Microarrays are one of the trailblazing technologies of the last two decades and have displayed their importance in all the associated fields of biology. They are widely explored to screen, identify, and gain insights on the characteristics traits of biomolecules (individually or in complex solutions). A wide variety of biomolecule-based microarrays (DNA microarrays, protein microarrays, glycan microarrays, antibody microarrays, peptide microarrays, and aptamer microarrays) are either commercially available or fabricated in-house by researchers to explore diverse substrates, surface coating, immobilization techniques, and detection strategies.
- microarray
- DNA
- protein
- peptide
- glycan
1. Nucleic Acid Microarrays
2. Protein Microarrays
3. Peptide Microarrays
4. Glycan Microarrays
5. Antibody Microarrays
6. Aptamer Microarrays
This entry is adapted from the peer-reviewed paper 10.3390/biom13040602
References
- Minchin, S.; Lodge, J. Understanding biochemistry: Structure and function of nucleic acids. Essays Biochem. 2019, 63, 433–456.
- Devine, K.G.; Jheeta, S. De Novo Nucleic Acids: A Review of Synthetic Alternatives to DNA and RNA That Could Act as Bio-Information Storage Molecules. Life 2020, 10, 346.
- Kumar, A.; Pandey, S.C.; Samant, M. DNA-based microarray studies in visceral leishmaniasis: Identification of biomarkers for diagnostic, prognostic and drug target for treatment. Acta Trop. 2020, 208, 105512.
- Bumgarner, R. Overview of DNA microarrays: Types, applications, and their future. Curr. Protoc. Mol. Biol. 2013, 101, 22.1.21.
- Jaleh, B.; Amir Ata, S.; Yadollah, O. Impacts of DNA Microarray Technology in Gene Therapy. In Gene Therapy; Chunsheng, K., Ed.; IntechOpen: Rijeka, Croatia, 2011; Chapter 8.
- Fesseha, H.; Tilahun, H. Principles and Applications of Deoxyribonucleic Acid Microarray: A Review. Pathol. Lab. Med. Open J. 2020, 1, 54–62.
- Dufva, M. Fabrication of high quality microarrays. Biomol. Eng. 2005, 22, 173–184.
- Lietard, J.; Somoza, M.M. Spotting, Transcription and In Situ Synthesis: Three Routes for the Fabrication of RNA Microarrays. Comput. Struct. Biotechnol. J. 2019, 17, 862–868.
- Dufva, M. Fabrication of DNA Microarray. In DNA Microarrays for Biomedical Research: Methods and Protocols; Dufva, M., Ed.; Humana Press: Totowa, NJ, USA, 2009; pp. 63–79.
- Lekang, K.; Hadziavdic, K.; Sandnes Skaar, K.; Jonassen, I.; Thompson, E.M.; Troedsson, C. Development and testing of an 18S rRNA phylogenetic microarray for marine sediments. J. Microbiol. Methods 2018, 154, 95–106.
- Rivas, L.; Reuterswärd, P.; Rasti, R.; Herrmann, B.; Mårtensson, A.; Alfvén, T.; Gantelius, J.; Andersson-Svahn, H. A vertical flow paper-microarray assay with isothermal DNA amplification for detection of Neisseria meningitidis. Talanta 2018, 183, 192–200.
- Lee, D.; Kim, E.J.; Kilgore, P.E.; Kim, S.A.; Takahashi, H.; Ohnishi, M.; Anh, D.D.; Dong, B.Q.; Kim, J.S.; Tomono, J.; et al. Clinical Evaluation of a Loop-Mediated Isothermal Amplification (LAMP) Assay for Rapid Detection of Neisseria meningitidis in Cerebrospinal Fluid. PLoS ONE 2015, 10, e0122922.
- Yin, H.-B.; Chen, C.-H.; Katchman, B.; Newland, C.; May, M.; Patel, J. Rapid detection of Salmonella enterica in leafy greens by a novel DNA microarray-based PathogenDx system. Food Microbiol. 2022, 107, 104086.
- Taguchi, T.; Ishikawa, M.; Ichikawa, M.; Tadenuma, T.; Hirakawa, Y.; Yoshino, T.; Maeda, Y.; Takeuchi, H.; Nojima, D.; Tanaami, T.; et al. Amplification-free detection of bacterial genes using a signaling probe-based DNA microarray. Biosens. Bioelectron. 2021, 194, 113659.
- Thanthrige-Don, N.; Lung, O.; Furukawa-Stoffer, T.; Buchanan, C.; Joseph, T.; Godson, D.L.; Gilleard, J.; Alexander, T.; Ambagala, A. A novel multiplex PCR-electronic microarray assay for rapid and simultaneous detection of bovine respiratory and enteric pathogens. J. Virol. Methods 2018, 261, 51–62.
- Jiang, Y.; Nie, F.; Jiang, S.; Li, Y.; Wu, Y.; Yang, J.; Bao, Y.; Wang, Y.; Wang, G.; Li, X.; et al. Development of multiplex oligonucleotide microarray for simultaneous detection of six swine pathogens. J. Virol. Methods 2020, 285, 113921.
- Xiao, Q.; Wu, J.; Dang, P.; Ju, H. Multiplexed chemiluminescence imaging assay of protein biomarkers using DNA microarray with proximity binding-induced hybridization chain reaction amplification. Anal. Chim. Acta 2018, 1032, 130–137.
- Zong, C.; Wu, J.; Liu, M.; Yang, L.; Yan, F.; Ju, H. Chemiluminescence Imaging for a Protein Assay via Proximity-Dependent DNAzyme Formation. Anal. Chem. 2014, 86, 9939–9944.
