
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Stefano Stracquadanio | + 4360 word(s) | 4360 | 2021-05-26 05:44:45 | | | |
| 2 | Camila Xu | Meta information modification | 4360 | 2021-05-26 10:37:35 | | |
Video Upload Options
Bacterial internalization is a strategy that non-intracellular microorganisms use to escape the host immune system and survive inside the human body.
1. Background
During the first steps of growth, remodeling and recovery of the bone, different cell types co-exist and cooperate to form the extracellular bone matrix (EBM) [1][2]. Among these, osteoblasts are the cells that form the bone and, together with osteoclasts, preserve its physiological homeostasis [3].
Pathological conditions, such as bacterial infections, are responsible for altered osteoblast activity. In detail, surgical procedures, especially in the presence of medical (orthopedic) devices, are responsible for an increased susceptibility of osteoblasts to osteomyelitis [4][5][6][7] and, in this context, Staphylococcus aureus represents a frequent intra- and extracellular pathogen [8].
The host–pathogen interaction between osteoblasts and S. aureus occurs through the recognition of pathogen-associated molecular patterns (PAMPs) by the pattern recognition receptors (PRRs) exposed on the extracellular surface of the osteoblasts. The consequent production of chemokines and cytokines is responsible for the recruitment and subsequent activation of innate and adaptive immune cells, typical of the cellular inflammatory response [9]. At the same time, the overstimulation of osteoblasts by S. aureus causes an increase in osteoclastogenesis with consequent osteoblast death, as well as an alteration of bone homeostasis (Figure 1) [10][11].
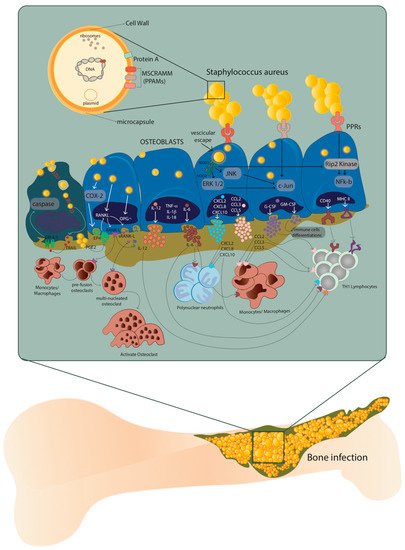
Figure 1. Host–pathogen interaction between osteoblasts and Staphylococcus aureus. After internalization, S. aureus escapes from the vesicle and interacts with extracellular receptors TLR2 and TNFR-1, as well as with intracellular receptors TLR9 and NODs through α5β1 integrin and actin filaments of the osteoblasts. This interaction increases the expression of cytokines IL-1β, IL-18, TNF-α, the production and release of IL-6, IL-12 and the expression and release of chemokines CXCL2, CXCL8, CXCL10, CCL2, CCL3, CCL5 and growth factors G-CSF and GM-CSF. At the same time, the expression and production of CD40 and MHC II increase. Illustration by A. Costantino (co-author).
The presence of proteins and glycans—such as type I collagen, bone sialoprotein, osteopontin and fibronectin—make the EBM a perfect niche for S. aureus that binds these EBM components through multiple adhesins known as microbial surface components which recognize adhesive matrix molecules (MSCRAMMs) [8][12]. Indeed, the S. aureus attachment to the EBM represents a key step in the onset of osteomyelitis, where type I collagen represents approximately 90–95% of the organic fraction of the EBM directly interacting with this pathogen (Figure 1).
Recently, it was demonstrated that the ability of S. aureus to internalize inside osteoblasts is a key strategy to protect itself and maintain the infection. On the contrary, osteoblasts respond to S. aureus internalization by secreting inflammatory factors—such as cytokines, chemokines and growth factors—which, in turn, activate and recruit immune cells from the innate or adaptive immune systems (Figure 1) [13].
The excessive formation and activation of osteoclasts is caused by an increase in RANK-L expression and production through the COX-2/PGE2 pathway. This leads to severe bone resorption as well as to decreased production of osteoprotegerin (OPG). Finally, through the release of membrane-damaging virulence factors such as phenol-soluble modulins (PSMs), S. aureus can cause osteoblast necrosis and apoptosis through intrinsic and extrinsic caspase pathways. These processes can lead to the release of intracellular S. aureus, which can re-infect other osteoblasts.
Consequently, the ability of S. aureus to survive in osteoblasts after internalization also results in effective escape from the antibiotic therapy, which cannot penetrate inside the cells [14][15].
The hypothesis that increasing our understanding of the immune response, as well as intensifying the host’s defenses, could be a valuable avenue for developing new anti-infectious strategies dates back several years [16].
The use of murine or human in vitro culture models—including primary cells, induced osteoblasts from pluripotent stem cells and immortalized and malignant cell lines—has allowed a better understanding of osteoblast cell biology during infection processes [17].
To date, the progress of research in the field of orthopedic engineering, as well as the development of new therapies and biomaterials, increases the importance of these in vitro models. At the same time, a deeper knowledge of their phenotypic and genotypic status and their differences in relation to primary human osteoblast cells is needed, especially in order to choose the most appropriate experimental model.
In this regard, the results obtained from in vitro infections of osteoblasts grown as two-dimensional (2D) monolayers provided important information on the molecular mechanisms underlying bacterium–host cell interactions. Despite this, these models do not reproduce the dynamic aspects of this interaction, such as the organization of osteoblasts in healthy bone to provide strength and resistance and therefore to respond better to bacterial infections [18][19]. To overcome these limitations, animal models of osteomyelitis were considered the gold standard for the study of bone infections, but the different responses to bacteria between the mouse model and other animal models made the use of these models not exhaustive [20][21][22][23]. From here, more relevant in vitro models that physiologically mimic the human bone microenvironment have been developed and will be discussed in the last section of this review.
