The coffee plant Coffea spp. offers much more than the well-known drink made from the roasted coffee bean. During its cultivation and production, a wide variety of by-products are accrued, most of which are currently unused, thermally recycled, or used as animal feed.
- coffee by-products
- novel food
- coffee flower
- coffee leaves
- coffee pulp
- husk
- green coffee
- coffee silver skin
- spent coffee grounds
- cascara
The coffee plant is a special crop which, starting in Ethiopia, has now covered large areas of tropical and subtropical cultivation [11][1]. Coffee belongs to the family Rubiaceae and the tribe Coffeeae [12][2]. Currently, more than 100 species of the genus Coffea are known, with the two varieties C. arabica and C. canephora being the most economically exploited species [13][3]. The evergreen coffee plants produce white flowers which, after fertilization, produce mainly red fruits called coffee cherries [11][1]. About 3% of C. arabica also produce fully ripe yellow cherries. Other coffee species can also produce black or black-and-yellow-striped cherries. Most varieties need 210 days from blossom to ripe cherry (e.g., bourbon or catuai), whereas dwarf varieties (such as caturra and villa sarchi) require some days less, and tall and giant varieties (maragogype and pacamara) around 230–240 days. The structure of a coffee cherry is shown in Figure 21. Under the leathery epicarp (skin), which is green in the beginning and turns red with increasing ripeness, is the soft sweet mesocarp (pulp). Beneath this is the strongly developed endocarp (parchment), enclosing the two seeds (endosperm) called coffee beans, which are covered by their silver skin, a thin tegument [14][4].
After harvesting the coffee cherries, there are different ways to produce green coffee (Figure 32). In dry processing, the fruits are spread out immediately after harvesting and dried in the sun. Then, the dried pulp, parchment, and parts of the silver skin are removed with peeling machines. This waste is called husk. The much more complex wet method can be differentiated in three different sub-forms: i. pulped natural, ii. semi-washed, and iii. fully washed. In the first step of all wet processing methods, the pulp of the coffee cherries is squeezed off the parchment beans in a pulper. The differentiation between the sub-forms is directly connected to the treatment of the adhering mucilage. In the “pulped natural” method (sometimes also referred to as “semi-dry” method), all mucilage is left on the parchment and dried on it, in order to allow the sugars to enter the coffee bean by osmosis and create a very sweet coffee. In both the “semi-washed” and the “fully washed” method, the mucilage is removed; the semi-washed technique is a mechanical cleaning, i.e., using brushes and the friction of the parchment beans amongst themselves. In the “fully washed” method, the mucilage is broken down by microorganisms, which occur naturally in the environment of the coffee pulping stations and the coffee plants. Bacteria, yeast, and fungi break down the sugars and the pectins within the mucilage and produce acids and alcohols, thus creating a broader spectrum of aromas, while the sweetness is decreased. It is still uncommon to use controlled fermentation as done in the wine, beer, cheese, or bakery industry. In all cases, fermentation is a “spontaneous fermentation”, while the created flavors differ a lot depending on the weather and other influencing factors during harvest and processing.
During roasting of the coffee beans, which usually no longer takes place in the countries of origin, the remaining silver skin still adhering to the beans accrues as waste [15][5]. Spent coffee grounds are another type of waste produced either by coffee consumers or by the industrial production of instant coffee [11][1]. As a result, a large quantity of by-products is generated during the production of coffee, and their recycling is becoming more and more important. Parts of the coffee plant such as flowers, leaves, twigs and wood are equally classified as coffee by-products, because they arise during cultivation. The most important by-products for food production are listed in Table 1.
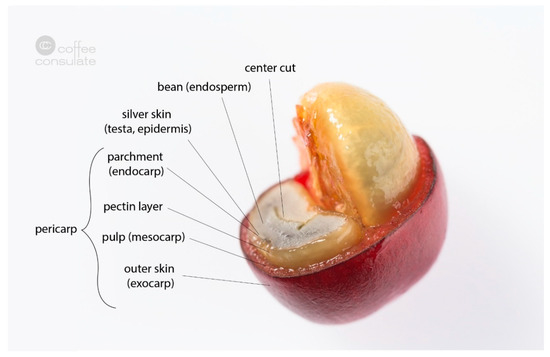
Figure 21. Cross section of a coffee cherry with its different layers.
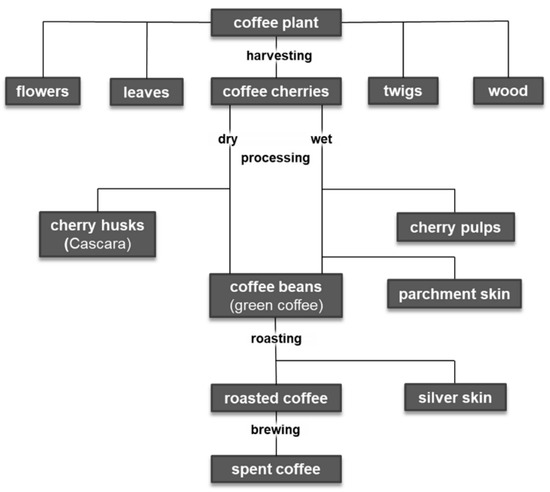
Figure 32. Coffee production-related by-products.
Table 1. Main uses of coffee by-products in the food sector.
Coffee By-Product | Possible Use as Food | |||||||||||||
Flowers | Beverages (tea): “coffee blossom tea” [16] | [6] |
||||||||||||
Leaves | Beverages (tea): “coffee leaf tea” [17–21] | |||||||||||||
Coffee pulp | Jam, juice, concentrate, jelly [22]; | [12]; | Coffee pulp flour for breads, cookies, muffins, squares, brownies, pastas, sauces and beverages [23]; | [ 13]; | spirits/ethanol [24] | [ ] |
||||||||
Husks, cascara, dried coffee cherries | Beverages (tea) [25–27]; | spirits [28,29]; | [ | qishr (mixture with spices) [30,31]; | [ ][21]; | dietary fiber source [11,32]; | [ ] [22]; | extraction of caffeine [24] | [ ] |
|||||
Green unroasted beans | Dietary supplement [33–35]; | beverages (tea): “white coffee” [36,37] | [ |
|||||||||||
Silver skin | Dietary fiber source [38–43]; | bakery products (breads, biscuits) [44–47]; | [ | beverages (tea) [48,49]; | [ ][39]; | smoke flavor additive | ||||||||
Spent coffee grounds | Historical: adulteration of coffee; | spirits [50]; | [ 40]; | bakery products [51–53]; | [ | dietary fiber source [54,55] | [ ] [45] |
|||||||
Defatted Coffea arabica seed powder (coffee flour) for savory and sweet recipes, in bakery, confectionary, snacks, ready-to-eat products [56] | [46] |
|||||||||||||
Parchment | Food preservative, antioxidant [24,57] | |||||||||||||
Flowers
Coffee plants form white, multi-flowered, cymose inflorescences. Every year, an adult coffee tree produces 30,000–40,000 flowers[6] [16] (according to the authors’ own observations, this number is probably for C. arabica, with C. canephora likely expected to have a higher number of flowers). Because of colleters, which produce a glue-like secretion, the flowers protect the plant from dehydration and insects [58][48]. When the flower is starting to wilt off the plant, the blossoms are plucked, so that the payload of coffee cherries is not affected [59][49]. After drying, the flowers could be brewed as tisane. Beside high values of total phenolic content, dried flowers obtained from C. canephora contain about 1 g caffeine/100 g dry weight and also about 1 g trigonelline/100 g dry weight [16][6]. Research in the field of coffee flowers is lacking, so there are no toxicologically data available.
