Adverse pregnancy outcomes are considered significant health risks for pregnant women and their offspring during pregnancy and throughout their lifespan. Maternal dietary intake of polyphenolic antioxidants is linked to a reduced risk of maternal obesity and cardio-metabolic disorders, positively affecting both the fetus and offspring. In this work, we will gather and critically appraise the current literature highlighting the effect/s of the naturally occurring polyphenol antioxidant resveratrol on oxidative stress, inflammation, and other molecular and physiological phenomena associated with pregnancy and pregnancy conditions, such as gestational diabetes, preeclampsia, and preterm labor.
- resveratrol
- pregnancy
- placenta
1. Resveratrol’s Mechanism of Action during Pregnancy Complications
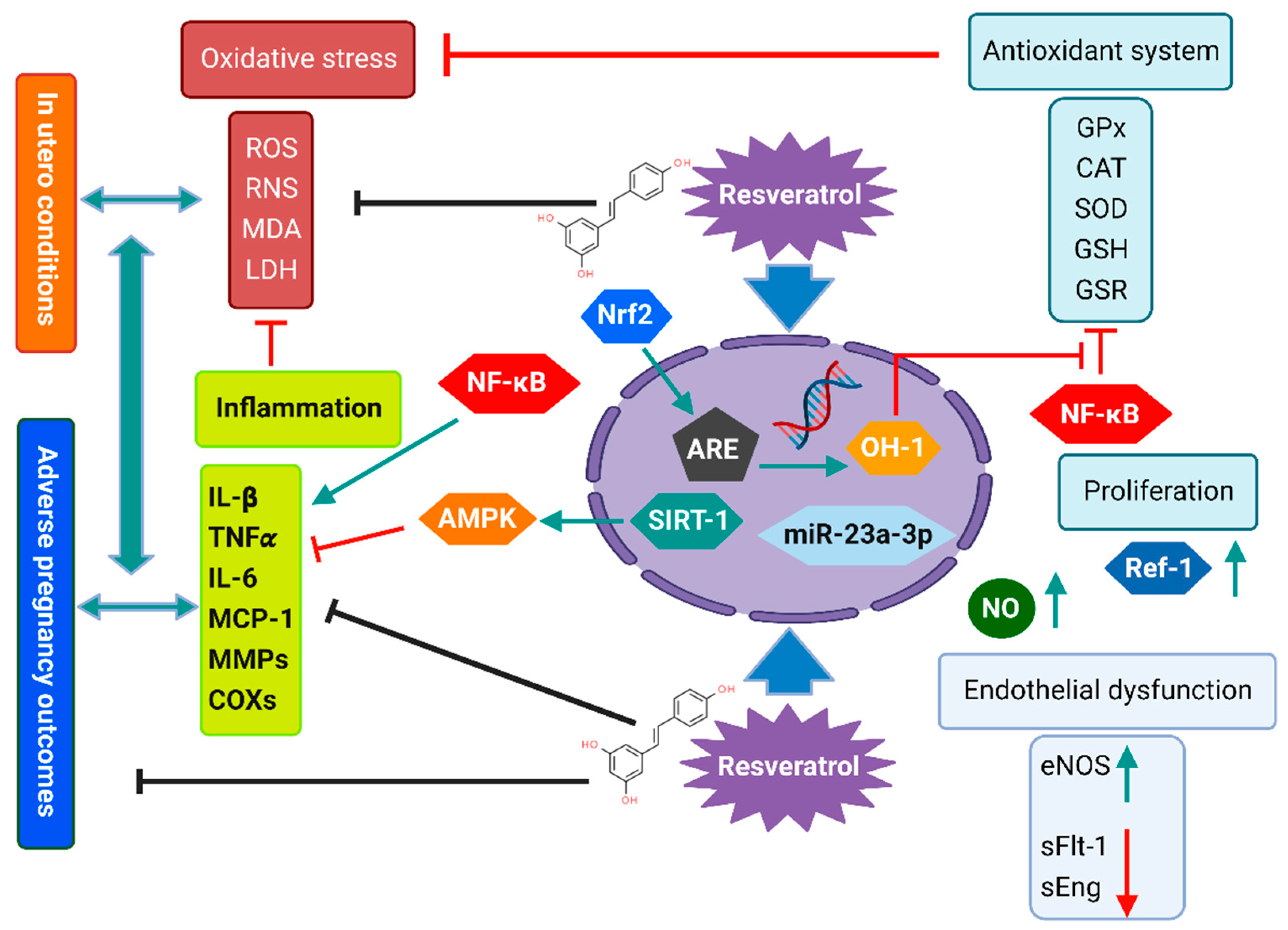
2. Maternal Pregnancy-Related Disorders
2.1. Resveratrol Effect in Pre-Eclampsia and Related Disorders
| Reference | Type of the Study | PE Experimental Model | Dose | Mechanism of Action | Outcomes of Supplementation in the Experimental Model |
|---|---|---|---|---|---|
| [58] | In vitro/Clinical trial | -ECs -Human |
200 mL of polyphenol rich-grape fruit/1 µM of trans-resveratrol | NO, HO-1, miRNA expression, GSH, ARE, ROS levels | ↑ ARE, NO, HO-1, GSH, ARE ↓ ROS, oxidative stress No studied effect in offspring |
| [60] | In vitro/Clinical trial | -ECs -Human |
200 mL of polyphenol rich-grape fruit/1µM of trans-resveratrol | SIRT1 expression | No significant effect No studied effect in offspring |
| [66] | In vitro/In vivo | -HTR-8/SVeno cell culture/ -NG-nitro-l-arginine methyl ester mice model (L-NAME) |
100 µmol/L resveratrol 20 mg/kg/day |
Expression of genes regulating migration, invasion, angiogenesis and EMT-related factors in trophoblasts Hypertension and proteinuria measurement. Endothelial dysfunction/ injury |
↑ E-cadherin, β-catenin, N-cadherin, and vimentin expression, ↓ Alteration of WNT-related gene expression, including WNT1, WNT3 and WNT5B. ↓ Hypertension and proteinuria No studied effect in offspring |
| [72] | In vitro | primary trophoblasts and HUVECs | 0–100 μM | -Angiogenesis activation (sFlt-1) (sEng) secretion Anti-inflammatory effect -NFκB , IL-6 and IL-1β -Antioxidant effect HO-1, NQO1, GCLC, (TXN) Endothelial dysfunction, VCAM Endothelin-1 eNOS |
↓ sFlt-1, sEng ↓ IL-6, IL-1β and TNF-α.‘ ↑, NQO1, GCLC TXN ↓ HO-1 protein in trophoblast. ↑ VCAM-1 ↓ Endotelin-1, ↑eNOS No studied effect in offspring |
| [73] | In vivo | NG-nitro-l-arginine methyl ester mice model (L-NAME) | 20 mg/kg/day | -Antioxidant effect, Apoptosis SOD, MDA |
↑ SOD, MDA No studied effect in offspring |
2.2. Resveratrol Effect in Gestational Diabetes and Related Metabolic Disorders
| Reference | Type of the Study | GDM Experimental Model | Dose | Mechanism of Action | Outcomes of Supplementation in the Experimental Model |