S. aureus vs. Mycobacterium tuberculosis
Before delving into different aspects of S. aureus internalization, a comparison between the behavior of S. aureus and that of an obligate intracellular bacterium, such as Mycobacterium tuberculosis—the etiological agent of tuberculosis (TBC)—could be useful.
While M. tuberculosis needs to replicate within human cells to disseminate to other individuals and cause disease, internalization of S. aureus by osteoblasts is a key element in the spread of the infection, as it allows S. aureus to persist inside osteoblasts protected from the immune system and it gives S. aureus the opportunity to sustain the infection [3], but it is not necessary for its replication.
M. tuberculosis spreads from person to person almost exclusively by aerosolized particles that can be trapped in the upper airway or oropharynx. Once in the lower respiratory tract, M. tuberculosis is primarily phagocytosed by macrophages and dendritic cells, but neutrophils are also infected [24]. Although M. tuberculosis usually infects macrophages, it was also found in non-myelocytic cells of TBC patient. As M. tuberculosis internalization in non-phagocytic cells is an actin-dependent process involving heparin-binding hemagglutinin, toll-like receptors (TLRs), surfactant proteins and complement and scavenger receptors [25], S. aureus internalization involves some cytoskeletal elements too, particularly actin microfilaments. It has been demonstrated that the internalization process of S. aureus can occur with dead bacteria but not with dead osteoblasts, suggesting that the internalization process is more of an active cellular mechanism than an active bacterial mechanism [26].
After M. tuberculosis or S. aureus internalization, infected cells trigger a local inflammatory response that attracts immune cells to the site of infection. Osteoblasts infected by S. aureus secrete inflammatory factors like cytokines, chemokines and growth factors, all of which can activate and recruit immune cells from the innate or adaptive immune systems [27], while M. tuberculosis promotes the buildup of cellular aggregates forming the granulomas, that represent a complex environment constituted by macrophages, multinucleated giant cells, epithelioid and foamy cells, granulocytes and lymphocytes [25].
We only reported the main common characteristics regarding the internalization process of both M. tuberculosis and S. aureus, as M. tuberculosis internalization in phagocytic cells needs more tricks to allow the bacteria to escape the phagosomal threat.
2. MG-63 Osteoblast-Like Cell Line as an Effective In Vitro Model to Investigate Host–Pathogen Mechanisms during S. aureus Infection
Although preclinical models are known to offer an essential prescreening method for testing new biomaterials useful in the treatment of orthopedic disorders, the increasingly restrictive regulations for the use of in vivo models and the ever-increasing demand for primary cells from healthy or sick donors have led to the development of “continuous” osteoblast cell models. Among these, human- and animal-derived primary cells [28][29][30][31][32], immortalized cell lines [33][34], malignant cells [35][36][37], and induced pluripotent stem cells (iPSs) are used not only in drug and biomaterial testing, but also in bone biology investigations.
Certainly, primary cells, deriving directly from patients, better reproduce the behavior of the original cellular niche, resulting in a preclinical model closer to clinical conditions. Over the years, however, researchers and physicians have realized that the phenotypic and often genotypic differences of these cells isolated from different patients make it difficult to study the disease under examination and consequently to establish the best therapeutic strategy.
An improvement in the knowledge of bone biology and, in particular, osteoblast cells has been achieved through the development of stabilized osteoblast cell lines as models for in vitro investigation of cell differentiation, cytokines and hormone regulation, matrix protein synthesis and secretion and molecular mechanisms of bone diseases. At the same time, these models were found to be useful for the evaluation of the cytocompatibility and osteogenicity of new biomaterials [17].
There is no evidence indicating the superiority of one model over the others; therefore, an evaluation of their respective advantages and disadvantages, on the basis of studies to be conducted, is important.
Primary cultures represent an in vitro model that uses cells directly obtained from tissue biopsies (~1 cm3) or organ dissections. These cells have the unique characteristic of maintaining their genetic, morphological and functional features. This makes them the best representative indicators of normal cell phenotype and early-stage disease progression, and as such they are commonly used as in vitro tools for preclinical and investigative biological research and toxicological studies, besides reducing the number of animals required for preclinical toxicology studies at an initial stage, making them cost-saving.
Even though primary human osteoblasts tend to preserve their differentiated phenotype in vitro, after a certain number of cell divisions, these cells have a limited lifespan and will stop dividing (or senesce) and may be more difficult to grow and maintain than a continuous (immortalized) cell line. Induced variability in primary cells obtained from donors and in subculture practices is one of the main challenges faced by researchers studying cell signaling pathways [17].
Specifically, it has been shown that the age of the donor influences the proliferative capacity of isolated cells, whose proliferation times are doubled if derived from patients over the age of 65 [38][39].
Furthermore, bone aging, defined as a change in the degree and distribution of bone mineralization, is also age-dependent. This is reflected in the physiology of isolated osteoblast cells, characterized not only by slower proliferation, but also by phenotypic modification [40][41]. Likewise, the expression of genes and the synthesis of proteins associated with the osteoblast phenotype are also influenced by the age of the donor, as well as by the anatomical site of isolation. For example, it is now known that FGFβ and IGFII gene expression is downregulated in osteoblast cells isolated from the mandible, and the synthesis of type I collagen and osteonectin shows higher expression in cells isolated from fetal to 20-year-old bone donors, while a 65% decrease in collagen levels was observed in cells from donors older than 20 years [42].
Moreover, when these cells are extracted ex vivo and transferred to a culture environment, they may lose their structural and functional characteristics. In this regard, cells having completely different morphology in vivo at the tissue level may show similar morphology in the culture environment [43][44].
Thus, donor age, site of isolation and the gender differences that we have discussed so far are just some of the factors that can influence the behavior of primary human osteoblast cells and, in turn, confer different times of phenotypic modification in vitro. As a result, in the absence of a homogenous target of patients/donors, obtaining cultures of osteoblasts suitable for the study of basic applied biology or particular mechanisms, such as infection, is not efficient.