Leaves
The shiny, waxed leaves on the side of the main stem of the coffee plant are typically green [60][50]. For some species or varieties, the young leaves can be bronzed-tipped and are green or dark-green when aging but never brown, except when they are stressed or when they fall down. The leaves of C. arabica and C. canephora experience a life time of 8 months and 7–10 month, respectively, until they reach a length of up to 15 cm[51] [61] (this literature value of 15 cm is probably for C. arabica, while C. canephora leaves can reach a length of 30 cm and those of Coffea liberica W. Bull ex Hiern even up to 50 cm).
The leaves of the coffee plants have been used for a long time to prepare tea-like drinks. Especially in West Sumatra, Ethiopia, Jamaica, India, Java, and South Sudan, the infusion is consumed as a traditional food [18,62,63][8][52][53]. Regarding species, C. arabica leaves have probably been used in Ethiopia, while C. canephora leaves have been used in India and West Sumatra. According to Novita et al. the drink is called “kahwa daun” or “kawa” in Sumatra [20][10]. In Yemen, it is called “giser” and in Ethiopia “kuti”, “jeno”, or ”jenuai” [64][54]. Before preparing the drink, there are different production methods to obtain coffee leaf tea. Most of them include leaf steaming, rolling, and drying [65][55]. However, some manufacturers work under a protective gas atmosphere to preserve the ingredients from oxidation [66–69][56][57][58][59]. Alternatively, the leaves can also be fermented [65][55]. In some production methods, the drying process is supplemented by a roasting process [70,71][60][61]. To produce a drink, the tea has to be extracted with water or aqueous solvents [19][9]. Chen et al. found that a variation of the production method (white, green, oolong, black; based on the production of tea from Camellia sinensis (L.) Kuntze) produced different phytochemical compositions and different flavors [61][51].
Besides their use as tea, coffee leaves are also used in ethnomedicine in the originating countries. The composition of coffee leaves was recently reviewed by Chen [61][51]. The leaves contain carbohydrates, protein and amino acids, organic acids as well as a large spectrum of ingredients, such as alkaloids, flavonoids, terpenes, tannins, xanthonoids, phenolic acids, flavonoids, phytosterols, amino acids, and carotenoids, which is held responsible for the diverse potential bioactive effects typically known from in vitro studies only, e.g., antioxidant, anti-inflammatory, antihypertensive, antibacterial, and antifungal activities [18,62,64,72–75][8][52][54][62][63][64][65]. C. arabica leaves contain 1.8 and 3.2 mg/g fresh weight of caffeine in mature and young leaves, respectively [17][7]. The content considerably changes during aging [76][66].
Besides the use as food, coffee leaves are being tested in several other new applications, as vehicle perfume [77][67], facial cleanser [78][68], tobacco substitute [79[69][70],80], animal feed [81][71], Lactobacillus proliferating agent [82[72][73],83], packaging material [84][74], absorbance pad [85][75], and deodorizer [86][76].
For risk assessment, a level of 7 mg/g of caffeine in dried leaves and 9.9–10.9 mg/L of caffeine in an aqueous drink prepared from 20 g/L of these leaves was determined by the EFSA in the context of a novel food notification as traditional food from a third country [87][77]. As specifications for a safe food, EFSA gives a reference of <100 mg/L for chlorogenic acid, <80 mg/L for caffeine, and <700 mg/L for epigallocatechin gallate. The maximum levels for caffeine and chlorogenic acid are based on the recommendations of the applicant, whereas the maximum level for epigallocatechin gallate is based on the observed safe level according to Hu et al. and the intake of green tea [88,89][78][79]. The xanthine mangiferin was only found in the leaves of C. arabica (5 mg/kg dry weight) and not in those of C. canephora [72][62]. Signs of exocrine pancreas toxicity of mangiferin in rodents were observed at a dose of 1000 mg/kg bodyweight (bw) [90][80]. Reddeman et al. found no evidence of genotoxicity for mangiferin in the Ames test, but a clastogenic effect could be detected in the chromosomal aberration test. However, this could not be confirmed in an in vivo mammalian micronucleus test with a dose up to 2000 mg/kg bw/day [91][81]. Thus, the EFSA considers the presence of mangiferin in coffee leaves to be safe [87][77].
Stems, Twigs, Wood
Stems, twigs, and wood are by-products without potential food uses; minor amounts are tolerated in coffee trade as “defects” and considered a contamination of green coffee [92][82]. For example, the German coffee regulation tolerates 2 g/kg of non-coffee compounds in roasted coffee [93][83].
Cherry Pulp
A large part of the coffee cherry is the cherry pulp, which also typically contains the skin. It represents around 29% of the dry matter. Cherry pulp is a by-product of all wet processing methods [11,94][1][84]. Cherry pulps contain 4%–12% of protein, 1%–2% of lipids, 6%–10% of minerals, and 45%–89% of total carbohydrates [95][85]. Phenolic compounds and caffeine (1.3%) are also present in considerable amounts [96][86]. Possible uses of cherry pulps are jam, juice, concentrate, and jelly. Coffee pulp flour can be used for breads, cookies, muffins, squares, brownies, pastas, and sauces [22,23][12][13]. Currently, there are no specific data about the toxicity of cherry pulps in the literature. It can be assumed that bioactive compounds, microbial spoilage, and mycotoxin contamination may pose a safety risk.
Parchment
Coffee parchment is a lignocellulosic material that has been rarely studied and not efficiently utilized [57][47]. Parchment was suggested as an antifungal additive with potential uses for food preservation [57][47]. Extracts of parchment have also been suggested as biocomponents with antioxidant activity [24][14]. Recent literature shows the potential of this by-product as a promising low-calorie functional ingredient for dietary fiber enrichment in foods to regulate blood glucose and reduce the concentration of serum lipids [97][87].
Assessing the food safety of parchment, Iriondo-DeHond et al. detected the mycotoxin ochratoxin A at 2.7 µg/kg[22] [32]. They also investigated the acute toxicity of raw parchment in rats with a single dose of 2000 mg/kg bw and observed no apparent signs of toxicity, abnormal behavior, or mortality [32][22].
Cherry Husk
During dry processing, coffee cherries are dried in the sun, and then the beans are mechanically removed from the dried husks. The remaining husks are composed of skin, pulp, and parchment [14][4]. They contain 8%–11% of protein, 0.5%–3% of lipids, 3%–7% of minerals, and 58%–85% of total carbohydrates [95][85]. The fiber amount contains 24.5% of cellulose, 29.7% of hemicellulose, and 23.7% of lignin [98][88]. Caffeine and tannins represent around 1% and 5% of the cherry husk, respectively [95][89].
The production of a refreshing and stimulating beverage called cascara (from Spanish “cáscara”, meaning husk) has been described. During the preparation, the husks are infused with hot water, and the result is commonly known as coffee cherry tea or cascara [26][16]. Traditional beverages from coffee cherries have been consumed in Yemen (called Qishr) and Ethiopia (called Hashara) [30,31,99][20][21][89]. Despite the Spanish word being commonly used worldwide for coffee husk or its derived beverage, the authors have not observed any traditional use of cascara in Latin America.
The beverage cascara contains 226 mg/L of caffeine and 283 mg/L of gallic acid equivalents, which represents the total polyphenol content [25][15]. While studying the in vitro biological activity of aqueous extracts of cherry husks, associations with biomarkers of inflammation, oxidative stress, adipogenesis, and insulin resistance were detected [100,101][90][91].
Cherry husks can also be used for the cultivation of microorganisms based on their high fiber content. Using fermentation, the production of citric acid, gibberellic acid, and enzymes has been realized [11,38][1][28]. Gouvea et al. claimed that cherry husks are a potential residue-based ethanol production source [102][92]. Additional uses in foods are in spirits[18][19] [28,29] and dietary fiber supplements [11,32][1][22].
