
- Barok, M.; Puhka, M.; Vereb, G.; Szollosi, J.; Isola, J.; Joensuu, H. Cancer-Derived Exosomes from HER2-Positive Cancer Cells Carry Trastuzumab-Emtansine into Cancer Cells Leading to Growth Inhibition and Caspase Activation. BMC Cancer 2018, 18, 504.
- Parolini, I.; Federici, C.; Raggi, C.; Lugini, L.; Palleschi, S.; De Milito, A.; Coscia, C.; Iessi, E.; Logozzi, M.; Molinari, A.; et al. Microenvironmental PH Is a Key Factor for Exosome Traffic in Tumor Cells. J. Biol. Chem. 2009, 284, 34211–34222.
- Kim, H.; Yun, N.; Mun, D.; Kang, J.-Y.; Lee, S.-H.; Park, H.; Park, H.; Joung, B. Cardiac-Specific Delivery by Cardiac Tissue-Targeting Peptide-Expressing Exosomes. Biochem. Biophys. Res. Commun. 2018, 499, 803–808.
- Haney, M.J.; Klyachko, N.L.; Zhao, Y.; Gupta, R.; Plotnikova, E.G.; He, Z.; Patel, T.; Piroyan, A.; Sokolsky, M.; Kabanov, A.V.; et al. Exosomes as Drug Delivery Vehicles for Parkinson’s Disease Therapy. J. Control. Release 2015, 207, 18–30.
- Kole, R.; Krainer, A.R.; Altman, S. RNA Therapeutics: Beyond RNA Interference and Antisense Oligonucleotides. Nat. Rev. Drug Discov. 2012, 11, 125–140.
- Guo, P. The Emerging Field of RNA Nanotechnology. Nat. Nanotechnol. 2010, 5, 833–842.
- Bramsen, J.B.; Kjems, J. Development of Therapeutic-Grade Small Interfering RNAs by Chemical Engineering. Front. Genet. 2012, 3, 154.
- Seow, Y.; Wood, M.J. Biological Gene Delivery Vehicles: Beyond Viral Vectors. Mol. Ther. 2009, 17, 767–777.
- Foged, C. SiRNA Delivery with Lipid-Based Systems: Promises and Pitfalls. Curr. Top. Med. Chem. 2012, 12, 97–107.
- El Andaloussi, S.; Mäger, I.; Breakefield, X.O.; Wood, M.J.A. Extracellular Vesicles: Biology and Emerging Therapeutic Opportunities. Nat. Rev. Drug Discov. 2013, 12, 347–357.
- Wahlgren, J.; Karlson, T.D.L.; Brisslert, M.; Vaziri Sani, F.; Telemo, E.; Sunnerhagen, P.; Valadi, H. Plasma Exosomes Can Deliver Exogenous Short Interfering RNA to Monocytes and Lymphocytes. Nucleic Acids Res. 2012, 40, e130.
- Shtam, T.A.; Kovalev, R.A.; Varfolomeeva, E.Y.; Makarov, E.M.; Kil, Y.V.; Filatov, M.V. Exosomes Are Natural Carriers of Exogenous SiRNA to Human Cells In Vitro. Cell Commun. Signal. 2013, 11, 88.
- Skog, J.; Würdinger, T.; van Rijn, S.; Meijer, D.H.; Gainche, L.; Sena-Esteves, M.; Curry, W.T.; Carter, B.S.; Krichevsky, A.M.; Breakefield, X.O. Glioblastoma Microvesicles Transport RNA and Proteins that Promote Tumour Growth and Provide Diagnostic Biomarkers. Nat. Cell Biol. 2008, 10, 1470–1476.
- Alvarez-Erviti, L.; Seow, Y.; Yin, H.; Betts, C.; Lakhal, S.; Wood, M.J.A. Delivery of SiRNA to the Mouse Brain by Systemic Injection of Targeted Exosomes. Nat. Biotechnol. 2011, 29, 341–345.
- Banizs, A.B.; Huang, T.; Dryden, K.; Berr, S.S.; Stone, J.R.; Nakamoto, R.K.; Shi, W.; He, J. In Vitro Evaluation of Endothelial Exosomes as Carriers for Small Interfering Ribonucleic Acid Delivery. Int. J. Nanomed. 2014, 9, 4223–4230.
- Zheng, M.; Huang, M.; Ma, X.; Chen, H.; Gao, X. Harnessing Exosomes for the Development of Brain Drug Delivery Systems. Bioconjugate Chem. 2019, 30, 994–1005.
