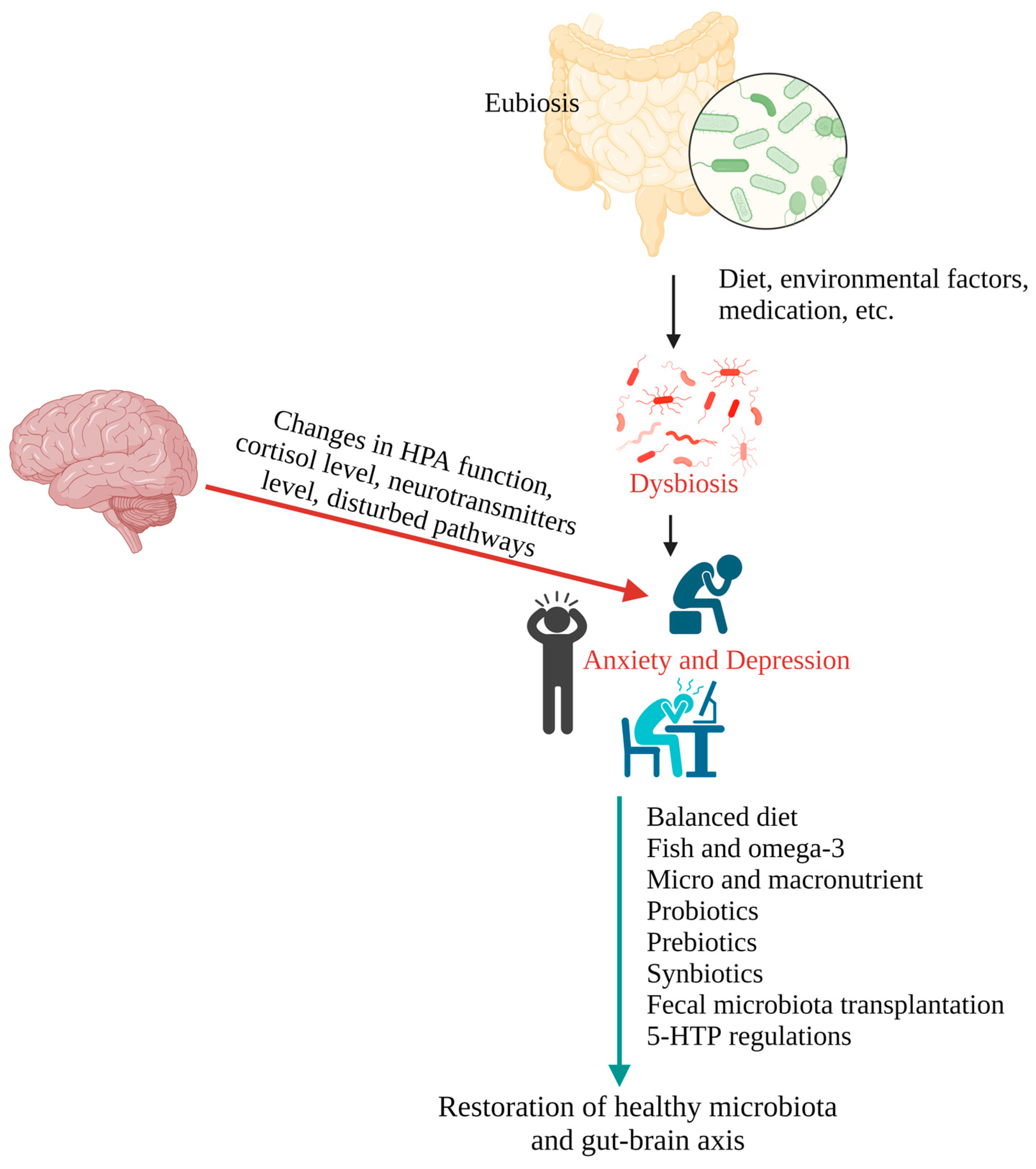
The gut microbiota is critical for maintaining human health and the immunological system. Several neuroscientific studies have shown the significance of microbiota in developing brain systems. The gut microbiota and the brain are interconnected in a bidirectional relationship, as research on the microbiome–gut–brain axis shows. Significant evidence links anxiety and depression disorders to the community of microbes that live in the gastrointestinal system. Modified diet, fish and omega-3 fatty acid intake, macro- and micro-nutrient intake, prebiotics, probiotics, synbiotics, postbiotics, fecal microbiota transplantation, and 5-HTP regulation may all be utilized to alter the gut microbiota as a treatment approach.