Mycotoxins are mainly produced during the drying process of coffee cherries and could be of toxicological concern. Paterson et al. described that ochratoxin A was formed in coffee cherry pericarp while sun-drying and found its highest contaminations in husks [103][93]. Iriondo-DeHond et al. also detected quantities of ochratoxin A (4.3 µg/kg) in the insoluble fraction of husks [32][22]. They also investigated the acute toxicity of raw husk in rats with a single dose of 2000 mg/kg bw and observed no apparent signs of toxicity, abnormal behavior, or mortality [32][22]. The EFSA determined a tolerable weekly intake for ochratoxin A of 120 ng/kg bw [104][94]. The preparation of cherry husks aiming to avoid microbial spoilage is very important to achieve low mycotoxin amounts. Nevertheless, the investigation of mycotoxins in dried husks is an essential measure for food safety.
Green Coffee
After coffee cherries have been cleaned of skin, pulp, mucilage, and parchment by dry or wet processing, green coffee beans remain, which are traded on the international market [3,14][95][4]. These beans are still mostly covered by their silver skin. In a further, optional polishing step, this silver skin can also be removed [105,106][96][97]. Green coffee has a mild, green, bean-like aroma [107][98].
The main constituents of green coffee are insoluble polysaccharides (~50%) such as celluloses and hemicelluloses. In addition to complex carbohydrates, it contains mono- and oligosaccharides, oils and waxes (8%–18%), proteins and amino acids (9%–12%), minerals (3%–5%), and polyphenolic compounds[4][97][99][100][101][102][103] [14,106,108–112]. The most common alkaloid in green coffee is caffeine (1%–4%), whose concentration strongly depends on variety and growing conditions, followed by trigonelline (~0.8%)[104][105][106] [113–115]. Trigonelline is partially degraded during coffee roasting, forming N-methylpyridinium ions by decarboxylation and nicotinic acid by demethylation[107][108][109] [116–118].
Green coffee is included in the context of coffee by-products as novel food because typically only roasted coffee is consumed by end consumers. Nevertheless, green coffee can be marketed as such (without roasting), and an infusion (from non-selective water extraction) can be prepared from the chopped beans [33–37][23][24][25][26][27]. Extracts, customized to the needs of the food industry, can generally also be produced by extraction with hot water [119][110], alcohol [120][111], or mixtures thereof [107][98]. Such selective extracts require a separate assessment as novel food. They are available in capsules as food supplements or are used for the preparation of beverages or chewing gum. Due to the high content of chlorogenic acids, which would be partially lost during roasting, the extracts are sold with reference to their potential, diverse, health-promoting effects, some of which have yet to be proven [35][25]. Watanabe et al. examined green coffee extract in a placebo-controlled human intervention study and found no serious side effects. The subjects received an extract containing 140 mg of chlorogenic acid per day for 12 weeks [121][112]. In a rat study of green coffee oil, which is rich in kahweol and cafestol, no signs of toxicity could be found. The acute administration of 2000 mg/kg bw and the subacute administration of up to 75 mg/kg bw exhibited no toxicological effects, but the relative weight of heart and thymus increased without histopathological changes [122][113].
Caffeine is known to affect the central nervous system and is therefore popular for its stimulating effect. However, overdosing can also have adverse effects, so that, especially when placing food supplements on the market, the recommendation for maximum daily intake of caffeine (400 mg/day) by the EFSA should be observed [123][114].
Especially in the production of extracts, it should be noted that mycotoxins can also be enriched. Vaclavik et al. found up to 136.9 μg/kg of ochratoxin A, 20.2 μg/kg of ochratoxin B, 415.0 μg/kg of fumonisin B1, and 395.0 μg/kg of mycophenolic acid in green coffee extracts [124][115].
Silver Skin
The coffee silver skin forms a thin tegument, which is located directly around the two beans of the coffee cherry. It accumulates in large amounts as a by-product of the roasting process. The coffee silver skin is composed to a large extent of dietary fiber (60%–80%) [44][34]. Pertinent fiber components are cellulose, hemicellulose, and lignin. The main monosaccharides in coffee silver skin hemicellulose are xylose, galactose, arabinose, and mannose. A relevant amount of the total dietary fiber is soluble, which gives coffee silver skin the potential to be used as raw material in the development of functional foods [43][33]. There is no significant difference of the dietary fiber content between C. arabica and C. canephora coffee silver skin [32][32]. The protein content in coffee silver skin is around 18% [41,43][31][33]. The ash value is high, which is attributed to minerals. Other nutrients such as fats and reducing carbohydrates are found in lower concentrations [41][31].
The coffee silver skin contains phenolic compounds and has a high antioxidant activity. Additionally, it contains Maillard reaction products, which are formed during the coffee roasting process [41][31]. The caffeine content in roasted coffee silver skin ranges between about 0.8 and 1.4 g/100 g [125][116]. Canephora coffee silver skin has a significantly higher caffeine content than Arabica coffee silver skin. Iriondo-DeHond et al. determined the caffeine content in extracts from C. arabica and C. canephora silver skin. Coffee silver skin extracts also contain other bioactive compounds such as 5-caffeoylquinic acid, a chlorogenic acid, and flavonoids such as rutin [126][117]. Thus, coffee silver skin has the potential to be used as a natural and sustainable ingredient in foods [32,126][22][117].
Primarily coffee silver skin could be deployed as a source of antioxidant dietary fiber in foods. For example, coffee silver skin extracts may be used as a natural colorant and as a source of dietary fiber in biscuits [45][35]. Furthermore, coffee silver skin may improve the quality, shelf life, and sensory quantities of barbari bread [44][34]. There are also applications of coffee silver skin in antioxidant beverages [49][39]. Due to its roasting-derived flavors, it could be used to impart the smoke aroma into other foods, e.g., as a smoked salt (authors’ own observation).
In addition to the abovementioned amounts of caffeine, coffee silver skin also contains acrylamide, as this contaminant is produced during roasting [32,45][22][35]. Contamination with mycotoxins, especially ochratoxin A (18.7–34.4 μg/kg) is also possible [127][118]. Iriondo-DeHond et al. investigated the acute toxicity of an aqueous extract of coffee silver skin in rats with a single dose of 2000 mg/kg bw and observed no apparent signs of toxicity, abnormal behavior, or mortality [32][22]. The extract also showed no cytotoxicity in the MTT assay on HepG2 cells. Also in the MTS and lactate dehydrogenase (LDH) assays in HaCaT and HFF-1 cells, no in vitro cytotoxic effects could be detected [128][119]. The comet assay for in vitro genotoxicity testing in HepG2 cells showed that at concentrations up to 1000 µg/mL of coffee silver skin extract, no relevant strand breaks or oxidized bases were induced [126][117]. In a subacute toxicity study, in which rats received 1 g/kg bw of aqueous coffee silver skin extract orally, no negative effects on hormone secretion and antioxidant or anti-inflammatory biomarkers were observed [129][120].
Spent Coffee Grounds
Spent coffee grounds are formed both during the extraction of coffee powder with hot water to produce a coffee beverage and during the production of instant coffee preparations [11][1]. For each kg of instant coffee, 2 kg of wet spent coffee grounds waste are produced, which corresponds to an annual amount of about 6,000,000 t worldwide [11,130][1][121].
In the food industry, coffee grounds could be used as a source of dietary fiber[44][45] [54,55] or in bakery products [51–53][41][42][43], as well as to produce alcoholic distillates [50][40]. After drying the spent coffee grounds, subsequent extraction of coffee oil with supercritical CO2, and sterilization, a “coffee flour” is produced, which is highly fibrous, high in protein, and gluten-free. It is intended for use as a novel food ingredient in savory and sweet recipes, bakery products, confectionery, snacks, and ready-to-eat products [56][46]. The novel food approval is currently pending.
