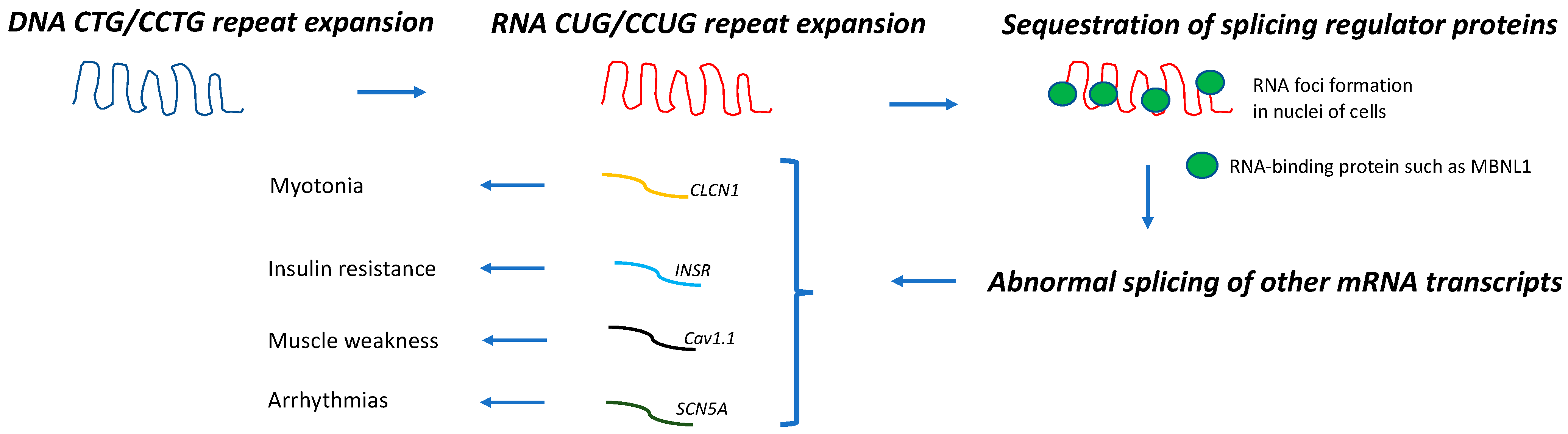
Myotonic dystrophy (DM) is the most common muscular dystrophy in adults. Dominantly inherited CTG and CCTG repeat expansions in dystrophia myotonica protein kinase (DMPK) and cellular nucleic acid-binding protein (CNBP) genes cause DM type 1 (DM1) and 2 (DM2), respectively. These genetic defects lead to the abnormal splicing of different mRNA transcripts, which are thought to be responsible for the multiorgan involvement of these diseases. Cancer frequency in patients with DM appears to be higher than in the general population or non-DM muscular dystrophy cohorts.
- cancer
- myotonic dystrophy
- repeat expansion
1. Introduction
Myopathies and Cancer
2. Carcinogenesis in DM
Although DM1 and DM2 appear to share the same pathogenic mechanism (Figure 1), the association between DM and cancer and its molecular basis has been mostly studied in DM1.
There are two types of studies that link DM1 and carcinogenesis: one type includes those that investigated the cancer risk and predisposition to specific cancer types in predominantly DM1 cohorts by using the general population (or healthy subjects) or a non-muscular dystrophy cohort as a comparator group, and the other type of study includes those that addressed the potential molecular mechanisms underlying DM carcinogenesis. Within the latter group, some investigated the length of the pathogenic repeat expansion in the tumoral tissue of patients with DM1, and a few of those demonstrated that a longer CTG expansion was present in tumoral cells when compared with non-tumoral cells from the same individual [14][15][16][17].
Table 1 summarizes the main findings from the studies that reported an increased risk of specific cancer types in the DM (mostly DM1) population [18][19][20][21][22][23][24][25].
