Electrospun polymer nanofibers constitute one of the most important nanomaterials with diverse applications. Nanofibers are classified as fibers with a ratio of length to thickness in the order of one thousand, or nanomaterials that have at least one dimension of 100 nm or less. A nanofiber with a diameter of ∼100 nm can have a specific surface area up to 1000 m2/g. Nanofibers can be produced by selecting the proper combination of polymers and additives, and using appropriate production techniques based on several essential characteristics that impact criteria of the intended particular application area. Electrospinning of polymer nanofibers is a widely used for investigation of their properties for uses in quite diverse applications. Attractive properties of electrospun nanofibers include the extremely high specific surface area, high porosity (typically 90%), light weight, controllable pore size, flexibility in surface functionalities, large permeability, excellent mechanical properties, high aspect ratio, and length up to many centimeters. Due to their exceptional characteristics, electrospun polymer nanofibers are used in many applications, which include biomedical technology, such as tissue engineering, wound healing and dressing, and drug delivery systems. In addition they have diverse uses in sensors and biosensors applications, air filtration, defense applications, energy devices and protective textiles.
- polymer nanofibers
- electrospinning
- polymer processing
- mechanical properties
- biomedical application
- energy storage separation
1. Polymers Used in Electrospinning
1.1. Natural and Synthetic Polymers
1.2. Composite Polymers/Copolymers
2. Electrospinning Process Parameters
-
Parameters related to the solution, such as polymer molecular weight, polymer concentration, surface tension, conductivity, solvent volatility, and viscosity.
-
Parameters related to processing such field strength, flow rate, tip-to-collector separation, applied voltage, placement and design of the needle tip, composition and geometry of the collector, and take-up velocity of the collector.
-
Properties related to environmental factors including temperature, humidity, and pressure.
2.1. Parameters Related to the Polymer Solution
2.1.1. Concentration of the Polymer
2.1.2. The Solvent
2.1.3. Electrical Conductivity
2.1.4. Viscosity
2.1.5. Molecular Weight
2.2. Parameters Related Electrospinning Equipment
2.2.1. Applied Voltage
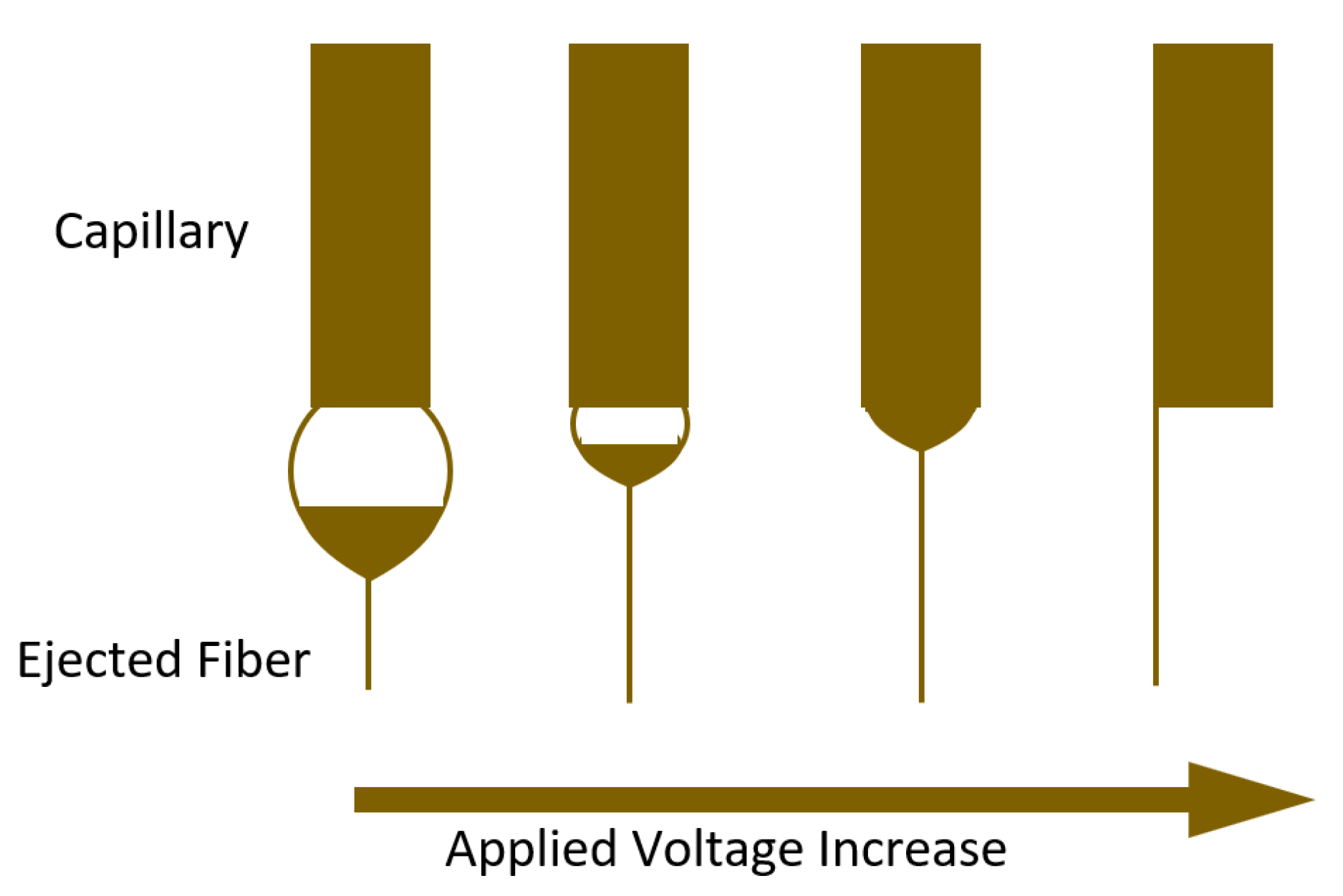
2.2.2. Feed Rate
2.2.3. Distance between Tip and Collector
2.3. Parameters Related to Environment
3. Properties of Polymer Nanofibers
3.1. Mechanical Properties
-
A manipulation system that precisely isolates, aligns, and grasps a single nanofiber on a frame without slipping or damaging.
-
