Trigonelline is a bioactive pyridine alkaloid that occurs naturally in high concentrations in coffee (up to 7.2 g/kg) and coffee by-products (up to 62.6 g/kg) such as coffee leaves, flowers, cherry husks or pulp, parchment, silver skin, and spent grounds. In the past, coffee by-products were mostly considered waste and discarded. However, the use of coffee by-products as food has attracted interest because of their economic and nutritional value and the environmental benefits of sustainable resource use. Their authorization as so-called novel foods in the European Union may lead to increased oral exposure of the general population to trigonelline.
- trigonelline
- coffee
- coffee by-products
- toxicity
- risk assessment
1. Introduction
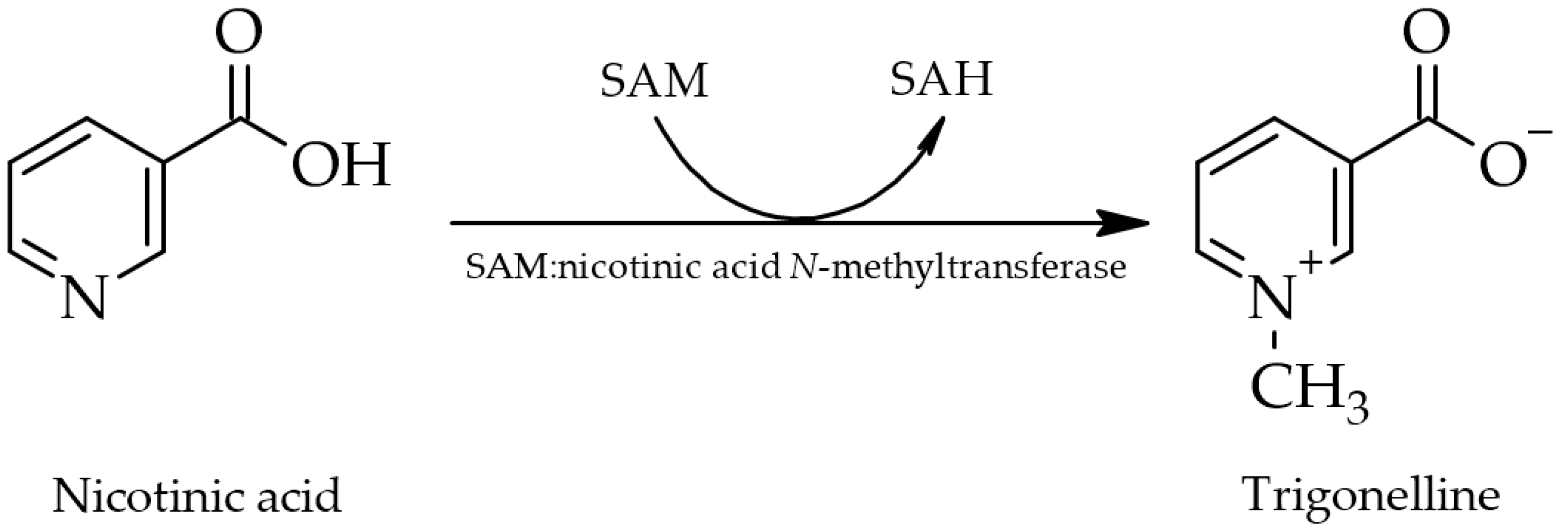

2. Estimation of Human Oral Exposure
3. Absorption, Distribution, Metabolism, and Excretion
4. Nutritional Information
Trigonelline, as a constituent of green coffee beans, decomposes readily when heated at roasting temperatures above 180 °C [14], giving rise to various volatile derivatives such as pyrroles, alkylpyridines, and phenolic compounds, thereby influencing the flavor and aroma of coffee products. Additionally, nicotinic acid is produced as one of two non-volatile derivatives following N-demethylation (the second being N-methylpyridinium as a decarboxylation product) [17][55][74]. Serving as a precursor for essential nicotinic acid (niacin or vitamin B3) in coffee beans, trigonelline thus has nutritional value [8][17][74]. However, it is unclear whether intact trigonelline ingested via foods is metabolized to nicotinic acid in the human body and thus acts as a provitamin. Metabolic transformation to nicotinic acid is only assumed to potentially occur [26].
