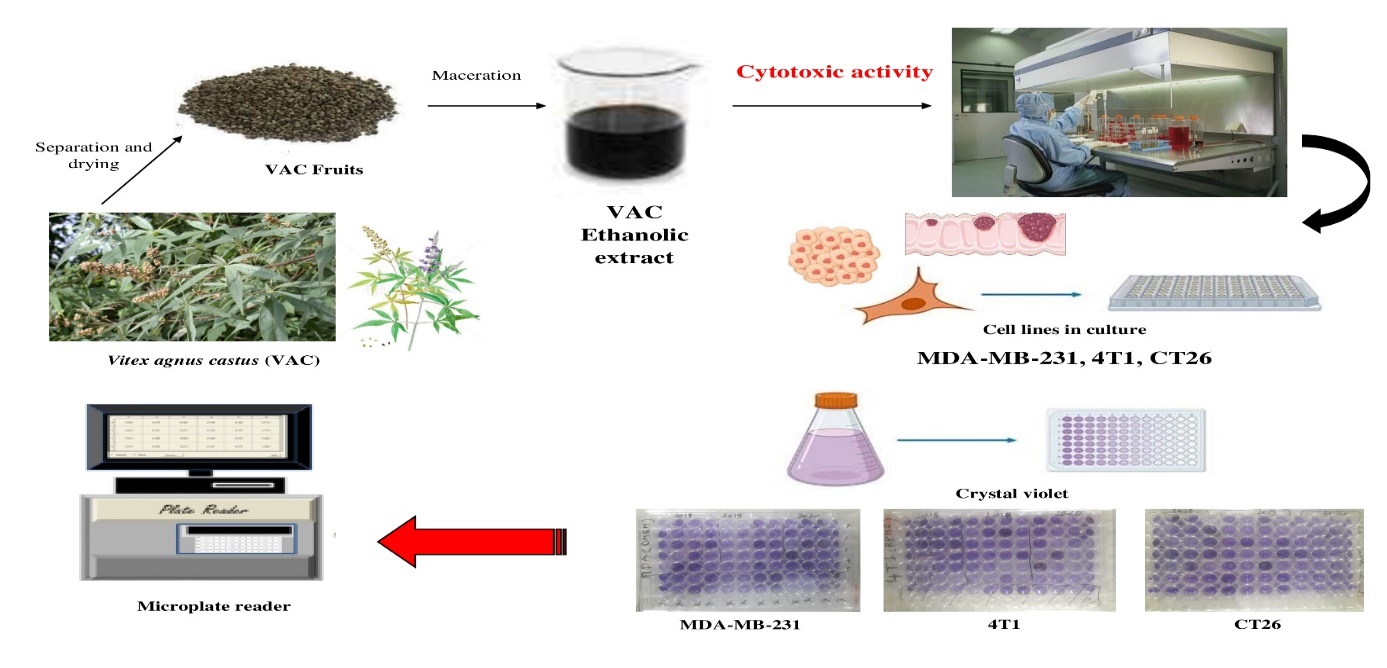
Vitex agnus-castus (VAC), native to the Mediterranean region, possesses antioxidant and radical scavenging properties that are involved in the development of carcinogenesis. The objective of our study was to investigate the cytotoxic activity of extracts from VAC fruits in vitro. Breast cancer (MDA-MB-231 and 4T1) and colon cancer (CT26) were selected for this research
- Vitex agnus-castus
- cytotoxic activity
- MDA-MB-231
- 4T1
- CT26
- Cell lines
In Vitro Cytotoxic Activity of Vitex Agnus Castus L. on MDA-MB-231, 4T1 and CT26 Cell Lines
Sarah El ayni 1*, Mouna Matouk 2, Soumaya kilani 2,3, Leila Chekir-Ghedira 2, Wahiba Douki 1
1Research laboratory of Vulnerability to Psychoses, LR05ES10, Faculty of medicine, University of Monastir, Avenue Avicenne, Monastir, 5019, Tunisia.
2Unit of Bioactive Natural Substances and Biotechnology, UR17ES49, Faculty of Dentistry, 5000 Monastir, Tunisia.
3Department of Pharmaceutical Sciences A, Faculty of Pharmacy, 5000 Monastir, Tunisia.
Mouna Matouk2
2 Unit of Bioactive Natural Substances and Biotechnology, UR17ES49, Faculty of Dentistry, 5000 Monastir, Tunisia.
E-mail : maatoukmouna@yahoo.fr
Telephone number: +21650584371
Soumaya Kilani2,3
2 Unit of Bioactive Natural Substances and Biotechnology, UR17ES49, Faculty of Dentistry, 5000 Monastir, Tunisia.
3Department of Pharmaceutical Sciences A, Faculty of Pharmacy, 5000 Monastir, Tunisia.