- gut–brain axis
- microbiota
- anxiety
- depression
1. Epidemiology of Depression and Anxiety
1.1. The Gut–Brain Axis in Depression and Anxiety
1.2. The Relationships between Epigenetics, Gut Microbiota, Depression, and Anxiety
2. Potential Therapy Involved in the Treatment of Depression and Anxiety
| Intervention | Subjects | Main Results | Ref. |
|---|---|---|---|
| L. rhamnosus | Mice | Lowered stress-induced anxiety and depressive-like behavior | [31] |
| Lactobacillus acidophilus, B. longum, B. lactis, B. bifidum, galacto-oligosaccharides (GOS), inulin and fructo-oligosaccharides (FOS) | Human | The synbiotic intervention reduced the depression and anxiety symptoms effectively | [39] |
| Bacillus coagulans | Human | Reduction of depression and irritable bowel syndrome symptoms | [40] |
| Bifidobacterium longum | Human | Improved the quality of life and lessened depression, but not anxiety, in patients with irritable bowel syndrome. | [41] |
| Clostridium butyricum | Human | Enhanced the effectiveness of traditional treatment for depression | [42] |
| L. acidophilus, L. casei, and B. bifidum | Human | Reduction of Beck’s Depression Inventory score | [43] |
| L. helveticus R0052 and B. longum | Human | Effectively reduced symptoms of depression and decreased anxiety | [44] |
| L. helveticus and B. longum |
Human | Improvement of Beck’s Depression Inventory score | [45][46] |
| L. plantarum | Human | Improvement in symptoms and psychological scores | [47] |
| B. longum subsp. longum BAMA-B05/Bau-B1024, B. lactis BAMA-B06/Bau-B0111, B. adolescentis, Streptococcus thermophiles, L. acidophilus, and L. delbrueckii subsp. bulgaricus |
Human | Decreased anxiety. | [48] |
| S. thermophiles, B. longum, B. breve, L. rhamnosus, L. bulgaricus, L. acidophilus, L. casei, and FOS | Human | Improved depressive symptoms in major depressive disorder | [49] |
| Short-chain FOS | Human | Improved depression and anxiety score in patients with irritable bowel syndrome | [50] |
| FOS and GOS | Mice | Improved depression and anxiety | [51] |
| Fecal microbiota transplantation | Mice | Reduced stress-associated depressive-like behavior | [52] |
| Oligosaccharides 3′sialyllactose (3′SL) or 6′sialyllactose (6′SL) | Mice | Lower nervous anxiety-related reactions and preventative impact on anxious behavior. | [53] |
| Polysaccharide of okra | Mice | Restored the gut microbiota | [54] |
2.1. Modified Diet
2.2. Fish and Omega-3 FA Intake
2.3. Micronutrient Intake
2.4. Macronutrient Intake
2.5. Probiotics
2.6. Prebiotics
2.7. Synbiotics
2.8. Postbiotics
2.9. Fecal Microbiota Transplantation (FMT)
2.10. Bifidobacterium and 5-HTP Regulation
This entry is adapted from the peer-reviewed paper 10.3390/ph16040565
References
- Alli, S.R.; Gorbovskaya, I.; Liu, J.C.W.; Kolla, N.J.; Brown, L.; Müller, D.J. The Gut Microbiome in Depression and Potential Benefit of Prebiotics, Probiotics and Synbiotics: A Systematic Review of Clinical Trials and Observational Studies. Int. J. Mol. Sci. 2022, 23, 4494.
- Smith, P.M.; Howitt, M.R.; Panikov, N.; Michaud, M.; Gallini, C.A.; Bohlooly-Y, M.; Glickman, J.N.; Garrett, W.S. The Microbial Metabolites, Short-Chain Fatty Acids, Regulate Colonic T Reg Cell Homeostasis. Science 2013, 341, 569–573.
- Cash, H.L.; Whitham, C.V.; Behrendt, C.L.; Hooper, L.V. Symbiotic Bacteria Direct Expression of an Intestinal Bactericidal Lectin. Science 2006, 313, 1126–1130.
- Shishov, V.A.; Kirovskaya, T.A.; Kudrin, V.S.; Oleskin, A.V. Amine Neuromediators, Their Precursors, and Oxidation Products in the Culture of Escherichia Coli k-12. Appl. Biochem. Microbiol. 2009, 45, 494–497.
- Barrett, E.; Ross, R.P.; O’Toole, P.W.; Fitzgerald, G.F.; Stanton, C. γ-Aminobutyric Acid Production by Culturable Bacteria from the Human Intestine. J. Appl. Microbiol. 2012, 113, 411–417.
- Silva, Y.P.; Bernardi, A.; Frozza, R.L. The Role of Short-Chain Fatty Acids From Gut Microbiota in Gut-Brain Communication. Front. Endocrinol. 2020, 11, 25.
- Wikoff, W.R.; Anfora, A.T.; Liu, J.; Schultz, P.G.; Lesley, S.A.; Peters, E.C.; Siuzdak, G. Metabolomics Analysis Reveals Large Effects of Gut Microflora on Mammalian Blood Metabolites. Proc. Natl. Acad. Sci. USA 2009, 106, 3698–3703.
- Yano, J.M.; Yu, K.; Donaldson, G.P.; Shastri, G.G.; Ann, P.; Ma, L.; Nagler, C.R.; Ismagilov, R.F.; Mazmanian, S.K.; Hsiao, E.Y. Indigenous Bacteria from the Gut Microbiota Regulate Host Serotonin Biosynthesis. Cell 2015, 161, 264–276.
- Rutsch, A.; Kantsjö, J.B.; Ronchi, F. The Gut-Brain Axis: How Microbiota and Host Inflammasome Influence Brain Physiology and Pathology. Front. Immunol. 2020, 11, 3237.
- Braniste, V.; Al-Asmakh, M.; Kowal, C.; Anuar, F.; Abbaspour, A.; Tóth, M.; Korecka, A.; Bakocevic, N.; Guan, N.L.; Kundu, P.; et al. The Gut Microbiota Influences Blood-Brain Barrier Permeability in Mice. Sci. Transl. Med. 2014, 6, 263ra158.
- Acharya, N.K.; Levin, E.C.; Clifford, P.M.; Han, M.; Tourtellotte, R.; Chamberlain, D.; Pollaro, M.; Coretti, N.J.; Kosciuk, M.C.; Nagele, E.P.; et al. Diabetes and Hypercholesterolemia Increase Blood-Brain Barrier Permeability and Brain Amyloid Deposition: Beneficial Effects of the LpPLA2 Inhibitor Darapladib. J. Alzheimer’s Dis. 2013, 35, 179–198.
- Lee, S.W.; Kim, W.J.; Choi, Y.K.; Song, H.S.; Son, M.J.; Gelman, I.H.; Kim, Y.J.; Kim, K.W. SSeCKS Regulates Angiogenesis and Tight Junction Formation in Blood-Brain Barrier. Nat. Med. 2003, 9, 900–906.
- Spadoni, I.; Fornasa, G.; Rescigno, M. Organ-Specific Protection Mediated by Cooperation between Vascular and Epithelial Barriers. Nat. Rev. Immunol. 2017, 17, 761–773.
- Shah, S.N.; Knausenberger, T.B.-A.; Connell, E.; Le Gall, G.; Hardy, T.A.J.; Randall, D.W.; McCafferty, K.; Yaqoob, M.M.; Solito, E.; Müller, M.; et al. Cerebrovascular Damage Caused by the Gut Microbe-Derived Uraemic Toxinp-Cresol Sulfate Is Prevented by Blockade of the Epidermal Growth Factor Receptor. bioRxiv 2022.
