The bromeliad Tillandsia cyanea Linden ex K. Koch, with its vibrant pink inflorescences, is a well-known ornamental plant all over the world. This epiphytic bromeliad possesses complex trichomes (peltate trichomes) that are capable of producing and exuding a polysaccharide-based secretion (mucilage) with high affinity for water. This observation indicates that T. cyanea - and probably many other bromeliads - might have more intricate strategies to cope with water availability than previously thought.
- scales
- colleter
- plant–environment interactions
- plant secretion
- gland
- cuticular pores
- desiccation
- absorbing trichomes
- herbivory
Mucilage Secretion by Bromeliad Trichomes
Bromeliad trichomes- also known as “scales” - have been investigated extensively due to their recognition as a key ecological and evolutionary feature of this plant group[1][2][3][4][5][6][7][8][9][10][11][12][13]. However, much remains unknown about such trichomes and only recently mucilage exudation was described for them in Aechmea blanchetiana (Baker) L. B.Sm., a species of the subfamily Bromelioideae [11]. A glandular function was also assigned to the leaf scales of the bromeliad Tillandsia usneoides (L.) L. due to the presence of an intracellular hydrophilic secretion accumulated in the uppermost cell of the trichome stalk [14]. Unlike A. blanchetiana, the secretion of T. usneoides is not released to the external environment but is also potentially associated with an increase in water and nutrient absorption by the scales [14]. The occurrence of mucilage exudation by the scales of T. cyanea comprises the first record of such secretion within the Tillandsioideae, the most diverse of bromeliad subfamilies [15]. Our findings confirm that the secretory activity of scales in bromeliad inflorescences is more common than previously considered. This constitute a feature with relevant ecological and evolutionary implications for Bromeliaceae.
Secretory Aspects and Structure of the Trichomes
The secretion in T. cyanea is observed throughout the adaxial surface of the inflorescence bracts, usually accumulating in the space between them and covering the axis and flower buds. In natural conditions, the secretion is hyaline, viscous, odorless, and it is kept in the space between successive bracts. Therefore, this secreted mucilage can only be observed on the surface of the inflorescences after manipulation. When artificially exposed to the external environment, the secretion drastically decreases in volume and turns into a solid film capable of rapid rehydration.
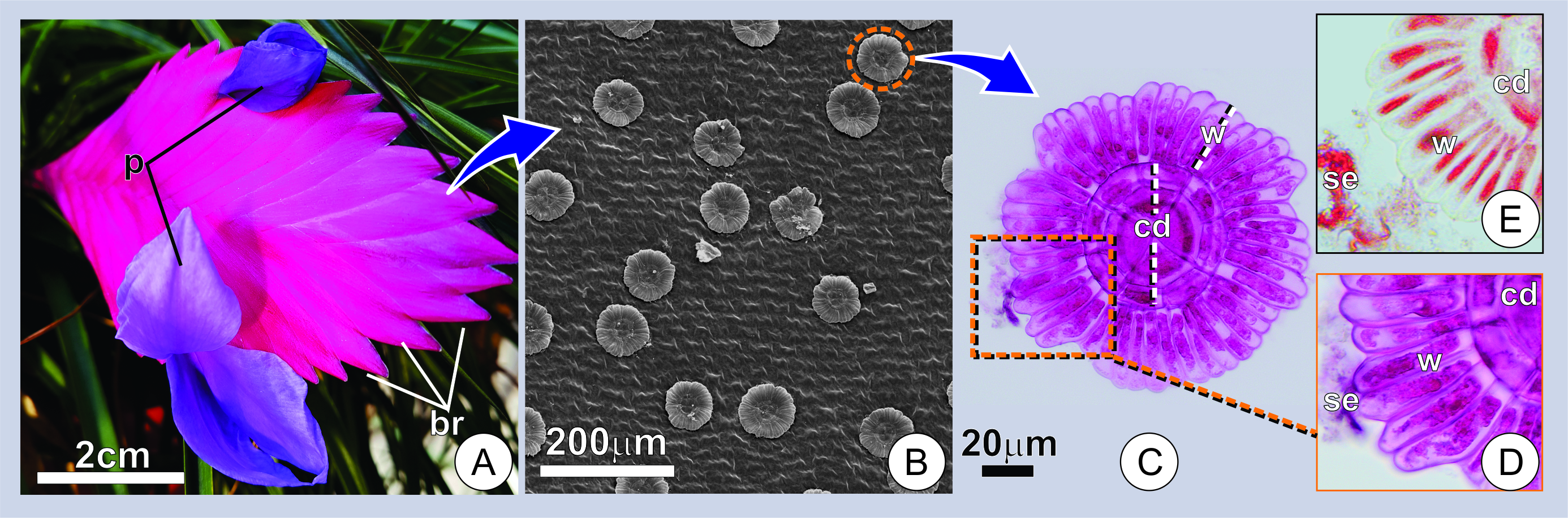
Anatomical and ultrastructural results indicate that the secretion is produced by the wing portion of typical peltate trichomes on the adaxial surface of the inflorescence bracts (Figure 1A-E).