5. Toxicological Information
Trigonelline did not exert any cytotoxic effects in concentrations up to 100 µmol/L in human neuroblastoma SK-N-SH cells [75], human hepatocellular carcinoma (Hep3B) cells [76], human immortalized dermal keratinocytes (HaCat) and human foreskin fibroblasts (Hs68) [77] after treatment for six days, 48 and 24 h, respectively. In fact, in a recent study, trigonelline protected UV-B radiation-exposed Hs68 cells against photodamage by restoring the physiological function of the endoplasmic reticulum, decreasing oxidative stress, and positively modulating calcium homeostasis and apoptosis in a dose-responsive manner (10, 25, 50, and 100 µmol/L) [77]. It also did not alter the morphology of Madin–Darby bovine kidney cells and African green monkey kidney cells (Vero cells) at concentrations of 1.6 µg/mL each after 48 h of exposure [78].
Three animal studies were identified with designs appropriate for the investigation of subchronic toxicity. In cats (strain and sex not provided), trigonelline administered at doses up to 3500 mg/kg bw for 62–70 days did not induce any visible effects [79]. Additionally, when albino Sabra rats were fed 50 mg/kg bw of trigonelline daily for 21 days and sacrificed after 42 days, no changes in the weight of the thyroid, thymus, kidneys, liver, adrenals, ovaries, and uterus were observed [80].
The first in vitro experiments on the potential mutagenicity of trigonelline were conducted by Fung et al. in 1988. The scholars showed that pure trigonelline did not produce any mutagenic activity in an L5178Y TK+/− mouse lymphoma assay and several Salmonella typhimurium test systems (i.e., Ames tests) [10]. Contrarily, in the S. typhimurium assays conducted by Wu and colleagues, heated trigonelline, applied to the plates (a) alone and (b) combined with certain amino acids caused substantial amounts of revertants, especially when in binary combination with serine (12,740 revertants/mmol) or threonine (11,270/mmol). Of all the 13 single compounds examined in that study in strain TA98, heated trigonelline applied to the plates alone produced the most revertants (8160/mmol) and exhibited the highest mutagenicity [7].
Trigonelline has been examined for its potential role in cancer in several in vitro experiments. In only one study was it shown to have effects that could increase the risk of tumor formation, as it significantly induced the proliferation of MCF-7 breast cancer cells in a dose-responsive manner. Noticeably increased total cell numbers were observed at considerably low concentrations of 10 pmol/L, with the most potent proliferative effect yielded at 100 pmol/L. The growth-promoting effect was due to trigonelline binding the estrogen receptors (ER) of the MCF-7 cells in a way similar to estradiol, thereby altering ER-mediated transcription. As discussed by the scholars, trigonelline has no structural similarity to estradiol or phytoestrogens. Thus, the scholars suggested that trigonelline did not bind the active ligand binding site of the ER, but probably another domain, changing the receptors’ conformation and enabling binding of unspecified transcriptional co-regulators. Hence, trigonelline exerted sound estrogenic activity [81]. This is a significant finding as increased estrogenic activity—either induced by pathological overexpression of ER or mediated by estrogen-like compounds encountered, for instance, via the diet—has been shown to be an important factor in some types of cancer (e.g., breast, colorectal, endometrial, and ovarian cancer [82]), as it stimulates cell growth and proliferation, promoting cancer initiation [83].
Except for one in vivo study from 2009, no data on reproductive toxicity or teratogenic effects are available. In that study, Aswar et al. aimed to investigate the effects of trigonelline on the estrous cycle and fertility of female Wistar rats. To examine effects on the estrous cycle (endpoint: vaginal cornification), the scholars administered doses of 75 mg/kg bw of trigonelline orally to ovariectomized, immature female Wister rats weighing 55–60 g twice on the first two days and once on the third and fourth day of the treatment. Afterwards, vaginal smears were collected and microscopically searched for cornified or nucleated epithelial cells. As no such cells were noticed, it was concluded that trigonelline did not exert estrogenic activity.
Doses up to 10 µmol/L ameliorated the atrophy of neuronal dendrites and axons in amyloid β-peptide-treated female, hemizygous transgenic 5XFAD Alzheimer’s disease mice model [73]. Additionally, trigonelline (30 µmol/L) improved functional neurite outgrowth in human neuroblastoma SK-N-SH cells after treatment for six days [75][84]. Furthermore, daily oral administration of 500 mg/kg bw for 15 days was demonstrated to improve memory retention in six-week-old amyloid β-peptide-treated male ddY mice [85]. Thus, trigonelline is believed to improve cognition in Alzheimer’s disease model laboratory animals [84][86]. Furthermore, it has been discussed that trigonelline may protect against cerebral ischemia [87] by positively modulating neuronal spike frequency [88], stimulating the release of dopamine [89], competitively inhibiting γ-aminobutyric acid (GABA) A-receptors [90], and weakly hampering acetylcholine esterase [91].