- Stachulski, A.V.; Knausenberger, T.B.A.; Shah, S.N.; Hoyles, L.; McArthur, S. A Host-Gut Microbial Amino Acid Co-Metabolite, p-Cresol Glucuronide, Promotes Blood-Brain Barrier Integrity in Vivo. Tissue Barriers 2023, 11, 2073175.
- Torrisi, S.A.; Lavanco, G.; Maurel, O.M.; Gulisano, W.; Laudani, S.; Geraci, F.; Grasso, M.; Barbagallo, C.; Caraci, F.; Bucolo, C.; et al. A Novel Arousal-Based Individual Screening Reveals Susceptibility and Resilience to PTSD-like Phenotypes in Mice. Neurobiol. Stress 2020, 14, 100286.
- Needham, B.D.; Funabashi, M.; Adame, M.D.; Wang, Z.; Boktor, J.C.; Haney, J.; Wu, W.L.; Rabut, C.; Ladinsky, M.S.; Hwang, S.J.; et al. A Gut-Derived Metabolite Alters Brain Activity and Anxiety Behaviour in Mice. Nature 2022, 602, 647–653.
- Hsiao, E.Y.; McBride, S.W.; Hsien, S.; Sharon, G.; Hyde, E.R.; McCue, T.; Codelli, J.A.; Chow, J.; Reisman, S.E.; Petrosino, J.F.; et al. Microbiota Modulate Behavioral and Physiological Abnormalities Associated with Neurodevelopmental Disorders. Cell 2013, 155, 1451–1463.
- Du, Y.; Gao, X.R.; Peng, L.; Ge, J.F. Crosstalk between the Microbiota-Gut-Brain Axis and Depression. Heliyon 2020, 6, e04097.
- Farooq, R.K.; Asghar, K.; Kanwal, S.; Zulqernain, A. Role of Inflammatory Cytokines in Depression: Focus on Interleukin-1β (Review). Biomed. Rep. 2017, 6, 15–20.
- Moulton, C.D.; Pavlidis, P.; Norton, C.; Norton, S.; Pariante, C.; Hayee, B.; Powell, N. Depressive Symptoms in Inflammatory Bowel Disease: An Extraintestinal Manifestation of Inflammation? Clin. Exp. Immunol. 2019, 197, 308–318.
- Schachter, J.; Martel, J.; Lin, C.S.; Chang, C.J.; Wu, T.R.; Lu, C.C.; Ko, Y.F.; Lai, H.C.; Ojcius, D.M.; Young, J.D. Effects of Obesity on Depression: A Role for Inflammation and the Gut Microbiota. Brain Behav. Immun. 2018, 69, 1–8.
- Kiecolt-Glaser, J.K.; Derry, H.M.; Fagundes, C.P. Inflammation: Depression Fans the Flames and Feasts on the Heat. Am. J. Psychiatry 2015, 172, 1075–1091.
- Martin-Subero, M.; Anderson, G.; Kanchanatawan, B.; Berk, M.; Maes, M. Comorbidity between Depression and Inflammatory Bowel Disease Explained by Immune-Inflammatory, Oxidative, and Nitrosative Stress; Tryptophan Catabolite; and Gut–Brain Pathways. CNS Spectr. 2016, 21, 184–198.
- Yang, C.; Wardenaar, K.J.; Bosker, F.J.; Li, J.; Schoevers, R.A. Inflammatory Markers and Treatment Outcome in Treatment Resistant Depression: A Systematic Review. J. Affect. Disord. 2019, 257, 640–649.
- Strawbridge, R.; Hodsoll, J.; Powell, T.R.; Hotopf, M.; Hatch, S.L.; Breen, G.; Cleare, A.J. Inflammatory Profiles of Severe Treatment-Resistant Depression. J. Affect. Disord. 2019, 246, 42–51.
- Guida, F.; Turco, F.; Iannotta, M.; de Gregorio, D.; Palumbo, I.; Sarnelli, G.; Furiano, A.; Napolitano, F.; Boccella, S.; Luongo, L.; et al. Antibiotic-Induced Microbiota Perturbation Causes Gut Endocannabinoidome Changes, Hippocampal Neuroglial Reorganization and Depression in Mice. Brain Behav. Immun. 2018, 67, 230–245.
- Kelly, J.R.; Borre, Y.; O’ Brien, C.; Patterson, E.; el Aidy, S.; Deane, J.; Kennedy, P.J.; Beers, S.; Scott, K.; Moloney, G.; et al. Transferring the Blues: Depression-Associated Gut Microbiota Induces Neurobehavioural Changes in the Rat. J. Psychiatr. Res. 2016, 82, 109–118.
- Mason, B.L.; Li, Q.; Minhajuddin, A.; Czysz, A.H.; Coughlin, L.A.; Hussain, S.K.; Koh, A.Y.; Trivedi, M.H. Reduced Anti-Inflammatory Gut Microbiota Are Associated with Depression and Anhedonia. J. Affect. Disord. 2020, 266, 394–401.
- Cryan, J.F.; Dinan, T.G. Mind-Altering Microorganisms: The Impact of the Gut Microbiota on Brain and Behaviour. Nat. Rev. Neurosci. 2012, 13, 701–712.
- Bravo, J.A.; Forsythe, P.; Chew, M.V.; Escaravage, E.; Savignac, H.M.; Dinan, T.G.; Bienenstock, J.; Cryan, J.F. Ingestion of Lactobacillus Strain Regulates Emotional Behavior and Central GABA Receptor Expression in a Mouse via the Vagus Nerve. Proc. Natl. Acad. Sci. USA 2011, 108, 16050–16055.
- Jokela, M.; Virtanen, M.; David Batty, G.; Kivimaki, M. Inflammation and Specific Symptoms of Depression. JAMA Psychiatry 2016, 73, 87–88.
