Plastic abandoned in the environment is prone to segregation, leading to the generation of microplastics (MPs) and nanoplastics (NPs), which can reach aquatic and terrestrial organisms. MPs/NPs in water can access fish’s bodies through the gills, triggering an inflammatory response in loco. Furthermore, from the gills, plastic fragments can be transported within the circulatory system altering blood biochemical parameters and hormone levels and leading to compromised immunocompetence and angiogenesis. In addition, it was also possible to observe an unbalanced ROS production, damage in vascular structure, and enhanced thrombosis. MPs/NPs led to cardiotoxicity, pericardial oedema, and impaired heart rate in fish cardiac tissue. MPs/NPs effects on aquatic organisms pose serious health hazards and ecological consequences because they constitute the food chain for humans. Once present in the mammalian body, plastic particles can interact with circulating cells, eliciting an inflammatory response, with genotoxicity and cytotoxicity of immune cells, enhanced haemolysis, and endothelium adhesion.
- microplastics
- nanoplastics
- environmental exposure
- cardiovascular system
1. Introduction
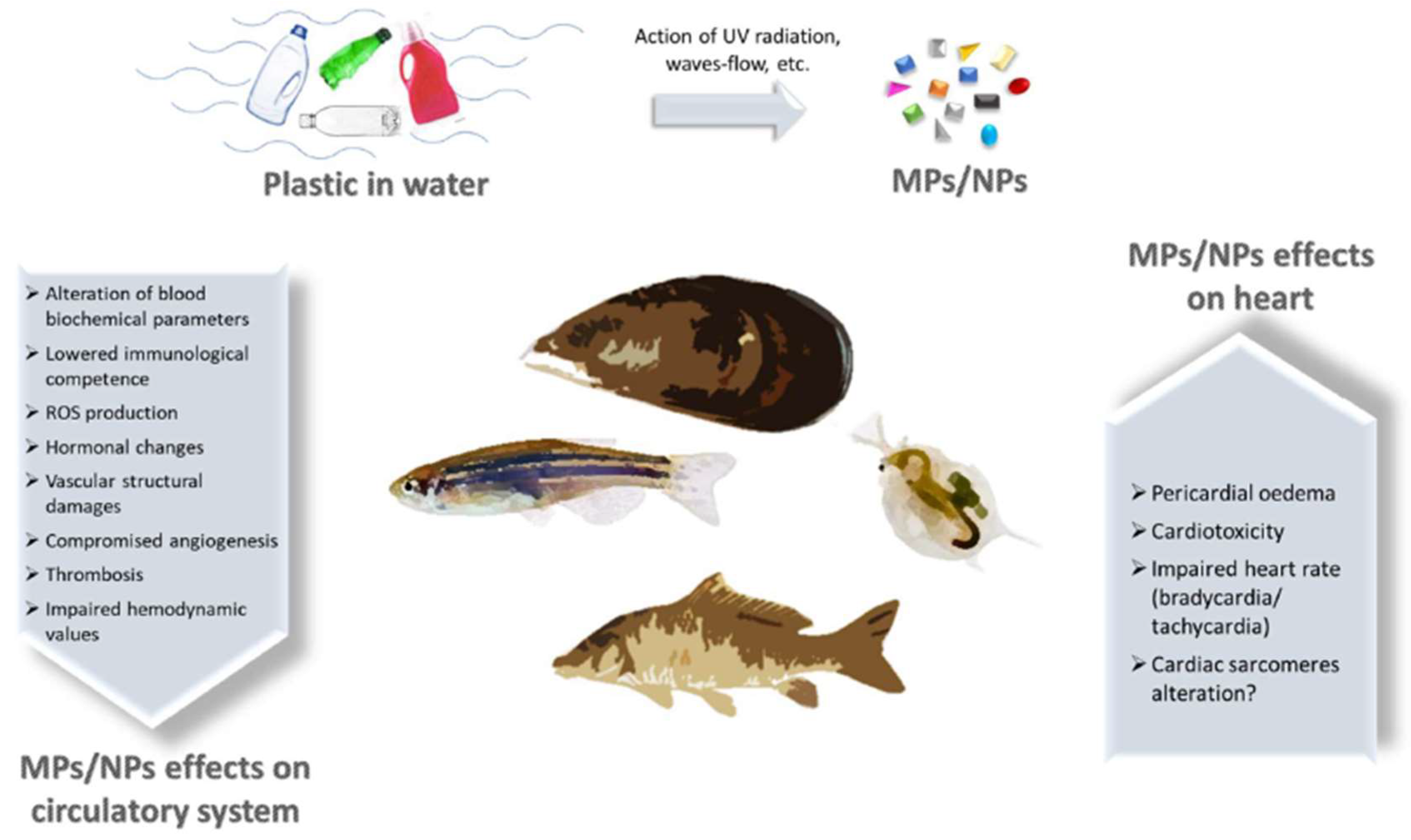
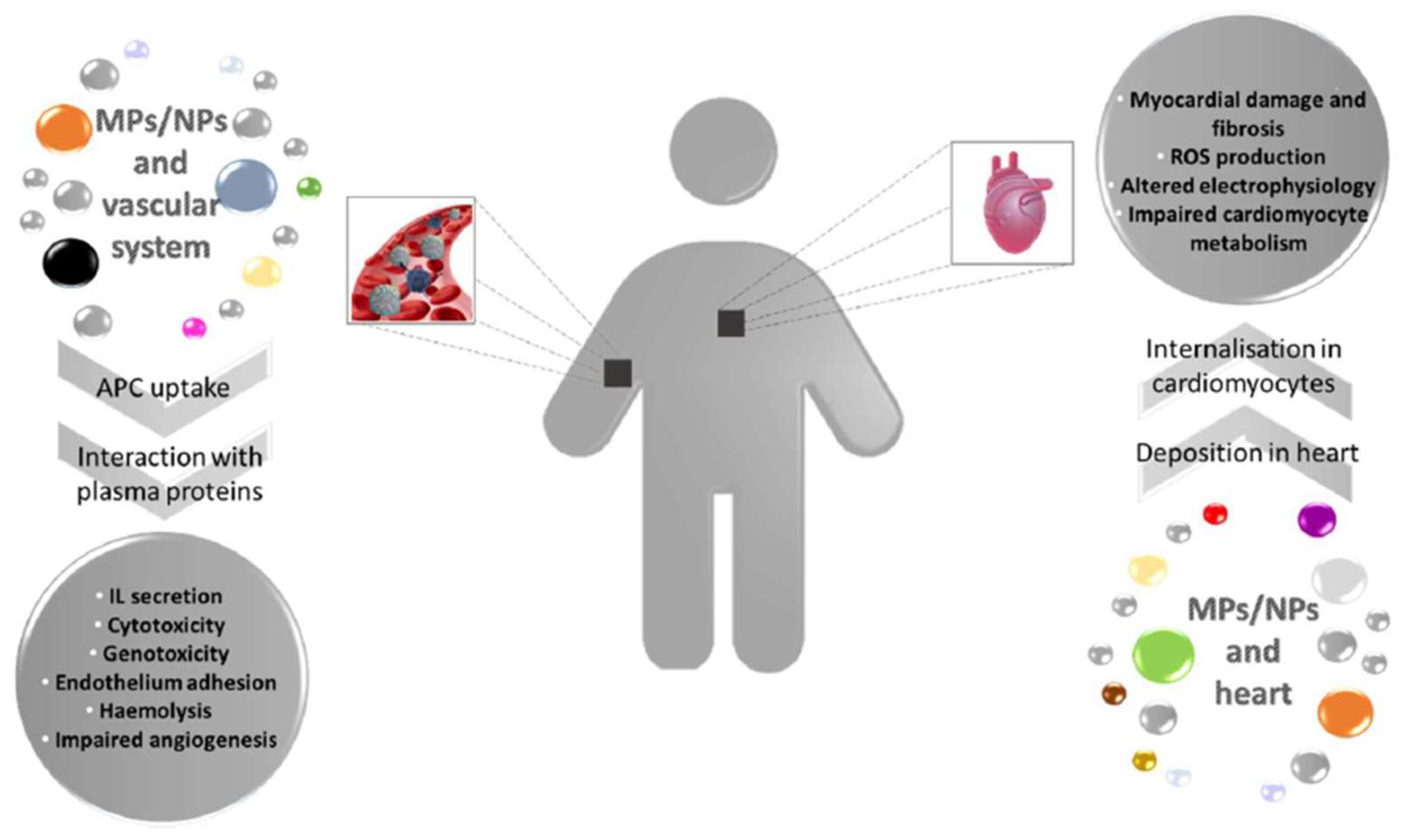
2. MP/NP Effects on Aquatic Fauna Vascular System
This entry is adapted from the peer-reviewed paper 10.3390/biomedicines11020264
References
- Jambeck, J.R.; Geyer, R.; Wilcox, C.; Siegler, T.R.; Perryman, M.; Andrady, A.; Narayan, R.; Law, K.L. Marine Pollution. Plastic Waste Inputs from Land into the Ocean. Science 2015, 347, 768–771.
- Eriksen, M.; Lebreton, L.C.M.; Carson, H.S.; Thiel, M.; Moore, C.J.; Borerro, J.C.; Galgani, F.; Ryan, P.G.; Reisser, J. Plastic Pollution in the World’s Oceans: More than 5 Trillion Plastic Pieces Weighing over 250,000 Tons Afloat at Sea. PLoS ONE 2014, 9, e111913.
- Zalasiewicz, J.; Waters, C.N.; Ivar do Sul, J.A.; Corcoran, P.L.; Barnosky, A.D.; Cearreta, A.; Edgeworth, M.; Gałuszka, A.; Jeandel, C.; Leinfelder, R.; et al. The Geological Cycle of Plastics and Their Use as a Stratigraphic Indicator of the Anthropocene. Anthropocene 2016, 13, 4–17.
