Nanobiotechnology has been used to bio-remediate or reclaim soil contaminated with organic and inorganic pollutants. The removal of pollutants from industrial wastes is a major challenge. The utilization of nanomaterials is gaining popularity, which might be accredited to their enhanced physical, chemical, and mechanical qualities. The development of advanced nanobiotechnological techniques involving the use of nanomaterials for the reclamation of polluted soils has indicated promising results and future hope for sustainable agriculture. By manufacturing environment-friendly nanomaterials, the industrial expenditure on decreasing the load of pollution might be reduced. A potential emerging domain of nanotechnology for eco-friendly production and cost reduction is “green biotechnology”, alongside the utilization of microorganisms in nanoparticle synthesis.
- bioremediation
- environmental pollution
- nanomaterials
- remediation
1. Introduction
2. An Evaluation of Nanobioremediation-Based Pollutant Reduction with a Focus on Microbe-Mediated Remediation
|
Nanoparticles |
Contaminant Remediated |
Factors of Performance and Removal Efficiency |
References |
|---|---|---|---|
|
Iron oxide nanoparticles with a polyvinyl pyrrolidone coating |
Cd, Pb |
The use of nanoparticles was combined with a bioremediation process driven by Halomonas sp. Halomonas sp. was inoculated for 48 h at 180 rpm and 28 °C in the Cd and Pb removal system. After 24 h, 100% removal was detected, whereas it took 48 h for Cd. |
[46] |
|
Industrial suspension of zero-valent iron (nZVI) at two dosages (1% and 10%) |
As |
The pH of the nZVI suspension was adjusted to 12.2 ± 0.1. Polyacrylic acid was utilized as a stabilizer to prevent the accumulation of nZVI in the suspension. The maximum amount of As immobilized in brownfield soil was 10% of nZVI. |
[47] |
|
Graphene oxide nanoparticles (nGOx) and nZVI |
Metals such as Cd, Pb, Zn, Cu, and As were found in As- and metal-contaminated soil. |
The application of nZVI and nGOx to contaminated soils had a significant influence on the availability of As and metals. nGOx immobilized Cu, Pb, and Cd while mobilizing As and P. In the case of nZVI, it successfully immobilized As and Pb (but not Cd) while increasing Cu’s availability. This study discovered that both NPs may work as techniques for immobilization and stabilization, which can then be used for phytoremediation. |
[48] |
|
Titanium oxide nanoparticles bonded to a chitosan nanolayer (NTiO2–NCh) |
Cd and Cu |
During the experiment, the pH was adjusted at 7.0. The elimination was aided by a microwave-enforced sorption technique that lasted 60–70 s. Cu and Cd were eliminated at a rate of 88.01% and 70.67%, respectively, when NTiO2–NCh was used. |
[49] |
|
Palladium (Pd), Pd NPs |
Cr |
Pd NPs were investigated as a bionanocatalyst. Pd NPs were shown to decrease Cr6+ completely in 12 h. To decrease 5.0 mol of Cr6+, 6.3 mg of Pd NPs was utilized. |
[50] |
|
Magnetic iron oxide nanoparticles (Fe3O4 NPs) were treated with Staphylococcus aureus and had their surfaces encapsulated in phthalic acid (n-Fe3O4–Phth–S. aureus) |
Cu, Ni, Pb |
The remediation efficiency of n-Fe3O4–Phth–S. aureus was reported to be 83.0–89.5% for Cu2+, 99.4–100% for Pb2+, and 92.6–7.5% for Ni2+. The researchers also discovered that n-Fe3O4–Phth–S. aureus was an effective biosorbent for removing pollutants. |
[51] |
|
ZnO NPs |
Cu, Cd, Cr, and Pb |
The maximum removal of Cr, Cu, and Pb by ZnO-NPs at 5 mg L−1 with Bacillus cereus and Lysinibacillus macroides was 60%, 70%, and 85%, respectively. The ideal pH for effective removal was 8.0. The elimination was reduced in the case of bacteria-mediated remediation, which was determined to be 83% and 70% with B. cereus, and 60% and 65% for L. macroides. |
[52] |
3. Soil Nanoremediation
4. Microorganism-Assisted Nanoremediation
5. Utilization of Nanomaterials for Micro-Remediation of Polluted Soils
This entry is adapted from the peer-reviewed paper 10.3390/su15010876
References
- Tian, H.; Qin, Y.; Niu, Z.; Wang, L.; Ge, S. Summer Maize Mapping by Compositing Time Series Sentinel-1A Imagery Based on Crop Growth Cycles. J. Indian Soc. Remote Sens. 2021, 49, 2863–2874.
