The skin microbiome is composed of a complex association of bacteria, fungi, and viruses. The maintenance of skin commensal microbes is essential for preventing the overgrowth of pathogenic microorganisms or already present opportunistic pathogens. Thus, the development of bioactive compounds capable of modulating skin microbiome has become an important topic for both researchers and the cosmetic industry. Increasingly, scientific evidence highlights that metabolites derived from probiotics have a great potential to prevent diseases affecting the skin. These compounds have recently been called postbiotics and are defined as a “preparation of inanimate microorganisms and/or their components that confers a health benefit on the host”. Postbiotics are obtained from fermentations performed almost exclusively by lactic acid bacteria and yeast. Short-chain fatty acids, bacteriocins, and organic acids are some examples of postbiotics. These compounds exhibit antimicrobial, immunomodulatory, antioxidant, and anti-inflammatory activities. In addition, postbiotic production possesses technological advantages, including high stability and increased safety, compared to viable probiotics.
- skin microbiome
- postbiotic production
- skin care
1. Overview of Postbiotics
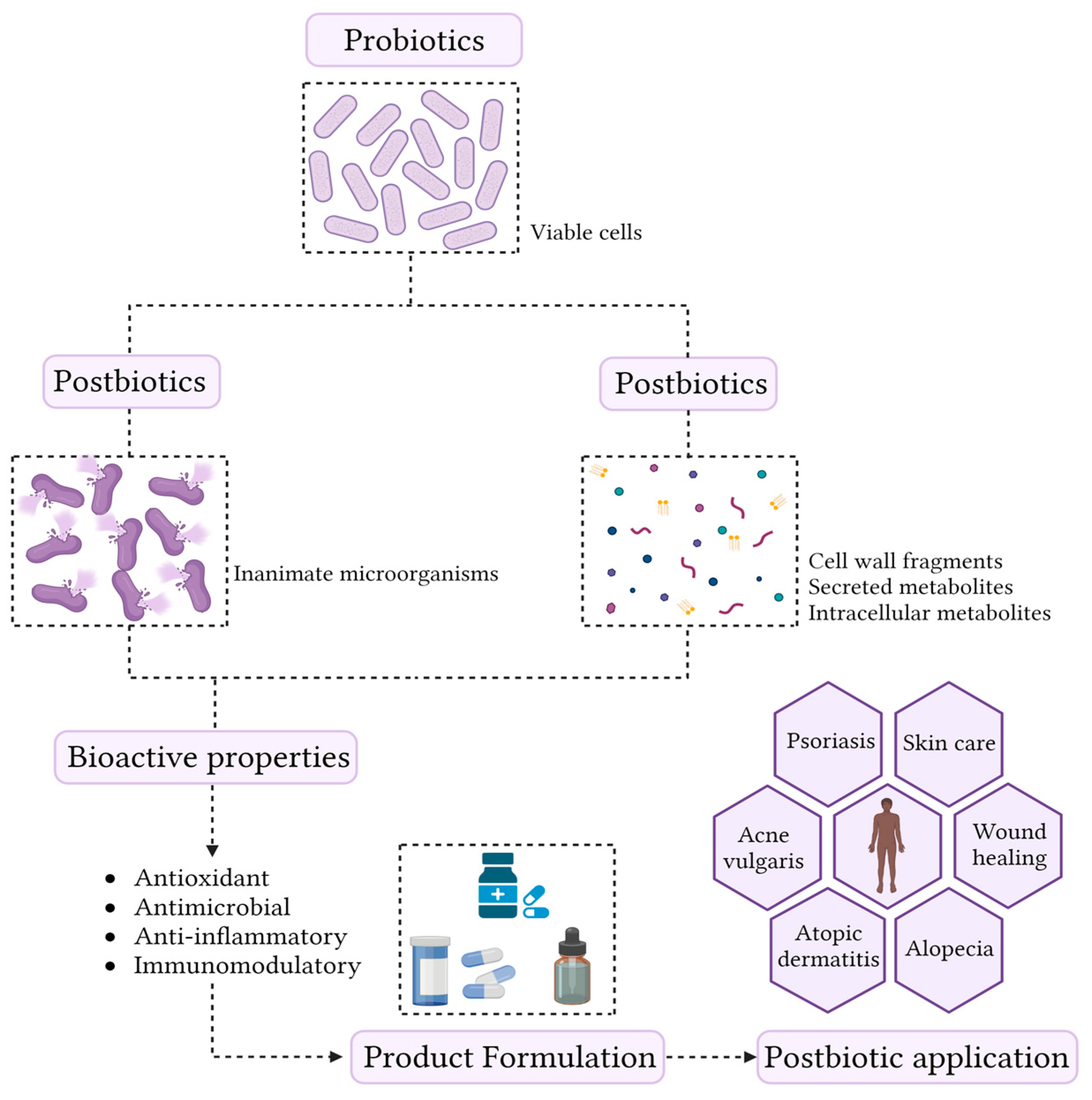
2. Postbiotic Production Process
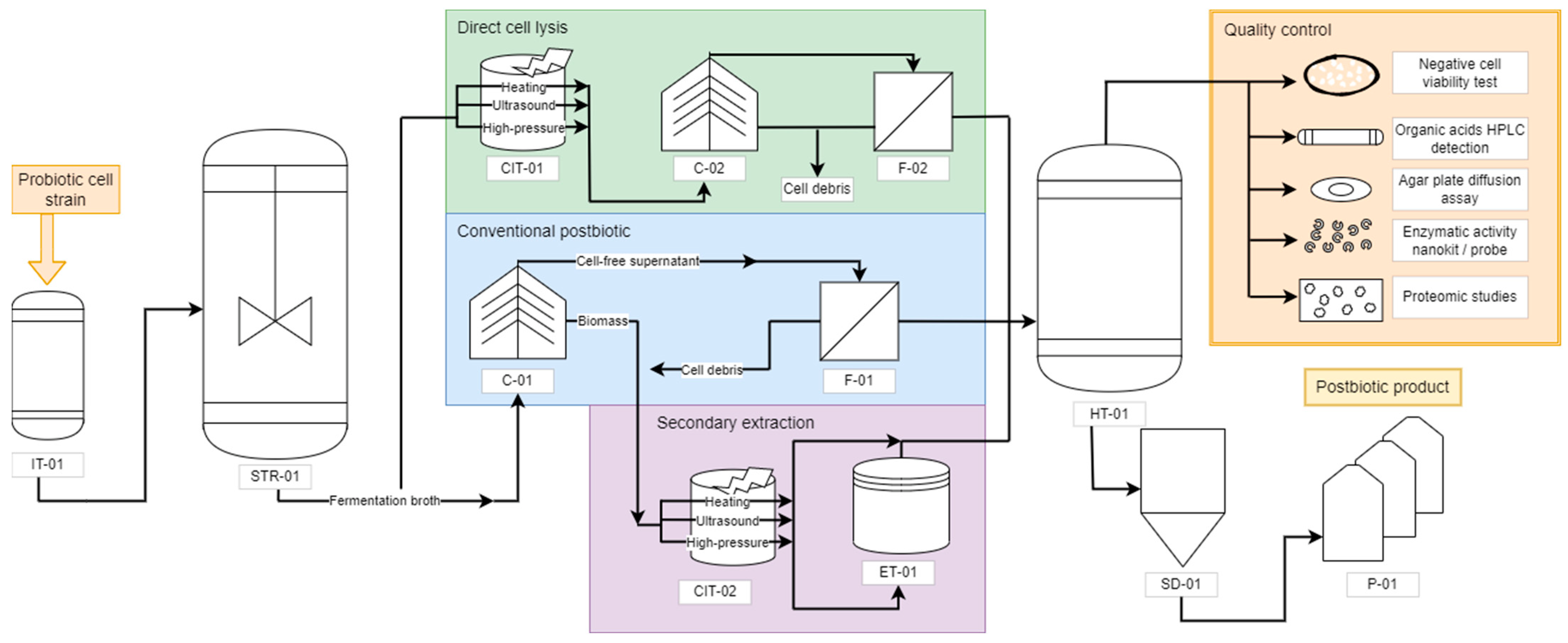
3. Postbiotics Formulation
4. Applications and Effects of Postbiotics in the Treatment of Skin Condition
| Strain | Derived Postbiotic | Product | Application | Reference |
|---|---|---|---|---|
| L. fermentum ATCC 9338 L. reuteri ATCC 23272 B. subtilis sp. Natto ATCC 15245 |