A proper monitoring system to verify that nanofibers are not harmed by characterization tools such as scanning electron microscopes or transmission electron microscopes.
-
A sensitive force transducer having a range of nano- to micro-Newton range (n/μN range) resolution that can measure applied force in the n/μN range.
-
An actuator that is capable to load nanofibers until fracture, with high resolution (load unit: μN).
3.2. Chemical Properties
3.3. Thermal Properties
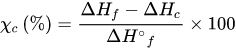
3.4. Electrical Properties
3.5. Optical Properties
4. Applications
4.1. Biomedical Applications
One of the most significant applications of polymer nanofibers is in the biomedical sector, particularly in the domains of medication delivery and tissue engineering. Given that biological molecules and nanoscale fibers have similar sizes, the latter are poised to perform well in simulating biological environments and natural extracellular matrices. Nanofibrous meshes exhibit enhanced biological activities, such as increased cell adhesion, differentiation, and proliferation, due to high porosity, large surface area to volume ratio, and interconnectivity of porous matrices comparable to macromolecular ones. Additionally, it is also conceivable for biological molecules to load for nutrients and wastes to exchange through pores [10][46]. The two main research directions in this field are explored below.
4.1.1. Tissue Engineering
Electrospun polymer nanofibers utilized as scaffolds in tissue engineering have attracted much attention. Due to comparable fibrous structure of nanofibrous made of biodegradable polymers with natural extracellular matrices, they operate so as to support cell proliferation, adhesion, and differentiation. As such, they possess great potential as scaffolds for tissue regeneration [47][48]. Collagen, keratin, elastin fibers and so on. obtained from extracellular matrix are the most often employed materials in this endeavor because they are inherently fibrous in nature and easily transformed into fibrous scaffolds.
4.1.2. Wound Healing and Dressing
In wound rehabilitation pursuit, the large porosity of electrospun fibers may provide additional structural space for accommodation of transplanted cells, promote cell migration and proliferation, and increase oxygen exchange and waste outflow. The tiny pore size of nanofibrous scaffolds can prevent dehydration and wound infection throughout the healing process. Additionally, tunable mechanical properties of electrospun nanofibers can maintain mechanical consistency between tissue engineering grafts and parent tissue and prevent the wound from wrinkling or shrinkage during implantation. A variety of synthetic biodegradable polymers such as polyglycolic acid, polylactic acid, polycaprolactone, and copolymers are generally used for skin tissue engineering because of their advantageous mechanical and biodegradable characteristics.