- Gündoğdu, S.; Yeşilyurt, İ.N.; Erbaş, C. Potential Interaction between Plastic Litter and Green Turtle Chelonia Mydas during Nesting in an Extremely Polluted Beach. Mar. Pollut. Bull. 2019, 140, 138–145.
- Chapron, L.; Peru, E.; Engler, A.; Ghiglione, J.F.; Meistertzheim, A.L.; Pruski, A.M.; Purser, A.; Vétion, G.; Galand, P.E.; Lartaud, F. Macro- and Microplastics Affect Cold-Water Corals Growth, Feeding and Behaviour. Sci. Rep. 2018, 8, 15299.
- Browne, M.A.; Crump, P.; Niven, S.J.; Teuten, E.; Tonkin, A.; Galloway, T.; Thompson, R. Accumulation of Microplastic on Shorelines Woldwide: Sources and Sinks. Environ. Sci. Technol. 2011, 45, 9175–9179.
- Andrady, A.L. Microplastics in the Marine Environment. Mar. Pollut. Bull. 2011, 62, 1596–1605.
- da Costa, J.P.; Santos, P.S.M.; Duarte, A.C.; Rocha-Santos, T. (Nano)Plastics in the Environment - Sources, Fates and Effects. Sci. Total Environ. 2016, 566–567, 15–26.
- Gigault, J.; Halle, A.T.; Baudrimont, M.; Pascal, P.-Y.; Gauffre, F.; Phi, T.-L.; El Hadri, H.; Grassl, B.; Reynaud, S. Current Opinion: What Is a Nanoplastic? Environ. Pollut. 2018, 235, 1030–1034.