- Saeedi, S.; Israel, S.; Nagy, C.; Turecki, G. The Emerging Role of Exosomes in Mental Disorders. Transl. Psychiatry 2019, 9, 1–11.
- Lamichhane, T.N.; Raiker, R.S.; Jay, S.M. Exogenous DNA Loading into Extracellular Vesicles via Electroporation Is Size-Dependent and Enables Limited Gene Delivery. Mol. Pharm. 2015, 12, 3650–3657.
- Faruqu, F.N.; Xu, L.; Al-Jamal, K.T. Preparation of Exosomes for SiRNA Delivery to Cancer Cells. J. Vis. Exp. 2018.
- Limoni, S.K.; Moghadam, M.F.; Moazzeni, S.M.; Gomari, H.; Salimi, F. Engineered Exosomes for Targeted Transfer of SiRNA to HER2 Positive Breast Cancer Cells. Appl. Biochem. Biotechnol. 2019, 187, 352–364.
- Zhang, D.; Lee, H.; Wang, X.; Rai, A.; Groot, M.; Jin, Y. Exosome-Mediated Small RNA Delivery: A Novel Therapeutic Approach for Inflammatory Lung Responses. Mol. Ther. 2018, 26, 2119–2130.
- Melzer, C.; Rehn, V.; Yang, Y.; Bähre, H.; von der Ohe, J.; Hass, R. Taxol-Loaded MSC-Derived Exosomes Provide a Therapeutic Vehicle to Target Metastatic Breast Cancer and Other Carcinoma Cells. Cancers 2019, 11, 798.
- Tian, Y.; Li, S.; Song, J.; Ji, T.; Zhu, M.; Anderson, G.J.; Wei, J.; Nie, G. A Doxorubicin Delivery Platform Using Engineered Natural Membrane Vesicle Exosomes for Targeted Tumor Therapy. Biomaterials 2014, 35, 2383–2390.
- Schindler, C.; Collinson, A.; Matthews, C.; Pointon, A.; Jenkinson, L.; Minter, R.R.; Vaughan, T.J.; Tigue, N.J. Exosomal Delivery of Doxorubicin Enables Rapid Cell Entry and Enhanced In Vitro Potency. PLoS ONE 2019, 14, e0214545.
- Mulcahy, L.A.; Pink, R.C.; Carter, D.R.F. Routes and Mechanisms of Extracellular Vesicle Uptake. J. Extracell. Vesicles 2014, 3.
- Finn, O.J. Immuno-Oncology: Understanding the Function and Dysfunction of the Immune System in Cancer. Ann. Oncol. 2012, 23, viii6-9.
- Kim, M.S.; Haney, M.J.; Zhao, Y.; Mahajan, V.; Deygen, I.; Klyachko, N.L.; Inskoe, E.; Piroyan, A.; Sokolsky, M.; Okolie, O.; et al. Development of Exosome-Encapsulated Paclitaxel to Overcome MDR in Cancer Cells. Nanomedicine 2016, 12, 655–664.
- Sun, D.; Zhuang, X.; Xiang, X.; Liu, Y.; Zhang, S.; Liu, C.; Barnes, S.; Grizzle, W.; Miller, D.; Zhang, H.-G. A Novel Nanoparticle Drug Delivery System: The Anti-Inflammatory Activity of Curcumin Is Enhanced When Encapsulated in Exosomes. Mol. Ther. 2010, 18, 1606–1614.
- Zhuang, X.; Xiang, X.; Grizzle, W.; Sun, D.; Zhang, S.; Axtell, R.C.; Ju, S.; Mu, J.; Zhang, L.; Steinman, L.; et al. Treatment of Brain Inflammatory Diseases by Delivering Exosome Encapsulated Anti-Inflammatory Drugs from the Nasal Region to the Brain. Mol. Ther. 2011, 19, 1769–1779.
- Canfrán-Duque, A.; Pastor, O.; Quintana-Portillo, R.; Lerma, M.; de la Peña, G.; Martín-Hidalgo, A.; Fernández-Hernando, C.; Lasunción, M.A.; Busto, R. Curcumin Promotes Exosomes/Microvesicles Secretion That Attenuates Lysosomal Cholesterol Traffic Impairment. Mol. Nutr. Food Res. 2014, 58, 687–697.
- Kalani, A.; Kamat, P.K.; Chaturvedi, P.; Tyagi, S.C.; Tyagi, N. Curcumin-Primed Exosomes Mitigate Endothelial Cell Dysfunction during Hyperhomocysteinemia. Life Sci. 2014, 107, 1–7.
