1000/1000
Hot
Most Recent

Ammonia use in maritime applications is considered promising, due to its high energy density, low flammability, easy storage and low production cost. Moreover, ammonia can be used as fuel in a variety of propulsors such as fuel cells and can be produced from renewable sources.
It is without a doubt a new era for the maritime industry, full of challenges that are poised to disrupt the status quo of common practices. The current coronavirus pandemic, the looming dangers of climate change and the novelties of smart shipping are the main contributing factors that catapult the maritime industry in an era of uncertainty. The severity of the challenges ahead is speculated to be as severe as those brought upon by the transition from sails to steam power [1]. Excluding the worst-case scenario of the impact of the pandemic to sea trade, the effects of the coronavirus are expected to have a short-term influence until 2024. From that point onwards, climate change and smart shipping will be the catalysts for change. Therefore, the systems that are currently under development, including new fuels, such as ammonia (NH3) and new power generation plants (e.g., fuel cells), are amongst the possible viable solutions to address these catalysts.
The shipping industry is accountable for a substantial part of global air pollution and the potential growth of seaborne transport can lead to an increase in carbon emissions [2]. Global shipping was responsible for more than 1 million tonnes of (Greenhouse Gas) GHG and CO2 emissions in 2018, indicating a significant increase of 9.6% and 9.3% respectively compared to the 2012 levels [3]. This leads to an increase in the share of shipping in the global anthropogenic emissions to the level of 2.89% [3]. Thus, in recent years, the shipping industry is facing great pressure to reduce environmental emissions and especially carbon emissions. This becomes even more important since it is forecasted that by 2050 carbon emissions could increase on a range of 80% to 130% compared to the 2008 levels [3]. In detail, by 2019 cruise ships emitted the most amount of CO2 per vessel (~79000 tons per vessel per year), followed by Liquified Natural Gas (LNG) carriers (~77000 tons per vessel per year). This highlights the increased effort required by the future designers and operators of these vessels. Similarly, on the same time-scale, Panama flagged vessels emitted the most amount of CO2 (~117E6 tons), followed by Liberian (~92E6 tons) and Marshall Islands (~79E6 tons) flagged vessels [4]. This represents the increased importance that the flag states have in enforcing IMO regulations through their regulatory control.
Due to the significant environmental impact of the shipping operations, the International Maritime Organisation (IMO) has imposed strict environmental regulations in the shipping industry. IMO has set the 2020 sulphur cap, which aims to decrease the sulphur global emissions to 0.5% compared to the previous level of 3.5%. Additional regulations to reduce GHG emissions, like the Efficiency Design Index (EEDI), Energy Efficiency Operations Index (EEOI) and the Ship Energy Efficiency Management Plan (SEEMP) [5][6]. Along with these efforts towards decarbonisation, a reduction of CO2 emissions around 90% is required from 2010 to 2050 for the shipping industry to contribute to the global target of keeping the temperature increase below 2 °C [7]. The IMO Marine Environmental Protection Committee (MEPC), acknowledging the great contribution of the shipping sector to the global CO2 emissions, on 2018 set a target to reduce the CO2 emissions from the shipping sector by 50% until 2050 [8].
Fuel cells represent a feasible solution for the decarbonisation of the maritime industry. They exhibit improved energy density compared with batteries, and are less pollutant and more efficient than traditional internal combustion engines [9]. In addition, fuel cells can be powered with green fuels (hydrogen, ammonia) which further increases their potential. Fuel cells applications on the maritime sector have been gaining attention, and as reviewed by [9], fuel cell systems can be used to reduce the ship emissions. A comparative life cycle assessment analysis on fuel cells comparing them with traditional diesel generators was performed by [10] and by [11], where it was identified that a hydrogen operated fuel cells and batteries configuration is the most environmentally promising alternative compared to a diesel or a diesel hybrid system. The simulation of a hybrid system that includes Solid Oxide Fuel Cells (SOFC) was presented by [12] and by [13], with considerations the reduction of carbon emissions in the former and the EEDI, as well as the availability of the system in the latter. Furthermore, the design optimisation of a waste heat recovery technology combined with fuel cells for electric energy production was performed with technical and energetic considerations [14]. The synthesis and design optimisation of the integrated ship systems with focus on the SOFCs and economic, environmental, as well as energy efficiency objectives was developed by [15]. A hybrid system including photovoltaic system was proposed by [16]. Finally, the technical analysis of fuel cells, in order to improve the energy systems environmental performance was addressed by [17].