- Yang, Y.; Chen, X.; Liu, L.; Li, T.; Dou, Y.; Qiao, J.; Wang, Y.; An, S.; Chang, S.X. Nitrogen fertilization weakens the linkage between soil carbon and microbial diversity: A global meta-analysis. Glob. Chang. Biol. 2022, 28, 6446–6461.
- Yang, Y.; Li, T.; Pokharel, P.; Liu, L.; Qiao, J.; Wang, Y.; An, S.; Chang, S.X. Global effects on soil respiration and its temperature sensitivity depend on nitrogen addition rate. Soil Biol. Biochem. 2022, 174, 108814.
- Khan, M.I.; Cheema, S.A.; Anum, S.; Niazi, N.K.; Azam, M.; Bashir, S.; Ashraf, I.; Qadri, R. Phytoremediation of agricultural pollutants. In Phytoremediation; Springer: Cham, Switzerland, 2020; pp. 27–81.
- Ghodszad, L.; Reyhanitabar, A.; Maghsoodi, M.R.; Lajayer, B.A.; Chang, S.X. Biochar affects the fate of phosphorus in soil and water: A critical review. Chemosphere 2021, 283, 131176.
- Karimi, H.; Mahdavi, S.; Asgari Lajayer, B.; Moghiseh, E.; Rajput, V.D.; Minkina, T.; Astatkie, T. Insights on the bioremediation technologies for pesticide-contaminated soils. Environ. Geochem. Health 2022, 44, 1329–1354.
- Souza, L.R.R.; Pomarolli, L.C.; da Veiga, M.A.M.S. From classic methodologies to application of nanomaterials for soil remediation: An integrated view of methods for decontamination of toxic metal (oid) s. Environ. Sci. Pollut. Res. 2020, 27, 10205–10227.
- Dai, J.; Feng, H.; Shi, K.; Ma, X.; Yan, Y.; Ye, L.; Xia, Y. Electrochemical degradation of antibiotic enoxacin using a novel PbO2 electrode with a graphene nanoplatelets inter-layer: Characteristics, efficiency and mechanism. Chemosphere 2022, 307, 135833.
- Keshavarzi, A.; Kumar, V. Spatial distribution and potential ecological risk assessment of heavy metals in agricultural soils of Northeastern Iran. Geol. Ecol. Landsc. 2020, 4, 87–103.
- Tan, Z.; Dong, B.; Xing, M.; Sun, X.; Xi, B.; Dai, W.; He, C.; Luo, Y.; Huang, Y. Electric field applications enhance the electron transfer capacity of dissolved organic matter in sludge compost. Environ. Technol. 2022, 1–11.
- Butu, M.; Stef, R.; Corneanu, M.; Butnariu, M. Mycoremediation: A Sustainable Approach for Pesticide Pollution Abatement. In Bioremediation and Biotechnology; Springer: Cham, Switzerland, 2020; Volume 2, pp. 73–96.
- Shahi Khalaf Ansar, B.; Kavusi, E.; Dehghanian, Z.; Pandey, J.; Asgari Lajayer, B.; Price, G.W.; Astatkie, T. Removal of organic and inorganic contaminants from the air, soil, and water by algae. Environ. Sci. Pollut. Res. 2022, 1–29.
- Kavusi, E.; Ansar, B.S.K.; Ebrahimi, S.; Sharma, R.; Ghoreishi, S.S.; Nobaharan, K.; Abdoli, S.; Dehghanian, Z.; Lajayer, B.A.; Senapathi, V. Critical review on phytoremediation of polyfluoroalkyl substances from environmental matrices: Need for global concern. Environ. Res. 2022, 217, 114844.