- Kim, S.M.; Yang, Y.; Oh, S.J.; Hong, Y.; Seo, M.; Jang, M. Cancer-Derived Exosomes as a Delivery Platform of CRISPR/Cas9 Confer Cancer Cell Tropism-Dependent Targeting. J. Control. Release 2017, 266, 8–16.
- Hou, S.; Niu, X.; Li, L.; Zhou, J.; Qian, Z.; Yao, D.; Yang, F.; Ma, P.X.; Fan, Y. Simultaneous Nano- and Microscale Structural Control of Injectable Hydrogels via the Assembly of Nanofibrous Protein Microparticles for Tissue Regeneration. Biomaterials 2019, 223, 119458.
- Pardridge, W.M. Drug Transport across the Blood-Brain Barrier. J. Cereb. Blood Flow Metab. 2012, 32, 1959–1972.
- Aspe, J.R.; Diaz Osterman, C.J.; Jutzy, J.M.S.; Deshields, S.; Whang, S.; Wall, N.R. Enhancement of Gemcitabine Sensitivity in Pancreatic Adenocarcinoma by Novel Exosome-Mediated Delivery of the Survivin-T34A Mutant. J. Extracell. Vesicles 2014, 3.
- Lai, R.C.; Tan, S.S.; Teh, B.J.; Sze, S.K.; Arslan, F.; de Kleijn, D.P.; Choo, A.; Lim, S.K. Proteolytic Potential of the MSC Exosome Proteome: Implications for an Exosome-Mediated Delivery of Therapeutic Proteasome. Int. J. Proteomics 2012, 2012, 971907.
- Viñas, J.L.; Burger, D.; Zimpelmann, J.; Haneef, R.; Knoll, W.; Campbell, P.; Gutsol, A.; Carter, A.; Allan, D.S.; Burns, K.D. Transfer of MicroRNA-486-5p from Human Endothelial Colony Forming Cell-Derived Exosomes Reduces Ischemic Kidney Injury. Kidney Int. 2016, 90, 1238–1250.
- Alhasan, A.H.; Patel, P.C.; Choi, C.H.J.; Mirkin, C.A. Exosome Encased Spherical Nucleic Acid Gold Nanoparticle Conjugates as Potent MicroRNA Regulation Agents. Small 2014, 10, 186–192.
- Saari, H.; Lázaro-Ibáñez, E.; Viitala, T.; Vuorimaa-Laukkanen, E.; Siljander, P.; Yliperttula, M. Microvesicle- and Exosome-Mediated Drug Delivery Enhances the Cytotoxicity of Paclitaxel in Autologous Prostate Cancer Cells. J. Control. Release 2015, 220, 727–737.
- Qu, M.; Lin, Q.; Huang, L.; Fu, Y.; Wang, L.; He, S.; Fu, Y.; Yang, S.; Zhang, Z.; Zhang, L.; et al. Dopamine-Loaded Blood Exosomes Targeted to Brain for Better Treatment of Parkinson’s Disease. J. Control. Release 2018, 287, 156–166.
- Vader, P.; Mol, E.A.; Pasterkamp, G.; Schiffelers, R.M. Extracellular Vesicles for Drug Delivery. Adv. Drug Deliv. Rev. 2016, 106, 148–156.
- Akuma, P.; Okagu, O.D.; Udenigwe, C.C. Naturally Occurring Exosome Vesicles as Potential Delivery Vehicle for Bioactive Compounds. Front. Sustain. Food Syst. 2019.
- Fuhrmann, G.; Serio, A.; Mazo, M.; Nair, R.; Stevens, M.M. Active Loading into Extracellular Vesicles Significantly Improves the Cellular Uptake and Photodynamic Effect of Porphyrins. J. Control. Release 2015, 205, 35–44.
- Luan, X.; Sansanaphongpricha, K.; Myers, I.; Chen, H.; Yuan, H.; Sun, D. Engineering Exosomes as Refined Biological Nanoplatforms for Drug Delivery. Acta Pharmacol. Sin. 2017, 38, 754–763.
- Vashisht, M.; Rani, P.; Onteru, S.K.; Singh, D. Curcumin Encapsulated in Milk Exosomes Resists Human Digestion and Possesses Enhanced Intestinal Permeability In Vitro. Appl. Biochem. Biotechnol. 2017, 183, 993–1007.