The ease of obtaining results in experiments and their repeatability, as well as the ease of maintenance, the unlimited number of cells without the need for isolation and the relative phenotypic stability of immortalized or continuous MG-63 cell lines has allowed researchers, in some respects, to overcome the limits imposed by cells primarily derived from the bone (HObs). Although these cell models differ in some respects from primary osteoblast cells, Czekanska et al. showed that MG-63 cells show some distinct similarities to HObs [45].
According to Billiau et al., the MG-63 cell line is derived from a juxtacortical osteosarcoma diagnosed in the distal diaphysis of the left femur of a 14-year-old male [35]. When cultured, these cells appear as rapidly proliferating aggregates without exhibiting contact inhibition [46].
The similarity between MG-63 and HObs was already studied several years ago, when Franceschi et al. observed the response of these cells to 1,25-dihydroxyvitamin D3 (1,25 (OH) 2D3) as an effect on cell morphology and on the phenotype comparable to normal osteoblasts [47].
More recent studies have shown that the cell growth kinetics of MG-63 were comparable to that of HObs as the exponential growth phase was observed from day 2 to day 6, followed by a plateau phase from day 6 to day 10 [45]. This result is confirmed by the ability of infinite proliferation typical of malignant cells, such as MG-63 cells, where the lack of intrinsic cell cycle control contributes to tumor progression.
Czekanska et al. also observed that the activity of alkaline phosphatase, an enzyme identifying mature osteoblasts, was lower in MG-63 cells than in primary cells [45]. This result confirmed the different degree of differentiation of MG-63 towards a more immature phenotype, compared to HObs.
The expression analysis of key osteoblast-specific genes [45] showed that the transcription factor Runx2—which regulates gene expression of the all-important bone matrix proteins (including ALP, OC, BSP and type I collagen)—is expressed more in MG-63 than in HObs, except on day 2 [48][49].
As previously introduced, type I collagen is essential for the function of osteoblast cells [50] and is overexpressed in the phase preceding matrix mineralization [51][52], in order to allow the formation of fibrils and a subsequent physiological maturation of the matrix. On the contrary, MG-63 cells show a low expression of type I collagen as well as of osteocalcin [45]. Consequently, the reported studies highlight the limitations of these cells as a model for the phenotypic development of osteoblasts as well as for the evaluation of the mineralization of the matrix and the properties of new biomaterials [45].
On the other hand, the MG-63 cell line proved to be a valid in vitro model for the study of bacterial infection mechanisms, especially during S. aureus internalization [14][53][54][55][56][57]. In 2010, Schroder and Tschopp demonstrated that the innate immune response against pathogens involves the activation of an inflammatory pathway known as the inflammasome activation pathway [58]. Inflammasomes are multiprotein signaling complexes that are assembled following the recognition of stress/pathogenic signals; among these, caspase-1 is the most involved [59]. Upon stimulation by pathogens, caspase-1 binds to an adapter molecule known as apoptosis- associated speck-like protein (ASC) [60]. This binding leads to the autocatalytic cleavage of caspase-1, the processing of pro-IL-1β and pro-IL-18 and the secretion of mature IL-1β and IL-18, triggering in some cases even an inflammatory form of cell death (pyroptosis) [61]). Recent studies have shown that this also applies to S. aureus and MG-63 cells [62][63].
Finally, in previously published works, we demonstrated that internalization, using MG-63 cells, is a pathophysiological pathway of some methicillin-resistant S. aureus (MRSA) which depends on the total number of cells infected and not on the number of bacterial cells that enter each osteoblast. Furthermore, even if our strains were not homogeneous in terms of genetic backgrounds and virulence factors, ST22-IVh and ST239-III S. aureus showed higher intracellular persistence in host cells, making them more prone to developing chronic and recurrent infections, and the different genetic background was also accompanied by a different modulation of inflammatory phenomena, metabolism and antioxidant machinery [64][65].
Take home message: We can conclude that although primary cell lines, and in particular HObs, have the advantage of maintaining their genetic, morphological and physiological features, they also have a limited life span and are difficult to grow and maintain in continuous cultures. MG-63, in our experience, is also a valid in vitro model for the study of S. aureus internalization and persistence, both as regards the mechanisms underlying the ability of S. aureus to adhere, invade and persist within osteoblasts and the host cell response to infection.
3. Multiplicity of Infection (MOI) and Invasiveness of Different Bacterial Strains
In the presence of prosthetic devices, complete eradication of bacterial infection is often a challenging task. Internalization in non-professional phagocytes is an important pathogenic mechanism actuated by bacteria to elude host defenses and medical therapies. The efficiency of invasion differs across bacterial species and adjustments to the titer of the microbial inocula used in the assay are often needed to enumerate intracellular bacteria.
There is a precise relation between the inoculum, in terms of multiplicity of infection (MOI), and the internalized bacteria. Furthermore, there is a relationship between MOI, internalized bacterium ratio and medical therapies [66][67].
Intracellular invasion occurs through a variety of pathogenic species. Some bacteria are obligate intracellular pathogens, while other only become intracellular to escape the host immune system. Among these, the following genera are the most representative: Mycobacterium; Escherichia; Salmonella; Listeria; Shigella; Legionella; Chlamydia; Yersinia; Streptococcus; Staphylococcus and Enterococcus (in particular E. faecalis). S. aureus is the only one capable of causing the onset of clinically relevant pathogenic mechanisms and consistently invades osteoblast cells [68].
Different MOIs have been adopted depending on the bacterial species tested. For example, an MOI of 100:1 (bacteria:host cells) is the inoculum titer most frequently used to test S. aureus. The inoculum level increases to 500:1 for S. epidermidis [55][69] and 1000:1 for S. lugdunensis [70]. The different MOIs used showed rapid and efficient internalization of S. aureus at low inoculum levels and inefficient internalization of other species at high inoculum levels.