4.1.3. Drug Delivery Systems
Various drug delivery systems such as polymer micelles, liposomes, and nanofibers are studied to diminish the toxicity of dosage and increase therapeutic efficacy [49][50][51][52]. There is a potential for electrospun nanofibers to provide significant benefit due to flexibility in selection of materials and medications, encapsulation efficiency, and delivery of therapeutic agents, among others, which makes them appealing candidates in drug delivery, particularly for topical chemotherapy after surgery and in wound casing materials [53]. Electrospun nanofibers are utilized in precise and localized drug delivery systems thanks to their main advantages of large surface to volume ratios and well-interlinked, open porous structure. Numerous attempts have been undertaken to integrate bioactive compounds after electrospinning them, either chemically or physically, into the scaffolds. Techniques such as blending, co-axial electrospinning, and surface modification are utilized to load drugs into nanofibers.
4.2. Sensors and Biosensors
Significant advancements have been achieved in the manufacturing of extremely biological and chemical sensitive sensors in response to increasing demands for high-precision reliable detections in different and evolving applications in medicine and sophisticated manufacturing for targeted industries. Electrospun polymer nanofibers provide a fertile source of utilization in sensing applications. An optical sensor that was developed by the electrospinning of fluorescent polymer nanofibers showed three-time improved sensitivity magnitude as compared to film sensors for detection of mercury ions and nitro and ferric compounds. Conductive electrospun polymer nanofibers such as polyaniline nanowires are strong candidates for sensing applications due to their outstanding electrical characteristics [54][55][56].
4.3. Air Filtration
Various studies have suggested that electrospun nanofibers have the ability to capture such volatile organic compounds in air. Electrospun polymer nanofiber membranes have shown faster adsorption and desorption of volatile organic compounds compared to conventional activated carbon. The performance of filter membranes is significantly influenced by the structural properties of electrospun fibrous membranes. Fiber diameter and distribution, pore size distribution, surface area, basis, and density constitute the determining factors for filtering process effectiveness. Polymer nanofibers with smaller diameter will have a more accessible surface area, which will reduce pressure drop. Therefore, selecting an optimal electrospun nanofiber diameter is essential to maximizing filtration performance.
4.4. Defense Applications
4.5. Energy Devices
It was reported that nanofibers perform better than typical materials in devices for energy storage, harvesting, and conversion, offering good alternative materials for use in energy devices such as lithium-ion batteries, nanogenerators, and solar cells [57]. Nanofibers used in solar cells have shown high photoelectric conversion efficiency due to separation, efficient charge transmission, and high light absorption mainly due to a large specific surface area and high porosity. The large ratio of surface area to volume in nanofibers enhances formation of the nonwoven structure, which improves conductivity and gives rise to possible utilization of NF in batteries and fuel cells as a separation medium. Nanofiber-based electrodes in solar cells have shown high cycling stability and specific capacity.
This entry is adapted from the peer-reviewed paper 10.3390/polym15010065
References
- Kai, D.; Liow, S.S.; Loh, X.J. Biodegradable polymers for electrospinning: Towards biomedical applications. Mater. Sci. Eng. C 2014, 45, 659–670.
- Ashammakhi, N.; Wimpenny, I.; Nikkola, L.; Yang, Y. Electrospinning: Methods and development of biodegradable nanofibres for drug release. J. Biomed. Nanotechnol. 2009, 5, 19.
- Sill, T.J.; Von Recum, H.A. Electrospinning: Applications in drug delivery and tissue engineering. Biomaterials 2008, 29, 1989–2006.
- Stojanovska, E.; Canbay, E.; Pampal, E.S.; Calisir, M.D.; Agma, O.; Polat, Y.; Simsek, R.; Gundogdu, N.S.; Akgul, Y.; Kilic, A. A review on non-electro nanofibre spinning techniques. RSC Adv. 2016, 6, 83783–83801.
