White light emitting diodes (WLEDs), as solid-state lighting sources, have attracted increasing attention in the past decades owing to their potential applications in displays and lighting. WLEDs are energy saving and environmentally friendly, and have higher luminous efficiency than conventional incandescent and fluorescent lamps. Moreover, WLEDs emit polychromatic light rather than monochromatic light that was emitted by traditional light emitting diodes (LEDs).
- metal–organic frameworks
- organic dye
- luminescence
- white light emitting diodes
1. Introduction
Metal-organic frameworks (MOFs) are a class of porous crystalline materials composed of inorganic and organic moieties via coordination bonds, which are known for tunable pore size, high surface areas, structure flexibility and multiple functionality. These extraordinary properties have made MOFs ideal candidates for catalysis, gas storage and separation, membranes, biomedical imaging and luminescence-based sensing and lighting [1][2][11,12]. Specially, MOFs offer a unique platform for the development of luminescent materials due to structural predictability, multifunctionality, nanoscale processability and well-defined environments for luminophores in crystalline states [3][4][13,14]. Luminescence in MOFs can arise from organic ligands, metal ions and charge transfers such as ligand-to-metal charge transfer (LMCT), metal-to-ligand charge transfer (MLCT), ligand-to-ligand charge transfer (LLCT) and metal-to-metal charge transfer (MMCT) [5][15]. In addition, some guests introduced into MOFs via supramolecular interactions can emit or induce luminescence, and white light can be easily obtained by rational structure design and luminescent guest selection. Overall, these various effects have naturally led to speculation that MOFs could find potential applications in WLEDs. The first attempt to obtain white light by using MOFs can be traced back to 2007 [6][16]. Since then, different color-emitting lanthanide metals, conjugated organic ligands and guest species such as dye molecules and quantum dots have been incorporated in MOFs to generate white light [7][8][17,18].
Encapsulation of emissive organic dyes is quite a simple way to obtain MOFs with multiple luminescence emissions [9][19]. Organic dyes are probably the most widespread fluorophores among the luminescent materials because of wide excitation band, large absorption coefficient, moderate-to-high quantum yields, short fluorescent lifetime and great availability [10][20]. However, there are two serious problems when directly applying organic dyes in WLEDs. One is the aggregation caused quenching (ACQ) effect induced by π-π stacking interactions of the organic dyes, which results in low fluorescence intensity in solid states in comparison with their bright solution states. Additionally, the other is the thermal and photo-stability of organic dyes [11][10]. MOFs are ideal supporting materials to prevent organic dyes aggregating in solid states [12][13][21,22], since MOFs are highly porous and able to encapsulate molecular dyes in confined pores, so they are capable of preventing aggregation-induced quenching and restricting internal molecular motions to inhibit non-radiative relaxation [14][23]. In addition, by carefully choosing fluorescent linkers and organic dyes, MOFs can serve as an antenna to transfer energy to the dyes. The emissions from encapsulated dyes can be easily adjusted by changing the component and content of dyes. Moreover, diverse luminescence properties can be achieved by engineering interactions between dyes and constituents of MOFs. Thus, encapsulation of dyes into MOFs is massively proposed as phosphor converters in white light emitting diodes [12][21].
There are three major methods to encapsulate organic dyes in MOFs [12][21]. The first is the two-step synthesis method, in which the pristine MOF is synthesized first and then immersed in a solution of fluorescent dyes. Despite the simplicity of this approach, the mismatch size between MOF aperture and organic dyes not only restricts the choice of dyes, but also causes guest leakage, which hiders the extensive application of this approach. The second is the in situ encapsulation method, where dyes are introduced during the crystal formation. Although this method is helpful in obtaining fluorescent MOFs with uniform distribution of fluorescent dyes, more factors including pore size, pore windows and structures of MOFs for desired organic dyes should be considered. The final method is to use fluorescent linkers incorporated in the frame of MOFs, in which permanent fluorescence can be easily obtained, although the steric hindrance caused by bulky ligands often reduces the yield of the fluorescent of MOFs. In practice, fluorescent ligands are often combined with dyes to induce dual emissions, and the ligand-to-dye energy transfer process can be controlled by changing the excitation energy.
MOFs materials with porosity, multifunctionality and crystallinity have aroused much interest since the debut of the “metal-organic frameworks” concept in 1995 [15][24], and the scope of this research has expanded from structure design and topology analysis to a wide range of applications in gas storage, catalysis and biomedicine [1][2][16][17][18][19][11,12,25,26,27,28]. A number of excellent reviews have summarized the properties and applications of luminescent metal–organic frameworks (LMOFs) [20][11][3][4][5][7][9][10][21][22][23][24][8,10,13,14,15,17,19,20,29,30,31,32], while the reviews that specifically and systemically discuss the encapsulation of organic dyes in MOFs (dye@MOFs) for WLEDs applications are still rare. This review mainly summarizes recent progress achieved in developing pc-WLEDs based on dye@MOFs, where white light can be generated by coating the dye encapsulated MOF hybrids on the corresponding blue-LED chip or UV-LED chip. The emphasis was put on the white light emitting phosphors fabrication. The origin of luminescence in dye@MOFs has been discussed to tune high-quality white light.
