1. Avian Influenza Virus
1.1. Etiology
The influenza virus is an RNA virus part of the
Orthomyxoviridae family with seven genera, namely
Influenzavirus A,
Influenzavirus B,
Influenzavirus C,
Influenzavirus D,
Thogotovirus, and
Isavirus and
Quarajavirus [
24,
25]; moreover,
Influenzavirus A has been identified in a wide range of hosts with the highest genetic variability and is the only one capable of infecting birds [
26,
27]. Moreover, due to the segmented nature of the viral genome, new strains can emerge through genetic reassortment and antigenic drift, further increasing the difficulty in its control and prevention [
28].
The AI virus subtypes depend on the antigen present on the surface of the influenza A virus; there are 16 hemagglutinin subtypes and 9 neuraminidase subtypes [
29,
30]. However, recent scientific studies reported new HA subtypes (18 in total) and NA (11 in total), which were isolated in bats [
31].
Additionally, there are two specific lineages of the HA subtype that phylogenetically divide into the Eurasian and North American lineages; these lineages have progressively evolved independently due to limited intercontinental contact between avian populations [
32].
1.2. Pathogenicity and Virulence
AI viruses generally cause gastrointestinal disturbances in birds with minimal clinical signs and are classified as LPAI viruses. LPAI virus subtypes H5 and H7 circulate naturally in domestic birds but can evolve and become highly pathogenic [
33].
Pathogenicity results from the accumulation of multiple basic amino acids at the HA cleavage site (termed the polybasic cleavage site or polybasic motif), allowing the HA molecule to develop outside the gastrointestinal tract and establish a systemic infection, causing an outbreak of HPAI that is characterized by rapid disease onset and progression associated with high mortality rates [
4,
32,
34]. Moreover, some H5 and H7 viruses of low and high pathogenicity show virulence in mammals, and the highly pathogenic viruses can cause systemic infection in animal models [
32,
35,
36].
1.2.1. Low Pathogenic Avian Influenza Virus (LPAI)
LPAI has a low mortality rate and ability to infect, causing little to no disease in birds, because they can only replicate in tracheal tissues and the small intestine [
24]. However, the H5/H7 subtypes of low pathogenicity (common in poultry and wild waterfowl) [
37] can mutate by insertion and recombination processes in the proteolytic cleavage site of HA [
6] until becoming HPAI viruses [
38].
1.2.2. Highly Pathogenic Avian Influenza Virus (HPAI)
The HPAI virus can cross respiratory and intestinal barriers, diffuse to the blood, and damage all tissues of the bird [
37]. HPAI refers to strains with an “intravenous pathogenicity index” (IVPI) greater than 1.2 or a mortality rate equal to or higher than 75% of the total number of poultry over a period of 10 days [
39].
The HPAI pathogenic strains of avian influenza belong to the H5 and H7 subtypes, with bird mortality that exceeds 90–100% during the 48 h after disease onset [
39,
40].
To date, subtypes H5 and H7 have been recognized as HPAI viruses capable of generating acute and considerable diseases in chickens, turkeys, and other economically significant birds. Moreover, H9 has been included as another subtype with pandemic risk because their high mutability could favor the evolution of viruses that allow sustained transmission in the human species, and H9 can cause zoonotic infections [
37].
1.3. Transmission Mechanisms
Bird-to-Bird Transmission
Wild waterfowl are natural reservoirs of the AI virus and play a role in spreading through their long-distance migratory routes [
41], infecting land birds and domesticated waterfowl via contaminated water sources or food [
14]. However, the oral–fecal path is the main transmission route between birds due to the high viral levels in the fecal matter of infected birds, and it can be transmissible for approximately 21 days [
17,
25].
Chatziprodromidou et al. described proximity to water as a significant risk factor for virus transmission because there may be a close interaction between migratory birds and commercial poultry activities, increasing disease transmission [
9].
AI virus can also be transmitted through secretions and body fluids, such as saliva, mucus, and urine [
5]. In the production systems, these fluids and feces contaminate the clothing and footwear of operators, cages, implements, and mechanical equipment for egg collection, among others. This route has been considered the principal vehicle for disease dissemination within flocks [
1], making commercial poultry responsible for epidemics registered worldwide [
42].
1.4. Interspecies Transmission
1.4.1. Transmission to Mammals
Direct contact is the main route of transmission because it has not been demonstrated that the virus can effectively infect mammals through aerosols [
43]. Transmission to other species generally occurs after virus circulation in densely populated infected avian species, indicating that AI viruses can adapt to promote the spread [
14]. For effective transmission and replication in mammals, the virus must evolve and mutate until it reaches compatibility with the new host environment; this is known as viral reassortment, which has been responsible for the appearance of almost all pandemic viruses in the past [
44,
45,
46].
Infections with avian influenza virus have been reported in cats, mice, and pigs with AI subtype H5N6 [
47,
48,
49]; in canines with subtype H3N8 [
50]; and in tigers and leopards with subtype H1N1 [
51,
52]. All of them have been epidemiologically related to avian influenza outbreaks. Furthermore, avian influenza subtypes have been isolated in ferrets and laboratory animals to evaluate their pathogenicity [
53,
54].
1.4.2. Zoonotic Transmission
Avian influenza viruses have demonstrated the capacity to cross the barrier between species for multifactorial reasons that have favored transmission. Certain mammals, such as bats [
55], pigs [
56], cats, dogs, horses, ferrets, sea lions, and bats [
57], can act as reservoirs, which allow genetic mixing between viruses that intend to infect humans and birds [
39]. Moreover, host susceptibility, exposure level to infected birds, viral mutations, and favorable environmental conditions form an ideal scenario for the zoonotic transmission of the avian influenza virus [
58].
