Micronutrients such as vitamins and trace elements are crucial for maintaining the health of all organisms. Micronutrients are involved in every cellular/biochemical process. They play roles in the proper heart and brain functioning, influence immunological response, and antioxidant defense systems. Therefore, prolonged deficiency in one or more of them leads to cardiovascular or neurodegenerative disorders. Keeping micronutrients at adequate levels is especially important for seniors. They are prone to deficiencies due to age-associated functional decline and often diet poor in nutrients. Moreover, the lack of micronutrients has an indirect impact on the genome. Their low levels reduce the activity of antioxidant enzymes and therefore inhibit the efficiency of defense against free radicals which may lead to the formation of DNA lesions. The more DNA damage in the genetic material, the faster aging at the cellular level and a higher risk of pathological processes (e.g. carcinogenesis). Supplementation of crucial antioxidative micronutrients such as selenium, zinc, vitamin C, and E seems to have the potential to positively influence the condition of an aging organism, including minimizing inflammation, enhancing antioxidative defense, and limiting the formation of DNA lesions. In consequence, it might lead to lowering the risk and incidence of age-related diseases such as cardiovascular diseases, neurodegenerative diseases, and malnutrition. In this article, we attempt to present the synergistic action of selected antioxidant micronutrients (vitamin C, vitamin E, selenium, and zinc) in inhibiting oxidative stress and DNA damage, which may impede the process of healthy aging.
- micronutrients
- aging
- DNA damage
- genome stability
- DNA repair
- neurodegenerative disorders
1. Introduction
Malnutrition, according to the definition, is an imbalance at the cellular level between the demand for nutrients and their intake. The fulfillment of nutritional needs supports proper growth and maintenance of the body’s vital functions [1]. Malnutrition should not be associated only with skinny people with anorectic appearance. It is a common clinical problem, with numerous causes, such as poverty, caring negligence, aging, chronic somatic diseases, or deliberate action to reduce weight. Therefore, it is difficult to estimate its actual scale. According to WHO, approximately 45% of children’s deaths (under age 5) are caused by malnutrition, mainly in destitute and middle-income countries. Interestingly, these countries have also an increasing percentage of overweight and obese children. In that case, eating high-energy but low-nutrient meals results in qualitative malnutrition. The same applies to adults for whom stressful lifestyle and increasingly inappropriate eating habits lead to dietary deficiencies. In obese people, malnutrition may result from a shortage of nutrients, vitamins, and microelements which are necessary for the proper functioning of the body. In addition, low-calorie or elimination diets (such as a vegan diet), if not adhered to a balanced eating plan, can cause malnutrition by increasing the risk of protein and vitamin deficiencies [2].
One of the more interesting forms of malnutrition is the so-called anorexia of aging—the loss of appetite associated with aging. It concerns approximately 25% of Europeans over 65 [3]. The risk of anorexia in seniors is higher due to physiological changes associated with aging, coexisting diseases, and medical treatments. Moreover, elderly people often struggle with psycho-social problems such as poverty or social isolation, which strongly predisposes them to loss of appetite. Anorexia is an independent risk factor for death in the older population [4]. As it is associated with qualitative and/or quantitative nutritional deficiencies, immune functions, metabolism, and antioxidative defense systems are weakened. Shortage of polyunsaturated fatty acids (PUFAs), vitamins, micro- and macroelements is partly responsible for geriatric syndromes such as frailty (i.e. drastic functional decline leading to multiorgan impairment). A special model of nutrition for longevity has not yet been identified, but a well-balanced diet with a sufficient quantity of nutrients promotes healthy aging in contrast to malnutrition which increases susceptibility to disease.
Although aging is a natural process and not a disease, older people are more prone to illness. The feeble and malnourished organism may suffer from, among others, impairment of the immune system. Inflammaging (low-grade chronic inflammation) develops with age and may speed up the deterioration processes and worsen other age-related disorders [5][6]. Age-specific conditions also coincide with chronic subliminal inflammation. Fagiolo et al. observed mononuclear peripheral blood cells in the elderly population compared to healthy young people [7]. Results show a higher concentration of tumor necrosis factor (TNF-α) and proinflammatory cytokines—interleukin 6 (IL-6) and interleukin 1 (IL-1) during 72h incubation with the mitogen. Additionally, elevated levels of IL-6 or TNF-α affect nutrition control centers, suppress appetite, change sensory sensations, and inhibit muscle protein synthesis [8], all of which may promote the development of anorexia. Subsequently, it might be a cause of inadequate nutrient intake or malfunctioning absorption.