- Wang, J.; Shi, L.; Zhai, L.; Zhang, H.; Wang, S.; Zou, J.; Shen, Z.; Lian, C.; Chen, Y. Analysis of the long-term effectiveness of biochar immobilization remediation on heavy metal contaminated soil and the potential environmental factors weakening the remediation effect: A review. Ecotoxicol. Environ. Saf. 2021, 207, 111261.
- Hu, X.; Huang, X.; Zhao, H.; Liu, F.; Wang, L.; Zhao, X.; Gao, P.; Li, X.; Ji, P. Possibility of using modified fly ash and organic fertilizers for remediation of heavy-metal-contaminated soils. J. Clean. Prod. 2021, 284, 124713.
- Dong, Y.; Lin, H.; Zhao, Y.; Menzembere, E.R.G.Y. Remediation of vanadium-contaminated soils by the combination of natural clay mineral and humic acid. J. Clean. Prod. 2021, 279, 123874.
- Gu, W.; Li, X.; Li, Q.; Hou, Y.; Zheng, M.; Li, Y. Combined remediation of polychlorinated naphthalene-contaminated soil under multiple scenarios: An integrated method of genetic engineering and environmental remediation technology. J. Hazard. Mater. 2021, 405, 124139.
- Delangiz, N.; Aliyar, S.; Pashapoor, N.; Nobaharan, K.; Lajayer, B.A.; Rodríguez-Couto, S. Can polymer-degrading microorganisms solve the bottleneck of plastics’ environmental challenges? Chemosphere 2022, 294, 133709.
- Delangiz, N.; Varjovi, M.B.; Lajayer, B.A.; Ghorbanpour, M. Beneficial microorganisms in the remediation of heavy metals. In Molecular Aspects of Plant Beneficial Microbes in Agriculture; Elsevier: Amsterdam, The Netherlands, 2020; pp. 417–423.
- Atigh, Z.B.Q.; Sardari, P.; Moghiseh, E.; Lajayer, B.A.; Hursthouse, A.S. Purified montmorillonite as a nano-adsorbent of potentially toxic elements from environment: An overview. Nanotechnol. Environ. Eng. 2021, 6, 12.
- Yousefi, S.R.; Alshamsi, H.A.; Amiri, O.; Salavati-Niasari, M. Synthesis, characterization and application of Co/Co3O4 nanocomposites as an effective photocatalyst for discoloration of organic dye contaminants in wastewater and antibacterial properties. J. Mol. Liq. 2021, 337, 116405.
- Mahdi, M.A.; Yousefi, S.R.; Jasim, L.S.; Salavati-Niasari, M. Green synthesis of DyBa2Fe3O7. 988/DyFeO3 nanocomposites using almond extract with dual eco-friendly applications: Photocatalytic and antibacterial activities. Int. J. Hydrogen Energy 2022, 47, 14319–14330.
- Maghsoodi, M.R.; Lajayer, B.A.; Hatami, M.; Mirjalili, M.H. Challenges and opportunities of nanotechnology in plant-soil mediated systems: Beneficial role, phytotoxicity, and phytoextraction. Adv. Phytonanotechnol. 2019, 2019, 379–404.
- Khalkhal, K.; Asgari Lajayer, B.; Ghorbanpour, M. An overview on the effect of soil physicochemical properties on the immobilization of biogenic nanoparticles. In Biogenic Nano-Particles and their Use in Agro-Ecosystems; Springer: Singapore, 2020; pp. 133–160.
- Tratnyek, P.G.; Johnson, R.L. Nanotechnologies for environmental cleanup. Nano Today 2006, 1, 44–48.
- Patil, S.S.; Shedbalkar, U.U.; Truskewycz, A.; Chopade, B.A.; Ball, A.S. Nanoparticles for environmental clean-up: A review of potential risks and emerging solutions. Environ. Technol. Innov. 2016, 5, 10–21.