This entry is adapted from the peer-reviewed paper 10.3390/molecules28083460
References
- Jahns, E. Ueber die Alkaloïde des Bockshornsamens. Ber. Dtsch. Chem. Ges. 1885, 18, 2518–2523.
- National Center for Biotechnology Information. PubChem Compound Summary for CID 5570, Trigonelline. Available online: https://pubchem.ncbi.nlm.nih.gov/compound/5570#section=Names-and-Identifiers (accessed on 25 September 2022).
- CAS Common Chemistry. CAS, a Division of the American Chemical Society. Trigonelline. Available online: https://commonchemistry.cas.org/detail?cas_rn=535-83-1 (accessed on 25 September 2022).
- Mazzafera, P. Trigonelline in coffee. Phytochemistry 1991, 30, 2309–2310.
- Viani, R.; Horman, I. Thermal Behavior of Trigonelline. J. Food Sci. 1974, 39, 1216–1217.
- Cangussu, L.B.; Melo, J.C.; Franca, A.S.; Oliveira, L.S. Chemical Characterization of Coffee Husks, a By-Product of Coffea arabica Production. Foods 2021, 10, 3125.
- Wu, X.; Skog, K.; Jägerstad, M. Trigonelline, a naturally occurring constituent of green coffee beans behind the mutagenic activity of roasted coffee? Mutat. Res. Genet. Toxicol. Environ. Mutagen. 1997, 391, 171–177.
- Ashihara, H. Metabolism of alkaloids in coffee plants. Braz. J. Plant Physiol. 2006, 18, 1–8.
- Zheng, X.; Ashihara, H. Distribution, Biosynthesis and Function of Purine and Pyridine Alkaloids in Coffea arabica Seedlings. Plant Sci. 2004, 166, 807–813.
- Fung, V.A.; Cameron, T.P.; Hughes, T.J.; Kirby, P.E.; Dunkel, V.C. Mutagenic activity of some coffee flavor ingredients. Mutat. Res. Genet. Toxicol. 1988, 204, 219–228.
- Evans, L.S.; Tramontano, W.A. Trigonelline and promotion of cell arrest in G2 of various legumes. Phytochemistry 1984, 23, 1837–1840.
- Mohamadi, N.; Sharififar, F.; Pournamdari, M.; Ansari, M. A Review on Biosynthesis, Analytical Techniques, and Pharmacological Activities of Trigonelline as a Plant Alkaloid. J. Diet. Suppl. 2018, 15, 207–222.
- Matsui, A.; Yin, Y.; Yamanaka, K.; Iwasaki, M.; Ashihara, H. Metabolic fate of nicotinamide in higher plants. Physiol. Plant. 2007, 131, 191–200.
- Taguchi, H.; Sakaguchi, M.; Shimabayashi, Y. Trigonelline Content in Coffee Beans and the Thermal Conversion of Trigonelline into Nicotinic Acid during the Roasting of Coffee Beans. Agric. Biol. Chem. 1985, 49, 3467–3471.
- Adepoju, A.F.; Adenuga, O.O.; Mapayi, E.F.; Olaniyi, O.O.; Adepoju, F.A. Coffee: Botany, Distribution, Diversity, Chemical Composition and Its Management. IOSR-JAVS 2017, 10, 57–62.
- Ky, C.L.; Louarn, J.; Dussert, S.; Guyot, B.; Hamon, S.; Noirot, M. Caffeine, Trigonelline, Chlorogenic acids and Sucrose Diversity in Wild Coffea arabica L. and C. canephora P. Accessions. Food Chem. 2001, 75, 223–230.
- Lang, R.; Yagar, E.F.; Eggers, R.; Hofmann, T. Quantitative Investigation of Trigonelline, Nicotinic Acid, and Nicotinamide in Foods, Urine, and Plasma by Means of LC-MS/MS and Stable Isotope Dilution Analysis. J. Agric. Food Chem. 2008, 56, 11114–11121.