Polysaccharides are the main component of spent coffee grounds, with hemicelluloses accounting for approximately 39 g/100 g dry weight and celluloses for 12 g/100 g dry weight [43,131,132][23][122][123]. Besides sugars, proteins, and minerals, spent coffee grounds also contain fat, which can be extracted as coffee oil and contains the diterpenes kahweol and cafestol [133,134][124][125]. Due to the good water solubility of caffeine, lower caffeine contents are measured in spent coffee grounds (0.007%–0.5%) than in roasted coffee. The caffeine content depends on coffee variety and extraction process [135,136][126][127].
The two food processing contaminants acrylamide and hydroxymethylfurfural result from the Maillard reaction during coffee roasting [137][128]. Spent coffee grounds contain 37.2 ± 0.4 μg/kg of acrylamide and 61.3 ± 0.4 mg/kg of hydroxymethylfurfural (dry weight), respectively [52][42]. Of alkyl pyrazines, which are formed during roasting by the Maillard reaction, about 70%–80% pass into the domestically prepared coffee beverage [138][129]. It can therefore be deduced that the rest remains in spent coffee grounds. In humans, alkyl pyrazines are metabolized to their corresponding carboxylic acids and almost completely excreted renally [139][130]. Spent coffee grounds are susceptible to contamination with mycotoxins, therefore Iriondo-DeHond et al. investigated this. They could not detect aflatoxin B1 and enniatin B but found ochratoxin A in an amount of 2.31 µg/kg. They also carried out an acute toxicity study in rats, in which no visible signs of toxic effects were observed at a dose of 2000 mg/kg bw of spent coffee grounds [134][125].
References
- Pushpa S. Murthy; M. Madhava Naidu; Sustainable management of coffee industry by-products and value addition—A review. Resources, Conservation and Recycling 2012, 66, 45-58, 10.1016/j.resconrec.2012.06.005.
- M.N. Clifford; T. Williams; D. Bridson; Chlorogenic acids and caffeine as possible taxonomic criteria in Coffea and Psilanthus. Phytochemistry 1989, 28, 829-838, 10.1016/0031-9422(89)80124-4.
- A. P. Davis; Rafaël Govaerts; Diane M. Bridson; Piet Stoffelen; An annotated taxonomic conspectus of the genus Coffea (Rubiaceae). Botanical Journal of the Linnean Society 2006, 152, 465-512, 10.1111/j.1095-8339.2006.00584.x.
- Belitz, H.-D.; Grosch, W.; Schieberle, P. Lehrbuch der Lebensmittelchemie, 6th ed.; Springer: Berlin/Heidelberg, Germany, 2008.
- Yusaku Narita; Kuniyo Inouye; Review on utilization and composition of coffee silverskin. Food Research International 2014, 61, 16-22, 10.1016/j.foodres.2014.01.023.
- Thi Minh Thu Nguyen; Eun Jin Cho; Younho Song; Chi Hoon Oh; Ryo Funada; Hyeun-Jong Bae; Use of coffee flower as a novel resource for the production of bioactive compounds, melanoidins, and bio-sugars.. Food Chemistry 2019, 299, 125120, 10.1016/j.foodchem.2019.125120.
- Ratanamarno, S.; Surbkar, S. Caffeine and catechins in fresh coffee leaf (Coffea arabica) and coffee leaf tea. Maejo Int. J. Sci. Technol. 2017, 11, 211–218.
- Campa, C.; Petitvallet, A. Beneficial compounds from coffee leaves. In Achieving Sustainable Cultivation of Coffee; Lashermes, P., Ed.; Burleigh Dodds Science Publishing Limited: Milton, UK, 2018; pp. 237–258.
- Kamiya, H. Manufacturing method of beverage raw material. Patent No. JP-2017153463-A, 1 March 2016.
- Rilma, N.; Anwar, K.; Tuty, A.; Deddi, P.P. Kahwa daun: Traditional knowledge of a coffee leaf herbal tea from West Sumatera, Indonesia. J. Ethn. Foods 2018, 5, 286–291.
- Yuwono, S.S.; Fibrianto, K.; Wahibah, L.Y.; Wardhana, A.R. Sensory attributes profiling of dampit robusta coffee leaf tea (Coffea canephora). Carpathian J. Food Sci. Technol. 2019, 164–175.
- Madahava Naidu, M.; Vijayanada, P.; Usha Devi, A.; Vijayalakshmi, M.R.; Ramalakshmi, K. Utilization of coffee by-products in food industry, preparation of jam using coffee pulp as raw material. In Plantation Crops Research and Development in the New Millennium: PLACROSYM XIV; Rethinam, P., Ed.; Indian Society for Plantation Crops: Kasaragod, India, 2004; pp. 201–203.
- Ramirez Velez, A.; Jaramillo Lopez, J.C. Process for Obtaining Honey and/or Flour of Coffee from the Pulp or Husk and the Mucilage of the Coffee Bean. U.S. Patent No. US20150017270A1, 14 December 2011.
- orres-Valenzuela, L.S.; Serna-Jiménez, J.A.; Martínez, K; Coffee by-products: Nowadays and perspectives. IntechOpen 2019, null, null, 10.5772/intechopen.89508.
- Andrea Heeger; Agnieszka Kosińska-Cagnazzo; Ennio Cantergiani; Wilfried Andlauer; Bioactives of coffee cherry pulp and its utilisation for production of Cascara beverage. Food Chemistry 2017, 221, 969-975, 10.1016/j.foodchem.2016.11.067.
- Brenna, C. What Is Cascara? Available online: https://www.freshcup.com/what-is-cascara/ (accessed on 16 March 2020).
- Velissariou, M.; Laudano, R.J.; Edwards, P.M.; Stimpson, S.M.; Jeffries, R.L. Beverage derived from the extract of coffee cherry husks and coffee cherry pulp. U.S. Patent No. US7833560B2, 18 March 2005
- Carlsen, Z. Magic in the Moonshine: Cascara Booze Is Here. Available online: https://sprudge.com/magic-in-the-moonshine-cascara-booze-is-here-115811.html (accessed on 16 March 2020).
- Discarded Spirits Co. Discarded Vermouth. Available online: https://www.discardedspirits.com/products/discarded-vermouth (accessed on 16 March 2020).
- Ota, K.; Coffee as a global beverage before 1700. . J. Int. Econ. Stud. 2018, 3, 43–55, 10.15002/00014518.
- Maxwell, G.W.; Poor man’s coffee? Yemeni qishr, qahwa, and qat.. ea Coffee Trade J. 1996, 168, 64–68.
- Amaia Iriondo De-Hond; Natalia Aparicio García; Beatriz Fernandez-Gomez; Eduardo Guisantes-Batán; Francisco Velázquez Escobar; Gracia Patricia Blanch; Manuel San Andres; Sebastián Sánchez-Fortún Rodríguez; María Dolores Del Castillo; Validation of coffee by-products as novel food ingredients. Innovative Food Science & Emerging Technologies 2019, 51, 194-204, 10.1016/j.ifset.2018.06.010.
- Igho J Onakpoya; Rohini Terry; Edzard Ernst; The Use of Green Coffee Extract as a Weight Loss Supplement: A Systematic Review and Meta-Analysis of Randomised Clinical Trials. Gastroenterology Research and Practice 2010, 2011, 1-6, 10.1155/2011/382852.
- Adriana Farah; Mariana Monteiro; Carmen. M Donangelo; Sophie Lafay; Chlorogenic Acids from Green Coffee Extract are Highly Bioavailable in Humans. The Journal of Nutrition 2008, 138, 2309-2315, 10.3945/jn.108.095554.