|---|---|---|---|---|---|
| [80] | In vivo | Streptozotocin GDM pregnant rats model | 120 and 240 mg/kg | Amelioration of blood glucose and blood lipids levels | ↑ Insulin levels, ↓ blood glucose levels Amelioration of lipidic profile No studied effect in offspring |
| [81] | In vivo | Streptozotocin GDM pregnant rats model | 500 μg/mL (CS-ZnO-RS) | Anti-diabetic effect Anti-inflammatory effect |
↓ blood glucose levels lipid content reduced the level of ↓ IL-6 and MCP-1, GRP78, p-IRE1α, p-eIF2α, and p-PERK Inhibition of α-glucosidase and α-amylase No studied effect in offspring |
| [3] | In vivo | Streptozotocin GDM pregnant rats model | 120 and 240 mg/kg | Amelioration of the lipids metabolic profile | ↑ HDL-C and adiponectin ↓ leptin, resistin, TNF-α, IL-6 levels, the body weight, TC, TG, and LDL-C No studied effect in offspring |
| [107] | In vivo | high-fat fed hamster model | 0.025% | Amelioration of the lipids metabolic profile | -HMG-CoA reductase expression No studied effect in offspring |
| [93] | In vivo | high-fat diet GDM mice model IR adipocyte model was established by dexamethasone-inducing |
0.2% | Amelioration of glucose and lipids metabolic profile Amelioration of IR in adipocytes |
↓ The bodyweight, serum glucose ↑ serum insulin Upregulations of miR-23a-3p/NOV axis ↑ Adiponectin, Leptin, p-PI3K, and p-Akt No studied effect in offspring |
| [82] | In vivo | female Sprague-Dawley rat model, fed with a high-fat and sucrose diet | 147 mg/kg/day | Protection against gestational diabetes-induced glucose intolerance and islet dysfunction | -Restored glucose tolerance, normoglycaemia and improved insulin secretion in offspring -Attenuation of hepatic steatosis, insulin resistance, glucose intolerance and dysregulated gluconeogenesis in offspring -Downregulation of lpl, ppara, g6p genes |
| [109] | In vivo | C57BL/KsJ-Lep (db/+) (db/+) genetic GDM pregnant mouse model | 10 mg/kg/day | Amelioration of glucose metabolic profile via SIRT1/AMPK pathway Amelioration of IR |
↑ Glucose metabolism, insulin tolerance and reproductive outcome of the pregnant db/+ females AMPK activation ↓ glucose-6-phosphatase in both pregnant db/+ females and their offspring |
This entry is adapted from the peer-reviewed paper 10.3390/antiox12020341
References
- Darby, J.R.T.; Dollah, M.H.B.M.; Regnault, T.R.H.; Williams, M.T.; Morrison, J.L. Systematic review: Impact of resveratrol exposure during pregnancy on maternal and fetal outcomes in animal models of human pregnancy complications-Are we ready for the clinic? Pharmacol. Res. 2019, 144, 264–278.
- Moraloglu, O.; Engin-Ustun, Y.; Tonguç, E.; Var, T.; Tapisiz, Ö.L.; Ergün, H.; Guvenc, T.; Gacar, A. The effect of resveratrol on blood pressure in a rat model of preeclampsia. J. Matern.-Fetal Neonatal Med. 2012, 25, 845–848.
