The moderator band (MB) is an intracavitary structure of the right ventricle composed of muscular fibers encompassing specialized Purkinje fibers, separated each other by collagen and adipose tissue. Premature ventricular complexes originating within the Purkinje network have been implicated in the genesis of life-threatening arrhythmias. However, right Purkinje network arrhythmias have been much less reported in the literature compared to the left counterpart. The MB has unique anatomical and electrophysiological properties, which may account for its arrhythmogenicity and may be responsible for a significant portion of idiopathic ventricular fibrillation.
- ventricular tachycardia
- electrical disorder
- moderator band
- Purkinje fibers
- ablation
- pharmacological treatment
1. Introduction
2. Anatomy of the Moderator Band
3. Electrical Properties of the Moderator Band
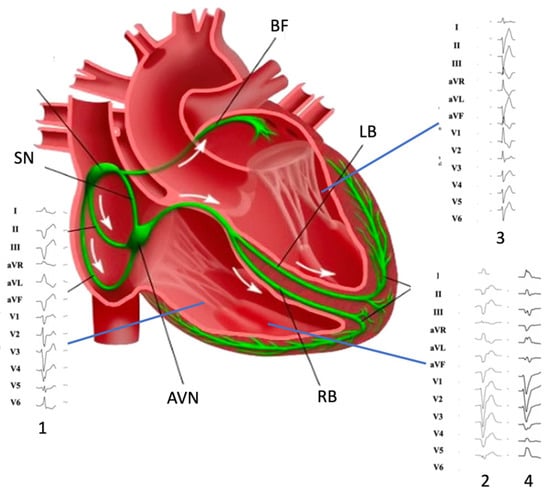
4. Moderator Band-Related Arrhythmias
4.1. Arrhythmogenic Mechanisms
4.2. Right Purkinje Network Arrhythmias: In Addition to the Moderator Band
This entry is adapted from the peer-reviewed paper 10.3390/jcdd10040159
References
- Loukas, M.; Klaassen, Z.; Tubbs, R.S.; Derderian, T.; Paling, D.; Chow, D.; Patel, S.; Anderson, R.H. Anatomical observations of the moderator band. Clin. Anat. 2010, 23, 443–450.
- Bojsen-Moller, F.; Tranum-Jensen, J. On nerves and nerve endings in the conducting sysetm of the moderator band (septomarginal trabecula). J. Anat. 1971, 108, 387–395.
- Belhassen, B.; Viskin, S. Idiopathic Ventricular Tachycardia and Fibrillation. J. Cardiovasc. Electrophysiol. 1993, 4, 356–368.
- Viskin, S.; Rosso, R.; Rogowski, O.; Belhassen, B. The “Short-Coupled” Variant of Right Ventricular Outflow Ventricular Tachycardia: A Not-So-Benign Form of Benign Ventricular Tachycardia? J. Cardiovasc. Electrophysiol. 2005, 16, 912–916.
- Boyden, P.A.; Dun, W.; Robinson, R.B. Cardiac Purkinje fibers and arrhythmias; The GK Moe Award Lecture 2015. Heart Rhythm. 2016, 13, 1172–1181.
- Haïssaguerre, M.; Shoda, M.; Jaïs, P.; Nogami, A.; Shah, D.C.; Kautzner, J.; Arentz, T.; Kalushe, D.; Lamaison, D.; Griffith, M.; et al. Mapping and Ablation of Idiopathic Ventricular Fibrillation. Circulation 2002, 106, 962–967.
- Haïssaguerre, M.; Shah, D.C.; Jaïs, P.; Shoda, M.; Kautzner, J.; Arentz, T.; Kalushe, D.; Kadish, A.; Griffith, M.; Gaita, F.; et al. Role of Purkinje conducting system in triggering of idiopathic ventricular fibrillation. Lancet 2002, 359, 677–678.
- Sadek, M.M.; Benhayon, D.; Sureddi, R.; Chik, W.; Santangeli, P.; Supple, G.E.; Hutchinson, M.D.; Bala, R.; Carballeira, L.; Zado, E.S.; et al. Idiopathic ventricular arrhythmias originating from the moderator band: Electrocardiographic characteristics and treatment by catheter ablation. Heart Rhythm. 2015, 12, 67–75.
- Anter, E.; Buxton, A.E.; Silverstein, J.R.; Josephson, M.E. Idiopathic Ventricular Fibrillation Originating from the Moderator Band. J. Cardiovasc. Electrophysiol. 2012, 24, 97–100.
- Nogami, A.; Sugiyasu, A.; Kubota, S.; Kato, K. Mapping and ablation of idiopathic ventricular fibrillation from the Purkinje system. Heart Rhythm. 2005, 2, 646–649.
- Walton, R.D.; Pashaei, A.; Martinez, M.E.; Constantin, M.; Duchateau, J.; Bear, L.; Cros, C.; Pascarel-Auclerc, C.; Guo, Y.; Benoist, D.; et al. Compartmentalized Structure of the Moderator Band Provides a Unique Substrate for Macroreentrant Ventricular Tachycardia. Circ. Arrhythmia Electrophysiol. 2018, 11, e005913.
- Atkinson, A.; Inada, S.; Li, J.; Tellez, J.O.; Yanni, J.; Sleiman, R.; Allah, E.A.; Anderson, R.H.; Zhang, H.; Boyett, M.R.; et al. Anatomical and molecular mapping of the left and right ventricular His–Purkinje conduction networks. J. Mol. Cell. Cardiol. 2011, 51, 689–701.
- Lee, J.Y.; Hur, M.S. Morphological classification of the moderator band and its relationship with the anterior papillary muscle. Anat. Cell Biol. 2019, 52, 38–42.