- Berk, M.; Williams, L.J.; Jacka, F.N.; O’Neil, A.; Pasco, J.A.; Moylan, S.; Allen, N.B.; Stuart, A.L.; Hayley, A.C.; Byrne, M.L.; et al. So Depression Is an Inflammatory Disease, but Where Does the Inflammation Come From? BMC Med. 2013, 11, 200.
- Abe-Higuchi, N.; Uchida, S.; Yamagata, H.; Higuchi, F.; Hobara, T.; Hara, K.; Kobayashi, A.; Watanabe, Y. Hippocampal Sirtuin 1 Signaling Mediates Depression-like Behavior. Biol. Psychiatry 2016, 80, 815–826.
- Higuchi, F.; Uchida, S.; Yamagata, H.; Abe-Higuchi, N.; Hobara, T.; Hara, K.; Kobayashi, A.; Shintaku, T.; Itoh, Y.; Suzuki, T.; et al. Hippocampal MicroRNA-124 Enhances Chronic Stress Resilience in Mice. J. Neurosci. 2016, 36, 7253–7267.
- Libert, S.; Pointer, K.; Bell, E.L.; Das, A.; Cohen, D.E.; Asara, J.M.; Kapur, K.; Bergmann, S.; Preisig, M.; Otowa, T.; et al. SIRT1 Activates MAO-A in the Brain to Mediate Anxiety and Exploratory Drive. Cell 2011, 147, 1459–1472.
- Begum, N.; Mandhare, A.; Tryphena, K.P.; Srivastava, S.; Shaikh, M.F.; Singh, S.B.; Khatri, D.K. Epigenetics in Depression and Gut-Brain Axis: A Molecular Crosstalk. Front. Aging Neurosci. 2022, 14, 1433.
- Fleshner, M.; Frank, M.; Maier, S.F. Danger Signals and Inflammasomes: Stress-Evoked Sterile Inflammation in Mood Disorders. Neuropsychopharmacology 2016, 42, 36–4525.
- Haghighat, N.; Rajabi, S.; Mohammadshahi, M. Effect of Synbiotic and Probiotic Supplementation on Serum Brain-Derived Neurotrophic Factor Level, Depression and Anxiety Symptoms in Hemodialysis Patients: A Randomized, Double-Blinded, Clinical Trial. Nutr. Neurosci. 2019, 24, 490–499.
- Majeed, M.; Nagabhushanam, K.; Arumugam, S.; Majeed, S.; Ali, F. Bacillus coagulans MTCC 5856 for the Management of Major Depression with Irritable Bowel Syndrome: A Randomised, Double blind, Placebo Controlled, Multi-Centre, Pilot Clinical Study. Food Nutr. Res. 2018, 62, 1218.
- Pinto-Sanchez, M.I.; Hall, G.B.; Ghajar, K.; Nardelli, A.; Bolino, C.; Lau, J.T.; Martin, F.P.; Cominetti, O.; Welsh, C.; Rieder, A.; et al. Probiotic Bifidobacterium Longum NCC3001 Reduces Depression Scores and Alters Brain Activity: A Pilot Study in Patients With Irritable Bowel Syndrome. Gastroenterology 2017, 153, 448–459.e8.
- Miyaoka, T.; Kanayama, M.; Wake, R.; Hashioka, S.; Hayashida, M.; Nagahama, M.; Okazaki, S.; Yamashita, S.; Miura, S.; Miki, H.; et al. Clostridium Butyricum MIYAIRI 588 as Adjunctive Therapy for Treatment-Resistant Major Depressive Disorder: A Prospective Open-Label Trial. Clin. Neuropharmacol. 2018, 41, 151–155.
- Akkasheh, G.; Kashani-Poor, Z.; Tajabadi-Ebrahimi, M.; Jafari, P.; Akbari, H.; Taghizadeh, M.; Memarzadeh, M.R.; Asemi, Z.; Esmaillzadeh, A. Clinical and Metabolic Response to Probiotic Administration in Patients with Major Depressive Disorder: A Randomized, Double-Blind, Placebo-Controlled Trial. Nutrition 2016, 32, 315–320.
- Messaoudi, M.; Violle, N.; Bisson, J.F.; Desor, D.; Javelot, H.; Rougeot, C. Beneficial Psychological Effects of a Probiotic Formulation (Lactobacillus helveticus R0052 and Bifidobacterium longum R0175) in Healthy Human Volunteers. Gut Microbes 2011, 2, 256–261.
- Romijn, A.R.; Rucklidge, J.J.; Kuijer, R.G.; Frampton, C. A Double-Blind, Randomized, Placebo-Controlled Trial of Lactobacillus helveticus and Bifidobacterium longum for the Symptoms of Depression. Aust. N. Z. J. Psychiatry 2017, 51, 810–821.
- Kazemi, A.; Noorbala, A.A.; Azam, K.; Eskandari, M.H.; Djafarian, K. Effect of Probiotic and Prebiotic vs Placebo on Psychological Outcomes in Patients with Major Depressive Disorder: A Randomized Clinical Trial. Clin. Nutr. 2019, 38, 522–528.
- Chong, H.X.; Yusoff, N.A.A.; Hor, Y.Y.; Lew, L.C.; Jaafar, M.H.; Choi, S.B.; Yusoff, M.S.B.; Wahid, N.; Abdullah, M.F.I.L.; Zakaria, N.; et al. Lactobacillus plantarum DR7 Alleviates Stress and Anxiety in Adults: A Randomised, Double-Blind, Placebo-Controlled Study. Benef. Microbes 2019, 10, 355–373.
- Qin, Q.; Liu, H.; Yang, Y.; Wang, Y.; Xia, C.; Tian, P.; Wei, J.; Li, S.; Chen, T. Probiotic Supplement Preparation Relieves Test Anxiety by Regulating Intestinal Microbiota in College Students. Dis. Markers 2021, 2021, e5597401.
- Ghorbani, Z.; Nazari, S.; Etesam, F.; Nourimajd, S.; Ahmadpanah, M.; Jahromi, S.R. The Effect of Synbiotic as an Adjuvant Therapy to Fluoxetine in Moderate Depression: A Randomized Multicenter Trial. Arch. Neurosci. 2018, 5, 60507.