- de Souza Machado, A.A.; Kloas, W.; Zarfl, C.; Hempel, S.; Rillig, M.C. Microplastics as an Emerging Threat to Terrestrial Ecosystems. Glob. Chang. Biol. 2018, 24, 1405–1416.
- Auta, H.S.; Emenike, C.U.; Fauziah, S.H. Distribution and Importance of Microplastics in the Marine Environment: A Review of the Sources, Fate, Effects, and Potential Solutions. Environ. Int. 2017, 102, 165–176.
- Galloway, T.S.; Cole, M.; Lewis, C. Interactions of Microplastic Debris throughout the Marine Ecosystem. Nat. Ecol. Evol. 2017, 1, 1–8.
- Lambert, S.; Scherer, C.; Wagner, M. Ecotoxicity Testing of Microplastics: Considering the Heterogeneity of Physicochemical Properties. Integr. Environ. Assess. Manag. 2017, 13, 470–475.
- Adam, V.; Yang, T.; Nowack, B. Toward an Ecotoxicological Risk Assessment of Microplastics: Comparison of Available Hazard and Exposure Data in Freshwaters. Environ. Toxicol. Chem. 2019, 38, 436–447.
- Akdogan, Z.; Guven, B. Microplastics in the Environment: A Critical Review of Current Understanding and Identification of Future Research Needs. Environ. Pollut. 2019, 254, 113011.
- Hartmann, N.B.; Rist, S.; Bodin, J.; Jensen, L.H.; Schmidt, S.N.; Mayer, P.; Meibom, A.; Baun, A. Microplastics as Vectors for Environmental Contaminants: Exploring Sorption, Desorption, and Transfer to Biota. Integr. Environ. Assess. Manag. 2017, 13, 488–493.
- Caruso, G. Microplastics as Vectors of Contaminants. Mar. Pollut. Bull. 2019, 146, 921–924.
- Sgier, L.; Freimann, R.; Zupanic, A.; Kroll, A. Flow Cytometry Combined with ViSNE for the Analysis of Microbial Biofilms and Detection of Microplastics. Nat. Commun. 2016, 7, 11587.
- Wu, X.; Pan, J.; Li, M.; Li, Y.; Bartlam, M.; Wang, Y. Selective Enrichment of Bacterial Pathogens by Microplastic Biofilm. Water Res. 2019, 165, 114979.
- Sl, W.; Rc, T.; Ts, G. The Physical Impacts of Microplastics on Marine Organisms: A Review. Environ. Pollut. 2013, 178.
- Mattsson, K.; Ekvall, M.T.; Hansson, L.-A.; Linse, S.; Malmendal, A.; Cedervall, T. Altered Behavior, Physiology, and Metabolism in Fish Exposed to Polystyrene Nanoparticles. Environ. Sci. Technol. 2015, 49, 553–561.
- Chae, Y.; Kim, D.; Kim, S.W.; An, Y.-J. Trophic Transfer and Individual Impact of Nano-Sized Polystyrene in a Four-Species Freshwater Food Chain. Sci. Rep. 2018, 8, 284.
- Llorca, M.; Farré, M.; Karapanagioti, H.K.; Barceló, D. Levels and Fate of Perfluoroalkyl Substances in Beached Plastic Pellets and Sediments Collected from Greece. Mar. Pollut. Bull. 2014, 87, 286–291.
- Cedervall, T.; Hansson, L.-A.; Lard, M.; Frohm, B.; Linse, S. Food Chain Transport of Nanoparticles Affects Behaviour and Fat Metabolism in Fish. PLoS ONE 2012, 7, e32254.
- Vethaak, A.D.; Legler, J. Microplastics and Human Health. Science 2021, 371, 672–674.
- Li, J.; Qu, X.; Su, L.; Zhang, W.; Yang, D.; Kolandhasamy, P.; Li, D.; Shi, H. Microplastics in Mussels along the Coastal Waters of China. Environ. Pollut. 2016, 214, 177–184.