- Singh, C.K.; Kumar, A.; LaVoie, H.A.; DiPette, D.J.; Singh, U.S. Diabetic complications in pregnancy: Is resveratrol a solution? Exp. Biol. Med. 2013, 238, 482–490.
- Shah, A.; Quon, A.; Morton, J.S.; Davidge, S.T. Postnatal resveratrol supplementation improves cardiovascular function in male and female intrauterine growth restricted offspring. Physiol. Rep. 2017, 5, e13109.
- Ding, J.; Kang, Y.; Fan, Y.; Chen, Q. Efficacy of resveratrol to supplement oral nifedipine treatment in pregnancy-induced preeclampsia. Endocr. Connect. 2017, 6, 595–600.
- Malvasi, A.; Kosmas, I.; Mynbaev, O.A.; Sparic, R.; Gustapane, S.; Guido, M.; Tinelli, A. Can trans resveratrol plus d-chiro-inositol and myo-inositol improve maternal metabolic profile in overweight pregnant patients? Clin. Ter. 2017, 168, e240–e247.
- Costa-Silva, J.H.; Simões-Alves, A.C.; Fernandes, M.P. Developmental Origins of Cardiometabolic Diseases: Role of the Maternal Diet. Front. Physiol. 2016, 7, 504.
- Lacerda, D.C.; Costa, P.C.T.; de Oliveira, Y.; de Brito Alves, J.L. The effect of resveratrol in cardio-metabolic disorders during pregnancy and offspring outcomes: A review. J. Dev. Orig. Health Dis. 2022, 14, 3–14.
- dos Santos, A.S.; de Albuquerque, T.M.R.; de Brito Alves, J.L.; de Souza, E.L. Effects of Quercetin and Resveratrol on in vitro Properties Related to the Functionality of Potentially Probiotic Lactobacillus Strains. Front. Microbiol. 2019, 10, 2229.
- Badran, A.; Nasser, S.A.; Mesmar, J.; El-Yazbi, A.F.; Bitto, A.; Fardoun, M.M.; Baydoun, E.; Eid, A.H. Reactive Oxygen Species: Modulators of Phenotypic Switch of Vascular Smooth Muscle Cells. Int. J. Mol. Sci. 2020, 21, 8764.
- Shaito, A.; Aramouni, K.; Assaf, R.; Parenti, A.; Orekhov, A.; El Yazbi, A.; Pintus, G.; Eid, A.H. Oxidative Stress-Induced Endothelial Dysfunction in Cardiovascular Diseases. Front. Biosci.-Landmark 2022, 27, 105.
- Mihu, D.; Sabau, L.; Costin, N.; Ciortea, R.; Malutan, A.; Mihu, C.M. Implications of maternal systemic oxidative stress in normal pregnancy and in pregnancy complicated by preeclampsia. J. Matern.-Fetal Neonatal Med. 2012, 25, 944–951.
- Tan, C.; Wei, H.; Sun, H.; Ao, J.; Long, G.; Jiang, S.; Peng, J. Effects of Dietary Supplementation of Oregano Essential Oil to Sows on Oxidative Stress Status, Lactation Feed Intake of Sows, and Piglet Performance. BioMed Res. Int. 2015, 2015, 525218.
- Pereira, A.C.; Martel, F. Oxidative stress in pregnancy and fertility pathologies. Cell Biol. Toxicol. 2014, 30, 301–312.
- Sanchez-Aranguren, L.C.; Prada, C.E.; Riano-Medina, C.E.; Lopez, M. Endothelial dysfunction and preeclampsia: Role of oxidative stress. Front. Physiol. 2014, 5, 372.
- Vega, C.C.; Reyes-Castro, L.A.; Rodríguez-González, G.L.; Bautista, C.J.; Vázquez-Martínez, M.; Larrea, F.; Chamorro-Cevallos, G.A.; Nathanielsz, P.W.; Zambrano, E. Resveratrol partially prevents oxidative stress and metabolic dysfunction in pregnant rats fed a low protein diet and their offspring. J. Physiol. 2016, 594, 1483–1499.
- Singh, C.K.; Kumar, A.; Hitchcock, D.B.; Fan, D.; Goodwin, R.; LaVoie, H.A.; Nagarkatti, P.; DiPette, D.J.; Singh, U.S. Resveratrol prevents embryonic oxidative stress and apoptosis associated with diabetic embryopathy and improves glucose and lipid profile of diabetic dam. Mol. Nutr. Food Res. 2011, 55, 1186–1196.
- Trejo-González, N.L.; Chirino-Galindo, G.; Palomar-Morales, M. Capacidad antiteratogénica del resveratrol en diabetes inducida por estreptozotocina en ratas. Rev. Peru. Med. Exp. Salud Pública 2015, 32, 457–463.
- Wang, L.; Tang, J.; Wang, L.; Tan, F.; Song, H.; Zhou, J.; Li, F. Oxidative stress in oocyte aging and female reproduction. J. Cell. Physiol. 2021, 236, 7966–7983.
- Homer, H.A. The role of oocyte quality in explaining “unexplained” infertility. Semin. Reprod. Med. 2020, 38, 021–028.
- Wang, T.; Zhang, M.; Jiang, Z.; Seli, E. Mitochondrial dysfunction and ovarian aging. Am. J. Reprod. Immunol. 2017, 77, e12651.