Lyophilization | Topical cold cream | Formulation of cold creams containing postbiotics to accelerate the wound healing process | [35] |
| L. acidophilus L-92 | Inactivated cell | Indeterminate | Postbiotic ingestion for the treatment of atopic dermatitis | [41] |
| B. coagulans MTCC 5856 | Cell-Free Supernatants | LactoSporin | Formulation of a cream containing (LactoSporin) for the treatment of Acne vulgaris | [37] |
| B. longum | Inactivated cell | Bifida Ferment Lysate | ||
| Indeterminate | Fragmented cells | Moisturizing Cream (CLS02021) | CLS02021 has shown beneficial effects on skin moisture and increased elasticity, wrinkle depth, and reduced pore size. The cream can be applied as an anti-aging skin treatment | [40] |
| L. kunkei | Bioactive peptides (PlnA) | TR-PRP plus-Celsi | Topical application for 3 months of a product containing a postbiotic resulted in a significant improvement in patients diagnosed with alopecia | [42] |
| L. Rhamnosus | Fragmented cells | Indeterminate | Administration of cell lysate in pediatric patients showed a marked improvement in eczema symptoms | [43] |
4.1. Alopecia
4.2. Acne Vulgaris
4.3. Atopic Dermatitis
4.4. Wound Healing
This entry is adapted from the peer-reviewed paper 10.3390/fermentation9030264
References
- Vallejo-Cordoba, B.; Castro-López, C.; García, H.S.; González-Córdova, A.F.; Hernández-Mendoza, A. Postbiotics and Paraprobiotics: A Review of Current Evidence and Emerging Trends. In Advances in Food and Nutrition Research; Academic Press Inc.: Cambridge, MA, USA, 2020; Volume 94, pp. 1–34. ISBN 9780128202180.