Two other parameters are used to express the potential of invasiveness of bacterial strains: (i) the number of internalized bacteria (NIB) at an established MOI, expressed in term of Colony-Forming Units (CFUs) per number of eukaryotic cells, influenced by the MOI used; and (ii) the percentage of internalized bacteria (PIB), which represents the percent fraction of the inoculum taken up by eukaryotic cells; this value is not affected by the MOI and can be used to express the degree of invasiveness of prokaryotic cells into eukaryotic cells.
Examining the correlation between MOI values and PIB values, it emerged that—over a broad range of inoculum levels—the MOI did not appear to affect PIB values [71]. PIB values can be used to compare strain invasiveness without fearing major effects resulting from varying MOIs.
However, a new parameter was proposed to express the invasiveness of bacterial strains: the internalization minimal inoculum (IMI), corresponding to the lowest MOI required for the internalization of a single bacterium. This value is inversely related to invasiveness and corresponds to the lowest concentration at which internalization occurs under the test condition used. Internalization at a 1:1 MOI inoculum (l1M) corresponds to the number of bacteria internalized when hypothetically exposing each eukaryotic cell to a single bacterium (i.e., using a 1:1 MOI). Its value is proportional to the degree of invasiveness of the strain given by the log10 of the IMI value (LIMI), obtained from the regression curve of log MOI vs. log (CFU).
In conclusion, the most used parameters to express the intracellular invasiveness of bacterial strains are the NIB and PIB values. PIB values can be used across a broad range of MOIs without fearing the influence of the inoculum size. Ultimately, PIB values do not depend on the MOI, whereas NIB values are strongly MOI-dependent.
Therefore, we can speculate that the internalization process can be influenced by several factors, such as: (i) bacterial sedimentation rate, influenced by the microbe size, shape and tendency to agglomerate and by the viscosity of the medium; (ii) composition of the culture medium; (iii) cell line used in the assay, considering its histological origin, phagocytic activity (professional or non-professional phagocytic cells in primary or secondary cultures), level of expression of integrins capable of interacting with adhesins; (iv) bacterial strain type (bacterial species and genes encoding for invasiveness).
As already mentioned, different MOIs have been used in several studies depending on the strains tested. These include e.g., S. aureus, S. epidermidis and S. lugdunensis, opportunistic pathogens causing implant-related infections.
These species can survive antibiotic therapies through different mechanisms related to genetic determinants, biofilm production and penetration into eukaryotic cells as the main causes of chronic infections. Furthermore, eukaryotic cells are impermeable to many antibiotics, such as rifampin, that are able to pass through the prokaryotic cell membrane [72][73].
Many staphylococcal species, other than S. aureus, are emerging as opportunistic pathogens capable of causing serious and persistent implant-associated infections. According to some authors, S. epidermidis is the foremost isolated staphylococcal species responsible for orthopedic infections and is able to internalize into osteoblasts, whilst others report S. epidermidis as the second staphylococcal species isolated during orthopedic infections (Khalil et al. 2007; Valour et al. 2013), but its antimicrobial resistance profile is usually not as severe as that of S. aureus. S. lugdunensis is an emergent pathogen responsible for periprosthetic infections [74].
The invasive potential of different bacterial species and their ability to internalize into the MG-63 cell line was evaluated using a method based on microtiter plates, where they were challenged with osteoblasts.
Campocia et al., in 2016, used different MOIs for each Staphylococcus species tested. The MOI value was always recorded in order to know the inoculum levels actually reached: MOI 560:1 for S. epidermidis and MOI 1844:1 for S. lugdunensis.
S. epidermidis has an extremely low rate of internalization, not comparable with that observed for S. aureus. Furthermore, the bacterial survival rate appeared rather marginal. Most S. epidermidis tested with MOI 500:1 showed a relatively low internalization (<50 CFUs), while other strains showed high internalization (>50 CFUs).
Some groups of bacteria appear homogeneous in terms of CFUs internalized regardless of the inoculum level, while others exhibit some heterogeneity in spite of similar inoculum levels. S. lugdunensis showed very low levels of internalization regardless of the level of inoculum, even though it was tested with a relatively high MOI (1000:1).
The species considered exhibited marginal rates of internalization compared to S. aureus, since S. aureus showed a higher rate of internalization at a lower MOI (100:1).
S. aureus requires a very low inoculum to reach a high internalization rate, whereas S. epidermidis cell invasiveness remains low and marginal. This finding suggests that the active mechanisms of invasion exhibited by S. aureus are either absent or much less efficient in S. epidermidis. Similarly, the clinical isolates of S. lugdunensis showed a low level of internalization (Figure 2) [70].
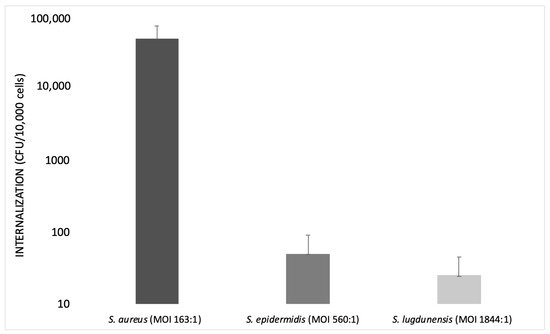
Figure 2. Bar graph illustrating the internalization of S. aureus, S. epidermidis and S. lugdunensis at different multiplicities of infection (MOIs) on a logarithmic scale (modified from Campocia et al., 2016).
To confirm the use of diverse MOIs in the internalization process, studies performed by Valour et al. (2013) compared the different behavior of S. aureus and S. epidermidis during internalization. As previously shown, S. epidermidis has a low rate of internalization, being an innocuous commensal of the human skin and mucous membranes, but it is considered a leading opportunistic pathogen [70].
The contrast between the low incidence of S. epidermidis orthopedic device infection and the highly prevalent S. epidermidis carriage suggests that S. epidermidis bone and joint infections may either correspond to accidental events due to colonizing strains or to a specific, more virulent sub-population of commensal isolates.
Two predominant mechanisms have been proposed to be involved in orthopedic device infections, i.e., bacterial invasion and persistence in non-professional phagocytes, such as osteoblasts [75]; and the bacterial ability to form biofilm [76][77].