- Rajput, V.D.; Minkina, T.; Upadhyay, S.K.; Kumari, A.; Ranjan, A.; Mandzhieva, S.; Sushkova, S.; Singh, R.K.; Verma, K.K. Nanotechnology in the Restoration of Polluted Soil. Nanomaterials 2022, 12, 769.
- Srivastav, A.; Yadav, K.K.; Yadav, S.; Gupta, N.; Singh, J.K.; Katiyar, R.; Kumar, V. Nano-phytoremediation of pollutants from contaminated soil environment: Current scenario and future prospects. In Phytoremediation; Springer: Cham, Switzerland, 2018; pp. 383–401.
- Romeh, A.A.; Saber, R.A.I. Green nano-phytoremediation and solubility improving agents for the remediation of chlorfenapyr contaminated soil and water. J. Environ. Manag. 2020, 260, 110104.
- Singh, R.; Behera, M.; Kumar, S. Nano-bioremediation: An innovative remediation technology for treatment and management of contaminated sites. In Bioremediation of Industrial Waste for Environmental Safety; Springer: Singapore, 2020; pp. 165–182.
- Tran, T.D.; Dao, N.T.; Sasaki, R.; Tu, M.B.; Dang, G.H.M.; Nguyen, H.G.; Dang, H.M.; Vo, C.H.; Inigaki, Y.; van Nguyen, N. Accelerated remediation of organochlorine pesticide-contaminated soils with phyto-Fenton approach: A field study. Environ. Geochem. Health 2020, 42, 3597–3608.
- Gamallo, M.; Fernández, L.; Feijoo, G.; Moreira, M. Nano-based technologies for environmental soil remediation. In Nanomaterials for Sustainable Energy and Environmental Remediation; Elsevier: Amsterdam, The Netherlands, 2020; pp. 307–331.
- Liao, Y.; Yang, J. Remediation of vanadium contaminated soil by nano-hydroxyapatite. J. Soils Sediments 2020, 20, 1534–1544.
- Liu, Y.; Xu, K.; Cheng, J. Different nanomaterials for soil remediation affect avoidance response and toxicity response in earthworm (Eisenia fetida). Bull. Environ. Contam. Toxicol. 2020, 104, 477–483.
- Lv, Y.; Huang, S.; Huang, G.; Liu, Y.; Yang, G.; Lin, C.; Xiao, G.; Wang, Y.; Liu, M. Remediation of organic arsenic contaminants with heterogeneous Fenton process mediated by SiO2-coated nano zero-valent iron. Environ. Sci. Pollut. Res. 2020, 27, 12017–12029.
- Ahmad, A.; Ghufran, R.; Al-Hosni, T.K. Bioavailability of zinc oxide nano particle with fly ash soil for the remediation of metals by Parthenium hysterophorus. J. Environ. Health Sci. Eng. 2019, 17, 1195–1203.
- Sundararaghavan, A.; Mukherjee, A.; Suraishkumar, G.K. Investigating the potential use of an oleaginous bacterium, Rhodococcus opacus PD630, for nano-TiO2 remediation. Environ. Sci. Pollut. Res. 2020, 27, 27394–27406.
- Sarkar, A.; Sengupta, S.; Sen, S. Nanoparticles for soil remediation. In Nanoscience and Biotechnology for Environmental Applications; Springer: Cham, Switzerland, 2019; pp. 249–262.
- Wang, Y.; Liu, Y.; Zhan, W.; Zheng, K.; Lian, M.; Zhang, C.; Ruan, X.; Li, T. Long-term stabilization of Cd in agricultural soil using mercapto-functionalized nano-silica (MPTS/nano-silica): A three-year field study. Ecotoxicol. Environ. Saf. 2020, 197, 110600.
- Misra, M.; Ghosh Sachan, S. Nanobioremediation of heavy metals: Perspectives and challenges. J. Basic Microbiol. 2022, 62, 428–443.
- Abdi, O.; Kazemi, M. A review study of biosorption of heavy metals and comparison between different biosorbents. J. Mater Env. Sci. 2015, 6, 1386–1399.