- Zong, C.; Wu, J.; Liu, M.; Yan, F.; Ju, H. High-throughput imaging assay of multiple proteins via target-induced DNA assembly and cleavage. Chem. Sci. 2015, 6, 2602–2607.
- Chu, W.; Chen, Y.; Liu, W.; Zhao, M.; Li, H. Paper-based chemiluminescence immunodevice with temporal controls of reagent transport technique. Sens. Actuators B 2017, 250, 324–332.
- Wei, W.; Zhang, C.; Qian, J.; Liu, S. Multianalyte immunoassay chip for detection of tumor markers by chemiluminescent and colorimetric methods. Anal. Bioanal. Chem. 2011, 401, 3269–3274.
- Takamiya, M.; Saigusa, K.; Dewa, K. DNA microarray analysis of hypothermia-exposed murine lungs for identification of forensic biomarkers. Legal Med. 2021, 48, 101789.
- Yao, Y.; Wang, J.; He, T.; Li, H.; Hu, J.; Zheng, M.; Ding, Y.; Chen, Y.-Y.; Shen, Y.; Wang, L.-L.; et al. Microarray assay of circular RNAs reveals cicRNA.7079 as a new anti-apoptotic molecule in spinal cord injury in mice. Brain Res. Bull. 2020, 164, 157–171.
- Govarthanan, K.; Gupta, P.K.; Ramasamy, D.; Kumar, P.; Mahadevan, S.; Verma, R.S. DNA methylation microarray uncovers a permissive methylome for cardiomyocyte differentiation in human mesenchymal stem cells. Genomics 2020, 112, 1384–1395.
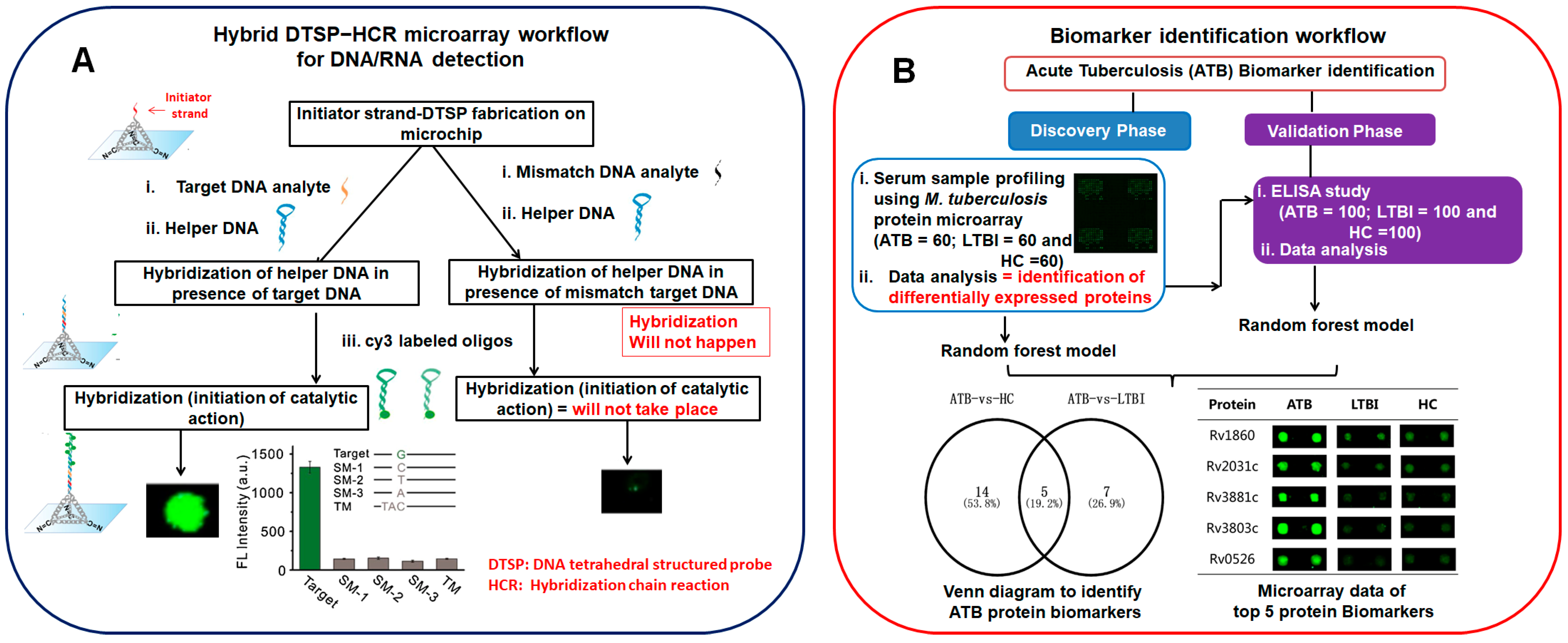
- Zhang, H.; Liu, X.; Zhang, C.; Xu, Y.; Su, J.; Lu, X.; Shi, J.; Wang, L.; Landry, M.P.; Zhu, Y.; et al. A DNA tetrahedral structure-mediated ultrasensitive fluorescent microarray platform for nucleic acid test. Sens. Actuators B 2020, 321, 128538.
- Jin, F.; Xu, D. A fluorescent microarray platform based on catalytic hairpin assembly for MicroRNAs detection. Anal. Chim. Acta 2021, 1173, 338666.
- Petralia, S.; Vigilanza, A.; Sciuto, E.; Maffia, M.; Romanini, A.; Conoci, S. The MC1R single nucleotide polymorphisms identification by DNA-microarray on miniaturized silicon chip. Sens. Actuators B 2021, 346, 130514.