- Wendy Marcason; What Is Green Coffee Extract?. Journal of the Academy of Nutrition and Dietetics 2013, 113, 364, 10.1016/j.jand.2012.12.004.
- Clarke, R.J. The Technology of converting green coffee into the beverage. In Coffee: Botany, Biochemistry and Production of Beans and Beverage; Clifford, M.N., Willson, K.C., Eds.; Springer: Boston, MA, USA, 1985; pp. 375–393.
- Lukas Macheiner; Anatol Schmidt; Matthias Schreiner; Helmut K. Mayer; Green coffee infusion as a source of caffeine and chlorogenic acid. Journal of Food Composition and Analysis 2019, 84, 103307, 10.1016/j.jfca.2019.103307.
- Pushpa S. Murthy; M. Madahava Naidu; Production and Application of Xylanase from Penicillium sp. Utilizing Coffee By-products. Food and Bioprocess Technology 2010, 5, 657-664, 10.1007/s11947-010-0331-7.
- Pushpa S. Murthy; M. Madhava Naidu; Recovery of Phenolic Antioxidants and Functional Compounds from Coffee Industry By-Products. Food and Bioprocess Technology 2010, 5, 897-903, 10.1007/s11947-010-0363-z.
- Anabela Guedes Da Costa; Rita C. Alves; Ana Vinha; Elísio Costa; Catarina S.G. Costa; Maria Antónia Nunes; Agostinho A. Almeida; Alice Santos-Silva; M. Beatriz P. P. Oliveira; Nutritional, chemical and antioxidant/pro-oxidant profiles of silverskin, a coffee roasting by-product. Food Chemistry 2018, 267, 28-35, 10.1016/j.foodchem.2017.03.106.
- Rosa Cinzia Borrelli; Fabrizio Esposito; Aurora Napolitano; Alberto Ritieni; Vincenzo Fogliano; Characterization of a New Potential Functional Ingredient: Coffee Silverskin. Journal of Agricultural and Food Chemistry 2004, 52, 1338-1343, 10.1021/jf034974x.
- Ana Jiménez-Zamora; Silvia Pastoriza; José Ángel Rufián-Henares; Revalorization of coffee by-products. Prebiotic, antimicrobial and antioxidant properties. LWT 2015, 61, 12-18, 10.1016/j.lwt.2014.11.031.
- Lina F. Ballesteros; José A. Teixeira; Solange I. Mussatto; Chemical, Functional, and Structural Properties of Spent Coffee Grounds and Coffee Silverskin. Food and Bioprocess Technology 2014, 7, 3493-3503, 10.1007/s11947-014-1349-z.
- Amir Pourfarzad; Hadi Mahdavian-Mehr; Naser Sedaghat; Coffee silverskin as a source of dietary fiber in bread-making: Optimization of chemical treatment using response surface methodology. LWT 2013, 50, 599-606, 10.1016/j.lwt.2012.08.001.
- Esther Garcia-Serna; Nuria Martinez-Saez; Marta Mesias; Francisco J. Morales; M. Castillo; Use of Coffee Silverskin and Stevia to Improve the Formulation of Biscuits. Polish Journal of Food and Nutrition Sciences 2014, 64, 243-251, 10.2478/pjfns-2013-0024.
- Gizem Ateş; Yeşim Elmaci; Coffee silverskin as fat replacer in cake formulations and its effect on physical, chemical and sensory attributes of cakes. LWT 2018, 90, 519-525, 10.1016/j.lwt.2018.01.003.
- Gizem Ateş; Yeşim Elmaci; Physical, chemical and sensory characteristics of fiber-enriched cakes prepared with coffee silverskin as wheat flour substitution. Journal of Food Measurement and Characterization 2018, 13, 755-763, 10.1007/s11694-018-9988-9.
- Martinez-Saez, N.; del Castillo, M.D. Development of sustainable novel foods and beverages based on coffee by-products for chronic diseases. In Encyclopedia of Food Security and Sustainability; Ferranti, P., Berry, E., Jock, A., Eds.; Elsevier: San Diego, CA, USA, 2018; pp. 307–315.
- Nuria Martinez-Saez; Mónica Ullate; María A. Martín-Cabrejas; Patricia Martorell; Salvador Genovés; Daniel Ramón; María-Dolores Mesa; A novel antioxidant beverage for body weight control based on coffee silverskin. Food Chemistry 2014, 150, 227-234, 10.1016/j.foodchem.2013.10.100.
- Armando Sampaio; Giuliano Dragone; M. Vilanova; José M. Oliveira; José A. Teixeira; Solange I. Mussatto; Production, chemical characterization, and sensory profile of a novel spirit elaborated from spent coffee ground. LWT 2013, 54, 557-563, 10.1016/j.lwt.2013.05.042.
- del Castillo, M.D.; Ibanez, M.E.; Amigo-Benavent, M.; Herrero, M.; Plaza, M.; Ullate, M. Aplicacion de Pruductos de la Cascarilla del Cafe en Cosmetica Antienvejecimiento y Alimentacion Funcional. Patent No. WO2013004873A1, 4 July 2011.
- Nuria Martinez-Saez; Alba Tamargo García; Inés Domínguez Pérez; Miguel Rebollo-Hernanz; Marta Mesias; Francisco J. Morales; María A. Martín-Cabrejas; María Dolores Del Castillo; Use of spent coffee grounds as food ingredient in bakery products. Food Chemistry 2017, 216, 114-122, 10.1016/j.foodchem.2016.07.173.
- Kenia Vázquez-Sánchez; Nuria Martinez-Saez; Miguel Rebollo-Hernanz; María Dolores Del Castillo; Marcela Gaytán-Martínez; Rocio Campos-Vega; In vitro health promoting properties of antioxidant dietary fiber extracted from spent coffee (Coffee arabica L.) grounds.. Food Chemistry 2018, 261, 253-259, 10.1016/j.foodchem.2018.04.064.
- Campos-Vega, R. Proseco de obtencion de fibra dietaria antioxidante natural de subproductos mediante calentamiento ohmico y compuesto alto en fibra dietaria antioxidante natural de cafe usado. Patent No. MX/a/2016008578, 2016.
- Campos-Vega, R.; Vázquez-Sánchez, K.; Martinez-Saez, N.; Castillo, M. Antioxidant coffee dietary fiber for gastrointestinal health and diabetes. In Proceedings of the 20th International Conference of FFC and 8th International Symposium of Academic Society of Functional Foods and Bioactive Compounds, Boston, MA, USA, 22–23 September 2016
- Kaffe Bueno ApS. Coffee Flour (Defatted Coffee Arabica Seed Powder). Available online: https://ec.europa.eu/food/sites/food/files/safety/docs/novel-food_sum_ongoing-app_2018-0698.pdf (accessed on 16 March 2020).
- Vicente A. Mirón-Mérida; Jorge Yáñez-Fernández; Brenda Montañez-Barragán; Blanca Estela Barragán-Huerta; Valorization of coffee parchment waste (Coffea arabica) as a source of caffeine and phenolic compounds in antifungal gellan gum films. LWT 2019, 101, 167-174, 10.1016/j.lwt.2018.11.013.
- Juliana Lischka Sampaio Mayer; Sandra Maria Carmello-Guerreiro; Paulo Mazzafera; A functional role for the colleters of coffee flowers. AoB PLANTS 2013, 5, plt029-plt029, 10.1093/aobpla/plt029.
- Hong, N. Today, We’re Drinking Flowers. Available online: https://www.drinkmagazine.asia/2017/06/16/today-drinking-flowers/ (accessed on 16 March 2020).