Figure 1. Distribution and aspect of secretory trichomes. (A) Overview of an inflorescence at the flowering stage. Note the compact arrangement of bracts (br) subtending the flowers with exposed petals (p) at anthesis. (B) Adaxial surface of a young bract showing numerous trichomes. (C) Surface view of a secreting trichome. Note the overall structure and wing cells with conspicuous protoplasts marked for mucilage (in pink). (D-E) Portions of the trichomes showing the histochemical characterization of trichomes and secretion in young bracts. (D) Note the positive reaction for mucilage in pink (D), and proteins in red (E). w=wing portion; cd=central disc; se=secretion.
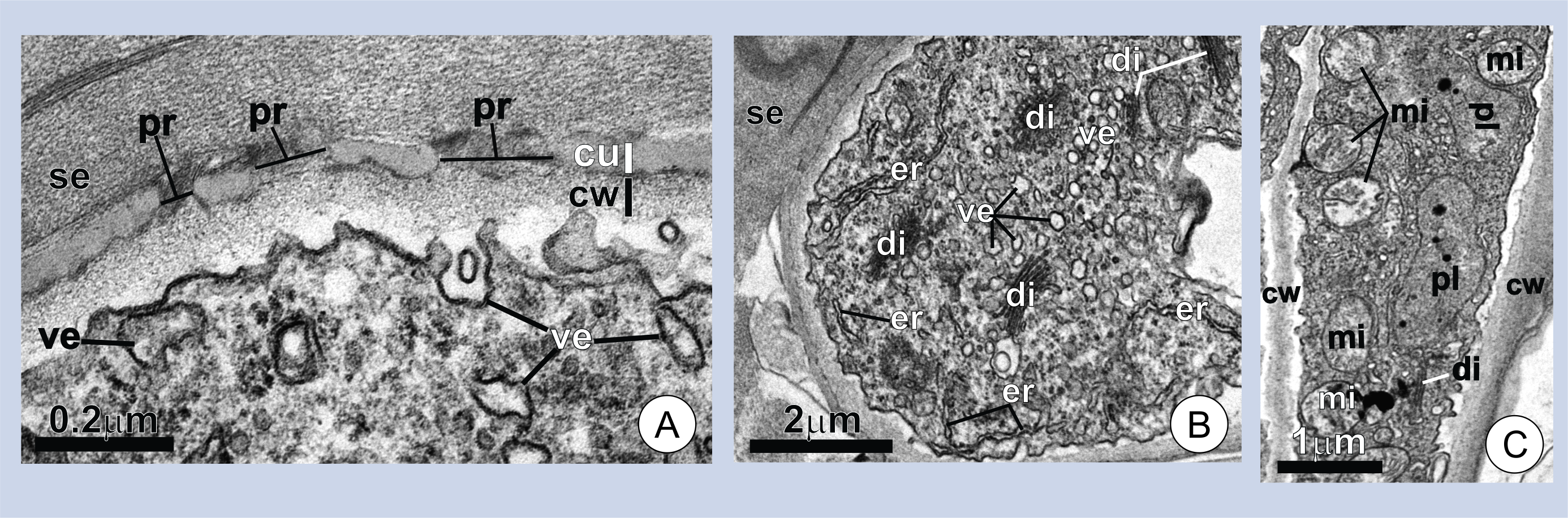
The secretory activity begins in the early stages of trichome expansion and ceases in mature bracts, where the trichomes have already fully expanded. The association between some plant secretions and juvenile organs is common in many secretory structures of plants[16][17][18][19]. Histochemical tests revealed polysaccharides and proteins in the secretion (Figure 1C-E). The cells producing the secretion characteristically display a porous cuticle and dense cytoplasm with numerous mitochondria and dictyosomes (Figure 2A-C). These features are common in the secretory cell of plants and are often considered as indicators of secretory activity [20][21][22][23].