Furthermore, in the quest to reduce the global carbon footprint, reach a carbon-neutral human activity and adapt to the future environmental challenges, one of the main efforts is to introduce zero-emission fuels [18]. It is assumed that the targets set by IMO for 2050 will only be reached if carbon-neutral fuels provide 30–40% of the total energy [19]. Finding a carbon-neutral solution that is safe, innovative and commercially viable consists a major challenge for the maritime industry. A possible solution to this problem is to focus on alternative fuels with favourable environmental impact, whilst keeping in mind their availability, compatibility, cost and compliance with international rules (IGF and IGC Code) [20].
There are numerous options for alternative marine fuels, including hydrogen (H2), alcohols (ethanol and methanol), Natural Gas (NG), biodiesel and NH3 [18]. However, none of the different options is a turn-key solution, as each potential application has different requirements and constraints. Among the possible alternatives, NH3 looks very promising since it has less complex and safer energy storage, compared to H2. It offers better energy density compared to H2, giving longer range and has a better environmental impact than NG. In addition, NH3 compared to other low carbon emission fuels is already produced in high quantities and transported around the globe, therefore it has established large-scale distribution infrastructures. An important issue when introducing a novel fuel is the power generation plant. NH3 is a flexible fuel that can be used both by the traditional marine engines and the more energy and environmental efficient fuel cells.
Large number of research studies have shown interest for ways to improve the environmental and carbon footprint of shipping by using zero-carbon fuels. Hydrogen has gained attention over the years, with various sources examining its potential as an energy vector [21][22]. In addition, the status-quo and existing issues of the hydrogen refuelling infrastructure has been discussed in various sources [23]. Authors have recently focused on ammonia, among them a review of the countermeasures to reduce the carbon emissions in shipping was performed in [24], where it is suggested among others that ammonia will be commercially viable alternative in the future. Others, presented an overview of the various hydrogen supply chains for the European ammonia production concluding that the production from electrolysis with non-fossil fuels has the highest overall emissions [25]. A review of the alternatives on the production and use of ammonia for hydrogen storage was presented [26]. However, there is a distinctive gap for a review discussing the advantages and challenges of ammonia-powered fuel cells for marine applications. Similarly, the topic of safe operations of ammonia-powered fuel cells has also not been thoroughly discussed, which is extremely important due to ammonia’s particular safety characteristics.
As previously mentioned, there are different alternative marine fuels available. As identified by Lloyd’s Register & UMAS in [27] and DNVGL in [28] the most viable options include hydrogen, ammonia, ethanol methanol, NG and methane. Biodiesel is not taken into consideration as it requires shipboard CO2 storage and related logistics which are time-consuming, complex and expensive to develop. The properties of the considered alternative fuels are shown in Table 1.
| Fuel | Energy Density LHV (MJ/kg) | Volumetric Energy Density (GJ/m3) | Renewable Synthetic Production Cost (MJ/MJ) | Storage Pressure (Bar) | Liquified Storage Temperature (°C) |
|---|---|---|---|---|---|
| Compressed hydrogen | 120 | 4.7 | 1.7 | 700 | 20 |
| Liquid hydrogen | 120 | 8.5 | 1.8 | 1 | −253 |
| Ethanol | 26.7 | 21.1 | 3.6 | 1 | 20 |
| Methanol | 19.9 | 15.8 | 2.6 | 1 | 20 |
| Liquid methane | 50 | 23.4 | 2.3 | 1 | −162 |
| Liquid ammonia | 18.6 | 12.7 | 1.8 | 1 or 10 | −34 or 20 |
Currently, the most competitive alternative to traditional marine fuels is Natural Gas (NG) which consists of more than 90% of methane, therefore the same properties of liquid methane are considered for NG. NG has the highest volumetric density compared to the other fuels (23.7 GJ/m3). Engines operating with natural gas have reduced NOX emissions by 85–90% and almost zero Particulate Matter (PM) emissions. In addition, NG has zero sulphur content and very low carbon content. Dual fuel engines operating with NG are an established technology, however, due to the fossil fuel origin of NG, it is considered that it will serve only as a transitional fuel or as a precursor to the generation of ammonia [29].