- Zheng, X.-Q.; Nagai, C.; Ashihara, H. Pyridine Nucleotide Cycle and Trigonelline (N-Methylnicotinic Acid) Synthesis in Developing Leaves and Fruits of Coffea arabica. Physiol. Plant. 2004, 122, 404–411.
- Acidri, R.; Sawai, Y.; Sugimoto, Y.; Handa, T.; Sasagawa, D.; Masunaga, T.; Yamamoto, S.; Nishihara, E. Phytochemical Profile and Antioxidant Capacity of Coffee Plant Organs Compared to Green and Roasted Coffee Beans. Antioxidants 2020, 9, 93.
- Li, X.; Zhang, X.; Tan, L.; Yan, H.; Yuan, Y. Heat-induced formation of N,N-dimethylpiperidinium (mepiquat) in Arabica and Robusta coffee. J. Food Sci. 2020, 85, 2754–2761.
- Tice, R.R. Trigonelline : Review of Toxicological Literature, Research Triangle Park, North Carolina 27709, 1997. Available online: https://ntp.niehs.nih.gov/ntp/htdocs/chem_background/exsumpdf/trigonelline_508.pdf (accessed on 16 April 2022).
- Guertin, K.A.; Moore, S.C.; Sampson, J.N.; Huang, W.-Y.; Xiao, Q.; Stolzenberg-Solomon, R.Z.; Sinha, R.; Cross, A.J. Metabolomics in nutritional epidemiology: Identifying metabolites associated with diet and quantifying their potential to uncover diet-disease relations in populations. Am. J. Clin. Nutr. 2014, 100, 208–217.
- Guertin, K.A.; Loftfield, E.; Boca, S.M.; Sampson, J.N.; Moore, S.C.; Xiao, Q.; Huang, W.-Y.; Xiong, X.; Freedman, N.D.; Cross, A.J.; et al. Serum biomarkers of habitual coffee consumption may provide insight into the mechanism underlying the association between coffee consumption and colorectal cancer. Am. J. Clin. Nutr. 2015, 101, 1000–1011.
- Playdon, M.C.; Sampson, J.N.; Cross, A.J.; Sinha, R.; Guertin, K.A.; Moy, K.A.; Rothman, N.; Irwin, M.L.; Mayne, S.T.; Stolzenberg-Solomon, R.; et al. Comparing metabolite profiles of habitual diet in serum and urine. Am. J. Clin. Nutr. 2016, 104, 776–789.
- Rothwell, J.A.; Fillâtre, Y.; Martin, J.-F.; Lyan, B.; Pujos-Guillot, E.; Fezeu, L.; Hercberg, S.; Comte, B.; Galan, P.; Touvier, M.; et al. New Biomarkers of Coffee Consumption Identified by the Non-Targeted Metabolomic Profiling of Cohort Study Subjects. PLoS ONE 2014, 9, e93474.
- Lang, R.; Wahl, A.; Skurk, T.; Yagar, E.F.; Schmiech, L.; Eggers, R.; Hauner, H.; Hofmann, T. Development of a Hydrophilic Liquid Interaction Chromatography−High-Performance Liquid Chromatography−Tandem Mass Spectrometry Based Stable Isotope Dilution Analysis and Pharmacokinetic Studies on Bioactive Pyridines in Human Plasma and Urine after Coffee Consumption. Anal. Chem. 2010, 82, 1486–1497.
- Joshi, J.G.; Handler, P. Biosynthesis of Trigonelline. J. Biol. Chem. 1960, 235, 2981–2983.
- Upmeier, B.; Gross, W.; Köster, S.; Barz, W. Purification and properties of S-adenosyl-l-methionine:Nicotinic acid-N-methyltransferase from cell suspension cultures of Glycine max L. Arch. Biochem. Biophys. 1988, 262, 445–454.
- Ashihara, H.; Ludwig, I.A.; Katahira, R.; Yokota, T.; Fujimura, T.; Crozier, A. Trigonelline and related nicotinic acid metabolites: Occurrence, biosynthesis, taxonomic considerations, and their roles in Planta and in human health. Phytochem. Rev. 2015, 14, 765–798.
- Mizuno, K.; Matsuzaki, M.; Kanazawa, S.; Tokiwano, T.; Yoshizawa, Y.; Kato, M. Conversion of Nicotinic acid to Trigonelline is Catalyzed by N-methyltransferase Belonged to Motif B′ Methyltransferase Family in Coffea arabica. Biochem. Biophys. Res. Commun. 2014, 452, 1060–1066.