- Banu, M.; Simion, M.; Popescu, M.C.; Varasteanu, P.; Kusko, M.; Farcasanu, I.C. Specific detection of stable single nucleobase mismatch using SU-8 coated silicon nanowires platform. Talanta 2018, 185, 281–290.
- Wolff, N.; Hendling, M.; Schönthaler, S.; Geiss, A.F.; Barišić, I. Low-cost microarray platform to detect antibiotic resistance genes. Sens. Bio-Sens. Res. 2019, 23, 100266.
- Xiang, H.; Wen, X.; Wen, Y.; Zhang, H.; Cao, S.; Huang, X.; Wu, R.; Zhao, Q. Development and application of a visual microarray for synchronously detecting H5N1, H7N9 and H9N2 avian influenza virus RNA. J. Virol. Methods 2022, 301, 114371.
- Gwida, M.; Awad, A.; El-Ashker, M.; Hotzel, H.; Monecke, S.; Ehricht, R.; Muller, E.; Reissig, A.; Barth, S.A.; Berens, C.; et al. Microarray-based detection of resistance and virulence factors in commensal Escherichia coli from livestock and farmers in Egypt. Vet. Microbiol. 2020, 240, 108539.
- Jin, F.; Li, H.; Xu, D. Enzyme-free fluorescence microarray for determination of hepatitis B virus DNA based on silver nanoparticle aggregates-assisted signal amplification. Anal. Chim. Acta 2019, 1077, 297–304.
- Kappel, K.; Eschbach, E.; Fischer, M.; Fritsche, J. Design of a user-friendly and rapid DNA microarray assay for the authentication of ten important food fish species. Food Chem. 2020, 311, 125884.
- Huan, H.; Zhang, K.; He, J.; Zhang, J. A DNA microarray assay for authenticating five important marine mammal species in food and feed. Food Chem. 2021, 348, 129136.
- Sola, L.; Damin, F.; Chiari, M. Array of multifunctional polymers for localized immobilization of biomolecules on microarray substrates. Anal. Chim. Acta 2019, 1047, 188–196.
- Li, J.; Wang, Y.; Yan, L.; Zhang, C.; He, Y.; Zou, J.; Zhou, Y.; Zhong, C.; Zhang, X. Novel serological biomarker panel using protein microarray can distinguish active TB from latent TB infection. Microbes Infect. 2022, 24, 105002.
- Watford, M.; Wu, G. Protein. Adv. Nutr. 2018, 9, 651–653.
- Neagu, M.; Bostan, M.; Constantin, C. Protein microarray technology: Assisting personalized medicine in oncology (Review). World Acad. Sci. J. 2019, 15, 113–124.
- Sutandy, F.X.R.; Qian, J.; Chen, C.; Zhu, H. Overview of protein microarrays. Curr. Protoc. Protein Sci. 2013, 72, 27.21.1–27.21.16.
- Rosenberg, J.M.; Utz, P.J. Protein Microarrays: A New Tool for the Study of Autoantibodies in Immunodeficiency. Front. Immunol. 2015, 6, 138.
- Hall, D.A.; Ptacek, J.; Snyder, M. Protein microarray technology. Mech. Ageing Dev. 2007, 128, 161–167.
- Mueller, C.; Liotta, L.A.; Espina, V. Reverse phase protein microarrays advance to use in clinical trials. Mol. Oncol. 2010, 4, 461–481.
- Liotta, L.A.; Espina, V.; Mehta, A.I.; Calvert, V.; Rosenblatt, K.; Geho, D.; Munson, P.J.; Young, L.; Wulfkuhle, J.; Petricoin, E.F., III. Protein microarrays: Meeting analytical challenges for clinical applications. Cancer Cell 2003, 3, 317–325.
- Coarfa, C.; Grimm, S.L.; Rajapakshe, K.; Perera, D.; Lu, H.Y.; Wang, X.; Christensen, K.R.; Mo, Q.; Edwards, D.P.; Huang, S. Reverse-Phase Protein Array: Technology, Application, Data Processing, and Integration. J. Biomol. Tech. JBT 2021, 32, 15–29.
- Kuang, Z.; Huang, R.; Yang, Z.; Lv, Z.; Chen, X.; Xu, F.; Yi, Y.-H.; Wu, J.; Huang, R.-P. Quantitative screening of serum protein biomarkers by reverse phase protein arrays. Oncotarget 2018, 9, 32624–32641.
- Sun, S.; Zhang, X.; Ma, J.; Ni, C.; Ying, X.; Wang, J.; Li, L.; Yuan, W.; Heng, X.; Xia, J. Serum protein profile of yang-deficiency constitution in traditional Chinese medicine revealed by protein microarray analyses. J. Tradit. Chin. Med. 2019, 6, 67–74.
- Petersen, A.C.; Clampitt, J.M.; Minion, F.C. Analysis of swine antigen-specific antibody responses to Mycoplasma hyopneumoniae infection determined by protein microarray. Vet. Microbiol. 2019, 230, 195–201.
- Zhao, H.; Sha, J.; Wu, T.; Chen, T.; Chen, X.; Ji, H.; Wang, Y.; Zhu, H.; Xie, L.; Ma, Y. Spatial modulation of biomolecules immobilization by fabrication of hierarchically structured PEG-derived brush micropatterns: An versatile cellular microarray platform. Appl. Surf. Sci. 2020, 529, 147056.
