Your browser does not fully support modern features. Please upgrade for a smoother experience.
Please note this is an old version of this entry, which may differ significantly from the current revision.
Subjects:
Pathology
Stroke is one of the leading causes of morbidity and mortality worldwide. A main cause of brain damage by stroke is ischemia-reperfusion (IR) injury due to the increased production of reactive oxygen species (ROS) and energy failure caused by changes in mitochondrial metabolism. Reverse electron transfer (RET) has been implicated in excessive ROS production during IR injury in stroke and new insights into the mechanism of RET gained from various systems in recent studies will help understand stroke pathophysiology and inform therapy.
- mitochondrial complex I
- reverse electron transport (RET)
- reactive oxygen species (ROS)
1. Introduction
Stroke is the second leading cause of morbidity and mortality worldwideafter cancer. In almost 80% of the cases, stroke develops due to a cerebral artery obstruction and/or occlusion [1,2]. During ischemic stroke, the absence of blood supply deprives the brain cells of the glucose and oxygen nutrients they require, disrupting their cellular homeostasis and ultimately resulting in cell death [2]. Mitochondrial dysfunction and deleterious post-stroke ROS are considered hallmark stroke pathologies [3]. A number of pathophysiological processes, including oxidative stress, excitotoxicity, dysregulated endocrine signaling, inflammation, and apoptosis, are involved in the complex pathophysiological process known as cerebral ischemia/reperfusion injury (CIRI), which frequently causes neuronal injury, cell death, and permanent brain damage [3]. ROS-induced oxidative stress during cerebral ischemia leads to eventual cell death after reperfusion. Mitochondrial ROS (mito-ROS) plays a detrimental role in neuronal death during CIRI at several key stages: inflammation, blood brain barrier (BBB) disruption, mitochondrial respiratory chain complex I-III dysfunction, oedema formation, and apoptosis and autophagy [4,5,6] Currently, thrombolytic and endoscopic thrombectomy are the only therapeutic options, along with post-stroke conservative treatments. The tissue plasminogen activator (tPA) is the only Federal Drug Administration (FDA)-approved therapy for stroke treatment, but due to its various side effects and limited therapeutic window, it only benefits a small portion of stroke patients [7,8,9,10,11]. Hence, there is an urgent need to develop safer and novel therapeutic options for the treatment of ischemia reperfusion injuries. Due to reduced blood flow and hypoxic conditions, mitochondria are heavily affected by the low O2 and glucose environment. Since the 1960s, the effect of ischemia reperfusion injury on mitochondria has been a research focus and extensive efforts have been dedicated to developing therapeutic strategies to combat reperfusion injury.
2. ROS and Oxidative Stress in Stroke
2.1. ROS Generation during Stroke
Oxidative stress is the result of an imbalance between ROS production and antioxidant defense mechanisms in cells [3]. Because of its high and specific metabolic activity, neurons are particularly vulnerable to oxidative damage. High oxygen consumption, almost entirely oxidative phosphorylation, low energy reserves, high concentrations of peroxidizable lipids, and high levels of iron acting as prooxidants all contribute to this vulnerability [12]. As a result, neuronal cells are extremely vulnerable to metabolic/ischemic damage and the associated oxidative stress [13]. The macro- and micromolecular changes of neuronal cells ends in neurodegeneration in various neurological disorders, e.g., post-stroke dementia, Alzheimer’s disease (AD), vascular dementia, and others [14]. The progression of ischemic stroke pathology is closely connected to dysregulated ROS. The most common ROS are superoxide (O2−), hydrogen peroxide (H2O2), hydroxyl radical (HO−), hypochlorous acid (HOCl−), nitric oxide (NO), and peroxynitrite (ONOO−) that are produced by either intracellular responses (mitochondria) or extracellular inflammation [3,15]. The intracellular production of ROS is mainly due to the altered metabolic activity of the mitochondrial respiratory chain, whereas extracellularly it is a result of inflammasome activation and the immune response [16,17]. These reactive molecules can cause lipid peroxidation, protein oxidation, and DNA and RNA damage, resulting in cellular homeostatic failure and tissue damage (Figure 1). Nitric oxide synthetase (NOS) produces NO that is instrumental in the immune response; phagocytes produce large amounts of NO during ischemic brain injury [3,18]. There are several natural defense mechanisms in the cell to remove ROS or prevent oxidative damage [19]. These include catalase (CAT), heme oxygenase (HO), glutathione reductase, glutathione (GSH), glutathione peroxidase (GSH-Px), superoxide dismutase (SOD), and Vitamins E and C. Microglia and astrocytes are the primary producers of ROS and reactive nitrogen species (RNS), the latter is produced by endothelial NOS (eNOS), inducible NOS (iNOS), or neuronal NOS (nNOS) during ischemic brain injury, which together influence synaptic transmission as well as non-synaptic communication between neurons and glia [20,21]. ROS and RNS diffuse to the oligodendrocyte myelin sheath during periods of increased neuronal activity, activating the protein kinase C and post-translationally modifying the myelin basic protein, a key structural component of myelin [22,23].