- Azpiroz, F.; Dubray, C.; Bernalier-Donadille, A.; Cardot, J.M.; Accarino, A.; Serra, J.; Wagner, A.; Respondek, F.; Dapoigny, M. Effects of ScFOS on the Composition of Fecal Microbiota and Anxiety in Patients with Irritable Bowel Syndrome: A Randomized, Double Blind, Placebo Controlled Study. Neurogastroenterol. Motil. 2017, 29, e12911.
- Vulevic, J.; Juric, A.; Walton, G.E.; Claus, S.P.; Tzortzis, G.; Toward, R.E.; Gibson, G.R. Influence of Galacto-Oligosaccharide Mixture (B-GOS) on Gut Microbiota, Immune Parameters and Metabonomics in Elderly Persons. Br. J. Nutr. 2015, 114, 586–595.
- Rao, J.; Qiao, Y.; Xie, R.; Lin, L.; Jiang, J.; Wang, C.; Li, G. Fecal Microbiota Transplantation Ameliorates Stress-Induced Depression-like Behaviors Associated with the Inhibition of Glial and NLRP3 Inflammasome in Rat Brain. J. Psychiatr. Res. 2021, 137, 147–157.
- Tarr, A.J.; Galley, J.D.; Fisher, S.E.; Chichlowski, M.; Berg, B.M.; Bailey, M.T. The Prebiotics 3′Sialyllactose and 6′Sialyllactose Diminish Stressor-Induced Anxiety-like Behavior and Colonic Microbiota Alterations: Evidence for Effects on the Gut–Brain Axis. Brain Behav. Immun. 2015, 50, 166–177.
- Yan, T.; Nian, T.; Liao, Z.; Xiao, F.; Wu, B.; Bi, K.; He, B.; Jia, Y. Antidepressant Effects of a Polysaccharide from Okra (Abelmoschus esculentus (L) Moench) by Anti-Inflammation and Rebalancing the Gut Microbiota. Int. J. Biol. Macromol. 2020, 144, 427–440.
- Jacka, F.N. Nutritional Psychiatry: Where to Next? EBioMedicine 2017, 17, 24–29.
- Borovikova, L.V.; Ivanova, S.; Zhang, M.; Yang, H.; Botchkina, G.I.; Watkins, L.R.; Wang, H.; Abumrad, N.; Eaton, J.W.; Tracey, K.J. Vagus Nerve Stimulation Attenuates the Systemic Inflammatory Response to Endotoxin. Nature 2000, 405, 458–462.
- Fülling, C.; Dinan, T.G.; Cryan, J.F. Gut Microbe to Brain Signaling: What Happens in Vagus…. Neuron 2019, 101, 998–1002.
- Knudsen, J.K.; Michaelsen, T.Y.; Bundgaard-Nielsen, C.; Nielsen, R.E.; Hjerrild, S.; Leutscher, P.; Wegener, G.; Sørensen, S. Faecal Microbiota Transplantation from Patients with Depression or Healthy Individuals into Rats Modulates Mood-Related Behaviour. Sci. Rep. 2021, 11, 21869.
- Tardy, A.L.; Pouteau, E.; Marquez, D.; Yilmaz, C.; Scholey, A. Vitamins and Minerals for Energy, Fatigue and Cognition: A Narrative Review of the Biochemical and Clinical Evidence. Nutrients 2020, 12, 228.
- Sánchez-Villegas, A.; Delgado-Rodríguez, M.; Alonso, A.; Schlatter, J.; Lahortiga, F.; Serra-Majem, L.; Martínez-González, M.A. Association of the Mediterranean Dietary Pattern With the Incidence of Depression: The Seguimiento Universidad de Navarra/University of Navarra Follow-up (SUN) Cohort. Arch. Gen. Psychiatry 2009, 66, 1090–1098.
- Féart, C.; Samieri, C.; Rondeau, V.; Amieva, H.; Portet, F.; Dartigues, J.F.; Scarmeas, N.; Barberger-Gateau, P. Adherence to a Mediterranean Diet, Cognitive Decline, and Risk of Dementia. JAMA 2009, 302, 638–648.
- Ν Georgousopoulou, E.; Kastorini, C.-M.; Milionis, H.J.; Ntziou, E.; Kostapanos, M.; Nikolaou, V.; Vemmos, K.; Goudevenos, J.; Panagiotakos, D.B. Association Between Mediterranean Diet and Non-Fatal Cardiovascular Events, in the Context of Anxiety and Depression Disorders: A Case/Case-Control Study. Hell. J. Cardiol. 2014, 55, 24–31.
- Sánchez-Villegas, A.; Henríquez, P.; Bes-Rastrollo, M.; Doreste, J. Mediterranean Diet and Depression. Public Health Nutr. 2006, 9, 1104–1109.
- Merra, G.; Noce, A.; Marrone, G.; Cintoni, M.; Tarsitano, M.G.; Capacci, A.; de Lorenzo, A. Influence of Mediterranean Diet on Human Gut Microbiota. Nutrients 2020, 13, 7.
- Konishi, K. Associations between Healthy Japanese Dietary Patterns and Depression in Japanese Women. Public Health Nutr. 2021, 24, 1753–1765.
- Lin, H.V.; Frassetto, A.; Kowalik, E.J.; Nawrocki, A.R.; Lu, M.M.; Kosinski, J.R.; Hubert, J.A.; Szeto, D.; Yao, X.; Forrest, G.; et al. Butyrate and Propionate Protect against Diet-Induced Obesity and Regulate Gut Hormones via Free Fatty Acid Receptor 3-Independent Mechanisms. PLoS ONE 2012, 7, e35240.
- Burokas, A.; Arboleya, S.; Moloney, R.D.; Peterson, V.L.; Murphy, K.; Clarke, G.; Stanton, C.; Dinan, T.G.; Cryan, J.F. Targeting the Microbiota-Gut-Brain Axis: Prebiotics Have Anxiolytic and Antidepressant-like Effects and Reverse the Impact of Chronic Stress in Mice. Biol. Psychiatry 2017, 82, 472–487.