- Dobrzynski, H.; Anderson, R.H.; Atkinson, A.; Borbas, Z.; D’Souza, A.; Fraser, J.F.; Inada, S.; Logantha, S.J.; Monfredi, O.; Morris, G.M.; et al. Structure, function and clinical relevance of the cardiac conduction system, including the atrioventricular ring and outflow tract tissues. Pharmacol. Ther. 2013, 139, 260–288.
- Sommer, J.R.; Johnson, E.A. Cardiac muscle: A comparative study of Purkinje fibers and ventricular fibers. J. Cell Biol. 1968, 36, 497–526.
- Schmid, P.G.; Greif, B.J.; Lund, D.D.; Roskoski, R., Jr. Regional choline acetyltransferase activity in the guinea pig heart. Circ. Res. 1978, 42, 657–660.
- Xiao, L.; Koopmann, T.T.; Ördög, B.; Postema, P.G.; Verkerk, A.O.; Iyer, V.; Sampson, K.J.; Boink, G.J.; Mamarbachi, M.A.; Varro, A.; et al. Unique cardiac Purkinje fiber transient outward current β-subunit composition: A potential molecular link to idiopathic ventricular fibrillation. Circ. Res. 2013, 112, 1310–1322.
- Myerburg, R.J.; Stewart, J.W.; Hoffman, B.F. Electrophysiological Properties of the Canine Peripheral A-V Conducting System. Circ. Res. 1970, 26, 361–378.
- He, B.; Lu, Z.; He, W.; Huang, B.; Yu, L.; Wu, L.; Cui, B.; Hu, X.; Jiang, H. The effects of atrial ganglionated plexi stimulation on ventricular electro-physiology in a normal canine heart. J. Interv. Card. Electrophysiol. 2013, 37, 1–8.
- Antzelevitch, C.; Sicouri, S. Clinical relevance of cardiac arrhythmias generated by afterdepolarizations: Role of M cells in the generation of U waves, triggered activity and torsade de pointes. J. Am. Coll. Cardiol. 1994, 23, 259–277.
- Li, P.; Rudy, Y. A model of canine purkinje cell electrophysiology and Ca(2+) cycling: Rate dependence, triggered activity, and comparison to ventricular myocytes. Circ. Res. 2011, 109, 71–79.
- Quinn, T.A.; Jin, H.; Lee, P.; Kohl, P. Mechanically Induced Ectopy via Stretch-Activated Cation-Nonselective Channels Is Caused by Local Tissue Deformation and Results in Ventricular Fibrillation if Triggered on the Repolarization Wave Edge (Commotio Cordis). Circ. Arrhythmia Electrophysiol. 2017, 10, e004777.
- Zeppenfeld, K.; Tfelt-Hansen, J.; de Riva, M.; Winkel, B.G.; Behr, E.R.; Blom, N.A.; Charron, P.; Corrado, D.; Dagres, N.; de Chillou, C.; et al. 2022 ESC Guidelines for the management of patients with ventricular arrhythmias and the prevention of sudden cardiac death. Eur. Heart J. 2022, 43, 3997–4126.
- Steinfurt, J.; Nazer, B.; Aguilar, M.; Moss, J.; Higuchi, S.; Zarse, M.; Trolese, L.; Gressler, A.; Faber, T.S.; Odening, K.E.; et al. Catheter ablation of short-coupled variant of torsade de pointes. Clin. Res. Cardiol. 2021, 111, 502–510.
- Crawford, T.; Mueller, G.; Good, E.; Jongnarangsin, K.; Chugh, A.; Pelosi, F.; Ebinger, M.; Oral, H.; Morady, F.; Bogun, F. Ventricular arrhythmias originating from papillary muscles in the right ventricle. Heart Rhythm. 2010, 7, 725–730.
- Enriquez, A.; Baranchuk, A.; Briceno, D.; Saenz, L.; Garcia, F. How to use the 12-lead ECG to predict the site of origin of idiopathic ventricular arrhythmias. Heart Rhythm. 2019, 16, 1538–1544.
- Efremidis, M.; Vlachos, K.; Kyriakopoulou, M.; Mililis, P.; Martin, C.A.; Bazoukis, G.; Dragasis, S.; Megarisiotou, A.; Unger, P.; Frontera, A.; et al. The RV1-V3 transition ratio: A novel electrocardiographic criterion for the differentiation of right versus left outflow tract premature ventricular complexes. Heart Rhythm. O2 2021, 2, 521–528.
- Fries, B.; Johnson, V.; Rutsatz, W.; Schmitt, J.; Bogossian, H. Lokalisation ventrikulärer Extrasystolen im 12-Kanal-EKG . Herzschrittmachertherapie Elektrophysiologie 2021, 32, 21–26.
- Santoro, F.; Di Biase, L.; Hranitzky, P.; Sanchez, J.E.; Santangeli, P.; Perini, A.P.; Burkhardt, J.D.; Natale, A. Ventricular Tachycardia Originating from the Septal Papillary Muscle of the Right Ventricle: Electrocardiographic and Electrophysiological Characteristics. J. Cardiovasc. Electrophysiol. 2014, 26, 145–150.
- Chen, H.; Shi, L.; Yang, B.; Ju, W.; Zhang, F.; Yang, G.; Gu, K.; Li, M.; Cao, K.; Ouyang, F.; et al. Electrophysiological Characteristics of Bundle Branch Reentry Ventricular Tachycardia in Patients Without Structural Heart Disease. Circ. Arrhythmia Electrophysiol. 2018, 11, e006049.