To verify the ability of internalization of S. epidermidis into the MG-63 cell line, an invasion assay of S. epidermidis was carried out using an MOI of 500:1 for S. epidermidis and an MOI of 100:1 for S. aureus (used as a control strain).
The results of this assay demonstrated that the number of internalized bacteria was MOI-dependent. There was a difference in bone cell invasion rates between S. epidermidis and S. aureus strains. S. epidermidis showed a lower rate of internalization. This could be due to several factors, such as the “cell line effect”—i.e., the use of the MG-63 cell line—and the acquisition of some phenotypic characteristics that may not reflect the in vivo reality [78].
To exclude a bias due to a “cell line effect”, the low internalization rate of S. epidermidis was confirmed using primary bone cells. For this reason, all assays were repeated using primary human osteoblasts [69].
Fibronectin-binding protein-like molecules are absent in S. epidermidis, therefore, the process of invasion is different from that of S. aureus.
Finally, the internalization of S. epidermidis in human osteoblasts is not a common pathophysiological mechanism in orthopedic device infections, contrary to what was observed in other clinical situations or with other strains (e.g., S. aureus).
MOI Values Were Selected Depending on the Strain Used and Several other Factors
In general, the best choice is to use as few bacteria as possible to reduce cell damage, as important strain-dependent differences may be missed if extended incubation periods or large inocula are used [79].
Hamza et al. (2014) performed an infection experiment using rat osteoblasts and S. aureus at different MOIs over different incubation times. They found that intracellular CFUs increased from MOI 100 to MOI 500 and that MOIs greater than 500 did not result in an increase in intracellular CFUs. Osteoblast viability did not change significantly in an MOI range of 100–1000. As a result, high intracellular CFUs and high osteoblast viability were reached at MOI 500 [80].
In the study carried out by Bongiorno et al. (2020), the frequency of internalization was evaluated in a cell culture model of infection using S. aureus and MG-63 osteoblasts at an MOI of 100:1. In order to assess this MOI, they first tested MG-63 infection with S. aureus ATCC 12598 at the following MOIs: 12:1, 50:1, 100:1 and 200:1. It was observed that, at MOI 12 and 50, the ability of S. aureus to internalize into non-specialized cells, such as the osteoblasts, was very limited, while with an MOI of 200, MG-63 cultured cells showed phenomena of cytotoxicity [64].
Take home message: Taken together, this information suggests that researchers should choose the right MOI carefully when designing an internalization experiment, strictly depending on the bacterial species (sometimes even the clone) and the cell line. Higher is not always better. It is critical to know if the bacteria are obligate or opportunistic intracellular species and if the cells are professional phagocytes. Furthermore, internalization experiments should consider other, less used, parameters, i.e., number of internalized bacteria (NIB), percentage of internalized bacteria (PIB) and internalization minimal inoculum (IMI), as these can help researchers to better describe and compare their results.
References
- Lew, D.P.; Waldvogel, F.A. Osteomyelitis. Lancet 2004, 364, 369–379.
- Hudson, M.C.; Ramp, W.K.; Nicholson, N.C.; Williams, A.S.; Nousiainen, M.T. Internalization of Staphylococcus aureus by cultured osteoblasts. Microb. Pathog. 1995, 19, 409–419.
- Josse, J.; Velard, F.; Gangloff, S.C. Staphylococcus aureus vs. Osteoblast: Relationship and Consequences in Osteomyelitis. Front. Cell Infect. Microbiol. 2015, 5, 85.
- Montanaro, L.; Speziale, P.; Campoccia, D.; Ravaioli, S.; Cangini, I.; Pietrocola, G.; Giannini, S.; Arciola, C.R. Scenery of Staphylococcus implant infections in orthopedics. Future Microbiol. 2011, 6, 1329–1349.
- Kim, P.H.; Leopold, S.S. Inbrief: Gustilo-Anderson classification. [corrected]. Clin. Orthop. Relat. Res. 2012, 470, 3270–3274.
- Hogan, A.; Heppert, V.G.; Suda, A.J. Osteomyelitis. Arch. Orthop. Trauma Surg. 2013, 133, 11831196.
- Shi, S.; Zhang, X. Interaction of Staphylococcus aureus with osteoblasts (Review). Exp. Ther. Med. 2012, 3, 367–370.
- Heilmann, C. Adhesion Mechanisms of Staphylococci. Advances in Experimental Medicine and Biology; Linke, D., Goldman, A., Eds.; Springer: Dordrecht, The Netherlands, 2011; Volume 715, pp. 105–123.
- Claro, T.; Widaa, A.; O’Seaghdha, M.; Miajlovic, H.; Foster, T.J.; O’Brien, F.J.; Kerrigan, S.V. Staphylococcus aureus protein A binds to osteoblasts and triggers signals that weaken bone in osteomyelitis. PLoS ONE 2011, 6, e18748.
- Tucker, K.A.; Reilly, S.S.; Leslie, C.S.; Hudson, M.C. Intracellular Staphylococcus aureus induces apoptosis in mouse osteoblasts. Fems Microbiol. Lett. 2000, 186, 151–156.
- Widaa, A.; Claro, T.; Foster, T.J.; O’Brien, F.J.; Kerrigan, S.W. Staphylococcus aureus protein a plays a critical role in mediating bone destruction and bone loss in osteomyelitis. PLoS ONE 2012, 7, e40586.
- Wen, Q.; Gu, F.; Sui, Z.; Su, Z.; Yu, T. The Process of Osteoblastic Infection by Staphylococcus aureus. Rev. Int. J. Med. Sci. 2020, 17, 1327–1332.
- Turner, M.D.; Nedjai, B.; Hurst, T.; Pennington, D.J. Cytokines and chemokines: At the crossroads of cell signalling and inflammatory disease. Bio-Chim. Biophys. Acta 2014, 1843, 2563–2582.