E-mail : soumaya.kilani@laposte.net
Telephone number: +21698977766
Leila Chekir-Ghedira2
2 Unit of Bioactive Natural Substances and Biotechnology, UR17ES49, Faculty of Dentistry, 5000 Monastir, Tunisia.
E-mail : leila.chekir@laposte.net
Wahiba Douki1
1 Research laboratory of Vulnerability to Psychoses, LR05ES10, Faculty of medicine, University of Monastir, Avenue Avicenne, Monastir, 5019, Tunisia.
E-mail : douki.wahiba@gmail.com
Telephone number: +21698404738
Correspondence
Sarah El ayni 1*
1 Research laboratory of Vulnerability to Psychoses, LR05ES10, Faculty of medicine, University of Monastir, Avenue Avicenne, Monastir, 5019, Tunisia.
Faculty of Pharmacy Rue Ibn Sina, 5000, Monastir, Tunisia
E-mail: sarahelayni281291@gmail.com
Telephone number: +216 22907325
ORCID: 0000-0002-5101-0474
Abstract
Vitex agnus-castus (VAC), native to the Mediterranean region, possesses antioxidant and radical scavenging properties that are involved in the development of carcinogenesis. The objective of our study was to investigate the cytotoxic activity of extracts from VAC fruits in vitro. Breast cancer (MDA-MB-231 and 4T1) and colon cancer (CT26) were selected for this research. The fruits grown in the three years 2018, 2019 and 2020 were dried at room temperature for one month. After grinding, they were cold macerated, evaporated and freeze-dried. Cytotoxicity evaluation was based on the detection of cell viability of the three lines MDA-MB-231, 4T1 and CT26 in the presence of different dilutions of the VAC extract by Crystal Violet. The absorbance (DO) was measured at 600 nm by plate spectrophotometry. The results showed significant inhibition of cell proliferation in all three lines for all extracts in a dose-dependent manner. Cell proliferation was significantly lower compared to untreated control cells with IC50s of 267; 162.5 and 196.8 µg/ml on average respectively. The VAC fruit extracts showed cytotoxic activity allowing to take advantage of these extracts as an anticancer drug which needs further anticancer studies in vivo.
Graphical abstract
Key words: Vitex agnus-castus, cytotoxic activity, MDA-MB-231, 4T1, CT26, Crystal Violet.
Abbreviations: VAC: Vitex agnus castus; IC50: half-maximal inhibitory concentration;
- Introduction
Current cancer treatment options, particularly surgery, chemotherapy and radiation therapy, or any combination of these approaches, are increasing in number and often work by intervening with DNA synthesis and mitosis. These drugs are non-selective because they damage normal healthy tissue and can cause serious, unexpected and undesirable side effects, such as decreased appetite, malaise and severe psychological consequences [1].
Traditional remedy and chemotherapy can reason severe aspect effects and lengthen a patient's existence by just a few years at exceptional. Consequently, it is vital to apply alternative standards [2]. Conventional therapy and chemotherapy can motive critical side consequences and lengthen an affected person's life by way of only a few years at first-rate. Consequently, it's far important to apply alternative ideas. In a constant quest to discover ingredients that may evidently assist heal ailment and keep health, humans are searching for safe haven on the supply, nature, an inexhaustible source of bioactive compounds. Therefore, there may be a want to apply alternative standards or techniques for cancer prevention which might be useful to human health without any apparent side outcomes, and these medicinal materials are usually derived from various wild plants [3].
Oxidative pressure and infection are related to the improvement and proliferation of most cancers cells [4] . Nevertheless, several authors have proven that natural supplements are rich in compounds that cope with this issue via their anticancer, antioxidant, and anti-inflammatory consequences [5].
Really worth mentioning is the genus Vitex, which belongs to the Verbenaceae family and consists of about 250 species disbursed in tropical and subtropical regions throughout the world [6]. Vitex agnus-castus L. (VAC) is a deciduous shrub that is native to tropical and temperate zones, the Mediterranean region, central Asia, and Southern Europe [7]. This plant facilitates with many illnesses together with PMS, sexual disorder and additionally allows with ache comfort [8].
In this case, we were interested in the fruit extracts of VAC in order to investigate the cytotoxic activity against Breast cancer (MDA-MB-231 and 4T1) and colon cancer (CT26) cells to current anticancer treatment and preventing cancer development.
- Results
All extracts from VAC fruits exhibited dose-dependent cytotoxic effects toward all cancer cell lines.





(a)
(b)
(c)
(d)
(e)
(f)
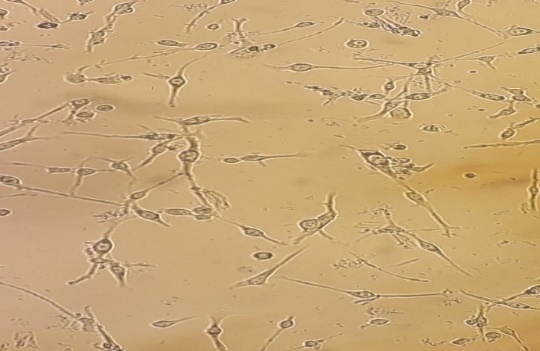
Figure 1. Morphologies of MDA-MB-231, 4T1 and CT26 cell lines untreated (a), (b), (c) and treated with 500 µg/ml VAC fruit extract (d), (e), (f) respectively.