2. Phosphors Excited by Blue-LED Chip
The combination of a blue-LED chip with yellow phosphors belongs in a partial conversion. The blue light emitting from LED chip is partially absorbed by the phosphor and refurbished into yellow light, while the remaining part of blue light is transmitted through the phosphor [25][3]. The blue and yellow light, as a pair of complementary colors, mix together to generate white light. Generally, compared to the UV chip WLEDs, the blue LED chips have higher theoretical efficiency, better reproducibility and lower input energy, so they are quite attractive for low-cost bright white-light sources [26][33]. However, these WLEDs often show low CRI and high CCT caused by red emitting deficiency, which limits their indoor use. In the past decades, the design and synthesis of new blue-light-excitable single-phase phosphors have emerged as a hot research area, and much progress has been made in improving color-rendering properties, especially benefiting from the development of MOF materials. From a fundamental point of view, the abundant luminescent behaviors and ordered structures of MOFs allow for the fine-tuning of emission color across the CIE diagram and improve luminescent intensity simultaneously.
An effective way to improve color-rendering properties is to broaden the emission spectra. Qian et al. [27][34] simultaneously encapsulated green-emitting coumarin 6 (Cou-6), yellow-emitting rhodamine 6G (R6G) and red-emitting rhodamine 101 (R101) into a MOF crystal to synthesize a yellow broadband phosphor ZJU-28⊃Cou-6 /R6G/R101 via ion exchange method. By coating the single-phase phosphor ZJU-28⊃Cou-6/R6G/R101 on commercial blue LED chips, the WLED lamp exhibits bright white light with luminous efficiency of 126 lm/W, CRI of 88 and CCT of 4446 K, and the total quantum yield (QY) can reach up to 82.9%. The good performance was ascribed to the high intrinsic quantum yields of dyes and fluorescence resonance energy transfer (FRET) process between them. In addition, the confinement effects of the MOFs can effectively inhibit the ACQ of dye molecules.
WLEDs can also be fabricated by combining blue chips with dye@MOFs and other commercialized phosphors [28][35]. Various concentration of rhodamine (Rh) dye was adopted to synthesize a series of Rh@bio-MOF-1 via cation exchange, and then the mixtures of the yellow-emitting Rh@bio-MOF-1, green (Ba,Sr) 2SiO 4: Eu 2+ and red CaAlSiN 3: Eu 2+ were coated on the blue LED chip to form phosphor film, which exhibits high luminous efficacy of 94–156 lm/W, CRI of 80–94 and excellent stability.
Unlike ion exchange, in situ encapsulation, in which fluorescent dyes are incorporated into the pores during the preparation of MOF crystals, have the advantage of uniform distribution of the fluorescent molecules, as long as the dyes can stand the synthesis conditions of MOFs. Li et al. [29][36] adopted the in situ encapsulation approach to avoid tedious ion-exchange synthesis and prevent dye leakage. Two yellow-emitting nanocomposites R6G@ZIF-8 and DBNT@UiO-66 with solubility compatibility and solution processability were synthesized, which can be excited by blue light to generate white light with absolute quantum yield of 63.1% and 22.7%, respectively. Similarly, Qian et al. [30][37] incorporated red, green and blue dyes into ZIF-8 by in situ self-assembly process to fabricate stable remote-type incandescent white-light device. They evaluated the thermostability and photostability of TPU-encapsulated ZIF-8⊃pm546/pm605/SRh101 phosphor in detail, and found the stability was greatly enhanced with TPU coating, which was ascribed to the protection against the oxygen and water invasion.
3. Phosphors Excited by UV-LED Chip
For WLEDs based on UV-LED and phosphors, all radiation from UV LED is converted into red, green and blue (RGB) light, which refers to full conversion. The phosphors excited by UV-LED chip must emit white light, so RGB phosphors are often adopted. As mentioned before, pc-WLEDs fabricated by blue LED coated with yellow phosphors may suffer such weaknesses as poor CRI and low stability of color temperature, due to deterioration of the chip or the phosphors. By contrast, UV-LED combined with mixed phosphors is one of the best approaches to generate white light for both high luminous efficiency and high CRI, at the expense of poorer efficacy owing to higher wavelength-conversion losses. Recently, developing single-phase white light phosphors is of great significance and different strategies have been adopted to improve UV-LED luminous efficacy. In general, luminescence in MOFs can be obtained from linkers, framework metal ions, and absorbed guests [21][29].