| Reference | N | Thyroid | Cutaneous Melanoma | Pancreas | Colon | Endometrium | Ovary | Prostate | Testes | Brain | Eye |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Gadalla et al., 2011 [19] | 1658 | ↑SIR 7.1 (1.8–19.3) |
––– | ↑SIR 3.2 (1.0–7.6) |
↑SIR 2.9 (1.5–5.1) |
↑SIR 7.6 (4.0–13.2) |
↑SIR 5.2 (2.3–10.2) |
SIR 0.7 (0.2–1.9) |
SIR 1.4 (0.1–6.8) |
↑SIR 5.3 (2.3–10.4) |
↑SIR 12.0 (2.0–39.6) |
| Win et al., 2012 [20] | 307 | ↑SIR 5.54 (1.80–12.93) |
SIR 2.05 (0.42–6.00) |
––– | SIR 1.09 (0.22–3.18) |
SIR 1.07 (0.03–5.98) |
SIR 1.66 (0.04–9.25) |
SIR 2.21 (0.95–4.35) |
SIR 5.09 (0.62–18.38) |
SIR 1.54 (0.04–8.57) |
↑SIR 27.54 (3.34–99.49) |
| Mohamed et al., 2013 [22] | 109 | ––– | RR 7.1 (0.8–25.8) |
––– | RR 5.0 (0.6–18.2) |
↑RR 21.7 (2.4–78.5) |
RR 9.3 (0.1–51.5) |
––– | ––– | ––– | ––– |
| Abbott et al., 2016 [21] | 281 | RR 3.78 (0.67–13.65) |
RR 0.89 (0.0–4.20) |
––– | RR 2.15 (0.11–11.99) |
↑RR 6.98 (1.24–25.22) |
RR 1.43 (0.25–5.16) |
↑RR 10.74 (1.91–38.79) |
––– | ––– | |
| Fernandez-Torron et al., 2016 [18] | 424 | ↑SIR 23.33 (9.38–48.08) |
SIR 1.72 (0.04–9.61) |
––– | SIR 2.06 (0.94–3.92) |
↑SIR 6.86 (2.23–16.02) |
↑SIR 8.33 (1.72–24.31) |
SIR 0.46 (0.06–1.67) |
SIR 14.25 (0.35–79.6) |
↑SIR 9.80 (3.18–22.88) |
|
| Wang et al., 2018 [23] | 1061 | ––– | HR: 2.40 (0.56–10.31) |
––– | ––– | ––– | ––– | ––– | ––– | ––– | ––– |
| Alsaggaf et al., 2018 [24] | 927 | ↑HR 15.93 (2.45–103.64) |
↑HR 5.98 (1.24–28.79) |
↑HR 2.96 (0.30–29.38) |
HR 1.82 (0.32–10.31) |
↑HR 14.88 (2.14–103.67) |
––– | ––– | HR 4.99 (0.46–53.78) |
––– | |
| HR 1.81 (0.21–15.23) |
HR 1.12 (0.37–3.45) |
HR 0.32 (0.04–2.62) |
HR 2.03 (0.23–17.68) |
||||||||
| Emparanza et al., 2018 [25] | 2779 (meta-analysis) |
↑pSIR = 8.52 (3.62–20.1) |
↑pSIR = 2.45 (1.31–4.58) |
––– | ↑pSIR = 2.2 (1.39–3.49) |
↑pSIR = 7.48 (4.72–11.8) |
↑pSIR = 5.56 (2.99–10.3) |
––– | ↑pSIR = 5.95 (2.34–15.1) |
––– | ––– |
This entry is adapted from the peer-reviewed paper 10.3390/jcm12051939
References
- Harper, P.S. Myotonic Dystrophy, 3rd ed.; W.B. Saunders: London, UK; New York, NY, USA, 2001.
- Johnson, N.E.; Butterfield, R.J.; Mayne, K.; Newcomb, T.; Imburgia, C.; Dunn, D.; Duval, B.; Feldkamp, M.L.; Weiss, R.B. Population Based Prevalence of Myotonic Dystrophy Type 1 Using Genetic Analysis of State-wide Blood Screening Program. Neurology 2021, 96, e1045–e1053.
- Fu, Y.H.; Pizzuti, A.; Fenwick, R.G.; King, J.; Rajnarayan, S.; Dunne, P.W.; Dubel, J.; Nasser, G.A.; Ashizawa, T.; de Jong, P.; et al. An Unstable Triplet Repeat in a Gene Related to Myotonic Muscular Dystrophy. Science 1992, 255, 1256–1258.
- Suominen, T.; Bachinski, L.L.; Auvinen, S.; Hackman, P.; Baggerly, K.A.; Angelini, C.; Peltonen, L.; Krahe, R.; Udd, B. Population frequency of myotonic dystrophy: Higher than expected frequency of myotonic dystrophy type 2 (DM2) mutation in Finland. Eur. J. Hum. Genet. 2011, 19, 776–782.
- Liquori, C.L.; Ricker, K.; Moseley, M.L.; Jacobsen, J.F.; Kress, W.; Naylor, S.L.; Day, J.W.; Ranum, L.P.W. Myotonic Dystrophy Type 2 Caused by a CCTG Expansion in Intron 1 of ZNF9. Science 2001, 293, 864–867.
- Brook, J.D.; McCurrach, M.E.; Harley, H.G.; Buckler, A.J.; Church, D.; Aburatani, H.; Hunter, K.; Stanton, V.P.; Thirion, J.-P.; Hudson, T.; et al. Molecular basis of myotonic dystrophy: Expansion of a trinucleotide (CTG) repeat at the 3′ end of a transcript encoding a protein kinase family member. Cell 1992, 68, 799–808.
- Udd, B.; Krahe, R. The myotonic dystrophies: Molecular, clinical, and therapeutic challenges. Lancet Neurol. 2012, 11, 891–905.
- Savkur, R.S.; Philips, A.V.; Cooper, T.A. Aberrant regulation of insulin receptor alternative splicing is associated with insulin resistance in myotonic dystrophy. Nat. Genet. 2001, 29, 40–47.
- Freyermuth, F.; Rau, F.; Kokunai, Y.; Linke, T.; Sellier, C.; Nakamori, M.; Kino, Y.; Arandel, L.; Jollet, A.; Thibault, C.; et al. Splicing misregulation of SCN5A contributes to cardiac-conduction delay and heart arrhythmia in myotonic dystrophy. Nat. Commun. 2016, 7, 11067.