- Valour, F.; Trouillet-Assant, S.; Riffard, N.; Tasse, J.; Flammier, S.; Rasigade, J.P.; Chidiac, C.; Vandenesch, F.; Ferry, T.; Laurent, F. Antimicrobial activity against intraosteoblastic Staphylococcus aureus. Antimicrob. Agents Chemother. 2015, 59, 2029–2036.
- Spellberg, B.; Lipsky, B.A. Systemic antibiotic therapy for chronic osteomyelitis in adults. Clin. Infect. Dis. 2012, 54, 393–407.
- Valour, F.; Rasigade, J.P.; Trouillet-Assant, S.; Gagnaire, J.; Bouaziz, A.; Karsenty, J.; Lacour, C.; Bes, M.; Lustig, S.; Bénet, T.; et al. Delta-toxin production deficiency in Staphylococcus aureus: A diagnostic marker of bone and joint infection chronicity linked with osteoblast invasion and biofilm formation. Clin. Microbiol. Infect. 2015, 21, 568.e1–568.e11.
- Czekanska, E.M.; Stoddart, M.J.; Richards, R.G.; Hayes, J.S. In search of an osteoblast cell model for in vitro research. Eur. Cells Mater. 2012, 24, 1–17.
- Baker, B.M.; Chen, C.S. Deconstructing the third dimension: How 3D culture microenvironments alter cellular cues. J. Cell Sci. 2012, 125 Pt 13, 3015–3024.
- Fitzgerald, K.A.; Malhotra, M.; Curtin, C.M.; O’ Brien, F.J.; O’ Driscoll, C.M. Life in 3D is never flat: 3D models to optimize drug delivery. J. Control. Release 2015, 215, 39–54.
- Mestas, J.; Hughes, C.C.W. Of Mice and Not Men: Differences between Mouse and Human Immunology. J. Immunol. 2004, 172, 2731–2738.
- Patel, M.; Rojavin, Y.; Jamali, A.A.; Wasielewski, S.J.; Salgado, C.J. Animal Models for the Study of Osteomyelitis. Semin. Plast. Surg. 2009, 23, 148–154.
- Reizner, W.; Hunter, J.G.; O’ Malley, N.T.; Southgate, R.D.; Schwarz, E.M.; Kates, S.L. A systematic review of animal models for Staphylococcus aureus osteomyelitis. Eur. Cells Mater. 2014, 27, 196–212.
- Shanks, N.; Greek, R.; Greek, J. Are animal models predictive for humans? Philos. Ethics Hum. Med. 2009, 4, 2.
- Eum, S.Y.; Kong, J.H.; Hong, M.S.; Lee, Y.J.; Kim, J.H.; Hwang, S.H.; Cho, S.N.; Via, L.E.; Barry, C.E., 3rd. Neutrophils are the predominant infected phagocytic cells in the airways of patients with active pulmonary TB. Chest 2010, 137, 122–128.
- Lerner, T.R.; Borel, S.; Gutierrez, M.G. The innate immune response in human tuberculosis. Cell Microbiol. 2015, 17, 1277–1285.
- Jevon, M.; Guo, C.; Ma, B.; Mordan, N.; Nair, S.P.; Harris, M.; Henderson, B.; Bentley, G.; Meghji, S. Mechanisms of internalization of Staphylococcus aureus by cultured human osteoblasts. Infect. Immun. 1999, 67, 2677–2681.
- Horn, J.; Stelzner, K.; Rudel, T.; Fraunholz, M. Inside job: Staphylococcus aureus host-pathogen interactions. Int. J. Med. Microbiol. 2018, 308, 607–624.
- Abe, Y.; Aida, Y.; Abe, T.; Hirofuji, T.; Anan, H.; Maeda, K. Development of mineralized nodules in fetal rat mandibular osteogenic precursor cells: Requirement for dexame-thasone but not for beta-glycerophosphate. Calcif. Tissue Int. 2000, 66, 66–69.
- Bellows, C.G.; Aubin, J.E.; Heersche, J.N.; Antosz, M.E. Mineralized bone nodules formed in vitro from enzymatically released rat calvaria cell populations. Calcif. Tissue Int. 1986, 38, 143–154.
- Cao, X.Y.; Yin, M.Z.; Zhang, L.N.; Li, S.P.; Cao, Y. Establishment of a new model for culturing rabbit osteoblasts in vitro. Biomed. Mater. 2006, 1, L16–L19.
- Ecarot-Charrier, B.; Glorieux, F.H.; van der Rest, M.; Pereira, G. Osteoblasts isolated from mouse calvaria initiate matrix mineralization in culture. J. Cell Biol. 1983, 96, 639–643.
- Collignon, H.; Davicco, M.J.; Barlet, J.P. Isolation of cells from ovine fetal long bone and characterization of their osteoblastic activities during in vitro mineralization. Arch. Physiol. Biochem. 1997, 105, 158–166.
- Wang, D.; Christensen, K.; Chawla, K.; Xiao, G.; Krebsbach, P.H.; Franceschi, R.T. Isolation and characterization of MC3T3-E1 preosteoblast; subclones with distinct in vitro and in vivo differentiation/mineralization potential. J. Bone Miner. Res. 1999, 14, 893–903.
- Harris, S.A.; Enger, R.J.; Riggs, B.L.; Spelsberg, T.C. Development and characterization of a conditionally immortalized human fetal osteoblastic cell line. J. Bone Miner. Res. 1995, 10, 178–186.
- Billiau, A.; Edy, V.G.; Heremans, H.; Van, D.J.; Desmyter, J.; Georgiades, J.A.; De, S.P. Human interferon: Mass production in a newly established cell line, MG-63. Antimicrob. Agents Chemother. 1977, 12, 11–15.
- Rodan, S.B.; Imai, Y.; Thiede, M.A.; Wesolowski, G.; Thompson, D.; Bar-Shavit, Z.; Shull, S.; Mann, K.; Rodan, G.A. Characterization of a human osteosarcoma cell line (Saos-2) with osteoblastic properties. Cancer Res. 1987, 47, 4961–4966.