- Kulkarni, A.; Bambole, V.; Mahanwar, P. Electrospinning of polymers, their modeling and applications. Polym. Plastics Technol. Eng. 2010, 49, 427–441.
- Doshi, J.; Reneker, D.H. Electrospinning process and applications of electrospun fibers. J. Electrost. 1995, 35, 151–160.
- Bae, H.-S.; Haider, A.; Selim, K.; Kang, D.-Y.; Kim, E.-J.; Kang, I.-K. Fabrication of highly porous PMMA electrospun fibers and their application in the removal of phenol and iodine. J. Polym. Res. 2013, 20, 1–7.
- Haider, A.H.; Kang, S. A Comprehensive Review Summarizing the Effect of Electrospinning Parameters and Potential Applications of Nanofibers in Biomedical and Biotechnology. Arab. J. Chem. 2015, 10, 24.
- Bhardwaj, N.; Kundu, S.C. Electrospinning: A fascinating fiber fabrication technique. Biotechnol. Adv. 2010, 28, 325–347.
- Ingavle, G.C.; Leach, J.K. Advancements in electrospinning of polymeric nanofibrous scaffolds for tissue engineering. Tiss. Eng. Part B Rev. 2014, 20, 277–293.
- Baji, A.; Mai, Y.-W.; Wong, S.-C.; Abtahi, M.; Chen, P. Electrospinning of polymer nanofibers: Effects on oriented morphology, structures and tensile properties. Compos. Sci. Technol. 2010, 70, 703–718.
- Zafar, M.; Najeeb, S.; Khurshid, Z.; Vazirzadeh, M.; Zohaib, S.; Najeeb, B.; Sefat, F. Potential of electrospun nanofibers for biomedical and dental applications. Materials 2016, 9, 73.
- Nagam Hanumantharao, S.; Rao, S. Multi-functional electrospun nanofibers from polymer blends for scaffold tissue engineering. Fibers 2019, 7, 66.
- Jayaraman, K.; Kotaki, M.; Zhang, Y.; Mo, X.; Ramakrishna, S. Recent advances in polymer nanofibers. J. Nanosci. Nanotechnol. 2004, 4, 52–65.
- Tan, E.; Lim, C. Mechanical characterization of nanofibers—A review. Compos. Sci. Technol. 2006, 66, 1102–1111.
- Bazbouz, M.B.; Stylios, G.K. The tensile properties of electrospun nylon 6 single nanofibers Part B Polymer physics. J. Polymer Science Part Polym. Phys. 2010, 48, 1719–1731.
- Zhou, X.; Ding, C.; Cheng, C.; Liu, S.; Duan, G.; Xu, W.; Liu, K.; Hou, H. Mechanical and thermal properties of electrospun polyimide/rGO composite nanofibers via in-situ polymerization and in-situ thermal conversion. Eur. Polymer J. 2020, 141, 110083.
- Hang, F.; Lu, D.; Bailey, R.J.; Jimenez-Palomar, I.; Stachewicz, U.; Cortes-Ballesteros, B.; Davies, M.; Zech, M.; Bödefeld, C.; Barber, A.H. In situ tensile testing of nanofibers by combining atomic force microscopy and scanning electron microscopy. Nanotechnology 2011, 22, 365708.
- Gu, S.Y.; Wu, Q.L.; Ren, J.; Vancso, G.J. Mechanical properties of a single electrospun fiber and its structures. Macromolecul. Rapid Commun. 2005, 26, 716–720.
- Yang, L.; Fitie, C.F.; Van Der Werf, K.O.; Bennink, M.L.; Dijkstra, P.J.; Feijen, J. Mechanical properties of single electrospun collagen type I fibers. Biomaterials 2008, 29, 955–962.
- Lee, K.; Kim, H.; Khil, M.; Ra, Y.; Lee, D. Characterization of nano-structured poly (ε-caprolactone) nonwoven mats via electrospinning. Polymer 2003, 44, 1287–1294.
- Hansen, L.M.; Smith, D.J.; Reneker, D.H.; Kataphinan, W. Water absorption and mechanical properties of electrospun structured hydrogels. J. Appl. Polym. Sci. 2005, 95, 427–434.