- Duarte, M.; Oliveira, A.L.; Oliveira, C.; Pintado, M.; Amaro, A.; Madureira, A.R. Current Postbiotics in the Cosmetic Market—An Update and Development Opportunities. Appl. Microbiol. Biotechnol. 2022, 106, 5879–5891.
- Cuevas-González, P.F.; Liceaga, A.M.; Aguilar-Toalá, J.E. Postbiotics and Paraprobiotics: From Concepts to Applications. Food Res. Int. 2020, 136, 109502.
- Martyniak, A.; Medyńska-Przęczek, A.; Wędrychowicz, A.; Skoczeń, S.; Tomasik, P.J. Prebiotics, Probiotics, Synbiotics, Paraprobiotics and Postbiotic Compounds in IBD. Biomolecules 2021, 11, 1903.
- Keshari, S.; Wang, Y.; Herr, D.R.; Wang, S.M.; Yang, W.C.; Chuang, T.H.; Chen, C.L.; Huang, C.M. Skin Cutibacterium acnes Mediates Fermentation to Suppress the Calcium Phosphate-induced Itching: A Butyric Acid Derivative with Potential for Uremic Pruritus. J. Clin. Med. 2020, 9, 312.
- Pereira, G.V.d.M.; de Oliveira Coelho, B.; Magalhães Júnior, A.I.; Thomaz-Soccol, V.; Soccol, C.R. How to Select a Probiotic? A Review and Update of Methods and Criteria. Biotechnol. Adv. 2018, 36, 2060–2076.
- Nam, Y.; Kim, J.H.; Baek, J.; Kim, W. Improvement of Cutaneous Wound Healing via Topical Application of Heat-Killed Lactococcus chungangensis Cau 1447 on Diabetic Mice. Nutrients 2021, 13, 2666.
- Ácsová, A.; Hojerová, J.; Martiniaková, S. Efficacy of Postbiotics against Free Radicals and UV Radiation. Chem. Pap. 2022, 76, 2357–2364.
- Kumar, D.; Kumar, S.; Kumar, A. Extraction and Characterization of Secondary Metabolites Produced by Bacteria Isolated from Industrial Wastewater. J. Water Process Eng. 2021, 40, 108.
- Tantratian, S.; Pradeamchai, M. Select a Protective Agent for Encapsulation of Lactobacillus plantarum. LWT 2020, 123, 109075.
- Willey, J.M.; Sherwood, L.M.; Woolverton, C.J. Prescott, Harley, and Klein’s Microbology, 7th ed.; Willey, J.M., Sherwood, L.M., Woolverton, C.J., Eds.; The McGraw-Hill Companies: New York, NY, USA, 2008; ISBN 978-0-07-299291-5.
- Tu, N.H.K.; Thien, P.V.M. Detection of N-Acetyl-D-Glucosamine in Hyaluronan by Thin Layer Chromatography. IFMBE Proc. 2013, 40 IFMBE, 174–177.
- Ibáñez, A.B.; Bauer, S. Analytical Method for the Determination of Organic Acids in Dilute Acid Pretreated Biomass Hydrolysate by Liquid Chromatography-Time-of-Flight Mass Spectrometry. Biotechnol. Biofuels 2014, 7, 145.
- Messi, P.; Bondi, M.; Sabia, C.; Battini, R.; Manicardi, G. Detection and Preliminary Characterization of a Bacteriocin (Plantaricin 35d) Produced by a Lactobacillus plantarum Strain. Artic. Int. J. Food Microbiol. 2001, 64, 193–198.
- Choeisoongnern, T.; Sivamaruthi, B.S.; Sirilun, S.; Peerajan, S.; Choiset, Y.; Rabesona, H.; Haertlé, T.; Chaiyasut, C. Screening and Identification of Bacteriocin-like Inhibitory Substances Producing Lactic Acid Bacteria from Fermented Products. Food Sci. Technol. 2019, 40, 571–579.