- Tang, Z.Z.; Yarotskyy, V.; Wei, L.; Sobczak, K.; Nakamori, M.; Eichinger, K.; Moxley, R.T.; Dirksen, R.T.; Thornton, C.A. Muscle weakness in myotonic dystrophy associated with misregulated splicing and altered gating of CaV1.1 calcium channel. Hum. Mol. Genet. 2012, 21, 1312–1324.
- Koehorst, E.; Ballester-Lopez, A.; Arechavala-Gomeza, V.; Martínez-Piñeiro, A.; Nogales-Gadea, G. The Biomarker Potential of miRNAs in Myotonic Dystrophy Type I. J. Clin. Med. 2020, 9, 3939.
- Gonzalez-Perez, P.; Amato, A. Myopathies and Cancer. In Effects of Cancer Treatment on the Nervous System, 1st ed.; Grisold, W., Ed.; Cambridge Scholars Publishing: Newcastle upon Tyne, UK, 2021; Volume 2, pp. 344–368.
- Tanboon, J.; Nishino, I. Update on dermatomyositis. Curr. Opin. Neurol. 2022, 35, 611–621.
- Osanai, R.; Kinoshita, M.; Hirose, K.; Homma, T.; Kawabata, I. CTG triplet repeat expansion in a laryngeal carcinoma from a patient with myotonic dystrophy. Muscle Nerve 2000, 23, 804–806.
- Bañuls, J.; Botella, R.; Palau, F.; Ramón, R.; Díaz, C.; Payá, A.; Carnero, L.; Vergara, G. Tissue and tumor mosaicism of the myotonin protein kinase gene trinucleotide repeat in a patient with multiple basal cell carcinomas associated with myotonic dystrophy. J. Am. Acad. Dermatol. 2004, 50 (Suppl. 2), S1–S3.
- Jinnai, K.; Sugio, T.; Mitani, M.; Hashimoto, K.; Takahashi, K. Elongation of (CTG)n repeats in myotonic dystrophy protein kinase gene in tumors associated with myotonic dystrophy patients. Muscle Nerve 1999, 22, 1271–1274.
- Kinoshita, M.; Igarashi, A.; Komori, T.; Tamura, H.; Hayashi, M.; Kinoshita, K.; Deguchi, T.; Hirose, K. Differences in CTG triplet repeat expansions in an ovarian cancer and cyst from a patient with myotonic dystrophy. Muscle Nerve 1997, 20, 622–624.
- Fernández-Torrón, R.; García-Puga, M.; Emparanza, J.I.; Maneiro, M.; Cobo, A.M.; Poza, J.J.; Espinal, J.B.; Zulaica, M.; Ruiz, I.; Martorell, L.; et al. Cancer risk in DM1 is sex-related and linked to miRNA-200/141 downregulation. Neurology 2016, 87, 1250–1257.
- Gadalla, S.M.; Lund, M.; Pfeiffer, R.M.; Gørtz, S.; Mueller, C.M.; Moxley, R.T.; Kristinsson, S.Y.; Björkholm, M.; Shebl, F.M.; Hilbert, J.E.; et al. Cancer Risk Among Patients With Myotonic Muscular Dystrophy. JAMA 2011, 306, 2480–2486.
- Win, A.K.; Perattur, P.G.; Pulido, J.S.; Pulido, C.M.; Lindor, N.M. Increased Cancer Risks in Myotonic Dystrophy. Mayo Clin. Proc. 2012, 87, 130–135.
- Abbott, D.; Johnson, N.E.; Cannon-Albright, L.A. A population-based survey of risk for cancer in individuals diagnosed with myotonic dystrophy. Muscle Nerve 2016, 54, 783–785.
- Mohamed, S.; Pruna, L.; Kaminsky, P. Increasing risk of tumors in myotonic dystrophy type 1. Presse Med. 2013, 42 Pt 1, e281–e284.
- Wang, Y.; Pfeiffer, R.M.; AlSaggaf, R.; Meeraus, W.; Gage, J.C.; Anderson, L.; Bremer, R.C.; Nikolenko, N.; Lochmuller, H.; Greene, M.H.; et al. Risk of skin cancer among patients with myotonic dystrophy type 1 based on primary care physician data from the U.K. Clinical Practice Research Datalink. Int. J. Cancer 2017, 142, 1174–1181.
- Alsaggaf, R.; George, D.M.M.S.; Zhan, M.; Pfeiffer, R.M.; Wang, Y.; Wagner, K.R.; Greene, M.H.; Amr, S.; Gadalla, S.M. Cancer Risk in Myotonic Dystrophy Type I: Evidence of a Role for Disease Severity. JNCI Cancer Spectr. 2018, 2, pky052.
- Emparanza, J.I.; de Munain, A.L.; Greene, M.H.; Matheu, A.; Fernández-Torrón, R.; Gadalla, S.M. Cancer phenotype in myotonic dystrophy patients: Results from a meta-analysis. Muscle Nerve 2018, 58, 517–522.