The cells of MDA-MB-231, 4T1 and CT26 underwent a change in morphology after the addition of the VAC fruit extract. Indeed, we observed an exchange inclusive of a shrinking in their shapes and a loss of cell contact, membrane discoloration and compaction (Figure 1). We also studied the have an impact on of VAC fruit extract awareness on cell viability.
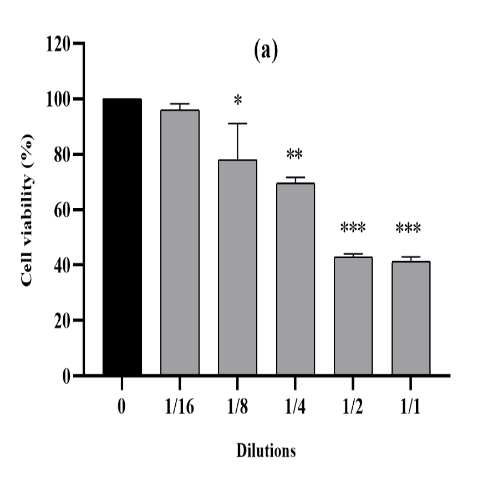
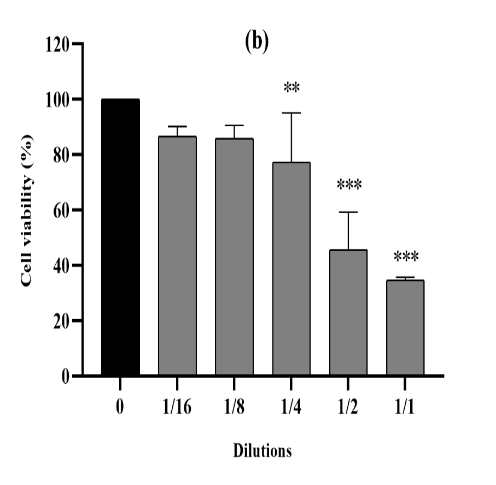
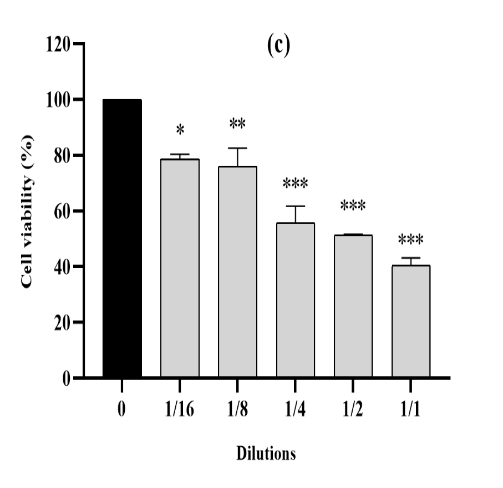
Figure 2. The cytotoxic effect of VAC ethanolic extract against MDA-MB-231 cells from the three consecutive seasons 2018 (a), 2019 (b) and 2020 (c) at different dilutions (1/1 to 1/16 from an initial concentration of 500 µg/ml). The effect was measured by the CV cell viability assay. The extract was cytotoxic to the cells in a dose-dependent manner. Each data point represents the mean of three independent experiments ± SD. *, were significantly different from the control (p < 0.05)
The shape of the curve suggests substantial inhibition of cell proliferation within the MDA-MB-231 mobile line for all ethanolic extracts of VAC 2018-2019 and 2020 in a dose-dependent manner (figure 2). Despite the fact that the inhibition of cellular viability became determined with all of the concentrations used, the extracts of the years 2018,2019 and 2020 had been able to lower the viability up to 41%, 34% and 40%, respectively on the concentration of 500 µg/ml as compared to the corresponding control group (untreated reference cells).