- Cicenia, A.; Scirocco, A.; Carabotti, M.; Pallotta, L.; Marignani, M.; Severi, C. Postbiotic Activities of Lactobacilli-Derived Factors. J. Clin. Gastroenterol. 2014, 48, S18–S22.
- Zhang, Y.; Wu, W.; Zhang, J.; Li, Z.; Ma, H.; Zhao, Z. Facile Method for Specifically Sensing Sphingomyelinase in Cells and Human Urine Based on a Ratiometric Fluorescent Nanoliposome Probe. Anal. Chem. 2021, 93, 11775–11784.
- MacFarlane, B. Common Cosmetic Ingredients: Chemistry, Actions, Safety and Products. In Cosmetic Formulation: Principles and Practice; Benson, H.A.E., Roberts, M.S., Leite-Silva, V.R., Walters, K.A., Eds.; CRC Press: Boca Raton, FL, USA, 2019; pp. 105–118. ISBN 9781482235395.
- Goyal, N.; Jerold, F. Biocosmetics: Technological Advances and Future Outlook. Environ. Sci. Pollut. Res. 2021, 1–22.
- Lourith, N.; Kanlayavattanakul, M. Natural Surfactants Used in Cosmetics: Glycolipids. Int. J. Cosmet. Sci. 2009, 31, 255–261.
- Nowak, K.; Jabłońska, E.; Ratajczak-Wrona, W. Controversy around Parabens: Alternative Strategies for Preservative Use in Cosmetics and Personal Care Products. Environ. Res. 2021, 198, 110488.
- Lane, M.E. Skin Penetration Enhancers. Int. J. Pharm. 2013, 447, 12–21.
- Mavranezouli, I.; Daly, C.H.; Welton, N.J.; Deshpande, S.; Berg, L.; Bromham, N.; Arnold, S.; Phillippo, D.M.; Wilcock, J.; Xu, J.; et al. A Systematic Review and Network Meta-Analysis of Topical Pharmacological, Oral Pharmacological, Physical and Combined Treatments for Acne Vulgaris. Br. J. Dermatol. 2022, 187, 639–649.
- Bakshi, H.; Nagpal, M.; Singh, M.; Dhingra, G.A.; Aggarwal, G. Treatment of Psoriasis: A Comprehensive Review of Entire Therapies. Curr. Drug Saf. 2020, 15, 82–104.
- Calabrese, G.; Licata, G.; Gambardella, A.; De Rosa, A.; Alfano, R.; Argenziano, G. Topical and Conventional Systemic Treatments in Atopic Dermatitis: Have They Gone Out of Fashion? Dermatol. Pract. Concept. 2022, 12, e2022155.
- Otlewska, A.; Baran, W.; Batycka-Baran, A. Adverse Events Related to Topical Drug Treatments for Acne Vulgaris. Expert Opin. Drug Saf. 2020, 19, 513–521.
- Frantz, T.; Wright, E.G.; Balogh, E.A.; Cline, A.; Adler-Neal, A.L.; Feldman, S.R. Topical and Oral Therapies for Childhood Atopic Dermatitis and Plaque Psoriasis. Children 2019, 6, 125.
- Schmid, B.; Künstner, A.; Fähnrich, A.; Bersuch, E.; Schmid-Grendelmeier, P.; Busch, H.; Glatz, M.; Bosshard, P.P. Dysbiosis of Skin Microbiota with Increased Fungal Diversity Is Associated with Severity of Disease in Atopic Dermatitis. J. Eur. Acad. Dermatol. Venereol. 2022, 36, 1811–1819.
- Moosbrugger-Martinz, V.; Hackl, H.; Gruber, R.; Pilecky, M.; Knabl, L.; Orth-Höller, D.; Dubrac, S. Initial Evidence of Distinguishable Bacterial and Fungal Dysbiosis in the Skin of Patients with Atopic Dermatitis or Netherton Syndrome. J. Investig. Dermatol. 2021, 141, 114–123.
- Dréno, B.; Dagnelie, M.A.; Khammari, A.; Corvec, S. The Skin Microbiome: A New Actor in Inflammatory Acne. Am. J. Clin. Dermatol. 2020, 21, 18–24.
- Li, C.X.; You, Z.X.; Lin, Y.X.; Liu, H.Y.; Su, J. Skin Microbiome Differences Relate to the Grade of Acne Vulgaris. J. Dermatol. 2019, 46, 787–790.
- Stehlikova, Z.; Kostovcik, M.; Kostovcikova, K.; Kverka, M.; Juzlova, K.; Rob, F.; Hercogova, J.; Bohac, P.; Pinto, Y.; Uzan, A.; et al. Dysbiosis of Skin Microbiota in Psoriatic Patients: Co-Occurrence of Fungal and Bacterial Communities. Front. Microbiol. 2019, 10, 438.