- Ayangbenro, A.S.; Babalola, O.O. A new strategy for heavy metal polluted environments: A review of microbial biosorbents. Int. J. Environ. Res. Public Health 2017, 14, 94.
- Usman, M.; Farooq, M.; Wakeel, A.; Nawaz, A.; Cheema, S.A.; ur Rehman, H.; Ashraf, I.; Sanaullah, M. Nanotechnology in agriculture: Current status, challenges and future opportunities. Sci. Total Environ. 2020, 721, 137778.
- Abebe, B.; Murthy, H.A.; Amare, E. Summary on adsorption and photocatalysis for pollutant remediation: Mini review. J. Encapsulation Adsorpt. Sci. 2018, 8, 225–255.
- Desiante, W.L.; Minas, N.S.; Fenner, K. Micropollutant biotransformation and bioaccumulation in natural stream biofilms. Water Res. 2021, 193, 116846.
- Cao, X.; Alabresm, A.; Chen, Y.P.; Decho, A.W.; Lead, J. Improved metal remediation using a combined bacterial and nanoscience approach. Sci. Total Environ. 2020, 704, 135378.
- Gil-Díaz, M.; Diez-Pascual, S.; González, A.; Alonso, J.; Rodríguez-Valdés, E.; Gallego, J.; Lobo, M.C. A nanoremediation strategy for the recovery of an As-polluted soil. Chemosphere 2016, 149, 137–145.
- Baragaño, D.; Forján, R.; Welte, L.; Gallego, J.L.R. Nanoremediation of As and metals polluted soils by means of graphene oxide nanoparticles. Sci. Rep. 2020, 10, 1896.
- Mahmoud, M.E.; Abou Ali, S.A.; Elweshahy, S.M. Microwave functionalization of titanium oxide nanoparticles with chitosan nanolayer for instantaneous microwave sorption of Cu (II) and Cd (II) from water. Int. J. Biol. Macromol. 2018, 111, 393–399.
- Ha, C.; Zhu, N.; Shang, R.; Shi, C.; Cui, J.; Sohoo, I.; Wu, P.; Cao, Y. Biorecovery of palladium as nanoparticles by Enterococcus faecalis and its catalysis for chromate reduction. Chem. Eng. J. 2016, 288, 246–254.
- Mahmoud, M.E.; Abdou, A.E.; Mohamed, S.M.; Osman, M.M. Engineered staphylococcus aureus via immobilization on magnetic Fe3O4-phthalate nanoparticles for biosorption of divalent ions from aqueous solutions. J. Environ. Chem. Eng. 2016, 4, 3810–3824.
- Akhtar, N.; Khan, S.; Rehman, S.U.; Rehman, Z.U.; Khatoon, A.; Rha, E.S.; Jamil, M. Synergistic effects of zinc oxide nanoparticles and bacteria reduce heavy metals toxicity in rice (Oryza sativa L.) plant. Toxics 2021, 9, 113.
- Machado, S.; Pinto, S.; Grosso, J.; Nouws, H.; Albergaria, J.T.; Delerue-Matos, C. Green production of zero-valent iron nanoparticles using tree leaf extracts. Sci. Total Environ. 2013, 445, 1–8.
- Karn, B.; Kuiken, T.; Otto, M. Nanotechnology and in situ remediation: A review of the benefits and potential risks. Environ. Health Perspect. 2009, 117, 1813–1831.
- Gillham, R.W.; O’Hannesin, S.F. Enhanced degradation of halogenated aliphatics by zero-valent iron. Groundwater 1994, 32, 958–967.
- Roehl, K.E.; Meggyes, T.; Simon, F.; Stewart, D. Long-Term Performance of Permeable Reactive Barriers; Gulf Professional Publishing: Houston, TX, USA, 2005.
- Shih, Y.-h.; Tai, Y.-t. Reaction of decabrominated diphenyl ether by zerovalent iron nanoparticles. Chemosphere 2010, 78, 1200–1206.