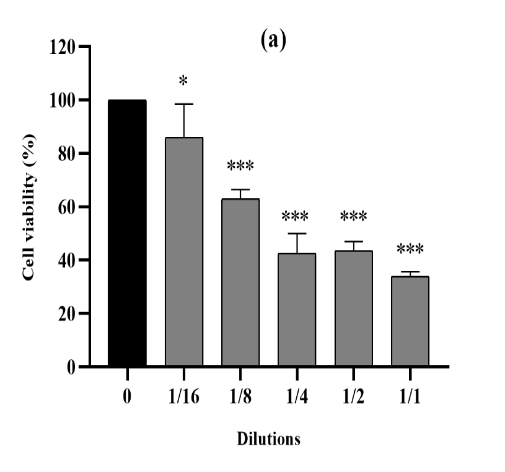
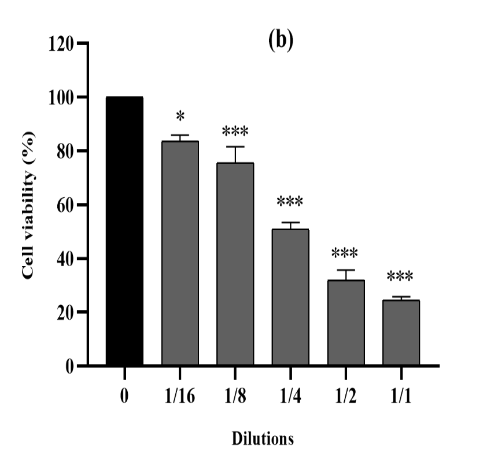
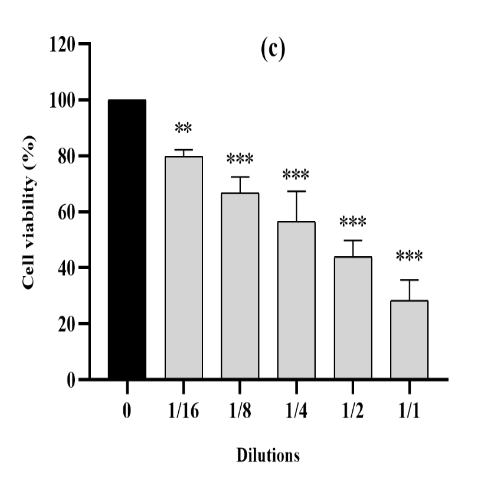
Figure 3. The cytotoxic effect of VAC ethanolic extract against 4T1 cells from the three seasons 2018 (a), 2019 (b) and 2020 (c) at different dilutions (1/1 to 1/16 from an initial concentration of 500 µg/ml). The effect was measured by the CV cell viability assay. The extract was cytotoxic to the cells in a dose-dependent manner. Each data point represents the mean of three independent experiments ± SD. *, were significantly different from the control (p < 0.05).
cytotoxic activity was also mentioned within the treatment of 4T1 cellular lines for all three extracts for the duration of 3 seasons (Figure 3), in comparison to untreated cells (p<0.0001). Moreover, for those cell lines, the three ethanolic extracts in 2018,2019 and 2020 have been capable of increase, respectively, their viability up to 34%, 25% and 28%. The maximum inhibition of proliferation become finished as much as 71% on common of the 3 years at the highest concentration (500 µg/mL).
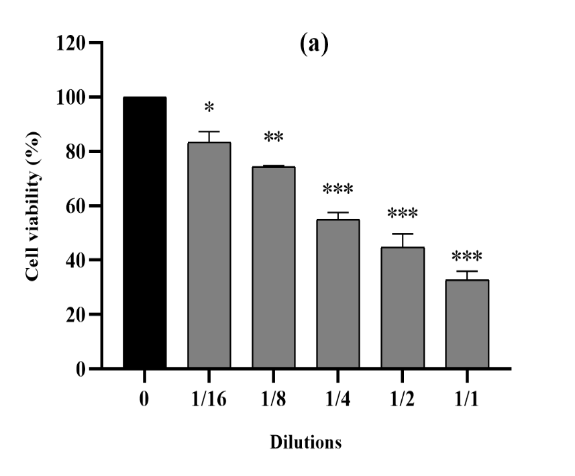


Figure 4. The cytotoxic effect of VAC ethanolic extract against CT26 cells from the three consecutive seasons 2018 (a), 2019 (b) and 2020 (c) at different dilutions (1/1 to 1/16 from an initial concentration of 500 µg/ml). The effect was measured by the CV cell viability assay. The extract was cytotoxic to the cells in a dose-dependent manner. Each data point represents the mean of three independent experiments ± SD. *, were significantly different from the control (p < 0.05).
For cell proliferation of the CT26 (Figure 4), we also revealed significant inhibition under the effect of VAC fruit extracts. Cell proliferation was significantly lower (p<0.0001) compared to untreated reference cells and we obtained a cell cytotoxicity that averaged 71% at the 500 µg/mL concentration for all three seasons.
The difference in cytotoxic activity between seasons is due to the change in soil climate and their differences in altitude (Table I).
Table I. Determination of IC50 of cytotoxic activity of the three lines treated with ethanolic extracts of VAC fruit grown in 2018, 2019 and 2020.
|
IC50 (µg/ml) |
|||
|
Cell lines |
2018 |
2019 |
2020 |
|
MDA-MB-231 |
263 ± 29 |
278. 6 ± 14.5 |
259.5 ± 39.46 |
|
4T1 |
178.8 ± 13.15 |
142.2 ± 13 |
166.7 ± 12.02 |
|
CT26 |
190.8 ± 17.82 |
187.3 ± 26.02 |
212.4 ± 55.15 |
The three cell lines showed distinct degrees of sensitivity towards the VAC fruit extract, nevertheless, cell line 4T1 was found to be the most sensitive with IC50s of 178.8 ±13.15; 142.2 ±13 and 166.7 ±12.02 µg/ml towards the extracts of the three years 2018,2019 and 2020 respectively.