- Ponten, J.; Saksela, E. Two established in vitro cell lines from human mesenchymal tumors. Int. J. Cancer 1967, 2, 434–447.
- Voegele, T.J.; Voegele-Kadletz, M.; Esposito, V.; Macfelda, K.; Oberndorfer, U.; Vecsei, V.; Schabus, R. The effect of different isolation techniques on human osteoblast-like cell growth. Anticancer Res. 2000, 20, 3575–3581.
- Jonsson, K.B.; Frost, A.; Nilsson, O.; Ljunghall, S.; Ljunggren, O. Three isolation techniques for primary culture of human osteoblast-like cells: A comparison. Acta Orthop. Scand. 1999, 70, 365–373.
- Bonjour, J.P.; Theintz, G.; Buchs, B.; Slosman, D.; Rizzoli, R. Critical years and stages of puberty for spinal and femoral bone mass accumulation during adolescence. J. Clin. Endocrinol. Metab. 1991, 73, 555–563.
- Bergot, C.; Wu, Y.; Jolivet, E.; Zhou, L.Q.; Laredo, J.D.; Bousson, V. The degree and distribution of cortical bone mineralization in the human femoral shaft change with age and sex in a microradiographic study. Bone 2009, 45, 435–442.
- Fedarko, N.S.; Vetter, U.K.; Weinstein, S.; Robey, P.G. Age-related changes in hyaluronan, proteoglycan, collagen, and osteonectin synthesis by human bone cells. J. Cell Physiol. 1992, 151, 215–227.
- Kasperk, C.; Wergedal, J.; Strong, D.; Farley, J.; Wangerin, K.; Gropp, H.; Ziegler, R.; Baylink, D.J. Human bone cell phenotypes differ depending on their skeletal site of origin. J. Clin. Endocrinol. Metab. 1995, 80, 2511–2517.
- Martinez, M.E.; Del Campo, M.T.; Medina, S.; Sanchez, M.; Sanchez-Cabezudo, M.J.; Esbrit, P.; Martinez, P.; Moreno, I.; Rodrigo, A.; Garces, M.V.; et al. Influence of skeletal site of origin and donor age on osteoblastic cell growth and differentiation. Calcif. Tissue Int. 1999, 64, 280–286.
- Czekanska, E.M.; Stoddart, M.J.; Ralphs, J.R.; Richards, R.G.; Hayes, J.S. A phenotypic comparison of osteoblast cell lines versus human primary osteoblasts for Biomaterials testing. J. Biomed. Mater. Res. A 2014, 102, 2636–2643.
- Heremans, H.; Billiau, A.; Cassiman, J.J.; Mulier, J.C.; de Somer, P. In vitro cultivation of human tumor tissues. II. Morphological and virological characterization of three cell lines. Oncology 1978, 35, 246–252.
- Franceschi, R.T.; Romano, P.R.; Park, K.Y. Regulation of type I collagen synthesis by 1,25-dihydroxyvitamin D3 in human osteosarcoma cells. J. Biol. Chem. 1988, 263, 18938–18945.
- Schinke, T.; Karsenty, G. Transcriptional Control of Osteoblast Differentiation and Function. Principles of Bone Biology; Academic Press: London, UK, 2002; pp. 83–92.
- Ducy, P.; Zhang, R.; Geoffroy, V.; Ridall, A.L.; Karsenty, G. Osf2/Cbfa1: A transcriptional activator of osteoblast differentiation. Cell 1997, 89, 747–754.
- Wenstrup, R.J.; Witte, D.P.; Florer, J.B. Abnormal differentiation in MC3T3-E1 preosteoblasts expressing a dominant-negative type I collagen mutation. Connect. Tissue Res. 1996, 35, 249–257.
- Ryoo, H.M.; Hoffmann, H.M.; Beumer, T.; Frenkel, B.; Towler, D.A.; Stein, G.S.; Stein, J.L.; van Wijnen, A.J.; Lian, J.B. Stage-specific expression of Dlx-5 during osteoblast differentiation: Involvement in regulation of osteocalcin gene expression. Mol. Endocrinol. 1997, 11, 1681–1694.
- Kirkham, G.; Cartmell, S. Genes and Proteins Involved in the Regulation of Osteogenesis. Top. Tissue Eng. 2007, 3, 3270–3274.
- Ahmed, S.; Meghji, S.; Williams, R.J.; Henderson, B.; Brock, J.H.; Nair, S.P. Staphylococcus aureus fibronectin binding proteins are essential for Internalization by osteoblasts but do not account for differences in intracellular levels of bacteria. Infect. Immun. 2001, 69, 2872–2877.
- Nair, S.P.; Bischoff, M.; Senn, M.M.; Berger Bächi, B. The sigma B regulon influences internalization of Staphylococcus aureus by osteoblasts. Infect. Immun. 2003, 71, 4167–4170.
- Khalil, H.; Williams, R.J.; Stenbeck, G.; Henderson, B.; Meghji, S.; Nair, S.P. Invasion of bone cells by Staphylococcus epidermidis. Microbes Infect. 2007, 9, 460–465.
- Testoni, F.; Montanaro, L.; Poggi, A.; Visai, L.; Campoccia, D.; Arciola, C.R. Internalization by osteoblasts of two Staphylococcus aureus clinical Isolates differing in their adhesin gene pattern. Int. J. Artif. Organs 2011, 34, 789–798.
- Jauregui, C.E.; Mansell, J.P.; Jepson, M.A.; Jenkinson, H.F. Differential interactions of Streptococcus gordonii and Staphylococcus aureus with cultured osteoblasts. Mol. Oral Microbiol. 2013, 28, 250–266.
- Schroder, K.; Tschopp, J. The inflammasomes. Cell 2010, 140, 821–832.
- Lamkanfi, M.; Dixit, V.M. The Inflammasomes. PLoS Pathog. 2009, 5, e1000510.