During the development of luminescent MOFs, the lanthanide MOFs have aroused extensive interest from the very beginning owing to high luminescence quantum yield, large Stokes shifts and sharp line-emissions [22][30]. Since f–f transition is parity-forbidden, lanthanide ions are often sensitized by organic ligands due to antenna effect. Qian [31][45] fabricated a phosphor for WLED by encapsulating blue dye within lanthanide MOF. EuBPT, TbBPT and Eu x Tb y BPT were synthesized by the solvothermal reaction. Owing to the energy transfer from BPT ligands to the lanthanide ions, the absolute quantum yields of red-emitting EuBPT and green-emitting TbBPT reached 37.11% and 73.68%, respectively. By optimizing the Eu 3+ /Tb 3+ ratio, Eu 0.05 Tb 0.95 BPT exhibits yellow light, and when combined with blue dye C460, white light emitting phosphor with absolute QY of 43.42% could be generated. The CRI and CCT values of the phosphors were estimated to be 90 and 6034 K, respectively. The WLED devices were fabricated by coating the prepared phosphor on a commercial UV-LED chip, and the luminous efficiency was measured to be 7.9 lm/W. Similarly, Saha [32][46] incorporated a single red emitting dye RhB into blue emitting gadolinium-based MOF to achieve perfect white light with high quantum yield.
Combining the emissions from linkers and dyes to generate single-phase white light phosphors is a hot research topic in recently years. In 2015, Qian [33][49] first encapsulated two dyes simultaneously into blue-emitting anionic MOFs via ion exchange. ZJU-28 exhibits blue emission under excitation at 365 nm, which ascribes to the H 3BTB ligand. ZJU-28⊃DSM/AF, as white lighting phosphor, can be easily prepared by soaking ZJU-28 into the mixed solution of red-emitting DSM and green-emitting AF, exhibiting broadband white emission with CIE coordinates of (0.34, 0.32), CRI value up to 91% and CCT of 5327 K. Since the confinement of MOFs can effectively suppress ACQ, the absolute QY could be improved to 17.4%. Substituting the H 3BTB ligand with carbazole-based ligand 4,4′,4′’-(9 H -carbazole-3,6,9-triyl)-tribenzoic acid (H 3L) [34][50], a white-light-emitting phosphor with same CRI value could be obtained, while the quantum yield could reach up to 39.4%.
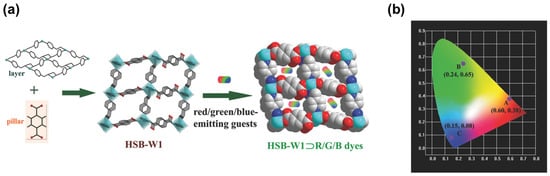
It is worth noting that efficient blue emission plays an important role in developing WLED, so strong blue fluorescent molecules are often introduced in company with red and green fluorescent molecules. Zhu [35][51] reported the incorporation of neutral and ionic RGB guest molecules into a neutral MOF HSB-W1 ( Figure 14 a). HSB-W1 exhibits blue emission upon excitation at 365 nm. HSB-W1⊃DCM, HSB-W1⊃C6 and HSB-W1⊃CBS-127 can be conveniently synthesized and exhibit red, green and blue emission, respectively, as shown in ( Figure 14 b). HSB-W1⊃DCM⊃C6⊃CBS-127 composite emits white-light with high quantum yield (up to 26%) and CRI (up to 92%). The results showed that incorporating RGB dyes into blue-emission MOFs is a useful strategy to design single-phase white-light phosphors.
4. Conclusions
Luminescent MOFs materials offer a promising platform for light-emitting diodes, chemical sensing, bioimaging and anti-counterfeiting codes. In the past decades, much attention has been focused on design of linkers and encapsulation of guest molecules instead of lanthanide metal-based MOFs for environment consideration.
Encapsulation of organic dyes into MOFs is a feasible and ingenious approach to construct pc-WLEDs, which combines the benefits from dyes and MOF structures. The porosity and crystallinity of MOFs are helpful to suppressing ACQ of dye molecules and thus improving both fluorescent intensity and quantum yield. Meanwhile, the organic dyes enrich the luminescent behaviors without sacrificing the strength of MOFs. Although the warm white light can be generated by encapsulating fluorescent dyes in luminescent MOFs with high performance, the luminous efficiency is still low. In addition, organic dyes leakage, stability and unsuitable size hinder the extensive application of this method. According to the previous study, most reported dye@MOFs are synthesized based on currently available organic dyes or MOFs. From fundamental views, it is necessary to develop novel organic dyes and MOF structures considering factors such as topology, luminescence, charge transfer and stability. Moreover, in-depth research on mechanism behand should be devoted in order to provide instructions for material design and synthesis. For the purpose of industrialization and commercialization, the stability of phosphors, including photo-stability and thermal stability is of vital significance, while currently the reports on the stability of dye-encapsulated MOF phosphors are still rare. It is predictable that more effort will be devoted to investigate the stability of LMOF materials.