- Ashihara, H. Trigonelline (N-methylnicotinic acid) biosynthesis and its biological role in plants. Nat. Prod. Commun. 2008, 3, 1934578X0800300906.
- Ashihara, H. Chapter 3—Plant biochemistry: Trigonelline biosynthesis in Coffea arabica and Coffea canephora. In Coffee in Health and Disease Prevention; Preedy, V.R., Ed.; Academic Press: San Diego, CA, USA, 2015; pp. 19–28. ISBN 978-0-12-409517-5.
- Minorsky, P.V. The functions of foliar nyctinasty: A review and hypothesis. Biol. Rev. 2019, 94, 216–229.
- Caetano-Anollés, G.; Gresshoff, P.M. Plant Genetic Control of Nodulation. Annu. Rev. Micobiol. 1991, 45, 345–382.
- Evans, L.S.; Almeida, M.S.; Lynn, D.G.; Nakanishi, K. Chemical characterization of a hormone that promotes cell arrest in g2 in complex tissues. Science 1979, 203, 1122–1123.
- Minorsky, P.V. The hot and the classic: Trigonelline: A diverse regulator in plants. Plant Physiol. 2002, 128, 7.
- Barz, W. Metabolism and Degradation of Nicotinic Acid in Plant Cell Cultures. In Primary and Secondary Metabolism of Plant Cell Cultures; Neumann, K.H., Barz, W., Reinhard, E., Eds.; Springer: Berlin/Heidelberg, Germany, 1985; pp. 186–195. ISBN 978-3-642-70717-9.
- de Melo Pereira, G.V.; de Carvalho Neto, D.P.; Magalhães Júnior, A.I.; Vásquez, Z.S.; Medeiros, A.B.P.; Vandenberghe, L.P.S.; Soccol, C.R. Exploring the impacts of postharvest processing on the aroma formation of coffee beans—A review. Food Chem. 2019, 272, 441–452.
- Lachenmeier, D.W.; Schwarz, S.; Rieke-Zapp, J.; Cantergiani, E.; Rawel, H.; Martín-Cabrejas, M.A.; Martuscelli, M.; Gottstein, V.; Angeloni, S. Coffee By-Products as Sustainable Novel Foods: Report of the 2nd International Electronic Conference on Foods—“Future Foods and Food Technologies for a Sustainable World”. Foods 2022, 11, 3.
- Lachenmeier, D.W.; Rajcic de Rezende, T.; Schwarz, S. An Update on Sustainable Valorization of Coffee By-Products as Novel Foods within the European Union. Biol. Life Sci. Forum 2021, 6, 37.
- Klingel, T.; Kremer, J.I.; Gottstein, V.; Rajcic de Rezende, T.; Schwarz, S.; Lachenmeier, D.W. A Review of Coffee By-Products Including Leaf, Flower, Cherry, Husk, Silver Skin, and Spent Grounds as Novel Foods within the European Union. Foods 2020, 9, 665.
- Hoseini, M.; Cocco, S.; Casucci, C.; Cardelli, V.; Corti, G. Coffee By-products Derived Resources. A Review. Biomass Bioenergy 2021, 148, 106009.
- Murthy, P.S.; Naidu, M.M. Sustainable management of coffee industry by-products and value addition—A review. Resour. Conserv. Recycl. 2012, 66, 45–58.
- Murthy, P.S.; Naidu, M.M. Recovery of Phenolic Antioxidants and Functional Compounds from Coffee Industry By-Products. Food Bioprocess Technol. 2012, 5, 897–903.
- Benitez, V.; Rebollo-Hernanz, M.; Hernanz, S.; Chantres, S.; Aguilera, Y.; Martin-Cabrejas, M.A. Coffee parchment as a new dietary fiber ingredient: Functional and physiological characterization. Food Res. Int. 2019, 122, 105–113.
- Martuscelli, M.; Esposito, L.; Di Mattia, C.D.; Ricci, A.; Mastrocola, D. Characterization of Coffee Silver Skin as Potential Food-Safe Ingredient. Foods 2021, 10, 1367.
- Tores de la Cruz, S.; Iriondo-DeHond, A.; Herrera, T.; Lopez-Tofiño, Y.; Galvez-Robleño, C.; Prodanov, M.; Velazquez-Escobar, F.; Abalo, R.; Del Castillo, M.D. An Assessment of the Bioactivity of Coffee Silverskin Melanoidins. Foods 2019, 8, 68.