- Mariathasan, S.; Newton, K.; Monack, D.M.; Vucic, D.; French, D.M.; Lee, W.P.; Roose-Girma, M.; Erickson, S.; Dixit, V.M. Differential activation of the inflammasome by caspase-1 adaptors ASC and Ipaf. Nature 2004, 430, 213–218.
- Strowig, T.; Henao-Mejia, J.; Elinav, E.; Flavell, R. Inflammasomes in health and disease. Nature 2012, 481, 278–286.
- Lima Leite, E.; Gautron, A.; Deplanche, M.; Nicolas, A.; Ossemond, J.; Nguyen, M.T.; do Carmo, F.L.R.; Gilot, D.; Azevedo, V.; Götz, F.; et al. Involvement of caspase-1 in inflammasomes activation and bacterial clearance in S. aureus-infected osteoblast-like MG-63 cells. Cell Microbiol. 2020, 22, e13204.
- Dinarello, C.A.; Simon, A.; van der Meer, J.W.M. Treating inflammation by blocking interleukin-1 in a broad spectrum of diseases. Nat. Rev. Drug Discov. 2012, 11, 633–652.
- Bongiorno, D.; Musso, N.; Lazzaro, L.M.; Mongelli, G.; Stefani, S.; Campanile, F. Detection of methicillin-resistant Staphylococcus aureus persistence in osteoblasts using imaging flow cytometry. MicrobiologyOpen 2020, 9, e1017.
- Musso, N.; Caruso, G.; Bongiorno, D.; Grasso, M.; Bivona, D.A.; Campanile, F.; Caraci, F.; Stefani, S. Different Modulatory Effects of Four Methicillin-Resistant Staphylococcus aureus Clones on MG-63 Osteoblast-Like Cells. Biomolecules 2021, 11, 72.
- Bhavsar, A.P.; Guttman, J.A.; Finlay, B.B. Manipulation of host-cell pathways by bacterial pathogens. Nature 2007, 449, 827–834.
- Ellington, J.K.; Harris, M.; Hudson, M.C.; Vishin, S.; Webb, L.X.; Sherertz, R. Intracellular Staphylococcus aureus and antibiotic resistance: Implications for treatment of staphylococcal osteomyelitis. J. Orthop. Res. 2006, 24, 87–93.
- Maali, Y.; Martins-Simões, P.; Valour, F.; Bouvard, D.; Rasigade, J.P.; Bes, M.; Haenni, M.; Ferry, T.; Laurent, F.; Trouillet-Assant, S. Pathophysiological mechanisms of Staphylococcus non-aureus bone and joint infection: Interspecies homogeneity and specific behavior of S. pseudintermedius. Front. Microbiol. 2016, 7, 1063.
- Valour, F.; Trouillet-Assant, S.; Rasigade, J.P.; Lustig, S.; Chanard, E.; Meugnier, H.; Tigaud, S.; Vandenesch, F.; Etienne, J.; Ferry, T.; et al. Staphylococcus epidermidis in orthopedic device infections: The role of bacterial internalization in human osteoblasts and biofilm formation. PLoS ONE 2013, 8, e67240.
- Campoccia, D.; Testoni, F.; Ravaioli, S.; Cangini, I.; Maso, A.; Speziale, P.; Montanaro, L.; Visai, L.; Arciola, C.R. Orthopedic implant infections: Incompetence of Staphylococcus epidermidis, Staphylococcus lugdunensis, and Enterococcus faecalis to invade osteoblasts. J. Biomed. Mater. Res. A 2016, 104, 788–801.
- Campoccia, D.; Montanaro, L.; Ravaioli, S.; Cangini, I.; Testoni, F.; Visai, L.; Arciola, C.R. New Parameters to Quantitatively Express the Invasiveness of Bacterial Strains from Implant-Related Orthopaedic Infections into Osteoblast Cells. Materials 2018, 11, 550.
- Stewart, P.S.; Costerton, J.W. Antibiotic resistance of bacteria in biofilms. Lancet 2001, 358, 135–138.
- Arciola, C.R.; Campoccia, D.; Speziale, P.; Montanaro, L.; Costerton, J.W. Biofilm formation in Staphylococcus implant infections. A review of molecular mechanisms and implications for biofilm-resistant materials. Biomaterials 2012, 33, 5967–5982.
- Arciola, C.R.; Campoccia, D.; Baldassarri, L.; Pirini, V.; Huebner, J.; Montanaro, L. The role of Enterococcus faecalis in orthopedic peri-implant infections demonstrated by automated ribotyping and cluster analysis. Biomaterials 2007, 28, 3987–3995.
- Ellington, J.K.; Reilly, S.S.; Ramp, W.K.; Smeltzer, M.S.; Kellam, J.F.; Hudson, M.C. Mechanisms of Staphylococcus aureus invasion of cultured osteoblasts. Microb. Pathog. 1999, 26, 317–323.
- Costerton, J.W.; Stewart, P.S.; Greenberg, E.P. Bacterial biofilms: A common cause of persistent infections. Science 1999, 284, 1318–1322.
- Brady, R.A.; Leid, J.G.; Calhoun, J.H.; Costerton, J.W.; Shirtliff, M.E. Osteomyelitis and the role of biofilms in chronic infection. FEMS Immunol. Med. Microbiol. 2008, 52, 13–22.
- Pautke, C.; Schieker, M.; Tischer, T.; Kolk, A.; Neth, P.; Mutschler, W.; Milz, S. Characterization of osteosarcoma cell lines MG- 63, Saos-2 and U-2 OS in comparison to human osteoblasts. Anticancer Res. 2004, 24, 3743–3748.
- Edwards, A.M.; Massey, R.C. How does Staphylococcus aureus escape the bloodstream? Trends Microbiol. 2011, 19, 184–190.
- Hamza, T.; Li, B. Differential responses of osteoblasts and macrophages upon Staphylococcus aureus infection. BMC Microbiol. 2014, 14, 207.