- discussion
Other authors have also suggested that ethanolic extract of dried ripe fruit of VAC developing in Israel exhibited cytotoxic interest towards human cancer tissue cellular traces: Human Uterine Cervical Duct Fibroblasts (HCF), Ovarian most cancers (SKOV-three), Uterine Cervical Carcinoma (SKG-3a), Breast Carcinoma (MCF-7), Gastric Signet Ring Carcinoma (KATO-III), Colon Carcinoma (COLO 201) and small cell Lung Carcinoma (Lu-134-A-H) [9].
The Proliferation of a human colon carcinoma cell line, COLO 201, was efficaciously suppressed by way of apoptosis inside the presence of flavonoids, an ethanolic extract of VAC fruit. The induction of apoptosis was not inhibited with the aid of the presence of an antioxidant, N-acetyl-l-cysteine, while simplest the expression levels of the HO-1 gene multiplied most of the other standard oxidative pressure-related genes examined after Vitex treatment. Those outcomes advocate that Vitex treatment turns on a pathway associated with HO-1 gene activation, leading to induction of apoptosis in COLO 201 [10].
Ribat et al. Defined the antiproliferative and anticancer properties of a methanolic extract of VAC fruit, displaying that ordinary rat embryonic fibroblasts have been much less touchy than AMN3 cells (a mouse breast most cancers cell line), with IC50 values of 1324 µg/ml and 129 μg/mL, respectively [11].
Some other have a look at showed that methanol and water extracts of VAC exhibited enormous cytotoxic, DNA-destroying and apoptotic consequences on MCF-7 human breast most cancers cells and those cytotoxic activities of the extracts extended in a concentration dependent manner [12].
Insofar as the chemical composition of the extracts of VAC fruits consists of the presence of flavonoids and iridoids [13], this antiproliferative effect could be attributed to those compounds. Certainly, this anti-cancer impact of those polyphenols has been suggested by numerous authors.
Casticin, as the main component of Vitex, plays a key role in Vitex-mediated cytotoxicity in HL-60 cells (a human leukemia cell line) and it was proven by Kikuchi and associates in an observation that Vitex extract in combination with a specific inhibitor appears to be a potent treatment in the prevention and fate of cancer patients [14]. In other work, casticin extracted from CAV was found to have cytotoxic and immunomodulatory properties [15]. In addition, the cytotoxic impact of agnuside (one of the predominant iridoids in the composition of VAC) was tested on a colon cancer cell line (COLO 320 DM), revealing a cytotoxic activity of 76.1% at two hundred µg/mL [16].
The cytotoxic activity is attributed to the presence of particular components which includes polyphenols and flavonoids. Our chemical observes confirmed that flavonoids are the primary additives of our extract and as their antiproliferative interest against different cell lines has been defined previously, we agree with that they're responsible, at least in part, for the antiproliferative ability of our extract.
The organic interest of VAC is associated with the following pathways. Data from a observe via Weisskopf and associates validated that VAC consists of components that inhibit proliferation and induce apoptosis in a human prostate epithelial cell line (VAD-FMK), indicating a caspase-established apoptotic cellular death [17]. Inside the same way, the casticin brought on launch of cytochrome c is swiftly launched from mitochondria into the cytoplasm, activating caspase-9 and -3, observed by DNA fragmentation. Furthermore, the pan-caspase inhibitor Z-VAD-FMK and the caspase 9 inhibitor Z-LEHD-FMK blocked casticin-triggered apoptosis. Consequently, the casticin induces mitochondrial dysfunction and a cytochrome c-mediated, caspase-dependent apoptosis in human cervical cancer cells [18].
Additionally, the casticin triggers apoptosis via modulating Bcl-2 and different pro-survivals through the intrinsic pathway of apoptosis by way of down-regulating Bcl-2, Bcl-xL, surviving, and up-regulating Bax as evidenced in numerous tumors of gallbladder most cancers, esophageal most cancers, colon cancer, leukemia, and glioblastoma [19]. Other studies team additionally confirmed that casticin decreased Bcl-2 expression ranges and downregulated the Bcl-2/Bax expression ratio in K562 cells [20].
The cancers suppressor p53 is an important issue influencing the cellular response to drugs, especially its impact on growth inhibition and induction of apoptosis. Research have proven that casticin induces apoptosis in breast cancer cell lines and in p53 mutant or p53 null cervical cancer HeLa cells. Consequently, Casticin-prompted apoptosis in these cells is independent of p53 protein [21–23]. Vitexin (a VAC flavonoid) additionally will increase apoptosis in A549 (lung cancer cells) in a dose-established way, accompanied via decreased Bcl-2/Bax ratio and extended expression of cleaved caspase-3 [24].