Apart from NG, alcohols like methanol and ethanol can be considered as viable fuel alternatives since they both show high volumetric energy density (15.8 and 21.1 GJ/m3, respectively). Methanol and ethanol can be produced from renewable sources and they appear as a promising substitute of marine fuel oils due to their high auto-ignition temperature and low viscosity [30]. In addition, they have negligible sulphur content, half of the NGs carbon content. Moreover, methanol operating marine engines have very low PM and NOX emissions [31]. However, due to the lower heating value of methanol (half of the NG), in order to have the same power output, the amount of fuel required is almost doubled compared to NG.
Hydrogen is a very attractive energy source with zero carbon emissions. It is produced from biomass, electrolysis and more often from NG, it is amply found in the universe, however hardly on its pure form [32]. It has low volumetric energy density, which leads to challenges in storing, hence, the storing technologies play an incremental role in the viable use of hydrogen in shipping [26]. Another limitation of hydrogen is that it has a low energy density (4.7 GJ/m3) in gaseous form compared to liquefied (8.5 GJ/m3), however, the liquefying process is energy consuming [22]. Hydrogen handling also causes serious concerns [33], due to the high flammability and the very low electro-conductivity rating [22]. Generally, there are safety issues regarding the fuel’s volatility that need to be addressed [34] as well as the use of hydrogen in large merchant ships [19]. The successful deployment of hydrogen as a marine fuel suffers limitations also due to the high fuel price. Finally, there are insufficient bunkering infrastructures as well as there is a lack of standardised design and fuelling procedures [35].
Ammonia is also a zero carbon emissions fuel, which if it is derived from renewable sources, can play a significant role as a solution to store renewable energy [36]. Ammonia can be used in fuel cells as well as in ICE [34]. However, due to ammonia’s high nitrogen content, its combustion in high temperatures leads to increased NOX emissions [26]. Ammonia is a widely traded commodity produced in large quantities by the chemical industry, approximately 200 million metric tonnes per year [18] and it is mostly used for fertilisers. Thus, compared to hydrogen, there is an existing extended distribution network [36] as well as available port loading infrastructures and experience in handling [34]. Liquid ammonia has a higher energy density (12.7 GJ/m3) both from liquid and compressed hydrogen [37], which benefits the fuel storage. In general, the storage of ammonia is much less complex than hydrogen [38]. It is usually stored either refrigerated (−34 °C) or under pressure at ambient conditions (20 °C) [18]. Also, ammonia has a flexible utilisation since it can be used as a fuel itself, however, due to the high hydrogen density of approximately 17.8 wt%, it could be used as hydrogen storage [26]. It is estimated that ammonia with a density of 653.1 kg/m3 contains more hydrogen than a cubic meter of liquid hydrogen [39]. Therefore, this avoids the required cryogenic system necessary for the transportation of liquid hydrogen [40], which is very costly [32]. In addition, compared to hydrogen, ammonia is a more cost-efficient option due to both the lower price of the fuel [39] and the fact that there are already existing infrastructures.