- Robajac, D.; Križáková, M.; Masnikosa, R.; Miljuš, G.; Šunderić, M.; Nedić, O.; Katrlík, J. Sensitive glycoprofiling of insulin-like growth factor receptors isolated from colon tissue of patients with colorectal carcinoma using lectin-based protein microarray. Int. J. Biol. Macromol. 2020, 144, 932–937.
- Pažitná, L.; Nemčovič, M.; Pakanová, Z.; Baráth, P.; Aliev, T.; Dolgikh, D.; Argentova, V.; Katrlík, J. Influence of media composition on recombinant monoclonal IgA1 glycosylation analysed by lectin-based protein microarray and MALDI-MS. J. Biotechnol. 2020, 314–315, 34–40.
- Wang, X.; Chen, Y.; Zhu, J.; Yang, Z.; Gong, X.; Hui, R.; Huang, G.; Jin, J. A comprehensive screening method for investigating the potential binding targets of doxorubicin based on protein microarray. Eur. J. Pharmacol. 2021, 896, 173896.
- Chang, C.-M.; Awanye, A.M.; Marsay, L.; Dold, C.; Pollard, A.J.; Rollier, C.S.; Feavers, I.M.; Maiden, M.C.J.; Derrick, J.P. Application of a Neisseria meningitidis antigen microarray to identify candidate vaccine proteins from a human Phase I clinical trial. Vaccine 2022, 40, 3835–3842.
- Volokitina, M.; Krutyakova, M.; Sirotov, V.; Larionov, M.; Tennikova, T.; Korzhikova-Vlakh, E. Protein biochips based on macroporous polymer supports: Material properties and analytical potential. J. Pharm. Biomed. Anal. 2019, 165, 242–250.
- Ho, T.-S.; Du, P.-X.; Su, W.-Y.; Santos, H.M.; Lin, Y.-L.; Chou, Y.-Y.; Keskin, B.B.; Pau, C.H.; Syu, G.-D. Development of SARS-CoV-2 variant protein microarray for profiling humoral immunity in vaccinated subjects. Biosens. Bioelectron. 2022, 204, 114067.
- Beck, S.; Nakajima, R.; Jasinskas, A.; Abram, T.J.; Kim, S.J.; Bigdeli, N.; Tifrea, D.F.; Hernandez-Davies, J.; Huw Davies, D.; Hedde, P.N.; et al. A Protein Microarray-Based Respiratory Viral Antigen Testing Platform for COVID-19 Surveillance. Biomedicines 2022, 10, 2238.
- Kober, C.; Niessner, R.; Seidel, M. Quantification of viable and non-viable Legionella spp. by heterogeneous asymmetric recombinase polymerase amplification (haRPA) on a flow-based chemiluminescence microarray. Biosens. Bioelectron. 2018, 100, 49–55.
- Wutz, K.; Meyer, V.K.; Wacheck, S.; Krol, P.; Gareis, M.; Nölting, C.; Struck, F.; Soutschek, E.; Böcher, O.; Niessner, R.; et al. New Route for Fast Detection of Antibodies against Zoonotic Pathogens in Sera of Slaughtered Pigs by Means of Flow-through Chemiluminescence Immunochips. Anal. Chem. 2013, 85, 5279–5285.
- Klüpfel, J.; Koros, R.C.; Dehne, K.; Ungerer, M.; Würstle, S.; Mautner, J.; Feuerherd, M.; Protzer, U.; Hayden, O.; Elsner, M.; et al. Automated, flow-based chemiluminescence microarray immunoassay for the rapid multiplex detection of IgG antibodies to SARS-CoV-2 in human serum and plasma (CoVRapid CL-MIA). Anal. Bioanal. Chem. 2021, 413, 5619–5632.
- Longworth, J.; Dittmar, G. An antigen microarray protocol for COVID-19 serological analysis. STAR Protoc. 2021, 2, 100815.
- Lai, D.-y.; Jiang, H.-w.; Li, Y.; Zhang, H.-n.; Tao, S.-c. SARS-CoV-2 proteome microarray for COVID-19 patient sera profiling. STAR Protoc. 2022, 3, 101238.
- Meng, X.; Wei, J.; Wang, Y.; Zhang, H.; Wang, Z. The role of peptide microarrays in biomedical research. Anal. Methods 2018, 10, 4614–4624.
- de la Rica, R.; Matsui, H. Applications of peptide and protein-based materials in bionanotechnology. Chem. Soc. Rev. 2010, 39, 3499–3509.
- Groß, A.; Hashimoto, C.; Sticht, H.; Eichler, J. Synthetic Peptides as Protein Mimics. Front. Bioeng. Biotechnol. 2016, 3, 211.
- Johansson, H.J.; Andaloussi, S.E.; Langel, U. Mimicry of protein function with cell-penetrating peptides. Methods Mol. Biol. 2011, 683, 233–247.
- Karoyan, P.; Vieillard, V.; Gómez-Morales, L.; Odile, E.; Guihot, A.; Luyt, C.-E.; Denis, A.; Grondin, P.; Lequin, O. Human ACE2 peptide-mimics block SARS-CoV-2 pulmonary cells infection. Commun. Biol. 2021, 4, 197.
- Wang, L.; Wang, N.; Zhang, W.; Cheng, X.; Yan, Z.; Shao, G.; Wang, X.; Wang, R.; Fu, C. Therapeutic peptides: Current applications and future directions. Signal Transduct. Target Ther. 2022, 7, 48.
- Fodor, S.P.; Read, J.L.; Pirrung, M.C.; Stryer, L.; Lu, A.T.; Solas, D. Light-directed, spatially addressable parallel chemical synthesis. Science 1991, 251, 767–773.