- Visser, M.J.E.; Kell, D.B.; Pretorius, E. Bacterial Dysbiosis and Translocation in Psoriasis Vulgaris. Front. Cell Infect. Microbiol. 2019, 9, 7.
- Salminen, S.; Collado, M.C.; Endo, A.; Hill, C.; Lebeer, S.; Quigley, E.M.M.; Sanders, M.E.; Shamir, R.; Swann, J.R.; Szajewska, H.; et al. The International Scientific Association of Probiotics and Prebiotics (ISAPP) Consensus Statement on the Definition and Scope of Postbiotics. Nat. Rev. Gastroenterol. Hepatol. 2021, 18, 649–667.
- Golkar, N.; Ashoori, Y.; Heidari, R.; Omidifar, N.; Abootalebi, S.N.; Mohkam, M.; Gholami, A. A Novel Effective Formulation of Bioactive Compounds for Wound Healing: Preparation, in Vivo Characterization, and Comparison of Various Postbiotics Cold Creams in a Rat Model. Evid.-Based Complement. Altern. Med. 2021, 2021, 8577116.
- Majeed, M.; Nagabhushanam, K.; Arumugam, S.; Ali, F. Method of Producing Partially Purified Extracellular Metabolite Products from Bacillus Coagulans and Biological Applications Thereof. U.S. Patent 9,596,861, 21 March 2017.
- Majeed, M.; Majeed, S.; Nagabhushanam, K.; Mundkur, L.; Rajalakshmi, H.R.; Shah, K.; Beede, K. Novel Topical Application of a Postbiotic, Lactosporin®, in Mild to Moderate Acne: A Randomized, Comparative Clinical Study to Evaluate Its Efficacy, Tolerability and Safety. Cosmetics 2020, 7, 70.
- Łętocha, A.; Miastkowska, M.; Sikora, E. Preparation and Characteristics of Alginate Microparticles for Food, Pharmaceutical and Cosmetic Applications. Polymers 2022, 14, 3834.
- Ashoori, Y.; Mohkam, M.; Heidari, R.; Abootalebi, S.N.; Mousavi, S.M.; Hashemi, S.A.; Golkar, N.; Gholami, A. Development and in Vivo Characterization of Probiotic Lysate-Treated Chitosan Nanogel as a Novel Biocompatible Formulation for Wound Healing. Biomed Res. Int. 2020, 2020, 8868618.
- Catic, T.; Pehlivanovic, B.; Pljakic, N.; Balicevac, A. The Moisturizing Efficacy of a Proprietary Dermo-Cosmetic Product (CLS02021) versus Placebo in a 4-Week Application Period. Med. Arch. 2022, 76, 108–114.
- Inoue, Y.; Kambara, T.; Murata, N.; Komori-Yamaguchi, J.; Matsukura, S.; Takahashi, Y.; Ikezawa, Z.; Aihara, M. Effects of Oral Administration of Lactobacillus acidophilus L-92 on the Symptoms and Serum Cytokines of Atopic Dermatitis in Japanese Adults: A Double-Blind, Randomized, Clinical Trial. Int. Arch. Allergy Immunol. 2014, 165, 247–254.
- Rinaldi, F.; Trink, A.; Pinto, D. Efficacy of Postbiotics in a PRP-Like Cosmetic Product for the Treatment of Alopecia Area Celsi: A Randomized Double-Blinded Parallel-Group Study. Dermatol. Ther. 2020, 10, 483–493.
- Hoang, B.X.; Shaw, G.; Pham, P.; Levine, S.A. Lactobacillus rhamnosus Cell Lysate in the Management of Resistant Childhood Atopic Eczema. Inflamm. Allergy-Drug Targets 2010, 9, 192–196.
- Darwin, E.; Hirt, P.; Fertig, R.; Doliner, B.; Delcanto, G.; Jimenez, J. Alopecia Areata: Review of Epidemiology, Clinical Features, Pathogenesis, and New Treatment Options. Int. J. Trichology 2018, 10, 51–60.
- Segal-Engelchin, D.; Shvarts, S. Does Severity of Hair Loss Matter? Factors Associated with Mental Health Outcomes in Women Irradiated for Tinea Capitis in Childhood. Int. J. Environ. Res. Public Health 2020, 17, 7388.
- Chiang, K.S.; Mesinkovska, N.A.; Piliang, M.P.; Bergfeld, W.F. Clinical Efficacy of Diphenylcyclopropenone in Alopecia Areata: Retrospective Data Analysis of 50 Patients. J. Investig. Dermatol. Symp. Proc. 2015, 17, 50–55.
- Gupta, A.K.; Carviel, J.; Abramovits, W. Treating Alopecia Areata: Current Practices versus New Directions. Am. J. Clin. Dermatol. 2017, 18, 67–75.