- Zhang, J.; Sun, X.; Liu, P.; Zhang, T.; Jelderks, J.A.; Corke, H. Preliminary Characterization of Phytochemicals and Polysaccharides in Diverse Coffee Cascara Samples: Identification, Quantification and Discovery of Novel Compounds. Foods 2022, 11, 1710.
- Stennert, A.; Maier, H.G. Trigonelline in coffee. Z. Lebensm. Unters. Forsch. 1994, 199, 198–200.
- Wu, H.; Gu, J.; Amrit, B.K.; Nawaz, M.A.; Barrow, C.J.; Dunshea, F.; Suleria, H. Effect of processing on bioaccessibility and bioavailability of bioactive compounds in coffee beans. Food Biosci. 2022, 46, 101373.
- Monteiro, Â.; Colomban, S.; Azinheira, H.G.; Guerra-Guimarães, L.; Do Céu Silva, M.; Navarini, L.; Resmini, M. Dietary Antioxidants in Coffee Leaves: Impact of Botanical Origin and Maturity on Chlorogenic Acids and Xanthones. Antioxidants 2019, 9, 6.
- Chen, X.; Ma, Z.; Kitts, D.D. Effects of Processing Method and Age of Leaves on Phytochemical Profiles and Bioactivity of Coffee Leaves. Food Chem. 2018, 249, 143–153.
- Montis, A.; Souard, F.; Delporte, C.; Stoffelen, P.; Stévigny, C.; van Antwerpen, P. Coffee Leaves: An Upcoming Novel Food? Planta Med. 2021, 87, 949–963.
- Stadler, R.H.; Varga, N.; Milo, C.; Schilter, B.; Vera, F.A.; Welti, D.H. Alkylpyridiniums. 2. Isolation and Quantification in Roasted and Ground Coffees. J. Agric. Food Chem. 2002, 50, 1200–1206.
- Lang, R.; Yagar, E.F.; Wahl, A.; Beusch, A.; Dunkel, A.; Dieminger, N.; Eggers, R.; Bytof, G.; Stiebitz, H.; Lantz, I.; et al. Quantitative Studies on Roast Kinetics for Bioactives in Coffee. J. Agric. Food Chem. 2013, 61, 12123–12128.
- Ziefuß, A.R.; Hupfeld, T.; Meckelmann, S.W.; Meyer, M.; Schmitz, O.J.; Kaziur-Cegla, W.; Tintrop, L.K.; Schmidt, T.C.; Gökce, B.; Barcikowski, S. Ultrafast cold-brewing of coffee by picosecond-pulsed laser extraction. NPJ Sci. Food 2022, 6, 19.
- Steger, M.C.; Rigling, M.; Blumenthal, P.; Segatz, V.; Quintanilla-Belucci, A.; Beisel, J.M.; Rieke-Zapp, J.; Schwarz, S.; Lachenmeier, D.W.; Zhang, Y. Coffee Leaf Tea from El Salvador: On-Site Production Considering Influences of Processing on Chemical Composition. Foods 2022, 11, 2553.
- Mehari, B.; Redi-Abshiro, M.; Chandravanshi, B.S.; Atlabachew, M.; Combrinck, S.; McCrindle, R. Simultaneous Determination of Alkaloids in Green Coffee Beans from Ethiopia: Chemometric Evaluation of Geographical Origin. Food Anal. Methods 2016, 9, 1627–1637.
- Arai, K.; Terashima, H.; Aizawa, S.; Taga, A.; Yamamoto, A.; Tsutsumiuchi, K.; Kodama, S. Simultaneous determination of trigonelline, caffeine, chlorogenic acid and their related compounds in instant coffee samples by HPLC using an acidic mobile phase Containing Octanesulfonate. Anal. Sci. 2015, 31, 831–835.
- Stadler, R.H.; Varga, N.; Hau, J.; Vera, F.A.; Welti, D.H. Alkylpyridiniums. 1. Formation in Model Systems via Thermal Degradation of Trigonelline. J. Agric. Food Chem. 2002, 50, 1192–1199.
- Jeszka-Skowron, M.; Frankowski, R.; Zgoła-Grześkowiak, A. Comparison of methylxantines, trigonelline, nicotinic acid and nicotinamide contents in brews of green and processed Arabica and Robusta coffee beans—Influence of steaming, decaffeination and roasting processes on coffee beans. LWT 2020, 125, 109344.