- Savignac, H.M.; Couch, Y.; Stratford, M.; Bannerman, D.M.; Tzortzis, G.; Anthony, D.C.; Burnet, P.W.J. Prebiotic Administration Normalizes Lipopolysaccharide (LPS)-Induced Anxiety and Cortical 5-HT2A Receptor and IL1-β Levels in Male Mice. Brain Behav. Immun. 2016, 52, 120–131.
- Ruusunen, A.; Lehto, S.M.; Mursu, J.; Tolmunen, T.; Tuomainen, T.P.; Kauhanen, J.; Voutilainen, S. Dietary Patterns Are Associated with the Prevalence of Elevated Depressive Symptoms and the Risk of Getting a Hospital Discharge Diagnosis of Depression in Middle-Aged or Older Finnish Men. J. Affect. Disord. 2014, 159, 1–6.
- Rienks, J.; Dobson, A.J.; Mishra, G.D. Mediterranean Dietary Pattern and Prevalence and Incidence of Depressive Symptoms in Mid-Aged Women: Results from a Large Community-Based Prospective Study. Eur. J. Clin. Nutr. 2013, 67, 75–82.
- Mamplekou, E.; Bountziouka, V.; Psaltopoulou, T.; Zeimbekis, A.; Tsakoundakis, N.; Papaerakleous, N.; Gotsis, E.; Metallinos, G.; Pounis, G.; Polychronopoulos, E.; et al. Urban Environment, Physical Inactivity and Unhealthy Dietary Habits Correlate to Depression among Elderly Living in Eastern Mediterranean Islands: The MEDIS (MEDiterranean ISlands Elderly) Study. J. Nutr. Health Aging 2010, 14, 449–455.
- Chatzi, L.; Melaki, V.; Sarri, K.; Apostolaki, I.; Roumeliotaki, T.; Georgiou, V.; Vassilaki, M.; Koutis, A.; Bitsios, P.; Kogevinas, M. Dietary Patterns during Pregnancy and the Risk of Postpartum Depression: The Mother-Child “Rhea” Cohort in Crete, Greece. Public Health Nutr. 2011, 14, 1663–1670.
- Réus, G.Z.; Maciel, A.L.; Abelaira, H.M.; de Moura, A.B.; de Souza, T.G.; dos Santos, T.R.; Darabas, A.C.; Parzianello, M.; Matos, D.; Abatti, M.; et al. ω-3 and Folic Acid Act against Depressive-like Behavior and Oxidative Damage in the Brain of Rats Subjected to Early- or Late-Life Stress. Nutrition 2018, 53, 120–133.
- Robertson, R.C.; Seira Oriach, C.; Murphy, K.; Moloney, G.M.; Cryan, J.F.; Dinan, T.G.; Paul Ross, R.; Stanton, C. Omega-3 Polyunsaturated Fatty Acids Critically Regulate Behaviour and Gut Microbiota Development in Adolescence and Adulthood. Brain Behav. Immun. 2017, 59, 21–37.
- Bastiaansen, J.A.; Munafò, M.R.; Appleton, K.M.; Oldehinkel, A.J. The Efficacy of Fish Oil Supplements in the Treatment of Depression: Food for Thought. Transl. Psychiatry 2016, 6, e975.
- Costantini, L.; Molinari, R.; Farinon, B.; Merendino, N. Impact of Omega-3 Fatty Acids on the Gut Microbiota. Int. J. Mol. Sci. 2017, 18, 2645.
- Pusceddu, M.M.; Kelly, P.; Ariffin, N.; Cryan, J.F.; Clarke, G.; Dinan, T.G. N-3 PUFAs Have Beneficial Effects on Anxiety and Cognition in Female Rats: Effects of Early Life Stress. Psychoneuroendocrinology 2015, 58, 79–90.
- Winther, G.; Pyndt Jørgensen, B.M.; Elfving, B.; Nielsen, D.S.; Kihl, P.; Lund, S.; Sorensen, D.B.; Wegener, G. Dietary Magnesium Deficiency Alters Gut Microbiota and Leads to Depressive-like Behaviour. Acta Neuropsychiatr. 2015, 27, 168–176.
- Kortman, G.A.M.; Raffatellu, M.; Swinkels, D.W.; Tjalsma, H. Nutritional Iron Turned inside out: Intestinal Stress from a Gut Microbial Perspective. FEMS Microbiol. Rev. 2014, 38, 1202–1234.
- Shah, H.E.; Bhawnani, N.; Ethirajulu, A.; Alkasabera, A.; Onyali, C.B.; Anim-Koranteng, C.; Mostafa, J.A.; Shah, H.E.; Bhawnani, N.; Ethirajulu, A.; et al. Iron Deficiency-Induced Changes in the Hippocampus, Corpus Striatum, and Monoamines Levels That Lead to Anxiety, Depression, Sleep Disorders, and Psychotic Disorders. Cureus 2021, 13, e18138.
- Rucklidge, J.J.; Kaplan, B.J. Broad-Spectrum Micronutrient Formulas for the Treatment of Psychiatric Symptoms: A Systematic Review. Expert Rev. Neurother. 2014, 13, 49–73.
- Miranda, P.M.; Bertolini, F.; Kadarmideen, H.N. Investigation of Gut Microbiome Association with Inflammatory Bowel Disease and Depression: A Machine Learning Approach. F1000Research 2019, 7, 702.
- Simpson, C.A.; Mu, A.; Haslam, N.; Schwartz, O.S.; Simmons, J.G. Feeling down? A Systematic Review of the Gut Microbiota in Anxiety/Depression and Irritable Bowel Syndrome. J. Affect. Disord. 2020, 266, 429–446.
- Coppen, A.; Bolander-Gouaille, C. Treatment of Depression: Time to Consider Folic Acid and Vitamin B12. J. Psychopharmacol. 2016, 19, 59–65.