Figure 2. Ultrastructure of wing cells in young trichomes. (A-B) Cross-sections of a wing cell. Note the pores (pr) in the cuticle (cu), the thick layer of secretion (se) on the cell surface in A, and the dense cytoplasm with numerous dictyosomes (di) and vesicles (ve) in B. (C) Longitudinal section of a wing cell. Note the numerous mitochondria (mi). er=endoplasmic reticulum; cw=cell wall; pl=plastids.
Functions
Further studies are necessary to fully understand the exact functional roles performed by the mucilage produced in T. cyanea inflorescences. However, the features of this secretory system suggest a few potential functions:
Protection against desiccation
The restriction of the secretory activity to young portions of the inflorescence and the hygroscopic exudate suggest a protective role against desiccation. This is especially important during the development of flower buds and in the later expansion of the delicate corolla. In the latter, secretion may also contribute as a lubricant, facilitating the extrusion of the petals. A role in protection against desiccation is a recognized function of some exudates in young reproductive axes of angiosperms[16][24][25][26][27][28]. For these plant portions, water supply via xylem may be inefficient, and secretions could avoid water loss due to excessive transpiration or increase water uptake through the cuticle [17].
Defense against herbivores and pathogens
While the mucilage in T. cyanea does not seem to provide effective immobilization of large insects and other herbivores, one cannot rule out possible protection against small arthropods and pathogens. The protein content found in the secretion constitutes a good piece of evidence in this direction, as the presence of proteins seems to be associated with defense against pathogen activity [29][30].
Water absorption
An additional role in water absorption must be considered concerning the trichomes of T. cyanea since they share many structural features with the scales of bromeliad leaves, which are well-known for their water uptake ability [8][9][10]. In this case, the mucilage produced by the trichomes of T. cyanea could aid in this process due to its high affinity for water.
References
- Schimper, A.F.W.; Die epiphytische Vegetation Amerikas. Bot. Mitt. Trop. 1888, 2, 162.
- Mez, C.; Physiologische Bromeliaceen-Studien. I. Die Wasser—Okonomie der extrem atmosphärischen Tillandsien. Jahrb. Wiss. Bot. 1904, 40 , 157–229.
- Tietze, M.; Physiologische Bromeliaceen Studien II. Die Entwicklung der wasseraufnehmenden Bromeliaceen Trichome. Z. f. Naturwiss. Z. Naturwiss. 1906, 78, 1-50.
- Wetzel, K.; Beitrag zur Anatomie der Saughaare von Bromeliaceen. Flora Oder Allg. Bot. Zeitung 1924, 117, 133-143.
- Dolzmann, P.; Elektronenmikroskopische Untersuchungen an den Saughaaren von Tillandsia Usneoides (Bromeliaceae)—I. Feinstruktur der Kuppelzelle. Planta 1964, 60, 461-472.
- Dolzmann, P.; II—Einige beobachtungen zur feinstruktur der plasmodesmen. Planta 1965, 64, 76-80.
- Tomlinson, P.B. . Anatomy of the Monocotyledons: III Commelinales-Zingiberales; Claredon Press: Oxford, UK, 1969; pp. ..
- Benzing, D.H.; Bromeliad trichomes: structure, function, and ecological significance. J. Marie Selby Bot. Gard. 1976, 1, 330-348.
- Benzing, D.H.; Henderson, K.; Kessel, B.; Sulak, J.; The absortive capacities of bromeliad trichomes. Am. J. Bot. 1976, 63, 1009–1014.
- Benzing, D.H.; Givnish, T.J.; Bermudes, D.; Absorptive trichomes in Brocchinia reducta (Bromeliaceae) and their evolutionary and systematic significance. Syst. Bot. 1985, 10, 81–91.
- Ballego-Campos, I; Paiva, E. A. S.; Mucilage secretion in the inflorescences of Aechmea blanchetiana: Evidence of new functions of scales in Bromeliaceae. Flora 2018, 246–247, 1-9, 10.1016/j.flora.2018.06.003.
- David H. Benzing; Foliar Permeability and the Absorption of Minerals and Organic Nitrogen by Certain Tank Bromeliads. International Journal of Plant Sciences 1970, 131, 23-31, 10.1086/336508.
- Thomas J. Givnish; Michael H.J. Barfuss; Benjamin Van Ee; Ricarda Riina; Katharina Schulte; Ralf Horres; Philip A. Gonsiska; Rachel S. Jabaily; Darren M. Crayn; J. Andrew C. Smith; et al.Klaus WinterGregory K. BrownTimothy M. EvansBruce K. HolstHarry LutherWalter TillGeorg ZizkaPaul E. BerryKenneth J. Sytsma Adaptive radiation, correlated and contingent evolution, and net species diversification in Bromeliaceae. Molecular Phylogenetics and Evolution 2014, 71, 55-78, 10.1016/j.ympev.2013.10.010.