- Elliott, D.W.; Lien, H.-L.; Zhang, W.-X. Degradation of lindane by zero-valent iron nanoparticles. J. Environ. Eng. 2009, 135, 317–324.
- Machado, S.; Pacheco, J.; Nouws, H.; Albergaria, J.T.; Delerue-Matos, C. Characterization of green zero-valent iron nanoparticles produced with tree leaf extracts. Sci. Total Environ. 2015, 533, 76–81.
- Shi, L.-n.; Zhang, X.; Chen, Z.-l. Removal of chromium (VI) from wastewater using bentonite-supported nanoscale zero-valent iron. Water Res. 2011, 45, 886–892.
- Trujillo-Reyes, J.; Peralta-Videa, J.; Gardea-Torresdey, J. Supported and unsupported nanomaterials for water and soil remediation: Are they a useful solution for worldwide pollution? J. Hazard. Mater. 2014, 280, 487–503.
- Wang, S.; Sun, H.; Ang, H.-M.; Tadé, M.O. Adsorptive remediation of environmental pollutants using novel graphene-based nanomaterials. Chem. Eng. J. 2013, 226, 336–347.
- Zhang, W. Nanoscale Iron Particles for Environmental Remediation: An Overview. J. Nanoparticle Res. 2003, 5, 323–332.
- Qiu, X.; Fang, Z.; Liang, B.; Gu, F.; Xu, Z. Degradation of decabromodiphenyl ether by nano zero-valent iron immobilized in mesoporous silica microspheres. J. Hazard. Mater. 2011, 193, 70–81.
- Xie, Y.; Cheng, W.; Tsang, P.E.; Fang, Z. Remediation and phytotoxicity of decabromodiphenyl ether contaminated soil by zero valent iron nanoparticles immobilized in mesoporous silica microspheres. J. Environ. Manag. 2016, 166, 478–483.
- Su, H.; Fang, Z.; Tsang, P.E.; Zheng, L.; Cheng, W.; Fang, J.; Zhao, D. Remediation of hexavalent chromium contaminated soil by biochar-supported zero-valent iron nanoparticles. J. Hazard. Mater. 2016, 318, 533–540.
- Gong, Y.; Liu, Y.; Xiong, Z.; Kaback, D.; Zhao, D. Immobilization of mercury in field soil and sediment using carboxymethyl cellulose stabilized iron sulfide nanoparticles. Nanotechnology 2012, 23, 294007.
- Mahanty, S.; Chatterjee, S.; Ghosh, S.; Tudu, P.; Gaine, T.; Bakshi, M.; Das, S.; Das, P.; Bhattacharyya, S.; Bandyopadhyay, S. Synergistic approach towards the sustainable management of heavy metals in wastewater using mycosynthesized iron oxide nanoparticles: Biofabrication, adsorptive dynamics and chemometric modeling study. J. Water Process Eng. 2020, 37, 101426.
- Govarthanan, M.; Jeon, C.-H.; Jeon, Y.-H.; Kwon, J.-H.; Bae, H.; Kim, W. Non-toxic nano approach for wastewater treatment using Chlorella vulgaris exopolysaccharides immobilized in iron-magnetic nanoparticles. Int. J. Biol. Macromol. 2020, 162, 1241–1249.
- Noman, M.; Shahid, M.; Ahmed, T.; Niazi, M.B.K.; Hussain, S.; Song, F.; Manzoor, I. Use of biogenic copper nanoparticles synthesized from a native Escherichia sp. as photocatalysts for azo dye degradation and treatment of textile effluents. Environ. Pollut. 2020, 257, 113514.
- Cheng, Y.; Dong, H.; Lu, Y.; Hou, K.; Wang, Y.; Ning, Q.; Li, L.; Wang, B.; Zhang, L.; Zeng, G. Toxicity of sulfide-modified nanoscale zero-valent iron to Escherichia coli in aqueous solutions. Chemosphere 2019, 220, 523–530.