- Liang, Q.-X.; Lin, Y.-H.; Zhang, C.-H.; Sun, H.-M.; Zhou, L.; Schatten, H.; Sun, Q.-Y.; Qian, W.-P. Resveratrol increases resistance of mouse oocytes to postovulatory aging in vivo. Aging 2018, 10, 1586–1596.
- Takeo, S.; Sato, D.; Kimura, K.; Monji, Y.; Kuwayama, T.; Kawahara-Miki, R.; Iwata, H. Resveratrol Improves the Mitochondrial Function and Fertilization Outcome of Bovine Oocytes. J. Reprod. Dev. 2014, 60, 92–99.
- Liu, M.-J.; Sun, A.-G.; Zhao, S.-G.; Liu, H.; Ma, S.-Y.; Li, M.; Huai, Y.-X.; Zhao, H.; Liu, H.-B. Resveratrol improves in vitro maturation of oocytes in aged mice and humans. Fertil. Steril. 2018, 109, 900–907.
- Yoon, J.; Juhn, K.-M.; Jung, E.-H.; Park, H.-J.; Yoon, S.-H.; Ko, Y.; Hur, C.-Y.; Lim, J.-H. Effects of resveratrol, granulocyte-macrophage colony-stimulating factor or dichloroacetic acid in the culture media on embryonic development and pregnancy rates in aged mice. Aging 2020, 12, 2659–2669.
- Liu, M.; Yin, Y.; Ye, X.; Zeng, M.; Zhao, Q.; Keefe, D.L.; Liu, L. Resveratrol protects against age-associated infertility in mice. Hum. Reprod. 2013, 28, 707–717.
- Igarashi, H.; Takahashi, T.; Abe, H.; Nakano, H.; Nakajima, O.; Nagase, S. Poor embryo development in post-ovulatory in vivo-aged mouse oocytes is associated with mitochondrial dysfunction, but mitochondrial transfer from somatic cells is not sufficient for rejuvenation. Hum. Reprod. 2016, 31, 2331–2338.
- Li, H.; Chian, R.-C. Mitochondrial Dysfunction and Age-related Oocyte Quality. Reprod. Dev. Med. 2017, 1, 45–54.
- Zheng, S.; Feng, Q.; Cheng, J.; Zheng, J. Maternal resveratrol consumption and its programming effects on metabolic health in offspring mechanisms and potential implications. Biosci. Rep. 2018, 38, BSR20171741.
- Park, E.-Y.; Park, J.-B. High glucose-induced oxidative stress promotes autophagy through mitochondrial damage in rat notochordal cells. Int. Orthop. 2013, 37, 2507–2514.
- Sebori, R.; Kuno, A.; Hosoda, R.; Hayashi, T.; Horio, Y. Resveratrol Decreases Oxidative Stress by Restoring Mitophagy and Improves the Pathophysiology of Dystrophin-Deficient mdx Mice. Oxidative Med. Cell. Longev. 2018, 2018, 9179270.
- Ding, W.-X.; Yin, X.-M. Mitophagy: Mechanisms, pathophysiological roles, and analysis. Biol. Chem. 2012, 393, 547–564.
- Zha, C.; Xiao, H.; Song, B.; Zheng, C.; Yang, X.; Wang, W.; Wang, L. Resveratrol promotes mammary cell proliferation and antioxidation capacity during pregnancy and lactation in mice. J. Appl. Microbiol. 2021, 130, 450–463.
- Phoswa, W.N.; Khaliq, O.P. The Role of Oxidative Stress in Hypertensive Disorders of Pregnancy (Preeclampsia, Gestational Hypertension) and Metabolic Disorder of Pregnancy (Gestational Diabetes Mellitus). Oxidative Med. Cell. Longev. 2021, 2021, 5581570.
- Rodrigo, R.; González, J.; Paoletto, F. The role of oxidative stress in the pathophysiology of hypertension. Hypertens. Res. 2011, 34, 431–440.
- Magee, L.A.; Kenny, L.; Ananth Karumanchi, S.; McCarthy, F.; Saito, S.; Hall, D.R.; Warren, C.E.; Adoyi, G.; Mohammed, S.I. TEMPORARY REMOVAL: The hypertensive disorders of pregnancy: ISSHP classification, diagnosis and management recommendations for international practice 2018. Pregnancy Hypertens. 2018, 72, 24–43.
- Agarwal, P.; Morriseau, T.S.; Kereliuk, S.M.; Doucette, C.A.; Wicklow, B.A.; Dolinsky, V.W. Maternal obesity, diabetes during pregnancy and epigenetic mechanisms that influence the developmental origins of cardiometabolic disease in the offspring. Crit. Rev. Clin. Lab. Sci. 2018, 55, 71–101.
- Yu, J.; Auwerx, J. The Role of Sirtuins in the Control of Metabolic Homeostasis. Ann. N. Y. Acad. Sci. 2009, 1173, E10–E19.
- Yu, J.; Auwerx, J. Protein deacetylation by SIRT1: An emerging key post-translational modification in metabolic regulation. Pharmacol. Res. 2010, 62, 35–41.