- Wei, X.; Xia, Z.; Wong, S.-C.; Baji, A. Modelling of mechanical properties of electrospun nanofibre network. Int. J. ExpeR. Comput. Biomechan. 2009, 1, 45–57.
- Domaschke, S.; Zündel, M.; Mazza, E.; Ehret, A.E. A 3D computational model of electrospun networks and its application to inform a reduced modelling approach. Int. J. Solids Struct. 2019, 158, 76–89.
- Dong, Y. Nanostructures: Properties, Production Methods and Applications; Nova Science Publishers, Inc.: New York, NY, USA, 2013.
- Uslu, E.; Gavgali, M.; Erdal, M.O.; Yazman, Ş.; Gemi, L. Determination of mechanical properties of polymer matrix composites reinforced with electrospinning N66, PAN, PVA and PVC nanofibers: A comparative study. Materials Today Commun. 2021, 26, 101939.
- Duan, G.; Liu, S.; Hou, H. Synthesis of polyacrylonitrile and mechanical properties of its electrospun nanofibers. e-Polymers 2018, 18, 569–573.
- Sadrjahani, M.; Hoseini, S.; Mottaghitalab, V.; Haghi, A. Development and characterization of highly oriented pan nanofiber. Braz. J. Chem. Eng. 2010, 27, 583–589.
- Ratner, B.; Chilkoti, A.; Castner, D. Contemporary methods for characterizing complex biomaterial surfaces. In Biologically Modified Polymeric Biomaterial Surfaces; Springer: Dordrecht, The Netherlands, 1992; pp. 25–36.
- Zhang, Y.; Huang, Z.-M.; Xu, X.; Lim, C.T.; Ramakrishna, S. Preparation of core− shell structured PCL-r-gelatin bi-component nanofibers by coaxial electrospinning. Chem. Mater. 2004, 16, 3406–3409.
- Li, H.; Ke, Y.; Hu, Y. Polymer nanofibers prepared by template melt extrusion. J. Appl. Polym. Sci. 2006, 99, 1018–1023.
- Unser, A.M.; Xie, Y. Electrospinning of nanofibers. The Nanobiotechnology Handbook; CRC Press: Boca Raton, FL, USA, 2012; pp. 293–320.
- Ma, P.X.; Zhang, R. Synthetic nano-scale fibrous extracellular matrix. J. Biomed. Mater. Res. Off. J. Soc. Biomater. Japan. Soc. Biomater. Aust. Soc. Biomater. 1999, 46, 60–72.
- Kim, S.H.; Nam, Y.S.; Lee, T.S.; Park, W.H. Silk fibroin nanofiber. Electrospinning, properties, and structure. Polym. J. 2003, 35, 185–190.
- Peresin, M.S.; Habibi, Y.; Zoppe, J.O.; Pawlak, J.J.; Rojas, O.J. Nanofiber composites of polyvinyl alcohol and cellulose nanocrystals: Manufacture and characterization. Biomacromolecules 2010, 11, 674–681.
- Luzio, A.; Canesi, E.V.; Bertarelli, C.; Caironi, M. Electrospun polymer fibers for electronic applications. Materials 2014, 7, 906–947.
- Srivastava, Y.; Marquez, M.; Thorsen, T. Multijet electrospinning of conducting nanofibers from microfluidic manifolds. J. Appl. Polym. Sci. 2007, 106, 3171–3178.
- Prabhakaran, M.P.; Ghasemi-Mobarakeh, L.; Jin, G.; Ramakrishna, S. Electrospun conducting polymer nanofibers and electrical stimulation of nerve stem cells. J. Biosci. Bioeng. 2011, 112, 501–507.
- Nune, M.; Kumaraswamy, P.; Maheswari Krishnan, U.; Sethuraman, S. Self-assembling peptide nanofibrous scaffolds for tissue engineering: Novel approaches and strategies for effective functional regeneration. Curr. Protein Peptide Sci. 2013, 14, 70–84.
- Zhang, Y.; Rutledge, G.C. Electrical conductivity of electrospun polyaniline and polyaniline-blend fibers and mats. Macromolecules 2012, 45, 4238–4246.