- Cornu, J.-Y.; Huguenot, D.; Jézéquel, K.; Lollier, M.; Lebeau, T. Bioremediation of copper-contaminated soils by bacteria. World J. Microbiol. Biotechnol. 2017, 33, 26.
- Wu, G.; Kang, H.; Zhang, X.; Shao, H.; Chu, L.; Ruan, C. A critical review on the bio-removal of hazardous heavy metals from contaminated soils: Issues, progress, eco-environmental concerns and opportunities. J. Hazard. Mater. 2010, 174, 1–8.
- Huang, D.-L.; Zeng, G.-M.; Feng, C.-L.; Hu, S.; Jiang, X.-Y.; Tang, L.; Su, F.-F.; Zhang, Y.; Zeng, W.; Liu, H.-L. Degradation of lead-contaminated lignocellulosic waste by Phanerochaete chrysosporium and the reduction of lead toxicity. Environ. Sci. Technol. 2008, 42, 4946–4951.
- Xu, P.; Zeng, G.M.; Huang, D.L.; Lai, C.; Zhao, M.H.; Wei, Z.; Li, N.J.; Huang, C.; Xie, G.X. Adsorption of Pb (II) by iron oxide nanoparticles immobilized Phanerochaete chrysosporium: Equilibrium, kinetic, thermodynamic and mechanisms analysis. Chem. Eng. J. 2012, 203, 423–431.
- Polti, M.A.; García, R.O.; Amoroso, M.J.; Abate, C.M. Bioremediation of chromium (VI) contaminated soil by Streptomyces sp. MC1. J. Basic Microbiol. 2009, 49, 285–292.
- He, Y.K.; Sun, J.G.; Feng, X.Z.; Czakó, M.; Márton, L. Differential mercury volatilization by tobacco organs expressing a modified bacterial merA gene. Cell Res. 2001, 11, 231–236.
- Zhang, Z.Y.; Pan, L.P.; Li, H.H. Isolation, identification and characterization of soil microbes which degrade phenolic allelochemicals. J. Appl. Microbiol. 2010, 108, 1839–1849.
- Ryoo, D.; Shim, H.; Canada, K.; Barbieri, P.; Wood, T.K. Aerobic degradation of tetrachloroethylene by toluene-o-xylene monooxygenase of Pseudomonas stutzeri OX1. Nat. Biotechnol. 2000, 18, 775–778.