Considering the above, a qualitative assessment of the alternative fuels together with an investigation of the relevant literature is necessary. From the discussed alternative fuels NG has the least potential as a long-term solution. This is caused by its uncompetitive characteristics, as it is prone to restrictions and higher prices. Moreover, from the alternative fuels, only methanol, hydrogen and ammonia can be produced from renewable electricity. And in more detail, only hydrogen and ammonia have the potential for zero net carbon emissions [27]. Similarly, it is observed, that ammonia and hydrogen have the lowest renewable synthetic production costs (Table 1). These are very important characteristics as they can influence the sustainability of the respective alternative fuel. From the above, and by also considering the insight from [28] ammonia and hydrogen are the two most promising alternative marine fuels.
For the following steps of the assessment, only hydrogen and ammonia are considered, due to their carbon-free emissions. To better understand their competitiveness, the Capital Expenditure (CAPEX) and Operating Expenditure (OPEX) of a hydrogen and ammonia power system is analysed. The CAPEX and OPEX of the two systems are also benchmarked against a traditional diesel system. In detail, the comparison is performed assuming a 2MW installation, an increasing CO2 tax [41], and a renewable electricity cost of 0.02 €/kWh [42][43]. Figure 1 shows the current, short-term and long-term (2030) CAPEX and OPEX for hydrogen and ammonia. As seen, the CAPEX for ammonia power system is slightly higher than hydrogen, however, it is predicted that this cost will decrease and reach a lower level than hydrogen. This behaviour is attributed to the increasing maturity of the technology and the comparatively reduced complexity of ammonia systems [44]. Similarly, the cost of ammonia fuel is predicted to decrease, owning to the maturity of the technology, with costs sustainably lower than hydrogen [45].
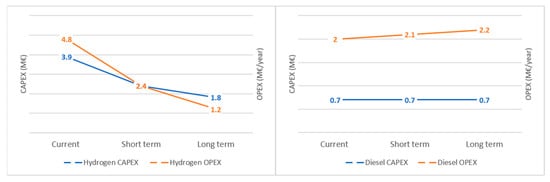
Figure 1. CAPEX and OPEX of hydrogen and ammonia system.
In a stark contrast with the aforementioned fuels, diesel powered systems face increased costs. The CAPEX on diesel powered systems is lower than the renewable counterparts, due to the maturity of the technology and the economies of scale. However, CO2 taxes and the IMO 2050 for decarbonisation will increase the cost of diesel fuel [44][46]. As a result, ammonia powered systems, are predicted to have the most favorable economic performance by 2030.
In addition, the largest benefit of ammonia as fuel, is that it is already a commonly traded commodity with established supply chains and availability in the proximity of ports globally. Therefore, even if the fuelling logistics must be worked out, the fuel prices can be expected to be lower than liquid hydrogen [47]. Also, due to ammonia’s existing infrastructure, there are already regulations and protocols regarding its transportation and handling. Another major benefit of ammonia compared to hydrogen are its superior safety characteristics. In detail, ammonia is not flammable during storage and transportation [26]. Moreover, it can be dissolved in water [47] and gaseous ammonia can be dissipated in the air due to the low density, thus reducing the risk of fire as well as explosion [26]. Furthermore, even though ammonia is toxic, its strong odour helps in identifying leaks [32], mitigating hazardous situations. Lastly, as shown in Table 1, ammonia has a less complex storage and handling systems which reduces operating and purchasing costs.
In the existing literature, the use of ammonia in shipping has been discussed in a limited extent. The role of ammonia as a mean to store the excess renewable energy produced was investigated [33]. In the study, different technologies were assessed, and it was inferred that the combination of ammonia and battery was amongst the most profitable. Also, in [48] a Life Cycle Assessment analysis was performed in order to investigate the environmental impact of hydrogen and ammonia fuelled marine transportation tankers and ships, compared to traditional fuel oil. Results indicated that ammonia can be used for marine engines either as supplementary fuel or as a main fuel leading to significantly lower global warming during ship operation. A techno-economic investigation was performed considering different fuels, including ammonia, hydrogen, natural gas and methanol [27]. From the analysis it was derived that ammonia has the lowest total cost of operation compared to the other fuels. Finally, the potential role of ammonia in long distance shipping was examined and from the results it is concluded that it is a favourable option, however using as a main fuel for a tanker ammonia might have an impact on the increase of the total mass of the ship by a 2.74% to accommodate the changes required [49]. These changes represent the net effect of the removed mass of the relating to heavy fuel oil (e.g., settling tanks, purifiers, heats) and the additional mass associated with the increased space required for ammonia due to its reduced energy density compared to traditional hydrocarbons. Nonetheless, ammonia has a preferable space-to-energy trade-off compared to non-carbon emitting alternative fuels [45].