- Wintgens, J.N. The coffee plant. In Coffee: Growing, Processing, Sustainable Production: A Guidebook for Growers, Processors, Traders and Researchers; Wintgens, J.N., Ed.; Wiley-VCH: Weinheim, Germany, 2008; pp. 1–24.
- Xiu-Min Chen; A review on coffee leaves: Phytochemicals, bioactivities and applications. Critical Reviews in Food Science and Nutrition 2018, 59, 1008-1025, 10.1080/10408398.2018.1546667.
- Ross, I.A. Medicinal plants of the world, volume 3. In Chemical Constituents, Traditional and Modern Medicinal Uses; Humana Press Inc.: Totowa, NJ, USA, 2005.
- Xiu-Min Chen; Zhili Ma; David D. Kitts; Effects of processing method and age of leaves on phytochemical profiles and bioactivity of coffee leaves. Food Chemistry 2018, 249, 143-153, 10.1016/j.foodchem.2017.12.073.
- Éva Brigitta Patay; Tímea Bencsik; Nóra Papp; Phytochemical overview and medicinal importance of Coffea species from the past until now. Asian Pacific Journal of Tropical Medicine 2016, 9, 1127-1135, 10.1016/j.apjtm.2016.11.008.
- Zhang, H.; Ling, X.; Bai, X.; Guo, T.; Li, J. Method for Making Granular Coffee Leaf Tea. Patent No. CN-105192194-A, 17 June 2014.
- Shan, Y. Making Method for Coffee Leave Tea. Patent No. CN-104719522-A, 9 April 2015.
- Ito, K. Preparation of Tea Extract. Patent No. JP-3115464-B2, 16 December 1993.
- Inoue, M. Coffee Leaf Tea and Its Preparation. Patent No. JP-H08173111-A, 22 December 1994
- Tagagaki, K.; Tsuzaki, S. Healthy Tea and Healthy Drink and Method of Manufacturing the Same. Patent No. JP-2002065227-A, 31 August 2000.
- Iwai, K.; Nakabayashi, Y. Coffee Leave Tea and Method for Producing the Same. Patent No. JP-2002191332-A, 27 December 2000.
- Hirose, Y.; Yoshimura, K.; Yamamoto, K. Manufacturing Method of Coffee Leave Tea Raw Material, and Coffee Leave Tea Beverage Using the Same. Patent No. JP-2013106536-A, 18 November 2011.
- Claudine Campa; Laurence Mondolot; Arsene Rakotondravao; Luc P. R. Bidel; Annick Gargadennec; Emmanuel Couturon; Philippe La Fisca; Jean-Jacques Rakotomalala; Christian Jay-Allemand; Aaron P. Davis; et al. A survey of mangiferin and hydroxycinnamic acid ester accumulation in coffee (Coffea) leaves: biological implications and uses. Annals of Botany 2012, 110, 595-613, 10.1093/aob/mcs119.
- Jyotshna; Puja Khare; Karuna Shanker; Yuling Chi; Mangiferin: A review of sources and interventions for biological activities. BioFactors 2016, 42, 504-514, 10.1002/biof.1308.
- P. Luczkiewicz; Adam Kokotkiewicz; A. Dampc; M. Luczkiewicz; Mangiferin: A promising therapeutic agent for rheumatoid arthritis treatment. Medical Hypotheses 2014, 83, 570-574, 10.1016/j.mehy.2014.08.021.
- Rohit Upadhyay; L. Jagan Mohan Rao; An Outlook on Chlorogenic Acids—Occurrence, Chemistry, Technology, and Biological Activities. Critical Reviews in Food Science and Nutrition 2013, 53, 968-984, 10.1080/10408398.2011.576319.
- H. Ashihara; A. M. Monteiro; F. M. Gillies; A. Crozier; Biosynthesis of Caffeine in Leaves of Coffee. Plant Physiology 1996, 111, 747-753, 10.1104/pp.111.3.747.
- Wang, S. Vehicle Perfume Useful for Preventing Sleep Comprises Lavender, Pelargonium Odoratissimum, Plum Blossom, Lily, Michelia, Laggera Pterodonta, Coffee Leaf, Ethanol, Plant Extracts, Citric Acid, Folic Acid and Sodium Chloride. Patent No. CN-107998268-A, 4 December 2017.
- Madya, A.P. Sutriyo Gel Composition Used for Manufacturing Gel-Shaped Product as Coagulant of Dead Skin Cells Protein, Contains Coffee Leaf Powder, Triethanolamine, Carbomer 940, Sodium Laureth Sulfate, Cocamidopropyl Betaine and Methyl Paraben. Patent No. ID-201606611-A, 6 May 2015.
- Teruel, E.B. Novel tobacco substitute. Patent No. US 2008/0017208 A1, 20 July 2006.
- Xu, H. Cigarette with Coffee Flavor and Its Preparing Method. Patent No. CN-1206948-C, 15 January 2003.
- Ye, H. Feed for Promoting Growth and Development of Broilers and Preparation Method Thereof. Patent No. CN-104431535-A, 10 December 2014.
- Furukawa, Y.; Hizaki, S.; Nii, H.; Yamauchi, M.; Yoshimur, H. Lactic Acid Proliferating Agent and Its Production. Patent No. JP-H06125771-A, 21 October 1992.
- Hizaki, S.; Yamauchi, M. Enteric-Coated Lactobacillus Granule. Patent No. JP-H06133735-A, 23 October 1992
- Ragot, P.; Barat, L.; Rousseau, C.; Pons, E. Reconstituted Plant Material and Its Use for Packaging, Wrapping and Food Appliances. Patent No. US-20170174404A1, 28 March 2014.
- Kim, T.H.; Lee, S.S.; Lee, H.H.; Bae, H.K.; Song, J.A.; Kim, J.M. Absorbent Pad Containing Coffee Leaves. Patent No. KR-101754481-B1, 31 August 2015.
- Ucc Ueshima Kohi, K. Deodoriser for Hospital Beds-Contains Adsorption Agent e.g., Dried Coffee Leaves or Tea Waste. Patent No. JP-10314286-A, 15 May 1997.
- EFSA. Technical Report on the Notification of Infusion from Coffee Leaves (Coffea Arabica L. and/or Coffea Canephora Pierre ex A. Froehner) as a Traditional Food from a Third Country Pursuant to Article 14 of Regulation (EU) 2015/2283; EFSA Supporting Publications; EFSA: Parma, Italy, 2020; Volume 17, EN-1783.
- Efsa Panel On Food Additives And Nutrient Sources Added To Food (Ans); Maged Younes; Peter Aggett; Fernando Aguilar; Riccardo Crebelli; Birgit Dusemund; Metka Filipič; Maria Jose Frutos; Pierre Galtier; David Gott; et al.Ursula Gundert‐RemyClaude LambréJean‐Charles LeblancInger Therese LillegaardPeter MoldéusAlicja MortensenAgneta OskarssonIvan StankovicIne Waalkens‐BerendsenRudolf Antonius WoutersenRaúl J AndradeCristina FortesPasquale MosessoPatrizia RestaniDavide ArcellaFabiola PizzoCamilla SmeraldiMatthew Wright Scientific opinion on the safety of green tea catechins. EFSA Journal 2018, 16, 5239, 10.2903/j.efsa.2018.5239.
- Jiang Hu; Nna Webster; Joyce Cao; Andrew Shao; The safety of green tea and green tea extract consumption in adults – Results of a systematic review. Regulatory Toxicology and Pharmacology 2018, 95, 412-433, 10.1016/j.yrtph.2018.03.019.
- Prado, Y.; Merino, N.; Acosta, J.; Herrera, J.A.; Luque, Y.; Hernández, I.; Prado, E.; Garrido, G.; Delgado, R.; Rodeiro, I.; et al. Acute and 28-day subchronic toxicity studies of mangiferin, a glucosyl xanthone isolated from Mangifera indica L. stem bark.. J. Pharm. Pharmacogn. Res. 2015, 3, 13–23.