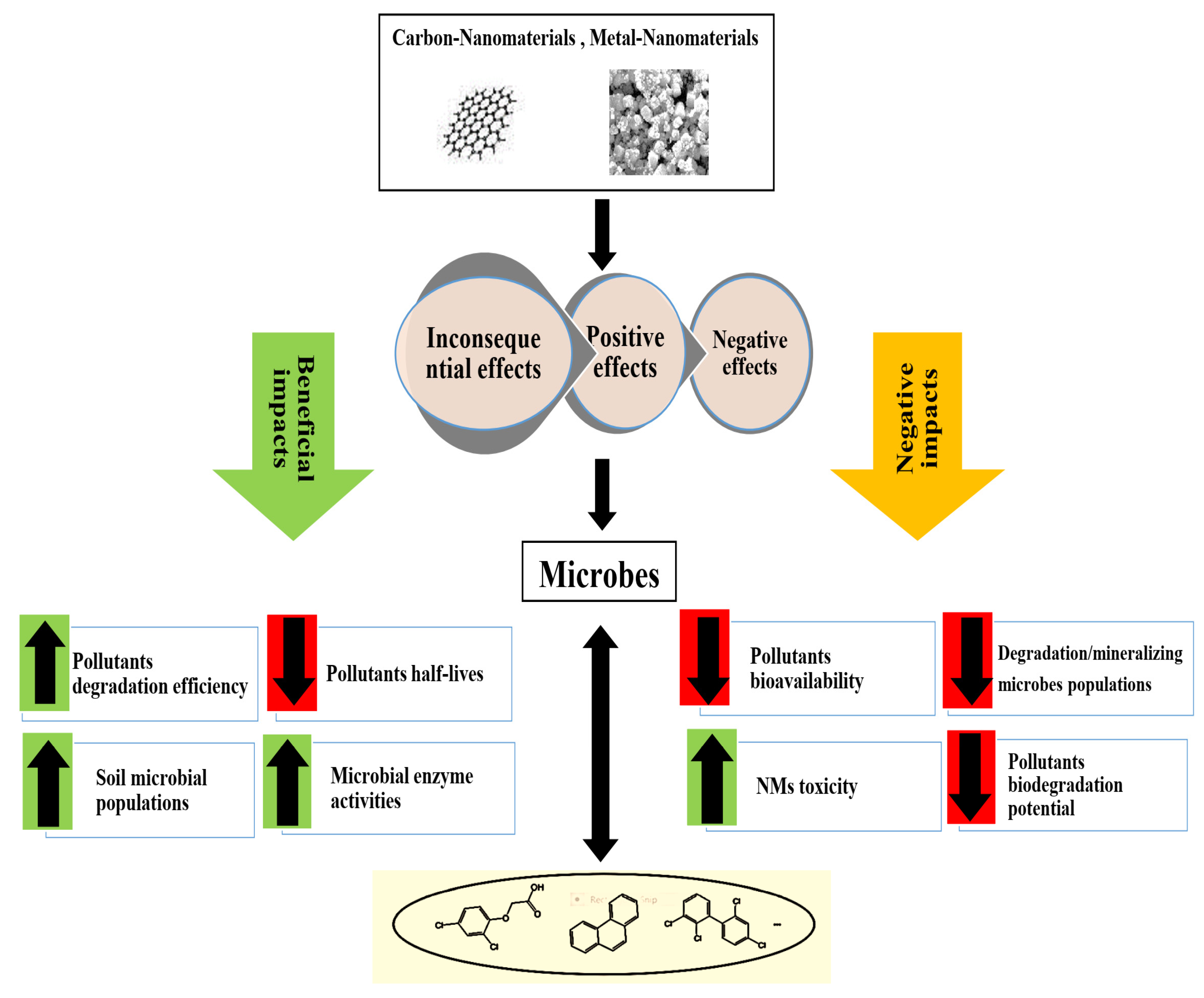
- Shrestha, B.; Acosta-Martinez, V.; Cox, S.B.; Green, M.J.; Li, S.; Cañas-Carrell, J.E. An evaluation of the impact of multiwalled carbon nanotubes on soil microbial community structure and functioning. J. Hazard. Mater. 2013, 261, 188–197.
- Fang, G.; Si, Y.; Tian, C.; Zhang, G.; Zhou, D. Degradation of 2, 4-D in soils by Fe3O4 nanoparticles combined with stimulating indigenous microbes. Environ. Sci. Pollut. Res. 2012, 19, 784–793.
- Tilston, E.L.; Collins, C.D.; Mitchell, G.R.; Princivalle, J.; Shaw, L.J. Nanoscale zerovalent iron alters soil bacterial community structure and inhibits chloroaromatic biodegradation potential in Aroclor 1242-contaminated soil. Environ. Pollut. 2013, 173, 38–46.
- Singh, J.; Lee, B.-K. Influence of nano-TiO2 particles on the bioaccumulation of Cd in soybean plants (Glycine max): A possible mechanism for the removal of Cd from the contaminated soil. J. Environ. Manag. 2016, 170, 88–96.
- Oyelami, A.O.; Semple, K.T. The impact of carbon nanomaterials on the development of phenanthrene catabolism in soil. Environ. Sci. Process. Impacts 2015, 17, 1302–1310.
- Xia, X.; Li, Y.; Zhou, Z.; Feng, C. Bioavailability of adsorbed phenanthrene by black carbon and multi-walled carbon nanotubes to Agrobacterium. Chemosphere 2010, 78, 1329–1336.
- Zhou, W.; Shan, J.; Jiang, B.; Wang, L.; Feng, J.; Guo, H.; Ji, R. Inhibitory effects of carbon nanotubes on the degradation of 14C-2, 4-dichlorophenol in soil. Chemosphere 2013, 90, 527–534.