Apart from using alternative fuels, the IMO decarbonisation targets can be met through the research and implementation of alternative power generations plants. One of the options that is currently investigated is the batteries. However, studies indicate that batteries will face challenges in long-distance shipping due to their size, weight and price [49]. In addition, even though batteries are considered one of the most efficient energy storage technologies, the high cost and low energy density constitutes a limitation [33]. Thus, currently, batteries are not a viable option for primary energy supply in long-distance shipping. Fuel Cells (FCs) present an alternative, promising and innovative technology for electric energy production, which manages to reduce the emissions as well as the noise and improve the energy efficiency of ship energy systems [50]. They are electrochemical conversion devices that output electricity, heat and waste from the chemical reaction of inputs (e.g., air and fuel) [51]. FCs require a constant source of fuel to produce electricity, which is their differentiating factor from the energy storing batteries [52].
There are numerous FC systems for marine applications, which vary in terms of their proprietary technologies, system architecture and used fuels. The selection of the appropriate FC system is not a turn-key solution, as each implementation of FC in ships is application specific. This is because different FC systems have varying operating parameters and requirements. To that end, the choice of the FC system also influences the environmental impact of the system, the hazards of the system and the operating economics [53].
In detail, there are five main FC types applicable to the maritime industry. These are the Low Temperature Polymer Electrolyte Membrane FC (LT-PEMFC), High Temperature Polymer Electrolyte Membrane FC (HT-PEMFC), Phosphoric Acid FC (PAFC), Molten Carbon FC (MCFC) and Solid Oxide FC (SOFC) [51]. LT-PEMFC can deliver high power density, have efficiency of up to 60% and can be directly fed with pure hydrogen [54]. They operate between 65–85 °C (low start-up, high durability), but have less tolerance to fuel impurities [55]. HT-PEMFC have an operating temperature envelope ranging between 120–180 °C [56] and electrical efficiency of up to 60% [54]. Also, HT-PEMFC require additional time to start-up and have a higher tolerance to fuel impurities [55]. PAFC operate at around 150–200 °C and have similar durability and start-up characteristics as HT-PEMFC However, they have low power density, limited durability, 40% electrical efficiency [54] have restricted their commercial shipping applications [57]. MCFCs are often used for large-scale power generation [58] due to their good power density and up to 50% electrical efficiency [54]. They operate between 700–800 °C (limited durability) which allows for the recovery of waste heat from the water. Lastly, SOFC operate between 700–1000 °C (low durability, high start-up) [59] and they can achieve very high power densities [51] and up to 60% electrical efficiency [54]. SOFC can be directly fed with NH3 as fuel and as such, they are deemed by the wider community as a promising candidate for sustainable energy conversion [60].
The discussed characteristics of the examined FCs are summarized in Table 2. As seen, the FC systems are evaluated in terms of their operating temperatures, durability, start-up time, Capital Expenditure (CAPEX), power density and electrical efficiency; criteria distilled from the work of [51][52]. It is observed that SOFC have very good power density and electrical efficiency. Also, due to their high operating temperature, they have simpler fuel feed systems as fuel (e.g., NH3) can be supplied directly without any pre-treatment. SOFC are ideal candidates for hybrid electric systems, and especially for larger vessels [61]. Due these advantages, SOFC for maritime applications have seen increased development.