- Neves, D.; Sobral, P.; Ferreira, J.L.; Pereira, T. Ingestion of Microplastics by Commercial Fish off the Portuguese Coast. Mar. Pollut. Bull. 2015, 101, 119–126.
- Karami, A.; Golieskardi, A.; Keong Choo, C.; Larat, V.; Galloway, T.S.; Salamatinia, B. The Presence of Microplastics in Commercial Salts from Different Countries. Sci. Rep. 2017, 7, 46173.
- Liebezeit, G.; Liebezeit, E. Non-Pollen Particulates in Honey and Sugar. Food Addit. Contam. Part A 2013, 30, 2136–2140.
- Hernandez, L.M.; Xu, E.G.; Larsson, H.C.E.; Tahara, R.; Maisuria, V.B.; Tufenkji, N. Plastic Teabags Release Billions of Microparticles and Nanoparticles into Tea. Environ. Sci. Technol. 2019, 53, 12300–12310.
- Pivokonsky, M.; Cermakova, L.; Novotna, K.; Peer, P.; Cajthaml, T.; Janda, V. Occurrence of Microplastics in Raw and Treated Drinking Water. Sci. Total Environ. 2018, 643, 1644–1651.
- Koelmans, A.A.; Mohamed Nor, N.H.; Hermsen, E.; Kooi, M.; Mintenig, S.M.; De France, J. Microplastics in Freshwaters and Drinking Water: Critical Review and Assessment of Data Quality. Water Res. 2019, 155, 410–422.
- Oßmann, B.E.; Sarau, G.; Holtmannspötter, H.; Pischetsrieder, M.; Christiansen, S.H.; Dicke, W. Small-Sized Microplastics and Pigmented Particles in Bottled Mineral Water. Water Res. 2018, 141, 307–316.
- Zuccarello, P.; Ferrante, M.; Cristaldi, A.; Copat, C.; Grasso, A.; Sangregorio, D.; Fiore, M.; Oliveri Conti, G. Exposure to Microplastics (<10 μm) Associated to Plastic Bottles Mineral Water Consumption: The First Quantitative Study. Water Res. 2019, 157, 365–371.
- Rist, S.; Carney Almroth, B.; Hartmann, N.B.; Karlsson, T.M. A Critical Perspective on Early Communications Concerning Human Health Aspects of Microplastics. Sci. Total Environ. 2018, 626, 720–726.
- Eyles, J.; Alpar, O.; Field, W.N.; Lewis, D.A.; Keswick, M. The Transfer of Polystyrene Microspheres from the Gastrointestinal Tract to the Circulation after Oral Administration in the Rat. J. Pharm. Pharmacol. 1995, 47, 561–565.
- Jani, P.; Halbert, G.W.; Langridge, J.; Florence, A.T. Nanoparticle Uptake by the Rat Gastrointestinal Mucosa: Quantitation and Particle Size Dependency. J. Pharm. Pharmacol. 1990, 42, 821–826.
- Wang, W.; Zhang, J.; Qiu, Z.; Cui, Z.; Li, N.; Li, X.; Wang, Y.; Zhang, H.; Zhao, C. Effects of Polyethylene Microplastics on Cell Membranes: A Combined Study of Experiments and Molecular Dynamics Simulations. J. Hazard. Mater. 2022, 429, 128323.
- Forte, M.; Iachetta, G.; Tussellino, M.; Carotenuto, R.; Prisco, M.; De Falco, M.; Laforgia, V.; Valiante, S. Polystyrene Nanoparticles Internalization in Human Gastric Adenocarcinoma Cells. Toxicol. Vitr. 2016, 31, 126–136.
- Zhou, J.; Wen, Y.; Marshall, M.R.; Zhao, J.; Gui, H.; Yang, Y.; Zeng, Z.; Jones, D.L.; Zang, H. Microplastics as an Emerging Threat to Plant and Soil Health in Agroecosystems. Sci. Total Environ. 2021, 787, 147444.