- Pascucci, L.; Coccè, V.; Bonomi, A.; Ami, D.; Ceccarelli, P.; Ciusani, E.; Viganò, L.; Locatelli, A.; Sisto, F.; Doglia, S.M.; et al. Paclitaxel Is Incorporated by Mesenchymal Stromal Cells and Released in Exosomes That Inhibit In Vitro Tumor Growth: A New Approach for Drug Delivery. J. Control. Release 2014, 192, 262–270.
- Chen, T.; Guo, J.; Yang, M.; Zhu, X.; Cao, X. Chemokine-Containing Exosomes Are Released from Heat-Stressed Tumor Cells via Lipid Raft-Dependent Pathway and Act as Efficient Tumor Vaccine. J. Immunol. 2011, 186, 2219–2228.
- Wang, L.-Y.; Shi, X.-Y.; Yang, C.-S.; Huang, D.-M. Versatile RBC-Derived Vesicles as Nanoparticle Vector of Photosensitizers for Photodynamic Therapy. Nanoscale 2013, 5, 416–421.
- Lamichhane, T.N.; Jeyaram, A.; Patel, D.B.; Parajuli, B.; Livingston, N.K.; Arumugasaamy, N.; Schardt, J.S.; Jay, S.M. Oncogene Knockdown via Active Loading of Small RNAs into Extracellular Vesicles by Sonication. Cell. Mol. Bioeng. 2016, 9, 315–324.
- Arun, B.; Narendar, D.; Veerabrahma, K. Development of Olmesartan Medoxomil Lipid-Based Nanoparticles and Nanosuspension: Preparation, Characterization and Comparative Pharmacokinetic Evaluation. Artif. Cells Nanomed. Biotechnol. 2018, 46, 126–137.
- Sato, Y.T.; Umezaki, K.; Sawada, S.; Mukai, S.; Sasaki, Y.; Harada, N.; Shiku, H.; Akiyoshi, K. Engineering Hybrid Exosomes by Membrane Fusion with Liposomes. Sci. Rep. 2016, 6, 21933.
- Johnsen, K.B.; Gudbergsson, J.M.; Skov, M.N.; Pilgaard, L.; Moos, T.; Duroux, M. A Comprehensive Overview of Exosomes as Drug Delivery Vehicles—Endogenous Nanocarriers for Targeted Cancer Therapy. Biochim. Biophys. Acta 2014, 1846, 75–87.
- Podolak, I.; Galanty, A.; Sobolewska, D. Saponins as Cytotoxic Agents: A Review. Phytochem. Rev. 2010, 9, 425–474.
- Reshke, R.; Taylor, J.A.; Savard, A.; Guo, H.; Rhym, L.H.; Kowalski, P.S.; Trung, M.T.; Campbell, C.; Little, W.; Anderson, D.G.; et al. Reduction of the Therapeutic Dose of Silencing RNA by Packaging It in Extracellular Vesicles via a Pre-MicroRNA Backbone. Nat. Biomed. Eng. 2020, 4, 52–68.
- Haney, M.J.; Zhao, Y.; Jin, Y.S.; Li, S.M.; Bago, J.R.; Klyachko, N.L.; Kabanov, A.V.; Batrakova, E.V. Macrophage-Derived Extracellular Vesicles as Drug Delivery Systems for Triple Negative Breast Cancer (TNBC) Therapy. J. Neuroimmune Pharmacol. 2020, 15, 487–500.
- Fu, S.; Wang, Y.; Xia, X.; Zheng, J.C. Exosome Engineering: Current Progress in Cargo Loading and Targeted Delivery. NanoImpact 2020, 20, 100261.
- Xu, M.; Yang, Q.; Sun, X.; Wang, Y. Recent Advancements in the Loading and Modification of Therapeutic Exosomes. Front. Bioeng. Biotechnol. 2020, 8, 586130.
- Meng, W.; He, C.; Hao, Y.; Wang, L.; Li, L.; Zhu, G. Prospects and Challenges of Extracellular Vesicle-Based Drug Delivery System: Considering Cell Source. Drug Deliv. 2020, 27, 585–598.
- Antimisiaris, S.G.; Mourtas, S.; Marazioti, A. Exosomes and Exosome-Inspired Vesicles for Targeted Drug Delivery. Pharmaceutics 2018, 10, 218.
- Jafari, D.; Shajari, S.; Jafari, R.; Mardi, N.; Gomari, H.; Ganji, F.; Forouzandeh Moghadam, M.; Samadikuchaksaraei, A. Designer Exosomes: A New Platform for Biotechnology Therapeutics. BioDrugs 2020, 34, 567–586.