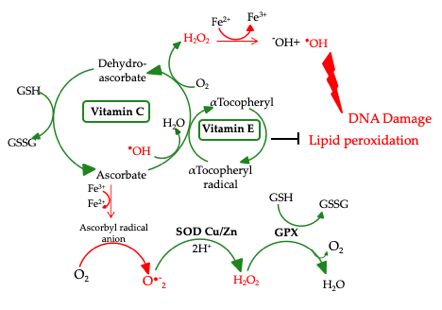
Malnourished older people may have deficits of most micronutrients, including zinc, selenium, vitamin C, vitamin E, riboflavin, electrolytes, and others. What’s important, micronutrients affect directly (e.g. vit. C and vit. E) or indirectly (e.g. selenium and zinc) the activity of antioxidant defense systems (e.g. antioxidant enzymes: superoxide dismutase (SOD), glutathione peroxidase (GPX) or catalase (CAT)) [9]. The proper operation of antioxidant systems is highly important for the whole organism. They protect the cell against endo- and exogenous pro-oxidative factors, including reactive oxygen species (ROS). The choice of the four presented micronutrients was dictated primarily by their antioxidant properties implemented through antioxidant enzymes. Selenium in the form of selenocysteine (Sec) is present in the active center of selenoproteins, including GPX, in which the main function is to neutralize H2O2 and organic peroxides. Vitamin E also neutralizes peroxides and its action is synergistic to vit. C, selenium, and zinc. Zinc is a component of enzymes from the group of SOD, which catalyze the dissolution reaction of the O2•- to H2O2 and O2. Vitamin C reduces ROS level (i.e. O2•-, •OH, and 1O2) but at the same time, it regenerates the oxidized form of vitamin E to its reduced form. Micronutrients selected for this review benefit from the presence of each other and sustain the overall effectiveness of antioxidant defense of the organism. The synergistic effect of the presented microelements on the antioxidant network is illustrated in Figure 1.
Figure 1. Antioxidant network.
SOD Cu/Zn: Cu/Zn superoxide dismutase; GPX: glutathione peroxidase; GSH: glutathione; GSSG: glutathione disulfide [9]
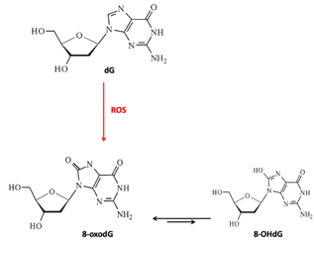
The proper operation of antioxidant systems is highly important for the whole organism. Antioxidants protect the cell against endo- and exogenous pro-oxidative factors, including ROS. Their levels increase especially in the case of physiological stress e.g. inflammatory processes, malnutrition, or anorexia [10]. Also, physiological decline connected with age may cause an increase in ROS levels. High ROS levels and lack of nutrients in the older body are even more dangerous as they accelerate aging and incidence of age-related disorders. ROS formed during physiological reactions are necessary for the proper gene expression and cell differentiation but in excess, ROS lead to the formation of DNA lesions and may impact the integrity of the nuclear and mitochondrial DNA (mtDNA). H2O2 does not pose a direct threat due to its moderate activity, but in certain conditions may be transformed into highly reactive •OH (Fig. 1) [11]. Due to its low redox potential guanine is the most susceptible to oxidation. The major type of guanine lesions is 8-oxo-7,8-dihydro-2’-deoxyguanosine (8-oxodG) and its enol form 8-hydroxy-2´-deoxyguanosine (8-OHdG)formed 105 times per day per cell (Fig. 2) [12].