- Frank, R.; Guler, S.; Krause, S.; Lindenmaier, W. Facile and rapid spot-synthesis of large numbers of peptides on membrane sheets. In Proceedings of the 21st European Peptide Symposium, Platja d’Aro, Spain, 2–8 September 1990; Volume 2, pp. 151–152.
- Szymczak, L.C.; Kuo, H.Y.; Mrksich, M. Peptide Arrays: Development and Application. Anal. Chem. 2018, 90, 266–282.
- Mikus, M.; Zandian, A.; Sjöberg, R.; Hamsten, C.; Forsström, B.; Andersson, M.; Greiff, L.; Uhlén, M.; Levin, M.; Nilsson, P.; et al. Allergome-wide peptide microarrays enable epitope deconvolution in allergen-specific immunotherapy. J. Allergy Clin. Immunol. 2021, 147, 1077–1086.
- Jian, M.; Su, M.; Gao, J.; Wang, Z. Peptide microarray-based fluorescence assay for quantitatively monitoring the tumor-associated matrix metalloproteinase-2 activity. Sens. Actuators B 2020, 304, 127320.
- Jian, M.; Zhang, H.; Li, X.; Wang, Z. Profiling of multiple matrix metalloproteinases activities in the progression of osteosarcoma by peptide microarray-based fluorescence assay on polymer brush coated zinc oxide nanorod substrate. Sens. Actuators B 2021, 330, 129361.
- Li, Y.; Lai, D.-Y.; Tao, S.-C. SARS-CoV-2 spike linear epitope scanning via a peptide microarray through sera profiling. STAR Protoc. 2021, 2, 100707.
- Sola, L.; Brambilla, D.; Mussida, A.; Consonni, R.; Damin, F.; Cretich, M.; Gori, A.; Chiari, M. A bi-functional polymeric coating for the co-immobilization of proteins and peptides on microarray substrates. Anal. Chim. Acta 2021, 1187, 339138.
- Wang, H.; Wu, X.; Zhang, X.; Hou, X.; Liang, T.; Wang, D.; Teng, F.; Dai, J.; Duan, H.; Guo, S.; et al. SARS-CoV-2 Proteome Microarray for Mapping COVID-19 Antibody Interactions at Amino Acid Resolution. ACS Cent. Sci. 2020, 6, 2238–2249.
- Zamecnik, C.R.; Rajan, J.V.; Yamauchi, K.A.; Mann, S.A.; Loudermilk, R.P.; Sowa, G.M.; Zorn, K.C.; Alvarenga, B.D.; Gaebler, C.; Caskey, M.; et al. ReScan, a Multiplex Diagnostic Pipeline, Pans Human Sera for SARS-CoV-2 Antigens. Cell Rep. Med. 2020, 1, 100123.
- Su, W.Y.; Du, P.X.; Santos, H.M.; Ho, T.S.; Keskin, B.B.; Pau, C.H.; Yang, A.M.; Chou, Y.Y.; Shih, H.C.; Syu, G.D. Antibody Profiling in COVID-19 Patients with Different Severities by Using Spike Variant Protein Microarrays. Anal. Chem. 2022, 94, 6529–6539.
- Gao, C.; Wei, M.; McKitrick, T.R.; McQuillan, A.M.; Heimburg-Molinaro, J.; Cummings, R.D. Glycan Microarrays as Chemical Tools for Identifying Glycan Recognition by Immune Proteins. Front. Chem. 2019, 7, 833.
- van Kooyk, Y.; Rabinovich, G.A. Protein-glycan interactions in the control of innate and adaptive immune responses. Nat. Immunol. 2008, 9, 593–601.
- Crocker, P.R.; Paulson, J.C.; Varki, A. Siglecs and their roles in the immune system. Nat. Rev. Immunol. 2007, 7, 255–266.
- Fukui, S.; Feizi, T.; Galustian, C.; Lawson, A.M.; Chai, W. Oligosaccharide microarrays for high-throughput detection and specificity assignments of carbohydrate-protein interactions. Nat. Biotechnol. 2002, 20, 1011–1017.
- Wang, D.; Liu, S.; Trummer, B.J.; Deng, C.; Wang, A. Carbohydrate microarrays for the recognition of cross-reactive molecular markers of microbes and host cells. Nat. Biotechnol. 2002, 20, 275–281.
- Hyun, J.Y.; Pai, J.; Shin, I. The Glycan Microarray Story from Construction to Applications. Acc. Chem. Res. 2017, 50, 1069–1078.
- Kim, Y.; Hyun, J.Y.; Shin, I. Glycan microarrays from construction to applications. Chem. Soc. Rev. 2022, 51, 8276–8299.
- Rillahan, C.D.; Paulson, J.C. Glycan microarrays for decoding the glycome. Annu. Rev. Biochem. 2011, 80, 797–823.
- Ruprecht, C.; Geissner, A.; Seeberger, P.H.; Pfrengle, F. Practical considerations for printing high-density glycan microarrays to study weak carbohydrate-protein interactions. Carbohydr. Res. 2019, 481, 31–35.
- Muthana, S.M. Glycan Microarray: Toward Drug Discovery and Development; Elsevier: Amsterdam, The Netherlands, 2022.
- Yu, Y.; Song, X.; Smith, D.F.; Cummings, R.D. Chapter 12—Applications of Glycan Microarrays to Functional Glycomics. In Comprehensive Analytical Chemistry; Simó, C., Cifuentes, A., García-Cañas, V., Eds.; Elsevier: Amsterdam, The Netherlands, 2014; Volume 63, pp. 281–303.
- Xu, B.; Liu, Y.; Xie, Y. Application of glycan microarrays in cancer research. Sheng Wu Gong Cheng Xue Bao 2020, 36, 2313–2326.