- Materials and methods
- Chemicals and reagents
Dublecco’s Modified Eagle Medium (DMEM) was obtained from PAN, BIOTECH; Royal Park Memorial Institue (RPMI 1640) from Gibco, UK ; trypsin-EDTA, L-glutamine, fetal bovine serum (FBS), Dimethyl sulfoxide (DMSO) and penicillin / streptomycin (pen/strep) were purchased from Sigma-Aldrich, Germany, Crystal violet (CV) , ethanol and distilled water were used as solvents for the preparation of the extracts.
-
- Plant materials
The plant material (fruits) of VAC was collected in Ichkeul national park (37° 10′ 00″ N, 9° 40′ 00″ E) at a maximum altitude of 510 m of altitude in Tinja in the Bizerte region of northern Tunisia during October for 3 seasons 2018-2019-2020 (Table II).
Table II. The climatic characteristics of the three years 2018- 2019-2020 in Tinja Bizerte
|
Years |
T° max (°C) |
T° min (°C) |
T° Med (°C) |
Humidity (%) |
Rainfall (mm) |
|
Year 2018 |
22 |
17 |
19 |
67 |
52 |
|
Year 2019 |
22 |
17 |
19 |
69 |
20.75 |
|
Year 2020 |
25 |
21 |
23 |
65 |
19.63 |
-
- Cell lines used
We used three types of cell lines; human and murine breast cancer cells MDA-MB-231 and 4T1, respectively, and murine colon cancer cells CT26. These cells were obtained from American Type Culture collection (ATCC, Manassas, VA).
-
- Preparation of Plant Extracts
- Preparation of samples
- Preparation of Plant Extracts

 The curator of the park Ichkeul identified the plant material (Figure 5). Voucher specimens (Figure 6) have deposited at the Pharmacognosy laboratory of the Faculty of Pharmacy of Monastir. The samples were air-dried at room temperature.
The curator of the park Ichkeul identified the plant material (Figure 5). Voucher specimens (Figure 6) have deposited at the Pharmacognosy laboratory of the Faculty of Pharmacy of Monastir. The samples were air-dried at room temperature.
Figure 6. The fruits of the VAC
Figure 5. VAC collected from Tinja region of Bizerte north of Tunisia
-
-
- Preparation of Fruits extracts
-
Fruit powder of each tree was macerated in a solvent (ethanol, distilled water 30:70 v/v). The hydroalcoholic extract obtained was filtered using Finisterre C8 solid-phase extraction columns for fast and efficient sample cleaning and concentration prior to analysis.
The extracts were evaporated to remove ethanol and then freeze-dried to obtain a powder without traces of water. The residue was stored in a dark glass bottle for further processing.
-
- Cell Preparation and Culturing
Human breast cancer cells MDA-MB-231 were maintained in DMEM, murine breast cancer cells 4T1 and murine colon cancer cells CT26 were maintained in RPMI 1640.
All media were supplemented with 10% FBS and 1% pen/strep to make complete growth medium. Cells were cultured in a humidified atmosphere with 5% CO2 at 37 °C.
-
- Cell Viability Assay
The evaluation of cytotoxicity is based on the detection of the Crystal Violet (CV) of cell viability by its binding to ribose type molecules such as DNA of viable cells to give them a violet coloration which subsequently will be measured by spectrophotometry [25]. Cells were seeded at 1 x 105 cells/well in 96-well plate and incubated for 24h. Sequential dilutions from ethanol extracts of VAC were prepared by dissolving the samples in DMSO and diluting it with the medium used for each cell lines. The final DMSO concentration in the medium was 0.1%. After 24h, cells were treated with different dilutions of ethanol extract of VAC for three seasons consecutives (1/1 to 1/16 from an initial concentration of 500 µg/ml). The control wells received 200 µl from the medium with the final DMSO concentration, while the samples were added to the well in quadruplicates, incubated at 37°C with 5% CO2 for 24h. After aspiration of the culture medium, the plates were washed with phosphate-buffered saline (PBS). The cell mat is fixed by 100µl of glutaraldehyde diluted in sterile H2O (1%) which will be removed after 10 min of incubation.
The cells are then incubated in the presence of a solution of CV (1 mg/ml) diluted in sterile H2O at a rate of 50 µl/well for 30 min at room temperature and the accumulation of the dye in the cells is regularly monitored under an inverse microscope. The CV solution is removed and the cell mat is washed with distilled water to remove the excess. After drying, the solubilization of the CV retained by the cells is performed by adding acetic acid (33%) at 100µl/well. Then, the absorbance is measured at 600 nm by plate spectrophotometry. It corresponds to the absorbance of the living cells present in each well. Each extract concentration was tested in triplicate. The concentration curve of the tested sample as a function of the percentage of cell viability allows us to determine the 50% inhibitory concentration of cell multiplication in the presence of the tested substance (IC50).