Figure 2. Guanosine and its oxidative modifications.
dG: 2´-deoxyguanosine; 8-oxodG: 8-oxo-7,8-dihydro-2’-deoxyguanosine; 8-OHdG: 8-hydroxy-2´-deoxyguanosine; ROS: reactive oxygen species
Oxidative lesions are detected and corrected mainly by base excision repair (BER) which is the only mechanism able to correct single nucleotide’s lesions. DNA repair mechanisms are also affected by nutritional deficiencies which may lead to decreased repair efficiency and subsequent mutations resulting in pathological conditions (e.g. carcinogenesis or neurodegeneration) [13].
The review focuses on selected micronutrients involved in the DNA repair processes and antioxidant protection of the body in the case of malnutrition and anorexia-related deficiencies in the elderly population.
2. Biochemical aspects of selected micronutrients
The most common biochemical problems that affect patients with anorexia nervosa are dehydration or electrolyte disturbances due to insufficient supply of micro- and macroelements in the diet. Physiologically, the concentration of K+ ions is much higher inside the cell, while the concentration of Na+, Ca2+, and Cl- ions is higher in the extracellular space. In anorectic patients, hypokalaemia is a frequent problem. It is dangerous due to the possible consequences: cardiac arrhythmias, abnormal nerve conduction or paralysis in striated and smooth muscles, etc. [14]. Moreover, hypophosphatemia was also observed in patients with the anorexia-related condition, which affects phosphate functions (including ATP synthesis) and impairs glucose metabolism, enzyme phosphorylation, and acid-base management. The biochemical imbalance may lead to the development of e.g. osteomalation, rhabdomyolysis, central nervous system disorders, or hemolysis [15]. Other most important microelements which regulate the overall well-being of the human organism are trace elements and vitamins such as selenium, zinc, vitamin E, and C, which are discussed in this article.
Selenium
Selenium (Se) is present in the active centers of many proteins and enzymes in the form of Sec residues. Selenoproteins play crucial roles in the proper functioning of the whole organism; selenoprotein K (SELENOK, SELK) participates in the construction of protein-protein complexes, selenoprotein M (SELENOM, SELM, SEPM) is involved in the protection of neurons against oxidative stress, selenoprotein N (SELENON, SELN, SEPN1) is involved in the regeneration of skeletal muscle tissue [16]. Iodothyronine deiodinases (DIO1-3) involved in the formation of thyroid hormones are also an example of selenoproteins. Selenium is associated with an immuno-inflammatory and proinflammatory response. Its deficiency in endothelial cells results in reduced production of prostaglandins (PGI2, PGE2, and PGF2α). Selenoproteins also regulate macrophages’ migration and phagocytosis [17].
Selenoproteins have antioxidant properties and are involved in the regulation of the antioxidant defense system. A low level of Se causes reduced cells’ resistance to free radicals. In this context, selenoprotein P (SELENOP, SELP, SEPP1), thioredoxin reductase (TXNRD1, TRXR1) and glutathione peroxidases (GPX) are the most important. SELP acts as an antioxidant, while TRXR1 provides proper cell growth, DNA synthesis, replication, and apoptosis’ inhibition [18][19]. GPX are involved in the protection against oxidative damage by conversion of H2O2 to water (Fig. 1). Interestingly, the level of glutathione (GSH), which is a cofactor for the antioxidant enzymes (GPX and glutathione transferase) is reduced in people suffering from anorexia. Therefore, their ability to detoxify electrophilic metabolites and neutralize ROS is impaired [20]. It may lead to peroxidation of membrane lipids, oxidation of unsaturated fatty acids, reduced membranes’ fluidity/permeability, and, as a consequence, to pathological conditions such as atherosclerosis, diabetes mellitus, or rheumatoid arthritis [21].
Severe selenium deficiency often manifests as cardiomyopathies and heart failures which are often seen in anorexia patients. Kashin-Beck disease, a musculoskeletal disorder with abnormal bone development, growth inhibition, joint pain, and edema with reduced mobility, is caused by the Se shortage in the unbalanced diet. Therefore, monitoring the selenium level in the patient’s body and fluids can be helpful and is recommended from the therapeutic point of view. Lack of Se may also result in neurological symptoms (e.g. depression), which are often observed in people with anorexia or malnutrition [22][23].