- Nietzold, C.; Dietrich, P.M.; Holzweber, M.; Lippitz, A.; Kamalakumar, A.; Blanchard, V.; Ivanov-Pankov, S.; Weigel, W.; Panne, U.; Unger, W.E.S. Surface chemical characterization of model glycan surfaces and shelf life studies of glycan microarrays using XPS, NEXAFS spectroscopy, ToF-SIMS and fluorescence scanning. Appl. Surf. Sci. 2018, 459, 860–873.
- Marcelo, F.; Supekar, N.; Corzana, F.; van der Horst, J.C.; Vuist, I.M.; Live, D.; Boons, G.-J.P.H.; Smith, D.F.; van Vliet, S.J. Identification of a secondary binding site in human macrophage galactose-type lectin by microarray studies: Implications for the molecular recognition of its ligands. J. Biol. Chem. 2019, 294, 1300–1311.
- Leviatan Ben-Arye, S.; Schneider, C.; Yu, H.; Bashir, S.; Chen, X.; von Gunten, S.; Padler-Karavani, V. Differential Recognition of Diet-Derived Neu5Gc-Neoantigens on Glycan Microarrays by Carbohydrate-Specific Pooled Human IgG and IgA Antibodies. Bioconjug. Chem. 2019, 30, 1565–1574.
- Di Maio, A.; Cioce, A.; Achilli, S.; Thépaut, M.; Vivès, C.; Fieschi, F.; Rojo, J.; Reichardt, N.-C. Controlled density glycodendron microarrays for studying carbohydrate–lectin interactions. Org. Biomol. Chem. 2021, 19, 7357–7362.
- Liu, C.; Yang, L.; Niu, Q.; Yu, G.; Li, G. Carbohydrate microarrays fabricated on poly(2-methylacrylic acid)-based substrates for analysis of carbohydrate–protein interactions. New J. Chem. 2022, 46, 4300–4306.
- Temme, J.S.; Crainic, J.A.; Walker, L.M.; Yang, W.; Tan, Z.; Huang, X.; Gildersleeve, J.C. Microarray-guided evaluation of the frequency, B-cell origins, and selectivity of human glycan-binding antibodies reveals new insights and novel antibodies. J. Biol. Chem. 2022, 298, 102468.
- Liu, C.; Li, C.; Niu, Q.; Cai, C.; Li, G.; Yu, G. Fabrication of carbohydrate microarrays on poly(2-hydroxyethyl methacrylate)-cyanuric chloride-modified substrates for the analysis of carbohydrate–lectin interactions. New J. Chem. 2019, 43, 9145–9151.
- Hu, J.; Xiao, Y.; Shao, S.-A.; Gu, R.; Shi, Q.-M.; Liu, Z.-H.; Yin, J. Construction and application of carbohydrate microarrays to detect foodborne bacteria. Chin. J. Nat. Med. 2020, 18, 219–225.
- Cao, C.; Yu, L.; Yan, J.; Fu, D.; Yuan, J.; Liang, X. Purification of natural neutral N-glycans by using two-dimensional hydrophilic interaction liquid chromatography × porous graphitized carbon chromatography for glycan-microarray assay. Talanta 2021, 221, 121382.
- Silva, L.M.; Correia, V.G.; Moreira, A.S.P.; Domingues, M.R.M.; Ferreira, R.M.; Figueiredo, C.; Azevedo, N.F.; Marcos-Pinto, R.; Carneiro, F.; Magalhães, A.; et al. Helicobacter pylori lipopolysaccharide structural domains and their recognition by immune proteins revealed with carbohydrate microarrays. Carbohydr. Polym. 2021, 253, 117350.