- Li, S.-W.; Yu, H.-R.; Sheen, J.-M.; Tiao, M.-M.; Tain, Y.-L.; Lin, I.C.; Lin, Y.-J.; Chang, K.-A.; Tsai, C.-C.; Huang, L.-T. A maternal high-fat diet during pregnancy and lactation, in addition to a postnatal high-fat diet, leads to metabolic syndrome with spatial learning and memory deficits: Beneficial effects of resveratrol. Oncotarget 2017, 8, 111998.
- Burton, G.J.; Jauniaux, E. Oxidative stress. Best Pract. Res. Clin. Obstet. Gynaecol. 2011, 25, 287–299.
- Lambert, G.; Brichant, J.F.; Hartstein, G.; Bonhomme, V.; Dewandre, P.Y. Preeclampsia: An update. Acta Anaesthesiol. Belg. 2014, 65, 137–149.
- Sibai, B.; Dekker, G.; Kupferminc, M. Pre-eclampsia. Lancet 2005, 365, 785–799.
- Pishko, A.M.; Levine, L.D.; Cines, D.B. Thrombocytopenia in pregnancy: Diagnosis and approach to management. Blood Rev. 2020, 40, 100638.
- Silvestro, S.; Calcaterra, V.; Pelizzo, G.; Bramanti, P.; Mazzon, E. Prenatal Hypoxia and Placental Oxidative Stress: Insights from Animal Models to Clinical Evidences. Antioxidants 2020, 9, 414.
- Hu, X.-Q.; Zhang, L. Hypoxia and Mitochondrial Dysfunction in Pregnancy Complications. Antioxidants 2021, 10, 405.
- Dolinsky, V.W.; Rueda-Clausen, C.F.; Morton, J.S.; Davidge, S.T.; Dyck, J.R. Continued postnatal administration of resveratrol prevents diet-induced metabolic syndrome in rat offspring born growth restricted. Diabetes 2011, 60, 2274–2284.
- Bourque, S.L.; Dolinsky, V.W.; Dyck, J.R.B.; Davidge, S.T. Maternal resveratrol treatment during pregnancy improves adverse fetal outcomes in a rat model of severe hypoxia. Placenta 2012, 33, 449–452.
- Baumwell, S.; Karumanchi, S.A. Pre-Eclampsia: Clinical Manifestations and Molecular Mechanisms. Nephron Clin. Pract. 2007, 106, c72–c81.
- Rosser, M.L.; Katz, N.T. Preeclampsia: An Obstetrician’s Perspective. Adv. Chronic Kidney Dis. 2013, 20, 287–296.
- Eiland, E.; Nzerue, C.; Faulkner, M. Preeclampsia 2012. J. Pregnancy 2012, 2012, 586578.
- Hansson, S.R.; Nääv, Å.; Erlandsson, L. Oxidative stress in preeclampsia and the role of free fetal hemoglobin. Front. Physiol. 2015, 5, 516.
- Sagrillo-Fagundes, L.; Laurent, L.; Bienvenue-Pariseault, J.; Vaillancourt, C. In Vitro Induction of Hypoxia/Reoxygenation on Placental Cells: A Suitable Model for Understanding Placental Diseases. In Preeclampsia: Methods and Protocols; Murthi, P., Vaillancourt, C., Eds.; Springer: New York, NY, USA, 2018; pp. 277–283.
- Aydın, S.; Benian, A.; Madazli, R.; Uludaǧ, S.; Uzun, H.; Kaya, S. Plasma malondialdehyde, superoxide dismutase, sE-selectin, fibronectin, endothelin-1 and nitric oxide levels in women with preeclampsia. Eur. J. Obstet. Gynecol. Reprod. Biol. 2004, 113, 21–25.
- Gupta, S.; Agarwal, A.; Sharma, R.K. The Role of Placental Oxidative Stress and Lipid Peroxidation in Preeclampsia. Obstet. Gynecol. Surv. 2005, 60, 807–816.
- Parsamanesh, N.; Asghari, A.; Sardari, S.; Tasbandi, A.; Jamialahmadi, T.; Xu, S.; Sahebkar, A. Resveratrol and endothelial function: A literature review. Pharmacol. Res. 2021, 170, 105725.
- Mann, G.E.; Niehueser-Saran, J.; Watson, A.; Gao, L.; Ishii, T.; de Winter, P.; Siow, R.C. Nrf2/ARE regulated antioxidant gene expression in endothelial and smooth muscle cells in oxidative stress: Implications for atherosclerosis and preeclampsia. Acta Physiol. Sin.-Chin. Ed. 2007, 59, 117.
- Caldeira-Dias, M.; Viana-Mattioli, S.; de Souza Rangel Machado, J.; Carlström, M.; de Carvalho Cavalli, R.; Sandrim, V.C. Resveratrol and grape juice: Effects on redox status and nitric oxide production of endothelial cells in in vitro preeclampsia model. Pregnancy Hypertens. 2021, 23, 205–210.
- Poudel, R.; Stanley, J.L.; Rueda-Clausen, C.F.; Andersson, I.J.; Sibley, C.P.; Davidge, S.T.; Baker, P.N. Effects of Resveratrol in Pregnancy Using Murine Models with Reduced Blood Supply to the Uterus. PLoS ONE 2013, 8, e64401.