-
- Statistical Analysis
Data are expressed as mean ± SD. For multiple comparisons, statistical analysis was performed by Two-way ANOVA analysis with the Tukey post hoc test. The concentration giving 50% inhibition (IC50) was calculated by linear regression. All statistics were realized by of Prism GraphPad version 8.0 for Windows (GraphPad Software, San Diego, CA, USA). Results were considered significant when p <0.05.
- Conclusion
The ethanolic extracts of VAC fruits showed significant antiproliferative activity on MDA-MB-231, 4T1 and CT26 cell lines. These data indicate a strong antiproliferative potential of VAC fruit extracts as anticancer substances and potential adjuvants in combination with chemotherapeutic agents in the treatment of cancer, thereby necessitating further in vivo anti-cancer studies.
Funding
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Acknowledgements
This work was supported by the Ichkeul National Park in Bizerte, Tunisia. Laboratory of chemical, galenic and pharmacological development of drugs (LR12ES09), Faculty of Pharmacy of Monastir, Laboratory of Biochemistry-Toxicology Research Laboratory "Vulnerability to Psychotic disorders" (LR05ES10), University Hospital of Monastir and The Unit of Bioactive Natural Substances and Biotechnology, UR17ES49, Faculty of Dentistry of Monastir in Tunisia.
Author Contributions
WD designed and supervised this work. SK supervised the experiments and data analysis. MM contributed to the experiments and activity determination. SE performed the analyses and wrote the manuscript. LCG contributed to the idea for this work.
Data availability statement
Not applicable.
Conflict of Interest
The authors declared that they have no conflict of interest.
References
1. Senapati, S.; Mahanta, A.K.; Kumar, S.; Maiti, P. Controlled Drug Delivery Vehicles for Cancer Treatment and Their Performance. Sig Transduct Target Ther 2018, 3, 1–19, doi:10.1038/s41392-017-0004-3.
2. Reddy, L.; Odhav, B.; Bhoola, K.D. Natural Products for Cancer Prevention: A Global Perspective. Pharmacology & Therapeutics 2003, 99, 1–13, doi:10.1016/S0163-7258(03)00042-1.
3. Grbović, F.; Stanković, M.S.; Ćurčić, M.; Đorđević, N.; Šeklić, D.; Topuzović, M.; Marković, S. In Vitro Cytotoxic Activity of Origanum Vulgare L. on HCT-116 and MDA-MB-231 Cell Lines. Plants 2013, 2, 371–378, doi:10.3390/plants2030371.
4. Reuter, S.; Gupta, S.C.; Chaturvedi, M.M.; Aggarwal, B.B. Oxidative Stress, Inflammation, and Cancer: How Are They Linked? Free Radical Biology and Medicine 2010, 49, 1603–1616, doi:10.1016/j.freeradbiomed.2010.09.006.
5. Makoshi, M.S.; Oladipo, O.O.; Gotep, J.G.; Forcados, G.E.; Shu, M.L.; Chinyere, C.N.; Yusuf, H.B.; Akanbi, B.O.; Samuel, A.L.; Ozele, N.; et al. Safety Evaluation of Acalypha Wilkesiana in Albino Rats and BHK-21 Cell Line. Comp Clin Pathol 2016, 25, 543–548, doi:10.1007/s00580-016-2224-2.
6. Rani, A.; Sharma, A.; Sharma, A.; Sharma, A. The Genus Vitex: A Review. Pharmacognosy Review 2013, 7, 188–198, doi:10.4103/0973-7847.120522.
7. Rashed, K.N. Antioxidant Activity of Different Extracts of Vitex Agnus-Castus (L.) and Phytochemical Profile. Research in Pharmacy 2013, 3.
8. Rafieian-Kopaei, M.; Movahedi, M. Systematic Review of Premenstrual, Postmenstrual and Infertility Disorders of Vitex Agnus Castus. Electron Physician 2017, 9, 3685–3689, doi:10.19082/3685.
9. Ohyama, K.; Akaike, T.; Hirobe, C.; Yamakawa, T. Cytotoxicity and Apoptotic Inducibility of Vitex Agnus-Castus Fruit Extract in Cultured Human Normal and Cancer Cells and Effect on Growth. Biological & Pharmaceutical Bulletin 2003, 26, 10–18, doi:10.1248/bpb.26.10.
10. Imai, M.; Kikuchi, H.; Denda, T.; Ohyama, K.; Hirobe, C.; Toyoda, H. Cytotoxic Effects of Flavonoids against a Human Colon Cancer Derived Cell Line, COLO 201: A Potential Natural Anti-Cancer Substance. Cancer Letters 2009, 276, 74–80, doi:10.1016/j.canlet.2008.10.036.
11. Ribat, Z.W.; Sahib, H.B.; Al-Shammari, A.M. Effect of Vitex Agnus Castus Fruits Methanol Extract against Murine Mammary Adenocarcinoma Cell Line (Amn3) and Rat Embryonic Fibroblast Normal Cell Line (Ref). 2019.