- Robin A. Reddeman; Róbert Glávits; John R. Endres; Amy E. Clewell; Gábor Hirka; Adél Vértesi; Erzsébet Béres; Ilona Pasics Szakonyiné; A Toxicological Evaluation of Mango Leaf Extract (Mangifera indica) Containing 60% Mangiferin.. Journal of Toxicology 2019, 2019, 4763015-14, 10.1155/2019/4763015.
- International Trade Centre. The Coffee Guide—Grading and Classification. Available online: http://www.thecoffeeguide.org/coffee-guide/world-coffee-trade/grading-and-classification/ (accessed on 25 March 2020).
- Bundesministerium für Verbraucherschutz, Ernährung und Landwirtschaft. Verordnung über Kaffee, Kaffee- und Zichorien-Extrakte vom 15. November 2001 (BGBl. I S. 3107), die zuletzt durch Artikel 6 der Verordnung vom 5. Juli 2017 (BGBl. I S. 2272) geändert worden ist: KaffeeV. Bundesgesetzblatt I 2017, G5702, 2272–2289.
- Verónica Alejandra Bonilla-Hermosa; Whasley Ferreira Duarte; Rosane Freitas Schwan; Utilization of coffee by-products obtained from semi-washed process for production of value-added compounds. Bioresource Technology 2014, 166, 142-150, 10.1016/j.biortech.2014.05.031.
- Franca, A.S.; Oliveira, L.S. Coffee processing solid wastes: Current uses and future perspectives (chapter 8). In Agricultural Wastes; Ashworth, G.S., Azevedo, P., Eds.; Nova Science: New York, NY, USA, 2009.
- M.N. Clifford; J.R. Ramirez-Martinez; Phenols and caffeine in wet-processed coffee beans and coffee pulp. Food Chemistry 1991, 40, 35-42, 10.1016/0308-8146(91)90017-i.
- Vanesa Benitez; Miguel Rebollo-Hernanz; Sara Hernanz; Silvia Chantres; Yolanda Aguilera; María A. Martín-Cabrejas; Coffee parchment as a new dietary fiber ingredient: Functional and physiological characterization. Food Research International 2019, 122, 105-113, 10.1016/j.foodres.2019.04.002.
- Samson Ayele Bekalo; H.W. Reinhardt; Fibers of coffee husk and hulls for the production of particleboard. Materials and Structures 2009, 43, 1049-1060, 10.1617/s11527-009-9565-0.
- Giuliano, J. What Is Cascara?—Exploring Coffee Cherry Tea. Available online: http://www.manualcoffeebrewing.com/what-is-cascara-how-to-brew-is-and-where-to-get-it-exploring-coffee-cherry-tea/ (accessed on 19 March 2020).
- Miguel Rebollo-Hernanz; Qiaozhi Zhang; Yolanda Aguilera; Mercedes M. Pedrosa; Elvira Gonzalez De Mejia; Phenolic compounds from coffee by-products modulate adipogenesis-related inflammation, mitochondrial dysfunction, and insulin resistance in adipocytes, via insulin/PI3K/AKT signaling pathways.. Food and Chemical Toxicology 2019, 132, 110672, 10.1016/j.fct.2019.110672.
- Miguel Rebollo-Hernanz; Qiaozhi Zhang; Yolanda Aguilera; Mercedes M. Pedrosa; Elvira Gonzalez De Mejia; Relationship of the Phytochemicals from Coffee and Cocoa By-Products with their Potential to Modulate Biomarkers of Metabolic Syndrome In Vitro.. Antioxidants 2019, 8, 279, 10.3390/antiox8080279.
- B. M. Gouvea; C. Torres; A. S. Franca; Leandro S. Oliveira; E. S. Oliveira; Feasibility of ethanol production from coffee husks. Biotechnology Letters 2009, 31, 1315-1319, 10.1007/s10529-009-0023-4.
- R. Russell M. Paterson; Nelson Lima; Marta H. Taniwaki; Coffee, mycotoxins and climate change. Food Research International 2014, 61, 1-15, 10.1016/j.foodres.2014.03.037.
- EFSA. Opinion of the scientific panel on contaminants in the food chain [CONTAM] related to ochratoxin A in food. EFSA J. 2006, 4, 365.
- Patricia Esquivel; Víctor M. Jiménez; Functional properties of coffee and coffee by-products. Food Research International 2012, 46, 488-495, 10.1016/j.foodres.2011.05.028.
- Thierry Joët; Andréina Laffargue; Frédéric Descroix; Sylvie Doulbeau; Benoît Bertrand; Alexandre De Kochko; Stéphane Dussert; Influence of environmental factors, wet processing and their interactions on the biochemical composition of green Arabica coffee beans. Food Chemistry 2010, 118, 693-701, 10.1016/j.foodchem.2009.05.048.
- A. Gustavo Gonzalez; F. Pablos; M.J. Martı́n; M. León-Camacho; M.S. Valdenebro; HPLC analysis of tocopherols and triglycerides in coffee and their use as authentication parameters. Food Chemistry 2001, 73, 93-101, 10.1016/s0308-8146(00)00282-x.
- M. Madhava Naidu; G. Sulochanamma; S.R. Sampathu; P. Srinivas; Studies on extraction and antioxidant potential of green coffee. Food Chemistry 2008, 107, 377-384, 10.1016/j.foodchem.2007.08.056.
- C Campa; S Doulbeau; S Dussert; S Hamon; M. Noirot; Qualitative relationship between caffeine and chlorogenic acid contents among wild species. Food Chemistry 2005, 93, 135-139, 10.1016/j.foodchem.2004.10.015.
- C.-L Ky; J Louarn; S Dussert; B Guyot; S Hamon; M Noirot; Caffeine, trigonelline, chlorogenic acids and sucrose diversity in wild Coffea arabica L. and C. canephora P. accessions. Food Chemistry 2001, 75, 223-230, 10.1016/s0308-8146(01)00204-7.
- F. Anthony; M. N. Clifford; M. Noirot; Biochemical diversity in the genus Coffea L.: chlorogenic acids, caffeine and mozambioside contents. Genetic Resources and Crop Evolution 1993, 40, 61-70, 10.1007/bf00052636.
- Clifford, M.N. Chemical and physical aspects of green coffee and coffee products. In Coffee: Botany, Biochemistry and Production of Beans and Beverage; Clifford, M.N., Willson, K.C., Eds.; Springer: Boston, MA, USA, 1985; pp. 305–374.
- Meenakshi Arya; L. Jagan Mohan Rao; An Impression of Coffee Carbohydrates. Critical Reviews in Food Science and Nutrition 2007, 47, 51-67, 10.1080/10408390600550315.
- Oestreich-Janzen, S. Chemistry of coffee. In Comprehensive Natural Products II: Chemistry and Biology; Mander, L., Ed.; Elsevier: Amsterdam, The Netherlands, 2010; pp. 1085–1117.
- Paulo Mazzafera; Maria Bernadete Silvarolla; Caffeine content variation in single green Arabica coffee seeds. Seed Science Research 2010, 20, 163-167, 10.1017/s0960258510000140.
- Yigzaw Dessalegn; Maryke Labuschagne; Gary Osthoff; Liezel Herselman; Genetic diversity and correlation of bean caffeine content with cup quality and green bean physical characteristics in coffee (Coffea arabicaL.). Journal of the Science of Food and Agriculture 2008, 88, 1726-1730, 10.1002/jsfa.3271.