- Viana-Mattioli, S.; Cinegaglia, N.; Bertozzi-Matheus, M.; Bueno-Pereira, T.O.; Caldeira-Dias, M.; Cavalli, R.C.; Sandrim, V.C. SIRT1-dependent effects of resveratrol and grape juice in an in vitro model of preeclampsia. Biomed. Pharmacother. 2020, 131, 110659.
- Khong, T.Y.; De Wolf, F.; Robertson, W.B.; Brosens, I. Inadequate maternal vascular response to placentation in pregnancies complicated by pre-eclampsia and by small-for-gestational age infants. BJOG Int. J. Obstet. Gynaecol. 1986, 93, 1049–1059.
- Labarrere, C.A.; Althabe, O.H. Inadequate maternal vascular response to placentation in pregnancies complicated by preeclampsia and by small-for-gestational-age infants. BJOG Int. J. Obstet. Gynaecol. 1987, 94, 1113–1115.
- Friedl, P.; Wolf, K. Tumour-cell invasion and migration: Diversity and escape mechanisms. Nat. Rev. Cancer 2003, 3, 362–374.
- Mao, X.-W.; Xiao, J.-Q.; Li, Z.-Y.; Zheng, Y.-C.; Zhang, N. Effects of microRNA-135a on the epithelial–mesenchymal transition, migration and invasion of bladder cancer cells by targeting GSK3β through the Wnt/β-catenin signaling pathway. Exp. Mol. Med. 2018, 50, e429.
- Kokkinos, M.I.; Murthi, P.; Wafai, R.; Thompson, E.W.; Newgreen, D.F. Cadherins in the human placenta—Epithelial–mesenchymal transition (EMT) and placental development. Placenta 2010, 31, 747–755.
- Zou, Y.; Li, S.; Wu, D.; Xu, Y.; Wang, S.; Jiang, Y.; Liu, F.; Jiang, Z.; Qu, H.; Yu, X.; et al. Resveratrol promotes trophoblast invasion in pre-eclampsia by inducing epithelial-mesenchymal transition. J. Cell. Mol. Med. 2019, 23, 2702–2710.
- Liu, Z.; Wang, C.; Pei, J.; Li, M.; Gu, W. SIRT1: A Novel Protective Molecule in Pre-eclampsia. Int. J. Med. Sci. 2022, 19, 993–1002.
- Romero, R.; Chaiworapongsa, T. Preeclampsia: A link between trophoblast dysregulation and an antiangiogenic state. J. Clin. Investig. 2013, 123, 2775–2777.
- Phipps, E.A.; Thadhani, R.; Benzing, T.; Karumanchi, S.A. Pre-eclampsia: Pathogenesis, novel diagnostics and therapies. Nat. Rev. Nephrol. 2019, 15, 275–289.
- Venkatesha, S.; Toporsian, M.; Lam, C.; Hanai, J.-i.; Mammoto, T.; Kim, Y.M.; Bdolah, Y.; Lim, K.-H.; Yuan, H.-T.; Libermann, T.A.; et al. Soluble endoglin contributes to the pathogenesis of preeclampsia. Nat. Med. 2006, 12, 642–649.
- Romero, R.; Nien, J.K.; Espinoza, J.; Todem, D.; Fu, W.; Chung, H.; Kusanovic, J.P.; Gotsch, F.; Erez, O.; Mazaki-Tovi, S.; et al. A longitudinal study of angiogenic (placental growth factor) and anti-angiogenic (soluble endoglin and soluble vascular endothelial growth factor receptor-1) factors in normal pregnancy and patients destined to develop preeclampsia and deliver a small for gestational age neonate. J. Matern.-Fetal Neonatal Med. 2008, 21, 9–23.
- Hannan, N.J.; Brownfoot, F.C.; Cannon, P.; Deo, M.; Beard, S.; Nguyen, T.V.; Palmer, K.R.; Tong, S.; Kaitu’u-Lino, T.u.J. Resveratrol inhibits release of soluble fms-like tyrosine kinase (sFlt-1) and soluble endoglin and improves vascular dysfunction—Implications as a preeclampsia treatment. Sci. Rep. 2017, 7, 1819.
- Zou, Y.; Zuo, Q.; Huang, S.; Yu, X.; Jiang, Z.; Zou, S.; Fan, M.; Sun, L. Resveratrol Inhibits Trophoblast Apoptosis through Oxidative Stress in Preeclampsia-Model Rats. Molecules 2014, 19, 20570–20579.
- Jiao, X.C.; Xiao, M.; Gao, Z.X.; Xie, J.; Liu, Y.; Yin, M.J.; Wu, Y.; Tao, R.X.; Zhu, P. Effects of comorbid gestational diabetes mellitus and depression on glucose metabolism during pregnancy and neonatal morphological outcome. Zhonghua Yu Fang Yi Xue Za Zhi 2020, 54, 968–973.
- Kim, J.; Piao, Y.; Pak, Y.K.; Chung, D.; Han, Y.M.; Hong, J.S.; Jun, E.J.; Shim, J.-Y.; Choi, J.; Kim, C.J. Umbilical Cord Mesenchymal Stromal Cells Affected by Gestational Diabetes Mellitus Display Premature Aging and Mitochondrial Dysfunction. Stem Cells Dev. 2014, 24, 575–586.