- Teply, L.J.; Prier, R.F. Nutrients in Coffee, Nutritional Evaluation of Coffee Including Niacin Bioassay. J. Agric. Food Chem. 1957, 5, 375–377.
- Honda, M.; Takezaki, D.; Tanaka, M.; Fukaya, M.; Goto, M. Effect of Roasting Degree on Major Coffee Compounds: A Comparative Study between Coffee Beans with and without Supercritical CO2 Decaffeination Treatment. J. Oleo Sci. 2022, 71, 1541–1550.
- Febvay, L.; Hamon, E.; Recht, R.; Andres, N.; Vincent, M.; Aoudé-Werner, D.; This, H. Identification of markers of thermal processing (“roasting”) in aqueous extracts of Coffea arabica L. seeds through NMR fingerprinting and chemometrics. Magn. Reson. Chem. 2019, 57, 589–602.
- Romualdo, G.R.; Rocha, A.B.; Vinken, M.; Cogliati, B.; Moreno, F.S.; García Chaves, M.A.; Barbisan, L.F. Drinking for protection? Epidemiological and experimental evidence on the beneficial effects of coffee or major coffee compounds against gastrointestinal and liver carcinogenesis. Food Res. Int. 2019, 123, 567–589.
- IARC Working Group on the Evaluation of Carcinogenic Risks to Humans. Drinking coffee, mate, and very hot beverages. IARC Monogr. Eval. Carcinog. Risks Hum. 2018, 116, 1–501.
- Yuyama, S.; Suzuki, T. The Excretion of N1-Methyl-2-Pyridone-5-Carboxylic Acid and Related Compounds in Human Subjects after Oral Administration of Nicotinic Acid, Trigonalline and N1-Methyl-2-Pyridone-5-Carboxylic Acid. In Kynurenine and Serotonin Pathways: Progress in Tryptophan Research; Schwarcz, R., Young, S.N., Brown, R.R., Eds.; Springer: Boston, MA, USA, 1991; pp. 475–479. ISBN 978-1-4684-5952-4.
- Yuyama, S.; Kawano, Y. Urinary Excretion of N1-Menthyl-2-Pyridone-5-Carboxylic Acid and the Fate of Remaining of Trigonelline. In Recent Advances in Tryptophan Research: Tryptophan and Serotonin Pathways; Filippini, G.A., Costa, C.V.L., Bertazzo, A., Eds.; Springer US: Boston, MA, USA, 1996; pp. 599–604. ISBN 978-1-4613-0381-7.
- Lang, R.; Wahl, A.; Stark, T.; Hofmann, T. Urinary N-methylpyridinium and trigonelline as candidate dietary biomarkers of coffee consumption. Mol. Nutr. Food. Res. 2011, 55, 1613–1623.
- Midttun, Ø.; Ulvik, A.; Nygård, O.; Ueland, P.M. Performance of Plasma Trigonelline as a Marker of Coffee Consumption in an Epidemiologic Setting. Am. J. Clin. Nutr. 2018, 107, 941–947.
- Bresciani, L.; Tassotti, M.; Rosi, A.; Martini, D.; Antonini, M.; Dei Cas, A.; Bonadonna, R.; Brighenti, F.; Del Rio, D.; Mena, P. Absorption, Pharmacokinetics, and Urinary Excretion of Pyridines After Consumption of Coffee and Cocoa-Based Products Containing Coffee in a Repeated Dose, Crossover Human Intervention Study. Mol. Nutr. Food. Res. 2020, 64, 2000489.
- Holford, N.; Yim, D.-S. Volume of Distribution. Transl. Clin. Pharmacol. 2016, 24, 74–77.
- Farid, M.M.; Yang, X.; Kuboyama, T.; Tohda, C. Trigonelline recovers memory function in Alzheimer’s disease model mice: Evidence of brain penetration and target molecule. Sci. Rep. 2020, 10, 16424.
- Buffo, R.A.; Cardelli-Freire, C. Coffee flavour: An overview. Flavour. Fragr. J. 2004, 19, 99–104.
- Tohda, C.; Nakamura, N.; Komatsu, K.; Hattori, M. Trigonelline-Induced Neurite Outgrowth in Human Neuroblastoma SK-N-SH Cells. Biol. Pharm. Bull. 1999, 22, 679–682.