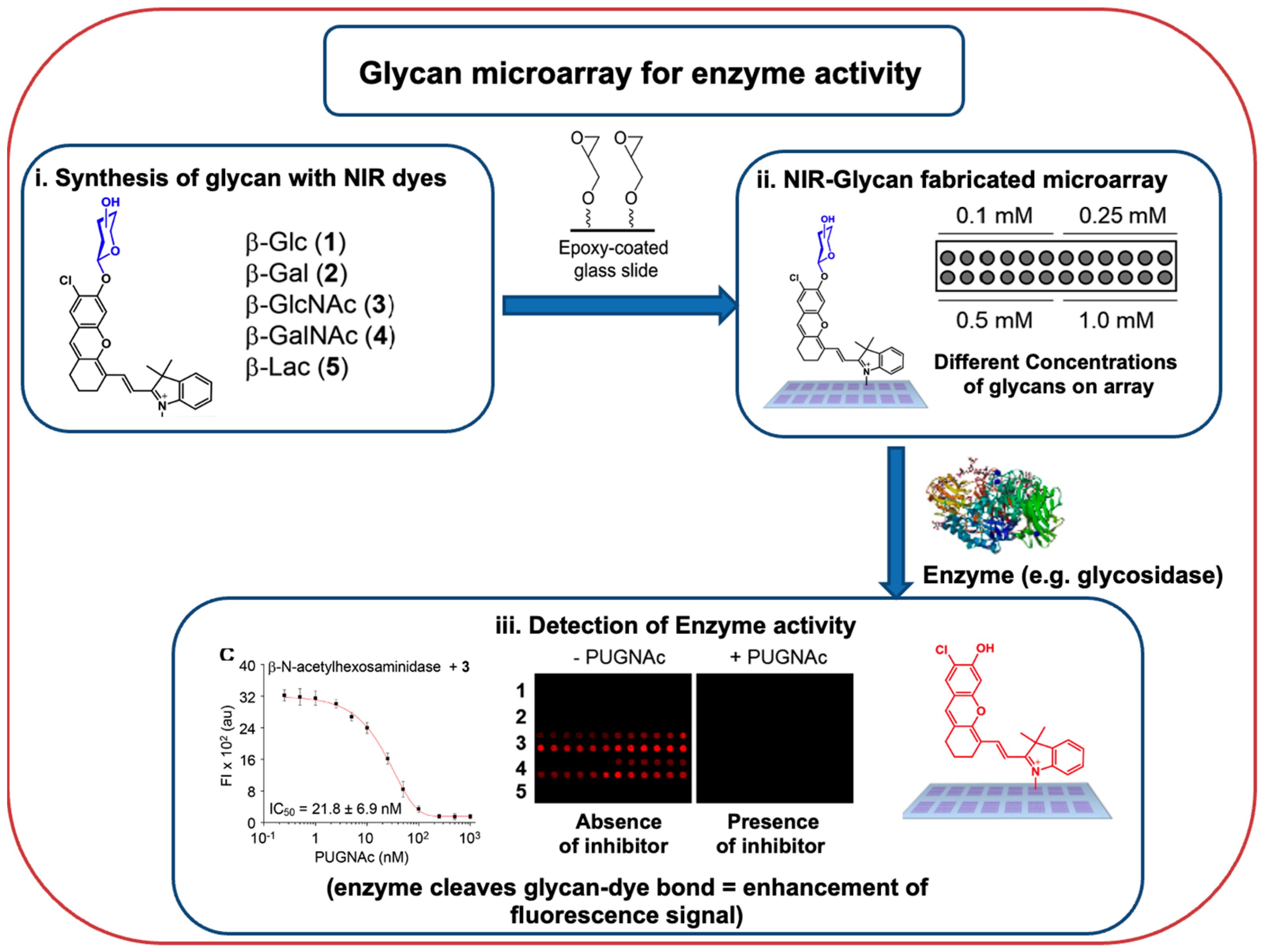
- Hyun, J.Y.; Kang, N.R.; Shin, I. Carbohydrate Microarrays Containing Glycosylated Fluorescent Probes for Assessment of Glycosidase Activities. Org. Lett. 2018, 20, 1240–1243.
- KÖHler, G.; Milstein, C. Continuous cultures of fused cells secreting antibody of predefined specificity. Nature 1975, 256, 495–497.
- Mahmuda, A.; Bande, F.; Abdulhaleem, N.; Al-Zihiry, K.J.K.; Majid, R.A.; Hamat, R.A.; Abdullah, W.O.; Zasmy, N. Monoclonal antibodies in immunodiagnostic assays: A review of recent applications. Sokoto J. Vet. Sci. 2017, 15, 1–10.
- Ball, H.J.; Finlay, D. Diagnostic Application of Monoclonal Antibody (MAb)-Based Sandwich ELISAs. In Mycoplasma Protocols; Miles, R., Nicholas, R., Eds.; Humana Press: Totowa, NJ, USA, 1998; pp. 127–132.
- Mahmoud, Z.; El-Gawad, A.R.; Abdel-Latef, T.; Hassan, M. Monoclonal Antibodies Production, Diagnostic and Therapeutic Applications. Sohag J. Sci. 2018, 22, 357–363.
- Lu, R.-M.; Hwang, Y.-C.; Liu, I.J.; Lee, C.-C.; Tsai, H.-Z.; Li, H.-J.; Wu, H.-C. Development of therapeutic antibodies for the treatment of diseases. J. Biomed. Sci. 2020, 27, 1.
- Wang, S.S.; Yan, Y.S.; Ho, K. US FDA-approved therapeutic antibodies with high-concentration formulation: Summaries and perspectives. Antib. Ther. 2021, 4, 262–272.
- Chen, Z.; Dodig-Crnković, T.; Schwenk, J.M.; Tao, S.-c. Current applications of antibody microarrays. Clin. Proteom. 2018, 15, 7.
- Gao, Y.; Fangel, J.U.; Willats, W.G.T.; Moore, J.P. Tracking polysaccharides during white winemaking using glycan microarrays reveals glycoprotein-rich sediments. Int. Food Res. J. 2019, 123, 662–673.
- Chen, M.; Yang, X.; Yang, M.; Zhang, W.; Li, L.; Sun, Q. Identification of a novel biomarker-CCL5 using antibody microarray for colorectal cancer. Pathol. Res. Pract. 2019, 215, 1033–1037.
- Gumanova, N.G.; Vasilyev, D.K.; Bogdanova, N.L.; Havrichenko, Y.I.; Kots, A.Y.; Metelskaya, V.A. Application of an antibody microarray for serum protein profiling of coronary artery stenosis. Biochem. Biophys. Res. Commun. 2022, 631, 55–63.
- Chen, B.-Y.; Hsu, C.-C.; Chen, Y.-Z.; Lin, J.-J.; Tseng, H.-H.; Jang, F.-L.; Chen, P.-S.; Chen, W.-N.; Chen, C.-S.; Lin, S.-H. Profiling antibody signature of schizophrenia by Escherichia coli proteome microarrays. Brain Behav. Immun. 2022, 106, 11–20.
- Liu, Q.; Zhou, X.; Wu, H.; Wu, L.; Zheng, B. A polydopamine patterned perfluoropolymer-based substrate for protein microarray applications. Sens. Actuators B 2019, 287, 306–311.
- Jeon, C.H.; Ha, T.H. Covalent and Oriented Immobilization of Antibodies through Systematic Modification of Photoactivatable RNA Hybrid Aptamers. Bioconjug. Chem. 2022, 33, 1527–1535.