- Choi, D.-J.; Yoon, C.-H.; Lee, H.; Ahn, S.-Y.; Oh, K.J.; Park, H.-Y.; Lee, H.Y.; Cho, M.C.; Chung, I.-M.; Shin, M.-S.; et al. The Association of Family History of Premature Cardiovascular Disease or Diabetes Mellitus on the Occurrence of Gestational Hypertensive Disease and Diabetes. PLoS ONE 2016, 11, e0167528.
- Wang, X.-M.; Gao, Y.; Eriksson, J.G.; Chen, W.; Chong, Y.S.; Tan, K.H.; Zhang, C.; Zhou, L.; Li, L.-J. Metabolic signatures in the conversion from gestational diabetes mellitus to postpartum abnormal glucose metabolism: A pilot study in Asian women. Sci. Rep. 2021, 11, 16435.
- Su, M.; Zhao, W.; Xu, S.; Weng, J. Resveratrol in Treating Diabetes and Its Cardiovascular Complications: A Review of Its Mechanisms of Action. Antioxidants 2022, 11, 1085.
- Do, G.-M.; Jung, U.J.; Park, H.-J.; Kwon, E.-Y.; Jeon, S.-M.; McGregor, R.A.; Choi, M.-S. Resveratrol ameliorates diabetes-related metabolic changes via activation of AMP-activated protein kinase and its downstream targets in db/db mice. Mol. Nutr. Food Res. 2012, 56, 1282–1291.
- Zhang, G.; Wang, X.; Ren, B.; Zhao, Q.; Zhang, F. The Effect of Resveratrol on Blood Glucose and Blood Lipids in Rats with Gestational Diabetes Mellitus. Evid.-Based Complement. Altern. Med. 2021, 2021, 2956795.
- Du, S.; Lv, Y.; Li, N.; Huang, X.; Liu, X.; Li, H.; Wang, C.; Jia, Y.-F. Biological investigations on therapeutic effect of chitosan encapsulated nano resveratrol against gestational diabetes mellitus rats induced by streptozotocin. Drug Deliv. 2020, 27, 953–963.
- Brawerman, G.M.; Kereliuk, S.M.; Brar, N.; Cole, L.K.; Seshadri, N.; Pereira, T.J.; Xiang, B.; Hunt, K.L.; Fonseca, M.A.; Hatch, G.M.; et al. Maternal resveratrol administration protects against gestational diabetes-induced glucose intolerance and islet dysfunction in the rat offspring. J. Physiol. 2019, 597, 4175–4192.
- Chen, S.; Zhao, Z.; Ke, L.; Li, Z.; Li, W.; Zhang, Z.; Zhou, Y.; Feng, X.; Zhu, W. Resveratrol improves glucose uptake in insulin-resistant adipocytes via Sirt1. J. Nutr. Biochem. 2018, 55, 209–218.
- Yi, L.; Chen, C.Y.; Jin, X.; Mi, M.T.; Yu, B.; Chang, H.; Ling, W.H.; Zhang, T. Structural requirements of anthocyanins in relation to inhibition of endothelial injury induced by oxidized low-density lipoprotein and correlation with radical scavenging activity. Febs. Lett. 2010, 584, 583–590.
- Shu, L.; Zhao, H.A.-O.; Huang, W.; Hou, G.; Song, G.; Ma, H. Resveratrol Upregulates mmu-miR-363-3p via the PI3K-Akt Pathway to Improve Insulin Resistance Induced by a High-Fat Diet in Mice. Diabetes Metab. Syndr. Obes. Targets Ther. 2020, 13, 391–403.
- Ferrante, M.; Conti, O.G. Environment and Neurodegenerative Diseases: An Update on miRNA Role. MicroRNA 2017, 6, 157–165.
- Mishra, S.; Yadav, T.; Rani, V. Exploring miRNA based approaches in cancer diagnostics and therapeutics. Crit. Rev. Oncol./Hematol. 2016, 98, 12–23.
- Tiwari, A.; Mukherjee, B.; Dixit, M. MicroRNA Key to Angiogenesis Regulation: MiRNA Biology and Therapy. Curr. Cancer Drug Targets 2018, 18, 266–277.
- Giordo, R.; Ahmed, Y.M.; Allam, H.; Abusnana, S.; Pappalardo, L.; Nasrallah, G.K.; Mangoni, A.A.; Pintus, G. EndMT regulation by small RNAs in diabetes-associated fibrotic conditions: Potential link with oxidative stress. Front. Cell Dev. Biol. 2021, 9, 683594.
- Wehbe, N.; Nasser, S.A.; Pintus, G.; Badran, A.; Eid, A.H.; Baydoun, E. MicroRNAs in cardiac hypertrophy. Int. J. Mol. Sci. 2019, 20, 4714.
- Giordo, R.; Wehbe, Z.; Posadino, A.M.; Erre, G.L.; Eid, A.H.; Mangoni, A.A.; Pintus, G. Disease-associated regulation of non-coding RNAs by resveratrol: Molecular insights and therapeutic applications. Front. Cell Dev. Biol. 2022, 10, 894305.
- Lozano-Bartolomé, J.; Llauradó, G.; Portero-Otin, M.; Altuna-Coy, A.; Rojo-Martínez, G.; Vendrell, J.; Jorba, R.; Rodríguez-Gallego, E.; Chacón, M.R. Altered Expression of miR-181a-5p and miR-23a-3p Is Associated With Obesity and TNFα-Induced Insulin Resistance. J. Clin. Endocrinol. Metab. 2018, 103, 1447–1458.