- Liao, J.C.; Lee, K.T.; You, B.J.; Lee, C.L.; Chang, W.T.; Wu, Y.C.; Lee, H.-Z. Raf/ERK/Nrf2 signaling pathway and MMP-7 expression involvement in the trigonelline-mediated inhibition of hepatocarcinoma cell migration. Food Nutr. Res. 2015, 59, 29884.
- Nazir Lone, A.; Tanveer Malik, A.; Shahid Naikoo, H.; Sharma Raghu, R.; Sheikh, A. Tasduq. Trigonelline, a naturally occurring alkaloidal agent protects ultraviolet-B (UV-B) irradiation induced apoptotic cell death in human skin fibroblasts via attenuation of oxidative stress, restoration of cellular calcium homeostasis and prevention of endoplasmic reticulum (ER) stress. J. Photochem. Photobiol. B Biol. 2020, 202, 111720.
- Özçelik, B.; Kartal, M.; Orhan, I. Cytotoxicity, antiviral and antimicrobial activities of alkaloids, flavonoids, and phenolic acids. Pharm. Biol. 2011, 49, 396–402.
- Faulkner, K.K.; Smith, J.W.H. Preliminary studies of the toxicity of locoweed extracts. Proc. Oklohoma Acad. Sci. 1950, 31, 48–50.
- Mishkinsky, J.S.; Goldschmied, A.; Joseph, B.; Ahronson, Z.; Sulman, F.G. Hypoglycaemic effect of Trigonella foenum graecum and Lupinus termis (leguminosae) seeds and their major alkaloids in alloxan-diabetic and normal rats. Arch. Int. Pharmacodyn. Ther. 1974, 210, 27–37.
- Allred, K.F.; Yackley, K.M.; Vanamala, J.; Allred, C.D. Trigonelline is a Novel Phytoestrogen in Coffee Beans. J. Nutr. 2009, 139, 1833–1838.
- Deli, T.; Orosz, M.; Jakab, A. Hormone Replacement Therapy in Cancer Survivors—Review of the Literature. Pathol. Oncol. Res. 2020, 26, 63–78.
- Harding, A.T.; Heaton, N.S. The Impact of Estrogens and Their Receptors on Immunity and Inflammation during Infection. Cancers 2022, 14, 909.
- Ghanem, S.S.; Fayed, H.S.; Zhu, Q.; Lu, J.-H.; Vaikath, N.N.; Ponraj, J.; Mansour, S.; El-Agnaf, O.M.A. Natural Alkaloid Compounds as Inhibitors for Alpha-Synuclein Seeded Fibril Formation and Toxicity. Molecules 2021, 26, 3736.
- Tohda, C.; Kuboyama, T.; Komatsu, K. Search for Natural Products Related to Regeneration of the Neuronal Network. Neurosignals 2005, 14, 34–45.
- Fahanik-Babaei, J.; Baluchnejadmojarad, T.; Nikbakht, F.; Roghani, M. Trigonelline protects hippocampus against intracerebral Aβ(1–40) as a model of Alzheimer’s disease in the rat: Insights into underlying mechanisms. Metab. Brain Dis. 2019, 34, 191–201.
- Lin, S.X.; Curtis, M.A.; Sperry, J. Pyridine Alkaloids with Activity in the Central Nervous System. Bioorg. Med. Chem. 2020, 28, 115820.
- Temraz, T.A.; Houssen, W.E.; Jaspars, M.; Woolley, D.R.; Wease, K.N.; Davies, S.N.; Scott, R.H. A pyridinium derivative from Red Sea soft corals inhibited voltage-activated potassium conductances and increased excitability of rat cultured sensory neurones. BMC Pharmacol. 2006, 6, 10.
- Walker, J.; Rohm, B.; Lang, R.; Pariza, M.W.; Hofmann, T.; Somoza, V. Identification of coffee components that stimulate dopamine release from pheochromocytoma cells (PC-12). Food Chem. Toxicol. 2012, 50, 390–398.
- Hossain, S.J.; Aoshima, H.; Koda, H.; Kiso, Y. Effects of Coffee Components on the Response of GABAA Receptors Expressed in Xenopus Oocytes. J. Agric. Food Chem. 2003, 51, 7568–7575.
- SatheeshKumar, N.; Mukherjee, P.K.; Bhadra, S.; Saha, B.P. Acetylcholinesterase enzyme inhibitory potential of standardized extract of Trigonella foenum graecum L and its constituents. Phytomedicine 2010, 17, 292–295.