- Shaffer, J.A.; Edmondson, D.; Wasson, L.T.; Falzon, L.; Homma, K.; Ezeokoli, N.; Li, P.; Davidson, K.W. Vitamin d Supplementation for Depressive Symptoms: A Systematic Review and Meta-Analysis of Randomized Controlled Trials. Psychosom. Med. 2014, 76, 190–196.
- Ekwaru, J.P.; Zwicker, J.D.; Holick, M.F.; Giovannucci, E.; Veugelers, P.J. The Importance of Body Weight for the Dose Response Relationship of Oral Vitamin D Supplementation and Serum 25-Hydroxyvitamin D in Healthy Volunteers. PLoS ONE 2014, 9, e111265.
- Tosunbayraktar, G.; Bas, M.; Kut, A.; Buyukkaragoz, A.H. Low Serum 25(OH)D Levels Are Assocıated to Hıgher BMI and Metabolic Syndrome Parameters in Adult Subjects in Turkey. Afr. Health Sci. 2016, 15, 1161–1169.
- Kimball, S.M.; Mirhosseini, N.; Rucklidge, J. Database Analysis of Depression and Anxiety in a Community Sample-Response to a Micronutrient Intervention. Nutrients 2018, 10, 152.
- Oh, J.; Yun, K.; Chae, J.H.; Kim, T.S. Association Between Macronutrients Intake and Depression in the United States and South Korea. Front. Psychiatry 2020, 11, 207.
- Fernstrom, J.D.; Wurtman, R.J.; Hammarstrom-Wiklund, B.; Rand, W.M.; Munro, H.N.; Davidson, C.S. Diurnal Variations in Plasma Neutral Amino Acid Concentrations among Patients with Cirrhosis: Effect of Dietary Protein. Am. J. Clin. Nutr. 1979, 32, 1923–1933.
- Wurtman, J.J. Depression and Weight Gain: The Serotonin Connection. J. Affect. Disord. 1993, 29, 183–192.
- Fernstrom, J.D.; Wurtman, R.J. Brain Serotonin Content: Increase Following Ingestion of Carbohydrate Diet. Science 1971, 174, 1023–1025.
- Maciej, S.; Becker, F.G.; Cleary, M.; Team, R.M.; Holtermann, H.; The, D.; Agenda, N.; Science, P.; Sk, S.K.; Hinnebusch, R.; et al. Systematic Review on Effects of Diet on Gut Microbiota in Relation to Metabolic Syndromes. J. Clin. Nutr. Metab. 2017, 1, 343–354.
- Breymeyer, K.L.; Lampe, J.W.; McGregor, B.A.; Neuhouser, M.L. Subjective Mood and Energy Levels of Healthy Weight and Overweight/Obese Healthy Adults on High-and Low-Glycemic Load Experimental Diets. Appetite 2016, 107, 253–259.
- Kris-Etherton, P.M.; Petersen, K.S.; Hibbeln, J.R.; Hurley, D.; Kolick, V.; Peoples, S.; Rodriguez, N.; Woodward-Lopez, G. Nutrition and Behavioral Health Disorders: Depression and Anxiety. Nutr. Rev. 2021, 79, 247.
- Chaiyasut, C.; Sivamaruthi, B.S. Influence of Probiotic Supplementation on Brain Function: Involvement of Gut Microbiome, Inflammation, and Stress Pathway. In Gut Microbiota–Brain Axis, 1st ed.; Evrensel, A., Unsalver, B.O., Eds.; Intechopen Limited: London, UK, 2018; Volume 1, pp. 19–33.
- Alkasir, R.; Li, J.; Li, X.; Jin, M.; Zhu, B. Human Gut Microbiota: The Links with Dementia Development. Protein Cell 2016, 8, 90–102.
- Sivamaruthi, B.S.; Prasanth, M.I.; Kesika, P.; Chaiyasut, C. Probiotics in Human Mental Health and Diseases—A Mini Review. Trop. J. Pharm. Res. 2021, 18, 889–895.
- Cheung, S.G.; Goldenthal, A.R.; Uhlemann, A.C.; Mann, J.J.; Miller, J.M.; Sublette, M.E. Systematic Review of Gut Microbiota and Major Depression. Front. Psychiatry 2019, 10, 34.
- Sherwin, E.; Dinan, T.G.; Cryan, J.F. Recent Developments in Understanding the Role of the Gut Microbiota in Brain Health and Disease. Ann. N. Y. Acad. Sci. 2018, 1420, 5–25.
- Saravana Babu, C.; Kesavanarayanan, K.S.; Kalaivani, P.; Ranju, V.; Ramanathan, M. A Simple Densitometric Method for the Quantification of Inhibitory Neurotransmitter Gamma-Aminobutyric Acid (GABA) in Rat Brain Tissue. Chromatogr. Res. Int. 2011, 2011, 732409.
- Katano, Y.; Fujinami, S.; Kawakoshi, A.; Nakazawa, H.; Oji, S.; Iino, T.; Oguchi, A.; Ankai, A.; Fukui, S.; Terui, Y.; et al. Complete Genome Sequence of Oscillibacter valericigenes Sjm18-20T (=NBRC 101213T). Stand. Genom. Sci. 2012, 6, 406.
- Allen, A.P.; Hutch, W.; Borre, Y.E.; Kennedy, P.J.; Temko, A.; Boylan, G.; Murphy, E.; Cryan, J.F.; Dinan, T.G.; Clarke, G. Bifidobacterium longum 1714 as a Translational Psychobiotic: Modulation of Stress, Electrophysiology and Neurocognition in Healthy Volunteers. Transl. Psychiatry 2016, 6, e939.
- Karen, C.; Shyu, D.J.H.; Rajan, K.E. Lactobacillus paracasei Supplementation Prevents Early Life Stress-Induced Anxiety and Depressive-Like Behavior in Maternal Separation Model-Possible Involvement of Microbiota-Gut-Brain Axis in Differential Regulation of MicroRNA124a/132 and Glutamate Receptors. Front. Neurosci. 2021, 15, 1115.
