1000/1000
Hot
Most Recent

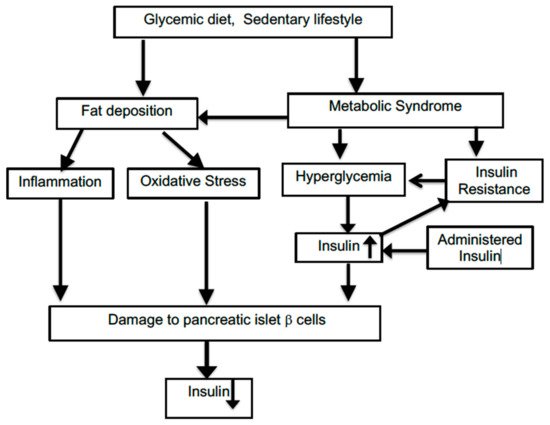
Insulin, via a series of kinase activations and transductions, causes the glucose type 4 transporter channels to become embedded in the cellular membrane, allowing an exponential increase of glucose entry into the cell. T2D is characterized by failure of the insulin receptors to respond to insulin, thus preventing glucose uptake from the bloodstream. Later in the disease, the production of insulin by pancreatic islet cells is also curtailed. The vast preponderance of diabetes cases (95%) in the United States constitute T2D.
| Goal of Treatment | Mechanism Involved | Treatment |
|---|---|---|
| Prevent hyperglycemia | Lower levels of circulating glucose | Insulin |
| Inhibition of the renal sodium-glucose co-transporter-2 (SGLT2) | ||
| Glucagon-like peptide-1 receptor agonists to stimulate insulin release | ||
| Reduced rate of carbohydrate absorption | Diminished consumption of refined or simple carbohydrates | |
| Reduced hepatic gluconeogenesis, increased glucose uptake by muscle | Metformin | |
| Reduction of levels of inflammation and oxidative stress | Increased antioxidant and anti-inflammatory milieu | Activation of Nrf/KEAP/ARE pathway |
| Antioxidant vitamins (e.g., lipoate, α-tocopherol, and ascorbate) | ||
| Use of phytochemicals with a broad range of properties (e.g., curcumin, resveratrol, and catechins) | ||
| Ketogenic diet | ||
| SIRT activation | Rapamycin | |
| Moderation of fat deposition | Lower accumulation of fat | Physical activity |
| Blocking of miR-208a with an antisense oligonucleotide | ||
| Modulation of gut biome | Ketogenic diet |