Selenium is considered as a factor of prolonged life expectancy. Recently, Hammad et al. demonstrated the relationship between Se and replication senescence in human embryonic fibroblasts (WI-38). The authors point out that lack of Se is related to increased ROS levels in aging cells and decreased antioxidant defense (including the activity of selenoproteins) [24]. The addition of selenium increases the number of cell divisions and reduces aging markers (β-galactosidase (SABG) and heterochromatin foci (SAHF)), while its deficiency accelerates senescence and reduces the cell’s proliferative capacity [25]. Mice’s diet enriched with Se enhances the activity/level of SOD, GPX, and total antioxidant capacity (T-AOC) [26]. Selenium may also reduce oxidative stress in peripheral blood lymphocytes and therefore improves healthy aging [27].
The elderly population usually has reduced selenium levels [28][29]. Adding Se to the seniors’ diet may be an important factor in preventing age-related diseases and improving their quality of life (QoL). A study on 347 elders (age >80) show that low plasma selenium levels are correlated with high levels of IL-6 and C-reactive protein (CRP) [30]. Moreover, all-cause mortality is higher in people with low selenium (≤105.3 µg/L). The authors suggest that higher selenium levels have a positive effect on age-related inflammation. Interestingly, the synergistic effect of Se and vit. E in the quenching of the free radicals is observed. Patients with selenium-related diseases often have vitamin E deficit (discussed in further sections) [31]. Having in mind that excess of Se can induce adverse effects (diarrhea, fatigue, hair loss, joint pain) [32], the carefully planned and advised diet enriched in selenium may potentially improve seniors’ QoL.
Zinc
Zinc (Zn) is essential for the proper functioning of the cells. It plays an important role in transcription regulation. Zn deficiency often occurs in a malnourished organism and can lead to growth retardation, delayed pubescence, impaired wound healing, dermatitis, decreased appetite, and mental lethargy [33]. Zn is involved in metabolic processes e.g. immune response, neurobehavioral and physical development. Its deficiency impacts antibody production, cytokine production (interleukin 2 (IL-2) and interferon γ (IFNg)), cell signaling, proliferation, and function of B, T helper, and natural killer (NK) cells [34][35].
Moreover, zinc is present in zinc-finger domains of many proteins such as transcription factors, and regulatory proteins. The presence of Zn2+ ions is also crucial for the stability of DNA binding proteins because the zinc-finger domain is directly involved in the binding process of the nucleic acid molecule [36]. Moreover, zinc neutralizes the O2•- as a component of the Zn/Cu-SOD and is a crucial element (as part of a catalytic domain) of the metalloproteinases [18]. Metallothioenins (MTs) are a family of highly conserved cysteine-rich metalloproteins [37]. MTs have strong antioxidant properties—they can scavenge ROS and detoxify heavy metals ions [38]. The availability of microelements such as selenium or zinc regulates MTs production and cellular accumulation. MTs’ expression increases during stress conditions (e.g. inflammation). Interestingly, the so-called redox cycle with MTs is described. The sulfone group confers redox activity to the Zn-MT complex and can be oxidized and reduced with simultaneous release and binding of Zn in an oxidoreductive environment. Zinc released from MT is available to other molecules. This process is modulated by GSH and glutathione disulfide (GSSG). A more oxidized state results in the release of zinc and a more reduced state promotes MT stabilization [39]. The reduction of oxidized MTs restores its ability to bind Zn. The MT genes have been characterized as one of the few longevity genes. Transgenic mice with MT overexpression live longer. Yang et al. observed that their cardiomyocytes inhibit age-related cytochrome C release and generate lower levels of superoxides compared to control mice. The authors point to MTs' direct impact on cardiac aging and lifespan [40].