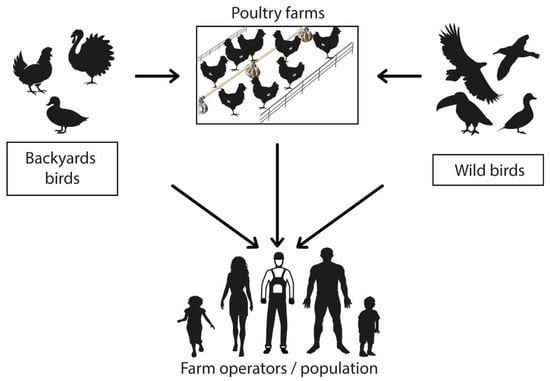
The main route of transmission between birds and humans is direct contact with the feces or secretions of infected animals and exposure to contaminated or virus-infected environments (
Figure 1) [
59,
60]. There is no evidence of human-to-human infection [
5,
14]. People within the poultry production chain (from farm to table) are at a higher infection risk than the general population due to prolonged exposure to the infectious agent [
4].
Figure 1. Avian influenza virus transmission mechanism. Graphic representation of virus zoonotic potential.
1.5. Virus Reservoirs
The avian influenza virus ecological niche or natural reservoir are waterfowl belonging to
Anseriformes (waterfowl, ducks, geese, and swans) and
Charadriiformes (gulls and shorebirds), which include more than 100 species of wild birds belonging to about 25 different families, indicating the global distribution of the virus in free-living waterfowl [
34,
61].
Subtype AI viruses (H5, H7, H6, and H9) can be found in both waterfowl and poultry [
62], and several papers have described avian influenza viruses identified in different mammals. For instance, the AI subtype (H3, H7) in equines, the AI subtype (H1, H3) in swine, and the AI subtype in aquatic mammals (H10, H4, H7, and H13) originate from the genetics inherent to viruses naturally found in wild waterfowl (H1–H16) [
25,
63].
1.6. Virus Survival
Studies have found that the virus is more resistant to low temperatures (below 28 °C) [
1,
64]. The avian influenza virus can survive for up to 200 days in the body fluids of infected birds, four days in feces at animal body temperature, 35 days in feces at temperatures below 4 °C, and about five weeks in the environment of the infected poultry house [
1,
64]. The virus can survive in carcasses, meat, and eggs (especially at low temperatures); therefore, upon suspicion or confirmation of positive cases of avian influenza, the products generated should be eliminated [
64]
2. Containment Measures for Confirmed Cases
2.1. Sanitary Slaughter of Infected Poultry
Within 24 to 48 h after confirmation of a positive case [
1], all birds within a 3 km radius of the infected area should be monitored and traced to establishments with direct or indirect links to the infected premises. A tracing period should be considered, and AI high-risk establishment identification should be performed to opt for stamping out [
40]. Sanitary slaughter could be an option for AI eradication, but it involves complex economic, ethical, environmental, and public health considerations due to the risk of zoonosis. However, if this procedure is chosen, animal welfare must be guaranteed [
77] and be under the current legislation [
78]. There are different procedures to be followed.
2.1.1. Environment Saturation with CO2
Slaughtering by releasing carbon dioxide is one of the most recommended methods because large animal volumes can be euthanized with minimal contact by the responsible personnel. For example, in optimum conditions, a 50 L tank is enough to sacrifice 20–30 thousand birds [
1]. This process can be carried out inside the same sheds where the animals are kept or by placing them in vans, cages, or airtight containers that ensure CO
2 concentration remains stable [
1]. Birds should first be exposed to concentrations below 40% CO
2 and once they lose consciousness due to the anesthetic and central nervous system depressant gas effect, the concentration values can be raised to above 60% [
79].
2.1.2. Carbon Dioxide Foam
The animals must be placed at ground level, and the foam should be concentrated at 1% CO
2 in water. The entire bird should be covered at approximately 1.50 m in height or 30 cm above their heads [
1,
80,
81]. This is a very efficient method to deal with emerging outbreaks because total depopulation can be carried out quickly by inducing hypoxia in the animals [
80,
81].
2.1.3. Cervical Dislocation
In case the previously mentioned methods are not available, euthanasia should be performed by manual or mechanical separation of the skull from the vertebral column after dislocating the neck of the bird [
69]. Cervical dislocation is a non-invasive method; however, it requires trained personnel and is inefficient for large flocks or birds, in addition to the long exposure time of the responsible personnel [
82].
2.2. Disposal
2.2.1. Burial
For the disposal of the slaughtered birds and all the implements that have been in contact with the infected area (protective clothing, bedding, feed, and eggs), burial is a cost-effective and efficient option [
1]. Heavy machinery and sufficient space should be available to dig the pits, occupying approximately 200–300 birds per cubic meter [
1]. Long trenches that are not too large should be considered, and the channels should be covered with a layer of soil of about 40 cm, followed by an even layer of calcium dioxide and, finally, another layer of soil [
40].
2.2.2. Incineration
This method is not recommended for large poultry populations due to its high costs, environmental contamination, and lack of certainty in the effectiveness of the disposal method due to the volatility of the remains [
1]. It is suggested to dig an 8 m long, 2 m wide, and 1 m deep trench; use firewood to completely burn the dead birds; and then add a layer of lime [
83]. The channels should be covered with a layer of soil, a layer of lime, and finally, another layer of soil [
40].
2.3. Infected Poultry Farms’ Disinfection
After slaughter and disposal of carcasses and other contaminated products, recommendations indicate that a three-step cleaning and disinfection protocol must be carried out [
1,
84]:
This entry is adapted from the peer-reviewed paper 10.3390/pathogens12040610