- Brambilla, D.; Sola, L.; Chiari, M. Advantageous antibody microarray fabrication through DNA-directed immobilization: A step toward use of extracellular vesicles in diagnostics. Talanta 2021, 222, 121542.
- Plotnikova, M.A.; Klotchenko, S.A.; Lebedev, K.I.; Lozhkov, A.A.; Taraskin, A.S.; Gyulikhandanova, N.E.; Ramsay, E.S.; Vasin, A.V. Antibody microarray immunoassay for screening and differential diagnosis of upper respiratory tract viral pathogens. J. Immunol. Methods 2020, 478, 112712.
- Martinaud, C.; Hejl, C.; Igert, A.; Bigaillon, C.; Bonnet, C.; Merens, A.; Wolf, A.; Foissaud, V.; Leparc-Goffart, I. Evaluation of the Quotient(R) MosaiQ COVID-19 antibody microarray for the detection of IgG and IgM antibodies to SARS-CoV-2 virus in humans. J. Clin. Virol. 2020, 130, 104571.
- Dawson, E.D.; Kuck, L.R.; Blair, R.H.; Taylor, A.W.; Toth, E.; Knight, V.; Rowlen, K.L. Multiplexed, microscale, microarray-based serological assay for antibodies against all human-relevant coronaviruses. J. Virol. Methods 2021, 291, 114111.
- Song, Y.; Ye, Y.; Su, S.-H.; Stephens, A.; Cai, T.; Chung, M.-T.; Han, M.K.; Newstead, M.W.; Yessayan, L.; Frame, D.; et al. A digital protein microarray for COVID-19 cytokine storm monitoring. Lab Chip 2021, 21, 331–343.
- Chen, X.; Tan, F.; Zhang, H.; Zhang, X.; Xu, F.; Yuan, J.; Sun, C.; Huang, L.; Guan, H.; Luo, C.; et al. Serum cytokine profiles in phlegm-dampness constitution and damp-heat constitution using proteomic antibody microarray. Eur. J. Integr. Med. 2022, 52, 102126.
- Lakhin, A.V.; Tarantul, V.Z.; Gening, L.V. Aptamers: Problems, solutions and prospects. Acta Nat. 2013, 5, 34–43.
- Collett, J.R.; Cho, E.J.; Ellington, A.D. Production and processing of aptamer microarrays. Methods 2005, 37, 4–15.
- Ni, S.; Zhuo, Z.; Pan, Y.; Yu, Y.; Li, F.; Liu, J.; Wang, L.; Wu, X.; Li, D.; Wan, Y.; et al. Recent Progress in Aptamer Discoveries and Modifications for Therapeutic Applications. ACS Appl. Mater. Interfaces 2021, 13, 9500–9519.
- Zhang, Y.; Lai, B.S.; Juhas, M. Recent Advances in Aptamer Discovery and Applications. Molecules 2019, 24, 941.
- Witt, M.; Walter, J.G.; Stahl, F. Aptamer Microarrays-Current Status and Future Prospects. Microarrays 2015, 4, 115–132.
- Kong, H.Y.; Byun, J. Nucleic Acid aptamers: New methods for selection, stabilization, and application in biomedical science. Biomol. Ther. 2013, 21, 423–434.
- Srivastava, S.; Abraham, P.R.; Mukhopadhyay, S. Aptamers: An Emerging Tool for Diagnosis and Therapeutics in Tuberculosis. Front. Cell. Infect. Microbiol. 2021, 11, 656421.
- Wang, T.; Chen, C.; Larcher, L.M.; Barrero, R.A.; Veedu, R.N. Three decades of nucleic acid aptamer technologies: Lessons learned, progress and opportunities on aptamer development. Biotechnol. Adv. 2019, 37, 28–50.
- Bennett, J.W.; Klich, M. Mycotoxins. Clin. Microbiol. Rev. 2003, 16, 497–516.
- Alshannaq, A.; Yu, J.H. Occurrence, Toxicity, and Analysis of Major Mycotoxins in Food. Int. J. Environ. Res. Public Health 2017, 14, 632.
- Liu, R.; Li, W.; Cai, T.; Deng, Y.; Ding, Z.; Liu, Y.; Zhu, X.; Wang, X.; Liu, J.; Liang, B.; et al. TiO2 Nanolayer-Enhanced Fluorescence for Simultaneous Multiplex Mycotoxin Detection by Aptamer Microarrays on a Porous Silicon Surface. ACS Appl. Mater. Interfaces 2018, 10, 14447–14453.
- Bhattu, M.; Verma, M.; Kathuria, D. Recent advancements in the detection of organophosphate pesticides: A review. Anal. Methods 2021, 13, 4390–4428.
- Kaushal, J.; Khatri, M.; Arya, S.K. A treatise on Organophosphate pesticide pollution: Current strategies and advancements in their environmental degradation and elimination. Ecotoxicol. Environ. Saf. 2021, 207, 111483.
- USUA Foreign Agricultural Service. China: National Food Safety Standard Maximum Residue Limits for Pesticides in Foods. 2019. Available online: https://www.fas.usda.gov/data/china-national-food-safety-standard-maximum-residue-limits-pesticides-foods (accessed on 17 February 2023).
- Wang, X.; Yang, Y.; Yin, Y.; Zeng, N.; Dong, Y.; Liu, J.; Wang, L.; Yang, Z.; Yang, C. High-Throughput Aptamer Microarrays for Fluorescent Detection of Multiple Organophosphorus Pesticides in Food. Anal. Chem. 2022, 94, 3173–3179.