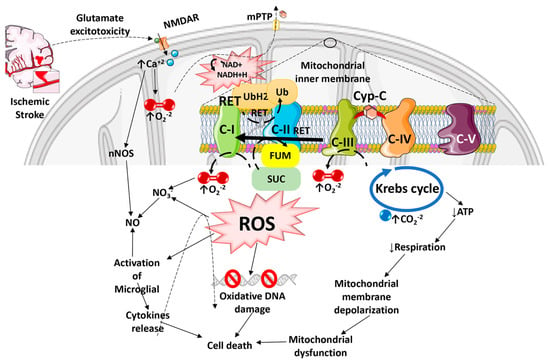
Figure 1. Diagram illustrating how Reactive Oxygen Species especially Reactive Oxygen Species generated by the Reverse Electron Transport process, and the ensuing oxidative stress contributes to mitochondrial dysfunction, cell death, and progressive ischemic stroke pathology. ROS can damage mitochondrial DNA because of the lack of a chromatin-like structure that would protect DNA against ROS insults. ROS can also damage lipids and protein structures in mitochondrial matrix and further exacerbate mitochondrial dysfunction. Mitochondrial ROS released into the cytosol can activate microglia and astrocytes, causing neuroinflammation and death of injured neurons.
2.2. ROS-Induced Damages during Stroke
The consequences of ROS imbalance in ischemic stroke are significant, and include apoptosis, disruption of the BBB, inflammation, edema formation, autophagy, and other pathophysiological events. O2, H2O2, and NO play critical roles in neuron-glia communication in the hippocampus [24]. The synaptic long-term potentiation (LTP), which is necessary for memory formation in the hippocampus, also becomes impaired by excessive ROS production during ischemia, resulting in post-stroke cognitive decline [25,26]. ROS-mediated injury can result in the formation of conjugated dienic hydroperoxides, which can be degraded into aldehydes, dienals, or alkanes that are extremely toxic to neurons and white matter, resulting in apoptosis followed by chronic neurodegeneration after ischemic brain damage [27]. The inflammation and oxidative stress that develop in the brain after a stroke have been linked more recently to tryptophan oxidation via the kynurenine pathway [28]. The release of glutamate is a crucial mechanism that determines tissue damage after cerebral ischemia [29]. On the other hand, when the a-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid (AMPA) receptors are activated, oxygen is produced. When oxygen combines with NO, it creates the extremely harmful ONOO. When one of the ROS species is inactivated, the vessel can expand due to the other reactant (ROS) escaping [30]. It has also been shown that increased kynurenic acid levels are linked to poorer outcomes, and the infarct volume is strongly correlated with a decreased ratio of 3-hydroxyanthranilic acid to anthranilic acid (a free radical generator) [31,32]. Nuclear DNA damage has been linked to two distinct mechanisms, including DNA fragmentation caused by endonucleases and oxidative modification. In ischemic stroke injury, poly (ADP-ribose) polymerase (PARP) activation occurs in two phases, starting in the neuronal components and localizing 3–4 days later in the infiltrating inflammatory cells [33]. It has also been suggested that there may be reductions in the nuclear protein apurinic apyrimidinic endonuclease (APE/Ref-1), which cleaves ROS-induced apyrimidinic sites in oxidized DNA [27]. Moreover, following transient global ischemia, the p53-upregulated modulator of apoptosis (PUMA) is upregulated in the hippocampal neurons [34]. ROS can also activate caspase-activated DNase (CAD), which cleaves DNA and causes apoptosis, resulting in progressive neurodegeneration and associated post-stroke disorders [35]. Thus, excessive ROS production can cause mitochondrial dysfunction leading to overall cellular dysfunction and cell death in ischemic stroke (Figure 1).
This entry is adapted from the peer-reviewed paper 10.3390/antiox12040895
This entry is offline, you can click here to edit this entry!