12. Sultan Aslantürk, Ö.; Aşkın Çelik, T. Antioxidant Activity and Anticancer Effect of Vitex Agnus-Castus L. (Verbenaceae) Seed Extracts on MCF–7 Breast Cancer Cells. Caryologia 2013, 66, 257–267, doi:10.1080/00087114.2013.850797.
13. Chen, S.-N.; Friesen, J.B.; Webster, D.; Nikolic, D.; van Breemen, R.B.; Wang, Z.J.; Fong, H.H.S.; Farnsworth, N.R.; Pauli, G.F. Phytoconstituents from Vitex Agnus-Castus Fruits. Fitoterapia 2011, 82, 528–533, doi:10.1016/j.fitote.2010.12.003.
14. Kikuchi, H.; Yuan, B.; Yuhara, E.; Imai, M.; Furutani, R.; Fukushima, S.; Hazama, S.; Hirobe, C.; Ohyama, K.; Takagi, N.; et al. Involvement of Histone H3 Phosphorylation via the Activation of P38 MAPK Pathway and Intracellular Redox Status in Cytotoxicity of HL-60 Cells Induced by Vitex Agnus-Castus Fruit Extract. International Journal of Oncology 2014, 45, 843–852, doi:10.3892/ijo.2014.2454.
15. Mesaik, M.A.; Azizuddin; Murad, S.; Khan, K.M.; Tareen, R.B.; Ahmed, A.; Atta-ur-Rahman; Choudhary, M.I. Isolation and Immunomodulatory Properties of a Flavonoid, Casticin from Vitex Agnus-Castus. Phytotherapy Research 2009, 23, 1516–1520, doi:https://doi.org/10.1002/ptr.2492.
16. Arokiyaraj; Kantharaj, P.; Pazhamalai, V.; Nyayiru Kannaian, U.P. Free Radical Scavenging and In Vitro Cytotoxiciity Activity of Agnuside from Vitex Agnus Castus (Verbenaceae). Journal of Pharmaceutical Research 2012, 5.
17. Weisskopf, M.; Schaffner, W.; Jundt, G.; Sulser, T.; Wyler, S.; Tullberg-Reinert, H. A Vitex agnus-castus Extract Inhibits Cell Growth and Induces Apoptosis in Prostate Epithelial Cell Lines. Planta Med 2005, 71, 910–916, doi:10.1055/s-2005-871235.
18. Chen, D.; Cao, J.; Tian, L.; Liu, F.; Sheng, X. Induction of Apoptosis by Casticin in Cervical Cancer Cells through Reactive Oxygen Species-Mediated Mitochondrial Signaling Pathways. Oncology Reports 2011, 26, 1287–1294, doi:10.3892/or.2011.1367.
19. Chan, E.W.C.; Wong, S.K.; Chan, H.T. Casticin from Vitex Species: A Short Review on Its Anticancer and Anti-Inflammatory Properties. Journal of Integrative Medicine 2018, 16, 147–152, doi:10.1016/j.joim.2018.03.001.
20. Wang, H.; Cai, B.; Cui, C.; Zhang, D.; Yang, B. [Vitexicarpin, a flavonoid from Vitex trifolia L., induces apoptosis in K562 cells via mitochondria-controlled apoptotic pathway]. Yao Xue Xue Bao 2005, 40, 27–31.
21. Csupor-Löffler, B.; Hajdú, Z.; Zupkó, I.; Réthy, B.; Falkay, G.; Forgo, P.; Hohmann, J. Antiproliferative Effect of Flavonoids and Sesquiterpenoids from Achillea Millefolium s.l. on Cultured Human Tumour Cell Lines. Phytotherapy Research 2009, 23, 672–676, doi:10.1002/ptr.2697.
22. Haïdara, K.; Zamir, L.; Shi, Q.-W.; Batist, G. The Flavonoid Casticin Has Multiple Mechanisms of Tumor Cytotoxicity Action. Cancer Letters 2006, 242, 180–190, doi:10.1016/j.canlet.2005.11.017.
23. Pirollo, K.F.; Bouker, K.B.; Chang, E.H. Does P53 Status Influence Tumor Response to Anticancer Therapies? Anti-Cancer Drugs 2000, 11, 419–432.
24. Liu, X.; Jiang, Q.; Liu, H.; Luo, S. Vitexin Induces Apoptosis through Mitochondrial Pathway and PI3K/Akt/MTOR Signaling in Human Non-Small Cell Lung Cancer A549 Cells. Biol Res 2019, 52, 7, doi:10.1186/s40659-019-0214-y.
25. Feoktistova, M.; Geserick, P.; Leverkus, M. Crystal Violet Assay for Determining Viability of Cultured Cells. Cold Spring Harb Protoc 2016, 2016, pdb.prot087379, doi:10.1101/pdb.prot087379.