- L. Brighigna; M.R. Palandri; M. Giuffrida; C. Macchi; G. Tani; Ultrastructural Features of theTillandsia UsneoidesL. Absorbing Trichome During Conditions Moisture and Aridity. Caryologia 1987, 41, 111-129, 10.1080/00087114.1988.10797853.
- Gouda, E.J.; Butcher, D. . A list of accepted Bromeliaceae names. Retrieved 2020-6-24
- Poliana Cardoso-Gustavson; Lisa M. Campbell; Solange C. Mazzoni-Viveiros; Fábio Cury De Barros; Floral colleters in Pleurothallidinae (Epidendroideae: Orchidaceae). American Journal of Botany 2014, 101, 587-597, 10.3732/ajb.1400012.
- Paiva, E.A.S; Occurrence, structure and functional aspects of the colleters of Copaifera langsdorffii Desf. (Fabaceae, Caesalpinioideae). Comptes Rendus. Biologies 2009, 332, 1078-1084, 10.1016/j.crvi.2009.08.003.
- Paiva, E.A.S; Colleters in Cariniana estrellensis (Lecythidaceae): structure, secretion and evidences for young leaf protection. The Journal of the Torrey Botanical Society 2011, 139, 1-8, 10.3159/torrey-d-11-00046.1.
- Thomas, V.; Structural, Functional and Phylogenetic Aspects of the Colleter. Annals of Botany 1991, 68, 287-305, 10.1093/oxfordjournals.aob.a088256.
- J. S. S. Mohan; J. A. Inamdar; Ultrastructure and Secretion of Extrafloral Nectaries of Plumeria rubra L.. Annals of Botany 1986, 57, 389-401, 10.1093/oxfordjournals.aob.a087121.
- Fahn, A. . Secretory Tissue in Plants; Academic Press: London, UK, 1979; pp. ..
- Fahn, A.; Structure and function of secretory cells. Adv. Bot. Res. 2000, 31, 36-75.
- Carolina Santos Oliveira; Alexandre Salino; Elder Antônio Sousa Paiva; Colleters in Thelypteridaceae: Unveiling mucilage secretion and its probable role in ferns. Flora 2017, 228, 65-70, 10.1016/j.flora.2017.01.009.
- Mayer, J. L. S.; Cardoso-Gustavson, P.; Appezzato-Da-Glória, B.; Colleters in monocots: New record for Orchidaceae. Flora 2011, 206, 185-190, 10.1016/j.flora.2010.09.003.
- Renata C. Ferrari; Aline Oriani; Floral anatomy and development of Saxofridericia aculeata (Rapateaceae) and its taxonomic and phylogenetic significance. Plant Systematics and Evolution 2016, 303, 187-201, 10.1007/s00606-016-1361-z.
- Cassola, F; Nunes, C. E. P.; Lusa, M. G.; Garcia, V. L.; Mayer, J. L. S.; Deep in the Jelly: Histochemical and Functional Aspects of Mucilage-Secreting Floral Colleters in the Orchids Elleanthus brasiliensis and E. crinipes.. Frontiers in Plant Science 2019, 10, 518, 10.3389/fpls.2019.00518.
- Almeida, A. L.; Paiva, E.A.S.; Colleters in Mabea fistulifera Mart. (Euphorbiaceae): Anatomy and biology of the secretory process. Flora 2019, 258, 151439, 10.1016/j.flora.2019.151439.
- Paiva, E.A.S; Martins, L.C.; Calycinal trichomes in Ipomoea cairica (Convolvulaceae): ontogenesis, structure and functional aspects. Australian Journal of Botany 2010, 59, 91, 10.1071/bt10194.
- Klein, D. E.; Gomes, V. M.; Da-Silva-Neto, S. J.; Da Cunha, Maura.; The Structure of Colleters in Several Species of Simira (Rubiaceae). Annals of Botany 2004, 94, 733-740, 10.1093/aob/mch198.
- Miguel, E. C.; Gomes, V. M.; De Oliveira, M.; Cunha, M.; Colleters in Bathysa nicholsonii K. Schum. (Rubiaceae): Ultrastructure, Secretion Protein Composition, and Antifungal Activity. Plant Biology 2006, 8, 715-722, 10.1055/s-2006-924174.