- McCullen, S.D.; Stevens, D.R.; Roberts, W.A.; Ojha, S.S.; Clarke, L.I.; Gorga, R.E. Morphological, electrical, and mechanical characterization of electrospun nanofiber mats containing multiwalled carbon nanotubes. Macromolecules 2007, 40, 997–1003.
- Chronakis, I.S.; Grapenson, S.; Jakob, A. Conductive polypyrrole nanofibers via electrospinning: Electrical and morphological properties. Polymer 2006, 47, 1597–1603.
- Nagata, S.; Atkinson, G.M.; Pestov, D.; Tepper, G.C.; Mcleskey, J.T. Electrospun polymer-fiber solar cell. Adv. Mater. Sci. Eng. 2011, 2013, 1000.
- Babel, A.; Li, D.; Xia, Y.; Jenekhe, S.A. Electrospun nanofibers of blends of conjugated polymers: Morphology, optical properties, and field-effect transistors. Macromolecules 2005, 38, 4705–4711.
- Balderas, U.; Falcony, C.; Moggio, I.; Arias, E.; Mondragón, M. A photoluminescence study of electrospun fibers of conjugated poly blended with poly (9-vinylcarbazole). Polymer 2013, 54, 2062–2066.
- Oliveira, J.E.; Mattoso, L.H.; Orts, W.J.; Medeiros, E.S. Structural and morphological characterization of micro and nanofibers produced by electrospinning and solution blow spinning: A comparative study. Adv. Mater. Sci. Eng. 2013, 2013, 409572.
- Moon, S.; Farris, R.J. Strong electrospun nanometer-diameter polyacrylonitrile carbon fiber yarns. Carbon 2009, 47, 2829–2839.
- Rahmati, M.; Mills, D.K.; Urbanska, A.M.; Saeb, M.R.; Venugopal, J.R.; Ramakrishna, S.; Mozafari, M. Electrospinning for tissue engineering applications. Progr. Mater. Sci. 2021, 117, 100721.
- Zahmatkeshan, M.; Gheybi, F.; Rezayat, S.M.; Jaafari, M.R. Improved drug delivery and therapeutic efficacy of PEgylated liposomal doxorubicin by targeting anti-HER2 peptide in murine breast tumor model. Eur. J. Pharm. Sci. 2016, 86, 125–135.
- Asyikin binti Abdul Aziz, Z.; Ahmad, A.; Hamidah Mohd-Setapar, S.; Hassan, H.; Lokhat, D.; Amjad Kamal, M. Recent advances in drug delivery of polymeric nano-micelles. Curr. Drug Metabol. 2017, 18, 16–29.
- Luraghi, A.; Peri, F.; Moroni, L. Electrospinning for drug delivery applications: A review. J. Control. Release 2021, 334, 463–484.
- Zelkó, R.; Lamprou, D.A.; Sebe, I. Recent Development Of Electrospinning For Drug Delivery; MDPI: Basel, Switzerland, 2019; Volume 12, p. 5.
- Hu, X.; Liu, S.; Zhou, G.; Huang, Y.; Xie, Z.; Jing, X. Electrospinning of polymeric nanofibers for drug delivery applications. J. Control. Release 2014, 185, 12–21.
- Aussawasathien, D.; Dong, J.-H.; Dai, L. Electrospun polymer nanofiber sensors. Synth. Met. 2005, 154, 37–40.
- Yilmaz, O.E.; Erdem, R. Evaluating hydrogen detection performance of an electrospun CuZnFe2O4 nanofiber sensor. Int. J. Hydr. Energy 2020, 45, 26402–26412.
- Wang, W.; Huang, H.; Li, Z.; Zhang, H.; Wang, Y.; Zheng, W.; Wang, C. Zinc oxide nanofiber gas sensors via electrospinning. J. Am. Ceramic Soc. 2008, 91, 3817–3819.
- Zahmatkeshan, M.; Adel, M.; Bahrami, S.; Esmaeili, F.; Rezayat, S.M.; Saeedi, Y.; Mehravi, B.; Jameie, S.B.; Ashtari, K. Polymer-based nanofibers: Preparation, fabrication, and applications. In Handbook of Nanofibers; Springer: Berlin/Heidelberg, Germany, 2019; pp. 215–261.