- Jahn-Bassler, K.; Bauer, W.M.; Karlhofer, F.; Vossen, M.G.; Stingl, G. Sequential High- and Low-Dose Systemic Corticosteroid Therapy for Severe Childhood Alopecia Areata. JDDG—J. Ger. Soc. Dermatol. 2017, 15, 42–47.
- Darwin, E.; Arora, H.; Hirt, P.A.; Wikramanayake, T.C.; Jimenez, J.J. A Review of Monochromatic Light Devices for the Treatment of Alopecia Areata. Lasers Med. Sci. 2018, 33, 435–444.
- Damsky, W.; King, B.A. JAK Inhibitors in Dermatology: The Promise of a New Drug Class. J. Am. Acad. Dermatol. 2017, 76, 736–744.
- Garg, S.; Manchanda, S. Platelet-Rich Plasma-an “Elixir” for Treatment of Alopecia: Personal Experience on 117 Patients with Review of Literature. Stem. Cell Investig. 2017, 4, 64.
- Rinaldi, F.; Marzani, B.; Pinto, D.; Sorbellini, E. Randomized Controlled Trial on a PRP-like Cosmetic, Biomimetic Peptides Based, for the Treatment of Alopecia Areata. J. Dermatol. Treat. 2019, 30, 588–593.
- Kimura-Ueki, M.; Oda, Y.; Oki, J.; Komi-Kuramochi, A.; Honda, E.; Asada, M.; Suzuki, M.; Imamura, T. Hair Cycle Resting Phase Is Regulated by Cyclic Epithelial FGF18 Signaling. J. Investig. Dermatol. 2012, 132, 1338–1345.
- Kawano, M.; Komi-Kuramochi, A.; Asada, M.; Suzuki, M.; Oki, J.; Jiang, J.; Imamura, T. Comprehensive Analysis of FGF and FGFR Expression in Skin: FGF18 Is Highly Expressed in Hair Follicles and Capable of Inducing Anagen from Telogen Stage Hair Follicles. J. Investig. Dermatol. 2005, 124, 877–885.
- Marx, R.E. Platelet-Rich Plasma: Evidence to Support Its Use. J. Oral Maxillofac. Surg. 2004, 62, 489–496.
- Giordano, S.; Romeo, M.; di Summa, P.; Salval, A.; Lankinen, P. A Meta-Analysis on Evidence of Platelet-Rich Plasma for Androgenetic Alopecia. Int. J. Trichology 2018, 10, 1–10.
- Dohan Ehrenfest, D.M.; Rasmusson, L.; Albrektsson, T. Classification of Platelet Concentrates: From Pure Platelet-Rich Plasma (P-PRP) to Leucocyte- and Platelet-Rich Fibrin (L-PRF). Trends Biotechnol. 2009, 27, 158–167.
- Marzani, B.; Pinto, D.; Minervini, F.; Calasso, M.; di Cagno, R.; Giuliani, G.; Gobbetti, M.; de Angelis, M. The Antimicrobial Peptide Pheromone Plantaricin A Increases Antioxidant Defenses of Human Keratinocytes and Modulates the Expression of Filaggrin, Involucrin, β-Defensin 2 and Tumor Necrosis Factor-α Genes. Exp. Dermatol. 2012, 21, 665–671.
- Pinto, D.; Marzani, B.; Minervini, F.; Calasso, M.; Giuliani, G.; Gobbetti, M.; de Angelis, M. Plantaricin A Synthesized by Lactobacillus plantarum Induces in Vitro Proliferation and Migration of Human Keratinocytes and Increases the Expression of TGF-Β1, FGF7, VEGF-A and IL-8 Genes. Peptides 2011, 32, 1815–1824.
- di Cagno, R.; Filannino, P.; Cantatore, V.; Gobbetti, M. Novel Solid-State Fermentation of Bee-Collected Pollen Emulating the Natural Fermentation Process of Bee Bread. Food Microbiol. 2019, 82, 218–230.
- Williams, H.C.; Dellavalle, R.P.; Garner, S. Acne Vulgaris. Lancet 2012, 379, 361–372.
- Bagatin, E.; Timpano, D.L.; Guadanhim, L.R.d.S.; Nogueira, V.M.A.; Terzian, L.R.; Steiner, D.; Florez, M. Acne Vulgaris: Prevalence and Clinical Forms in Adolescents from São Paulo, Brazil. An. Bras. Dermatol. 2014, 89, 428–435.
- Tan, J.K.L.; Bhate, K. A Global Perspective on the Epidemiology of Acne. Br. J. Dermatol. 2015, 172, 3–12.
- Toyoda, M.; Morohashi, M. Pathogenesis of Acne. Med. Electron. Microsc. 2001, 34, 29–40.