- Zheng, T.; Chen, H. Resveratrol ameliorates the glucose uptake and lipid metabolism in gestational diabetes mellitus mice and insulin-resistant adipocytes via miR-23a-3p/NOV axis. Mol. Immunol. 2021, 137, 163–173.
- Downs, D.S. Obesity in Special Populations: Pregnancy. Prim. Care Clin. Off. Pract. 2016, 43, 109–120.
- Reichetzeder, C. Overweight and obesity in pregnancy: Their impact on epigenetics. Eur. J. Clin. Nutr. 2021, 75, 1710–1722.
- Snelgrove-Clarke, E.; Macdonald, D.; Helwig, M.; Alsius, A. Women’s experiences of living with obesity during pregnancy, birthing, and postpartum: A qualitative systematic review protocol. JBI Evid. Synth. 2021, 19, 3183–3189.
- Álvarez, D.; Muñoz, Y.; Ortiz, M.; Maliqueo, M.; Chouinard-Watkins, R.; Valenzuela, R. Impact of Maternal Obesity on the Metabolism and Bioavailability of Polyunsaturated Fatty Acids during Pregnancy and Breastfeeding. Nutrients 2021, 13, 19.
- Mission, J.F.; Marshall, N.E.; Caughey, A.B. Pregnancy Risks Associated with Obesity. Obstet. Gynecol. Clin. North Am. 2015, 42, 335–353.
- Rodríguez-González, G.L.; Vargas-Hernández, L.; Reyes-Castro, L.A.; Ibáñez, C.A.; Bautista, C.J.; Lomas-Soria, C.; Itani, N.; Estrada-Gutierrez, G.; Espejel-Nuñez, A.; Flores-Pliego, A.; et al. Resveratrol Supplementation in Obese Pregnant Rats Improves Maternal Metabolism and Prevents Increased Placental Oxidative Stress. Antioxidants 2022, 11, 1871.
- Yu, H.-R.; Sheen, J.-M.; Tiao, M.-M.; Tain, Y.-L.; Chen, C.-C.; Lin, I.C.; Lai, Y.-J.; Tsai, C.-C.; Lin, Y.-J.; Tsai, C.-C.; et al. Resveratrol Treatment Ameliorates Leptin Resistance and Adiposity Programed by the Combined Effect of Maternal and Post-Weaning High-Fat Diet. Mol. Nutr. Food Res. 2019, 63, 1801385.
- Huang, Y.-C.; Huang, L.-T.; Sheen, J.-M.; Hou, C.-Y.; Yeh, Y.-T.; Chiang, C.-P.; Lin, I.C.; Tiao, M.-M.; Tsai, C.-C.; Lin, Y.-J.; et al. Resveratrol treatment improves the altered metabolism and related dysbiosis of gut programed by prenatal high-fat diet and postnatal high-fat diet exposure. J. Nutr. Biochem. 2020, 75, 108260.
- Ros, P.; Díaz, F.; Freire-Regatillo, A.; Argente-Arizón, P.; Barrios, V.; Argente, J.; Chowen, J.A. Resveratrol Intake During Pregnancy and Lactation Modulates the Early Metabolic Effects of Maternal Nutrition Differently in Male and Female Offspring. Endocrinology 2017, 159, 810–825.
- Sun, S.; Meng, Q.; Luo, Z.; Shi, B.; Bi, C.; Shan, A. Effects of dietary resveratrol supplementation during gestation and lactation of sows on milk composition of sows and fat metabolism of sucking piglets. J. Anim. Physiol. Anim. Nutr. 2019, 103, 813–821.
- Liao, E.; Li, Z.; Shao, Y. Resveratrol regulates the silent information regulator 1–nuclear factor-κB signaling pathway in intrahepatic cholestasis of pregnancy. Hepatol. Res. 2018, 48, 1031–1044.
- Chen, Z.; Hu, L.; Lu, M.; Shen, Z. Resveratrol reduces matrix metalloproteinases and alleviates intrahepatic cholestasis of pregnancy in rats. Can. J. Physiol. Pharmacol. 2015, 94, 402–407.
- Herrera, E.; Ortega-Senovilla, H. Disturbances in lipid metabolism in diabetic pregnancy—Are these the cause of the problem? Best Pract. Res. Clin. Endocrinol. Metab. 2010, 24, 515–525.
- Cho, I.J.; Ahn, J.Y.; Kim, S.; Choi, M.S.; Ha, T.Y. Resveratrol attenuates the expression of HMG-CoA reductase mRNA in hamsters. Biochem. Biophys. Res. Commun. 2008, 367, 190–194.
- Ofori, B.; Rey, E.; Bérard, A. Risk of congenital anomalies in pregnant users of statin drugs. Br. J. Clin. Pharmacol. 2007, 64, 496–509.
- Yao, L.; Wan, J.; Li, H.; Ding, J.; Wang, Y.; Wang, X.; Li, M. Resveratrol relieves gestational diabetes mellitus in mice through activating AMPK. Reprod. Biol. Endocrinol. 2015, 13, 118.