- Roman Lang; Erkan Firat Yagar; Rudolf Eggers; Thomas Hofmann; Quantitative Investigation of Trigonelline, Nicotinic Acid, and Nicotinamide in Foods, Urine, and Plasma by Means of LC-MS/MS and Stable Isotope Dilution Analysis. Journal of Agricultural and Food Chemistry 2008, 56, 11114-11121, 10.1021/jf802838s.
- R H Stadler; Natalia Varga; Christian Milo; Benoit Schilter; Francia Arce Vera; Dieter H. Welti; Alkylpyridiniums. 2. Isolation and Quantification in Roasted and Ground Coffees. Journal of Agricultural and Food Chemistry 2002, 50, 1200-1206, 10.1021/jf011235c.
- R H Stadler; Natalia Varga; Jörg Hau; Francia Arce Vera; Dieter H. Welti; Alkylpyridiniums. 1. Formation in Model Systems via Thermal Degradation of Trigonelline. Journal of Agricultural and Food Chemistry 2002, 50, 1192-1199, 10.1021/jf011234k.
- Atsushi Suzuki; Daiji Kagawa; Ryuji Ochiai; Ichiro Tokimitsu; Ikuo Saito; Green Coffee Bean Extract and Its Metabolites Have a Hypotensive Effect in Spontaneously Hypertensive Rats. Hypertension Research 2002, 25, 99-107, 10.1291/hypres.25.99.
- E Thom; The Effect of Chlorogenic Acid Enriched Coffee on Glucose Absorption in Healthy Volunteers and Its Effect on Body Mass When Used Long-term in Overweight and Obese People. Journal of International Medical Research 2007, 35, 900-908, 10.1177/147323000703500620.
- Takuya Watanabe; Yoichi Arai; Yuki Mitsui; Tatsuya Kusaura; Wataru Okawa; Yasushi Kajihara; Ikuo Saito; The Blood Pressure-Lowering Effect and Safety of Chlorogenic Acid from Green Coffee Bean Extract in Essential Hypertension. Clinical and Experimental Hypertension 2006, 28, 439-449, 10.1080/10641960600798655.
- Naila Albertina De Oliveira; Thaisa Meira Sandini; Heber Peleg Cornelio-Santiago; Elaine Cristina Lanzoni Martinelli; Leonila Ester Reinert Raspantini; Paulo Cesar Raspantini; Cláudia Momo; Alessandra Lopes De Oliveira; Heidge Fukumasu; Leonilda Ester Reinert Raspantini; et al. Acute and subacute (28 days) toxicity of green coffee oil enriched with diterpenes cafestol and kahweol in rats.. Regulatory Toxicology and Pharmacology 2019, 110, 104517, 10.1016/j.yrtph.2019.104517.
- EFSA Panel on Dietetic Products, Nutrition and Allergies. Scientific opinion on the safety of caffeine. EFSA J. 2015, 13, 4102.
- Lukas Vaclavik; Marta Vaclavikova; Timothy H. Begley; Alexander J. Krynitsky; Jeanne I. Rader; Determination of Multiple Mycotoxins in Dietary Supplements Containing Green Coffee Bean Extracts Using Ultrahigh-Performance Liquid Chromatography–Tandem Mass Spectrometry (UHPLC-MS/MS). Journal of Agricultural and Food Chemistry 2013, 61, 4822-4830, 10.1021/jf401139u.
- Napolitano, A.; Fogliano, V.; Tafuri, A.; Ritieni, A. Natural occurrence of ochratoxin A and antioxidant activities of green and roasted coffees and corresponding byproducts. J. Agric. Food Chem. 2007, 55, 10499–10504.
- Amaia Iriondo De-Hond; Ana I. Haza; Alicia Ávalos; María Dolores Del Castillo; Paloma Morales; Validation of coffee silverskin extract as a food ingredient by the analysis of cytotoxicity and genotoxicity. Food Research International 2017, 100, 791-797, 10.1016/j.foodres.2017.08.012.
- Toschi, T.G.; Cardenia, V.; Bonaga, G.; Mandrioli, M.; Rodriguez-Estrada, M.T. Coffee silverskin: Characterization, possible uses, and safety aspects. J. Agric. Food Chem. 2014, 62, 10836–10844.
- Rodrigues, F.; Gaspar, C.; Palmeira-de-Oliveira, A.; Sarmento, B.; Helena Amaral, M.; P P Oliveira, M.B. Application of coffee silverskin in cosmetic formulations: Physical/antioxidant stability studies and cytotoxicity effects. Drug Dev. Ind. Pharm. 2016, 42, 99–106.
- Iriondo-DeHond, A.; Rios, M.B.; Herrera, T.; Rodriguez-Bertos, A.; Nuñez, F.; San Andres, M.I.; Sanchez-Fortun, S.; Del Castillo, M.D. Coffee silverskin extract: Nutritional value, safety and effect on key biological functions. Nutrients 2019, 11, 2693
- Mussatto, S.I.; Carneiro, L.M.; Silva, J.P.A.; Roberto, I.C.; Teixeira, J.A. A study on chemical constituents and sugars extraction from spent coffee grounds. Carbohydr. Polym. 2011, 83, 368–374
- Del Castillo, M.D.; Fernandez-Gomez, B.; Martinez-Saez, N.; Iriondo-DeHond, A.; Mesa, M.D. Coffee By-products (chapter 12). In Coffee: Production, Quality and Chemistry; Farah, A., Ed.; Royal Society of Chemistry: London, UK, 2019; pp. 309–334.
- Janissen, B.; Huynh, T. Chemical composition and value-adding applications of coffee industry by-products: A. review. Resour. Conserv. Recycl. 2018, 128, 110–117.
- Campos-Vega, R.; Loarca-Piña, G.; Vergara-Castañeda, H.A.; Oomah, B.D. Spent coffee grounds: A review on current research and future prospects. Trends Food Sci. Technol. 2015, 45, 24–36.
- Iriondo-DeHond, A.; Cornejo, F.S.; Fernandez-Gomez, B.; Vera, G.; Guisantes-Batan, E.; Alonso, S.G.; Andres, M.I.S.; Sanchez-Fortun, S.; Lopez-Gomez, L.; Uranga, J.A.; et al. Bioaccessibility, metabolism, and excretion of lipids composing spent coffee grounds. Nutrients 2019, 11, 1411.
- Ramalakshmi, K.; Rao, L.J.M.; Takano-Ishikawa, Y.; Goto, M. Bioactivities of low-grade green coffee and spent coffee in different in vitro model systems. Food Chem. 2009, 115, 79–85.
- Andrade, K.S.; Gonçalvez, R.T.; Maraschin, M.; Ribeiro-do-Valle, R.M.; Martínez, J.; Ferreira, S.R.S. Supercritical fluid extraction from spent coffee grounds and coffee husks: Antioxidant activity and effect of operational variables on extract composition. Talanta 2012, 88, 544–552.
- Lachenmeier, D.W.; Schwarz, S.; Teipel, J.; Hegmanns, M.; Kuballa, T.; Walch, S.G.; Breitling-Utzmann, C.M. Potential antagonistic effects of acrylamide mitigation during coffee roasting on furfuryl alcohol, furan and 5-hydroxymethylfurfural. Toxics 2018, 7, 1.
- Pickard, S.; Wilms, H.; Richling, E. Alkylpyrazine contents of coffee beverages using stable isotope dilution gas chromatography–mass spectrometry. LWT Food Sci. Technol. 2014, 58, 188–193.
- Kremer, J.I.; Pickard, S.; Stadlmair, L.F.; Glaß-Theis, A.; Buckel, L.; Bakuradze, T.; Eisenbrand, G.; Richling, E. Alkylpyrazines from coffee are extensively metabolized to pyrazine carboxylic acids in the human body. Mol. Nut. Food Res. 2019, e1801341