- Chilicka, K.; Rusztowicz, M.; Rogowska, A.M.; Szyguła, R.; Asanova, B.; Nowicka, D. Efficacy of Hydrogen Purification and Cosmetic Acids in the Treatment of Acne Vulgaris: A Preliminary Report. J. Clin. Med. 2022, 11, 6269.
- Chilicka, K.; Rogowska, A.M.; Szyguła, R.; Rusztowicz, M.; Nowicka, D. Efficacy of Oxybrasion in the Treatment of Acne Vulgaris: A Preliminary Report. J. Clin. Med. 2022, 11, 3824.
- See, J.A.; Goh, C.L.; Hayashi, N.; Suh, D.H.; Casintahan, F.A. Optimizing the Use of Topical Retinoids in Asian Acne Patients. J. Dermatol. 2018, 45, 522–528.
- Fluhr, J.W.; Degitz, K. Antibiotika, Azelainsäure Und Benzoylperoxid in Der Topische Aknetherapie. JDDG—J. Ger. Soc. Dermatol. 2010, 8, S24–S30.
- Chng, K.R.; Tay, A.S.L.; Li, C.; Ng, A.H.Q.; Wang, J.; Suri, B.K.; Matta, S.A.; McGovern, N.; Janela, B.; Wong, X.F.C.C.; et al. Whole Metagenome Profiling Reveals Skin Microbiome-Dependent Susceptibility to Atopic Dermatitis Flare. Nat. Microbiol. 2016, 1, 16106.
- Nast, A.; Dréno, B.; Bettoli, V.; Degitz, K.; Erdmann, R.; Finlay, A.Y.; Ganceviciene, R.; Haedersdal, M.; Layton, A.; López-Estebaranz, J.L.; et al. European Evidence-Based (S3) Guidelines for the Treatment of Acne. J. Eur. Acad. Dermatol. Venereol. 2012, 26, 1–29.
- Strauss, J.S.; Krowchuk, D.P.; Leyden, J.J.; Lucky, A.W.; Shalita, A.R.; Siegfried, E.C.; Thiboutot, D.M.; van Voorhees, A.S.; Beutner, K.A.; Sieck, C.K.; et al. Guidelines of Care for Acne Vulgaris Management. J. Am. Acad. Dermatol. 2007, 56, 651–663.
- Simonart, T. Newer Approaches to the Treatment of Acne Vulgaris. Am. J. Clin. Dermatol. 2012, 13, 357–364.
- Majeed, M.; Majeed, S.; Nagabhushanam, K.; Lawrence, L.; Arumugam, S.; Mundkur, L. Skin Protective Activity of Lactosporin-the Extracellular Metabolite from Bacillus coagulans Mtcc 5856. Cosmetics 2020, 7, 76.
- Lambrechts, I.A.; de Canha, M.N.; Lall, N. Exploiting Medicinal Plants as Possible Treatments for Acne Vulgaris. In Medicinal Plants for Holistic Health and Well-Being; Elsevier: Amsterdam, The Netherlands, 2017; pp. 117–143. ISBN 9780128124758.
- Chung, H.J.; Lee, H.; Kim, M.; Lee, J.W.; Saeed, M.; Lee, H.; Jung, S.H.; Shim, J.J.; Lee, J.L.; Heo, K.; et al. Development and Metabolic Profiling of a Postbiotic Complex Exhibiting Antibacterial Activity against Skin Microorganisms and Anti-Inflammatory Effect on Human Keratinocytes. Food Sci. Biotechnol. 2022, 31, 1325–1334.
- Kim, J.; Kim, B.E.; Leung, D.Y.M. Pathophysiology of Atopic Dermatitis: Clinical Implications. Allergy Asthma Proc. 2019, 40, 84–92.
- Kim, B.E.; Leung, D.Y.M. Epidermal Barrier in Atopic Dermatitis. Allergy Asthma Immunol. Res. 2012, 4, 12–16.
- Silverberg, J.I. Public Health Burden and Epidemiology of Atopic Dermatitis. Dermatol. Clin. 2017, 35, 283–289.
- Guttman-Yassky, E.; Suárez-Fariñas, M.; Chiricozzi, A.; Nograles, K.E.; Shemer, A.; Fuentes-Duculan, J.; Cardinale, I.; Lin, P.; Bergman, R.; Bowcock, A.M.; et al. Broad Defects in Epidermal Cornification in Atopic Dermatitis Identified through Genomic Analysis. J. Allergy Clin. Immunol. 2009, 124, 1235–1244.e58.
- Blicharz, L.; Rudnicka, L.; Czuwara, J.; Waśkiel-Burnat, A.; Goldust, M.; Olszewska, M.; Samochocki, Z. The Influence of Microbiome Dysbiosis and Bacterial Biofilms on Epidermal Barrier Function in Atopic Dermatitis—An Update. Int. J. Mol. Sci. 2021, 22, 8403.