Table 2. FC types comparison.
| LT-PEMFC | HT-PEMFC | PAFC | MCFC | SOFC | |
|---|---|---|---|---|---|
| Operating Temperature (°C) | 65–85 | 120–180 | 150–200 | 700–800 | 700–1000 |
| Durability | High | Medium | Medium | Low | Low |
| Start-up Time | Low | Medium | Medium | High | Very High |
| CAPEX | High | Medium | High | Low | Medium |
| Complexity | High | Medium | Medium | Medium | Low |
| Power Density | Medium | Medium | Low | High | Very High |
| Electrical Efficiency (%) | 60 | 60 | 40 | 50 | 60 |
From the preceding discussion it is inferred that ammonia is a promising energy carrier with merits regarding the environmental footprint, production, and transportation, while avoiding the challenges associated with hydrogen. In this section, a review of the application and techniques of ammonia powered fuel cells is presented.
In the existing literature there has been great attention on potential catalysts for the ammonia oxidation [62][63][64][65] and specifically for SOFCs [66][67]. The performance of ammonia fuel cell for land-based energy production or for other systems has attracted attention in the literature, as presented in Table 3. Three different operation modes, direct ammonia supply, external decomposition supply and autothermal decomposition supply, of the ammonia SOFC performance were investigated and the stability of the stack was evaluated for up to 1000 h in [66][68] indicating no significant degradation. A direct ammonia FC was investigated and the results indicate that a high peak power density can be used when using an ammonia-tolerant catalysts [69]. An integrated system that recovers waste heat from an ammonia molten alkaline fuel cell was proposed and investigated to meet the different energy demands of a passenger railway transportation, concluding to improved efficiency of the system and zero carbon emissions [70]. In [47] it was demonstrated that ammonia can be used directly on SOFC as the sole source of hydrogen. The results indicated that the performance of the ammonia powered SOFCs were similar to the one operating solely with hydrogen. Two alternative hydrogen carriers, biogas and ammonia, were examined as a potential fuel of SOFCs, achieving high efficiencies and stability [71].
The performance and durability investigation of an ammonia powered solid oxide fuel cell stack was presented in [72] indicating amongst others that ammonia is a promising fuel for SOFCs. The SOFC operation with ammonia was compared in the same conditions with using hydrogen and results in the nominal conditions indicated the same degradation in both cases, also with no detection of ammonia on the off gasses or significant nitrification of anode [36]. Similar results were found on other studies, it was also indicated that no severe deterioration was recognised after a long operating period [73]. The exergy and energy performance of ammonia fuel cells was investigated in [74][75] and the potential of using a blend of ammonia and hydrogen was considered in [74], it can be inferred from the results that the mixture of the two fuels improves the efficiency of the fuel cells. The feasibility of ammonia combined use with biomethane in an internal reforming SOFC was presented in [76] and the overall system demonstrated an efficiency of 48%.
Furthermore, the potential of ammonia powered fuel cells in shipping has been also investigated in the studies presented in Table 3. An economic and environmental comparative assessment of an innovative ammonia powered system with the traditional power systems on a container feeder ship was performed in [32]. The results indicated that ammonia could lead to a carbon-free shipping operation, however, even though SOFC is the most environmentally friendly technology, it experiences a high life cycle cost.
Table 3. Ammonia powered fuel cells.
| Source | Fuels | Power Plant | Technology | |
|---|---|---|---|---|
| [67][69] | Ammonia | Land-based | SOFC | |
| [70] | Ammonia | Vehicle | Direct FC | |
| [71] | Ammonia | Railway | Molten alkaline fuel cell | |
| [36][74] | Ammonia | Hydrogen | Land-based | SOFC |
| [77] | Ammonia | Biomethane | Land-based | SOFC |
| [75] | Ammonia | Hydrogen | Alkaline fuel cells | |
| [72] | Ammonia | Biogas | Land-based | SOFC |
| [76] | Ammonia | vehicle | SOFC | |
| [32] | Ammonia | HFO | Container ship | SOFC |
| PEMFC | ||||
| Diesel engine | ||||
| Diesel electric | ||||
| [73] | Ammonia | hydrogen | Land-based | SOFC |
| [48] | Ammonia | hydrogen | Land-based | SOFC |