- Dong, C.-D.; Chen, C.-W.; Chen, Y.-C.; Chen, H.-H.; Lee, J.-S.; Lin, C.-H. Polystyrene Microplastic Particles: In Vitro Pulmonary Toxicity Assessment. J. Hazard. Mater. 2020, 385, 121575.
- Lu, K.; Lai, K.P.; Stoeger, T.; Ji, S.; Lin, Z.; Lin, X.; Chan, T.F.; Fang, J.K.-H.; Lo, M.; Gao, L.; et al. Detrimental Effects of Microplastic Exposure on Normal and Asthmatic Pulmonary Physiology. J. Hazard. Mater. 2021, 416, 126069.
- Li, X.; Zhang, T.; Lv, W.; Wang, H.; Chen, H.; Xu, Q.; Cai, H.; Dai, J. Intratracheal Administration of Polystyrene Microplastics Induces Pulmonary Fibrosis by Activating Oxidative Stress and Wnt/β-Catenin Signaling Pathway in Mice. Ecotoxicol. Environ. Saf. 2022, 232, 113238.
- Lu, Y.; Zhang, Y.; Deng, Y.; Jiang, W.; Zhao, Y.; Geng, J.; Ding, L.; Ren, H. Uptake and Accumulation of Polystyrene Microplastics in Zebrafish (Danio Rerio) and Toxic Effects in Liver. Environ. Sci. Technol. 2016, 50, 4054–4060.
- Ding, J.; Zhang, S.; Razanajatovo, R.M.; Zou, H.; Zhu, W. Accumulation, Tissue Distribution, and Biochemical Effects of Polystyrene Microplastics in the Freshwater Fish Red Tilapia (Oreochromis Niloticus). Environ. Pollut. 2018, 238, 1–9.
- Browne, M.A.; Dissanayake, A.; Galloway, T.S.; Lowe, D.M.; Thompson, R.C. Ingested Microscopic Plastic Translocates to the Circulatory System of the Mussel, Mytilus Edulis (L). Environ. Sci. Technol. 2008, 42, 5026–5031.
- Cajaraville, M.P.; Pal, S.G. Morphofunctional Study of the Haemocytes of the Bivalve Mollusc Mytilus Galloprovincialis with Emphasis on the Endolysosomal Compartment. Cell Struct. Funct. 1995, 20, 355–367.
- Banaee, M.; Soltanian, S.; Sureda, A.; Gholamhosseini, A.; Haghi, B.N.; Akhlaghi, M.; Derikvandy, A. Evaluation of Single and Combined Effects of Cadmium and Micro-Plastic Particles on Biochemical and Immunological Parameters of Common Carp (Cyprinus Carpio). Chemosphere 2019, 236, 124335.
- Wang, J.; Li, Y.; Lu, L.; Zheng, M.; Zhang, X.; Tian, H.; Wang, W.; Ru, S. Polystyrene Microplastics Cause Tissue Damages, Sex-Specific Reproductive Disruption and Transgenerational Effects in Marine Medaka (Oryzias Melastigma). Environ. Pollut. 2019, 254, 113024.
- Sun, M.; Ding, R.; Ma, Y.; Sun, Q.; Ren, X.; Sun, Z.; Duan, J. Cardiovascular Toxicity Assessment of Polyethylene Nanoplastics on Developing Zebrafish Embryos. Chemosphere 2021, 282, 131124.
- Canesi, L.; Ciacci, C.; Bergami, E.; Monopoli, M.P.; Dawson, K.A.; Papa, S.; Canonico, B.; Corsi, I. Evidence for Immunomodulation and Apoptotic Processes Induced by Cationic Polystyrene Nanoparticles in the Hemocytes of the Marine Bivalve Mytilus. Mar. Environ. Res. 2015, 111, 34–40.