- Brunner, P.M.; Leung, D.Y.M.; Guttman-Yassky, E. Immunologic, Microbial, and Epithelial Interactions in Atopic Dermatitis. Ann. Allergy Asthma Immunol. 2018, 120, 34–41.
- Langan, S.M.; Irvine, A.D.; Weidinger, S. Atopic Dermatitis. Lancet 2020, 396, 345–360.
- Liang, Y.; Chang, C.; Lu, Q. The Genetics and Epigenetics of Atopic Dermatitis—Filaggrin and Other Polymorphisms. Clin. Rev. Allergy Immunol. 2016, 51, 315–328.
- Renert-Yuval, Y.; Guttman-Yassky, E. New Treatments for Atopic Dermatitis Targeting beyond IL-4/IL-13 Cytokines. Ann. Allergy Asthma Immunol. 2020, 124, 28–35.
- Wollenberg, A.; Oranje, A.; Deleuran, M.; Simon, D.; Szalai, Z.; Kunz, B.; Svensson, A.; Barbarot, S.; von Kobyletzki, L.; Taieb, A.; et al. ETFAD/EADV Eczema Task Force 2015 Position Paper on Diagnosis and Treatment of Atopic Dermatitis in Adult and Paediatric Patients. J. Eur. Acad. Dermatol. Venereol. 2016, 30, 729–747.
- Torii, S.; Torii, A.; Itoh, K.; Urisu, A.; Terada, A.; Fujisawa, T.; Yamada, K.; Suzuki, H.; Ishida, Y.; Nakamura, F.; et al. Effects of Oral Administration of Lactobacillus acidophilus L-92 on the Symptoms and Serum Markers of Atopic Dermatitis in Children. Int. Arch. Allergy Immunol. 2011, 154, 236–245.
- Ishida, Y.; Nakamura, F.; Kanzato, H.; Sawada, D.; Yamamoto, N.; Kagata, H.; Oh-Ida, M.; Takeuchi, H.; Fujiwara, S. Effect of Milk Fermented with Lactobacillus acidophilus Strain L-92 on Symptoms of Japanese Cedar Pollen Allergy: A Randomized Placebo-Controlled Trial. Biosci. Biotechnol. Biochem. 2005, 69, 1652–1660.
- Kuwana, R.; Yamamoto, N. Increases in GroES and GroEL from Lactobacillus acidophilus L-92 in Response to a Decrease in Medium PH, and Changes in Cytokine Release from Splenocytes: Transcriptome and Proteome Analyses. J. Biosci. Bioeng. 2012, 114, 9–16.
- Shah, M.M.; Miyamoto, Y.; Yamada, Y.; Yamashita, H.; Tanaka, H.; Ezaki, T.; Nagai, H.; Inagaki, N. Orally Supplemented Lactobacillus acidophilus Strain L-92 Inhibits Passive and Active Cutaneous Anaphylaxis as Well as 2,4-Dinitroflurobenzene and Mite Fecal Antigen Induced Atopic Dermatitis-like Skin Lesions in Mice. Microbiol. Immunol. 2010, 54, 523–533.
- Kanzato, H.; Fujiwara, S.; Ise, W.; Kaminogawa, S.; Sato, R.; Hachimura, S. Lactobacillus acidophilus Strain L-92 Induces Apoptosis of Antigen-Stimulated T Cells by Modulating Dendritic Cell Function. Immunobiology 2008, 213, 399–408.
- Shah, M.M.; Saio, M.; Yamashita, H.; Tanaka, H.; Takami, T.; Ezaki, T.; Inagaki, N. Lactobacillus acidophilus Strain L-92 Induces CD4+CD25+Foxp3+ Regulatory T Cells and Suppresses Allergic Contact Dermatitis. Biol. Pharm. Bull. 2012, 35, 612–616.
- Tsiouris, C.G.; Tsiouri, M.G. Human Microflora, Probiotics and Wound Healing. Wound Med. 2017, 19, 33–38.
- Yu, R.; Zhang, H.; Guo, B. Conductive Biomaterials as Bioactive Wound Dressing for Wound Healing and Skin Tissue Engineering. Nanomicro Lett. 2022, 14, 1.
- Holmes, C.J.; Plichta, J.K.; Gamelli, R.L.; Radek, K.A. Dynamic Role of Host Stress Responses in Modulating the Cutaneous Microbiome: Implications for Wound Healing and Infection. Adv. Wound Care 2015, 4, 24–37.
- Percival, S.L.; McCarty, S.M.; Lipsky, B. Biofilms and Wounds: An Overview of the Evidence. Adv. Wound Care 2015, 4, 373–381.