- Sonali, S.; Ray, B.; Tousif, H.A.; Rathipriya, A.G.; Sunanda, T.; Mahalakshmi, A.M.; Rungratanawanich, W.; Essa, M.M.; Qoronfleh, M.W.; Chidambaram, S.B.; et al. Mechanistic Insights into the Link between Gut Dysbiosis and Major Depression: An Extensive Review. Cells 2022, 11, 1362.
- Kumar, A.; Prajapati, B.; Pramanik, J. Prebiotics as Adjunctive Therapy in Diabetes: A Review of Prebiotics in Diabetes. Curr. Nutrac. 2022, 3, e180822207618.
- Schmidt, K.; Cowen, P.J.; Harmer, C.J.; Tzortzis, G.; Errington, S.; Burnet, P.W.J. Prebiotic Intake Reduces the Waking Cortisol Response and Alters Emotional Bias in Healthy Volunteers. Psychopharmacology 2015, 232, 1793–1801.
- Dinan, T.G.; Cryan, J.F. The Microbiome-Gut-Brain Axis in Health and Disease. Gastroenterol. Clin. N. Am. 2017, 46, 77–89.
- Xiao, Q.; Shu, R.; Wu, C.; Tong, Y.; Xiong, Z.; Zhou, J.; Yu, C.; Xie, X.; Fu, Z. Crocin-I Alleviates the Depression-like Behaviors Probably via Modulating “Microbiota-Gut-Brain” Axis in Mice Exposed to Chronic Restraint Stress. J. Affect. Disord. 2020, 276, 476–486.
- Markowiak, P.; Ślizewska, K. Effects of Probiotics, Prebiotics, and Synbiotics on Human Health. Nutrients 2017, 9, 1021.
- Kleniewska, P.; Pawliczak, R. Influence of Synbiotics on Selected Oxidative Stress Parameters. Oxid. Med. Cell. Longev. 2017, 2017, e9315375.
- Schröder, J.; Dören, M.; Schneider, B.; Oettel, M. Are the Antioxidative Effects of 17β-Estradiol Modified by Concomitant Administration of a Progestin? Maturitas 1996, 25, 133–139.
- Salminen, S.; Collado, M.C.; Endo, A.; Hill, C.; Lebeer, S.; Quigley, E.M.M.; Sanders, M.E.; Shamir, R.; Swann, J.R.; Szajewska, H.; et al. The International Scientific Association of Probiotics and Prebiotics (ISAPP) Consensus Statement on the Definition and Scope of Postbiotics. Nat. Rev. Gastroenterol. Hepatol. 2021, 18, 649–667.
- Taverniti, V.; Guglielmetti, S. The Immunomodulatory Properties of Probiotic Microorganisms beyond Their Viability (Ghost Probiotics: Proposal of Paraprobiotic Concept). Genes Nutr. 2011, 6, 261–274.
- Maehata, H.; Kobayashi, Y.; Mitsuyama, E.; Kawase, T.; Kuhara, T.; Xiao, J.Z.; Tsukahara, T.; Toyoda, A. Heat-Killed Lactobacillus Helveticus Strain MCC1848 Confers Resilience to Anxiety or Depression-like Symptoms Caused by Subchronic Social Defeat Stress in Mice. Biosci. Biotechnol. Biochem. 2019, 83, 1239–1247.
- Kambe, J.; Watcharin, S.; Makioka-Itaya, Y.; Inoue, R.; Watanabe, G.; Yamaguchi, H.; Nagaoka, K. Heat-Killed Enterococcus Fecalis (EC-12) Supplement Alters the Expression of Neurotransmitter Receptor Genes in the Prefrontal Cortex and Alleviates Anxiety-like Behavior in Mice. Neurosci. Lett. 2020, 720, 134753.
- Nishida, K.; Sawada, D.; Kuwano, Y.; Tanaka, H.; Rokutan, K. Health Benefits of Lactobacillus Gasseri CP2305 Tablets in Young Adults Exposed to Chronic Stress: A Randomized, Double-Blind, Placebo-Controlled Study. Nutrients 2019, 11, 1859.
- Meyyappan, A.C.; Forth, E.; Wallace, C.J.K.; Milev, R. Effect of Fecal Microbiota Transplant on Symptoms of Psychiatric Disorders: A Systematic Review. BMC Psychiatry 2020, 20, 299.
- Cai, T.; Shi, X.; Yuan, L.Z.; Tang, D.; Wang, F. Fecal Microbiota Transplantation in an Elderly Patient with Mental Depression. Int. Psychogeriatr. 2019, 31, 1525–1526.
- Nirmal, J.; Babu, C.S.; Harisudhan, T.; Ramanathan, M. Evaluation of Behavioural and Antioxidant Activity of Cytisus Scoparius Link in Rats Exposed to Chronic Unpredictable Mild Stress. BMC Complement. Altern. Med. 2008, 8, 15.
- Pu, Y.; Zhang, Q.; Tang, Z.; Lu, C.; Wu, L.; Zhong, Y.; Chen, Y.; Hashimoto, K.; Luo, Y.; Liu, Y. Fecal Microbiota Transplantation from Patients with Rheumatoid Arthritis Causes Depression-like Behaviors in Mice through Abnormal T Cells Activation. Transl. Psychiatry 2022, 12, 223.
- Wu, L.; Ran, L.; Wu, Y.; Liang, M.; Zeng, J.; Ke, F.; Wang, F.; Yang, J.; Lao, X.; Liu, L.; et al. Oral Administration of 5-Hydroxytryptophan Restores Gut Microbiota Dysbiosis in a Mouse Model of Depression. Front. Microbiol. 2022, 13, 1419.
- Tian, P.; Wang, G.; Zhao, J.; Zhang, H.; Chen, W. Bifidobacterium with the Role of 5-Hydroxytryptophan Synthesis Regulation Alleviates the Symptom of Depression and Related Microbiota Dysbiosis. J. Nutr. Biochem. 2019, 66, 43–51.