Zinc lowers the level of proinflammatory cytokines and markers of oxidative stress. Studies conducted on healthy adults (age 55-87) show that monocytic cells of zinc supplemented people generated significantly less TNF. 6-month supplementation led to a significant reduction in TNF levels (1897±1004 pg/mL to 1411±786 pg/mL) compared to the placebo group (1728±498 pg/mL to 2698±785 pg/mL). Moreover, there was a significant decrease in plasma oxidative stress markers (malondialdehyde (MDA), 4-hydroxyalkenals (HAE), and 8-OHdG) in the supplemented group, with no change in the placebo group (8-OHdG: 0.63±0.16 ng/mL to 0.50±0.14 ng/mL (p=0.030) in the supplemented group vs. 0.66±0.13 ng/mL to 0.68±0.13 ng/mL in the placebo group; MDA+HAE: 1.66±0.343 µmol/L to 1.35±0.18 µmol/L (p=0.0002) in the supplemented group vs. 1.70±0.30 µmol/L to 1.71±0.35 µmol/L in the placebo group) [41]. The authors conclude that zinc, as a non-mutagenic, relatively non-toxic, effective anti-inflammatory and antioxidative agent, may be beneficial in preventing chronic disorders associated with oxidative stress in the elderly population.
Vitamin E
Vitamins E is a group of fat-soluble compounds with strong antioxidant properties as they inhibit lipid peroxidation. Vitamin E (α-tocopherol, α-T, vit. E) has a synergistic effect with vitamin C, selenium, and zinc. As a constituent of the cellular membranes, vitamin E is the main antioxidant of PUFA. It inhibits oxidation of cellular macromolecules as donating electron interrupts the chain reaction of phospholipids’ oxidation in membranes at the propagation stage (Fig. 1). It can be postulated that by protecting cell membranes, vit. E delays cellular aging [42] and has a beneficial effect on vascular and cardiac function [43]. Moreover, its moiety is built into the ceramides that are part of the intercellular spaces of the stratum corneum and due to strong antioxidant properties, it protects the epidermis and therefore increases its resistance to UV radiation [44].
The shortage of vitamin E harms external cell bilayer and may lead to cancer, cardiovascular diseases, infectious and inflammatory processes. Vitamin E deficiency is well characterized by an isolated lack of vit. E in Ataxia with vitamin E deficiency (AVED). AVED is an autosomal recessive neurodegenerative disorder caused by a mutation in the α-tocopherol protein transfer gene (α-TTP) with clinical manifestations being progressive spinocerebellar ataxia, loss of deep sensation (proprioceptivity), and areflexia. However, high doses of vit. E (800 mg/day) inhibit the symptoms’ progression and may even reverse some neurological symptoms [45]. It the case of anorectic and malnourished people level of vit. E is often reduced e.g. due to insufficient intake [46][47]. Vit. E deficiency may result in neuropathies, progressive necrosis of the nervous system, and muscles. Patients with anorexia may also experience cardiovascular complications, arrhythmias, peripheral edema, and even sudden cardiac arrest [48].
There is no consistent evidence that a diet enriched with vitamin E protects against chronic diseases or cancer [49][50]. On the other hand, studies conducted on a population with very low levels of micronutrients due to poor living conditions indicate that vitamin-mineral supplementation (including vit. E) potentially reduces the risk of cancer [51]. It may seem that supplementation is effective in population with low intake and concentration of antioxidant nutrients such as older people; the potential benefits possibly outweigh the side effects [52].
Vitamin C
It is important to supply it with food as the human body cannot synthesize vitamin C (vit. C, ascorbic acid) due to the lack of L-gulonolactone oxidase. Vit. C is primarily involved in the synthesis of collagen, catecholamines, and L-carnitine [53]. It is a cofactor for numerous enzymes (e.g. hydrolases, oxygenases, and dioxygenases) and is involved in many metabolic processes (e.g. synthesis of adrenaline from tyrosine). Ascorbic acid, as a water-soluble antioxidant, acts as the body's primary defense against ROS occurring in the water phase. Vit. C leads to the formation of well-soluble ferrous salts, by reducing Fe3+ to Fe2+, which can be more easily absorbed from the gastrointestinal tract [54]. At the intracellular level, ascorbic acid might be considered an ideal antioxidant—it is present in the cell in the right quantity (it varies in different types of cells, fluids, and tissues [55]), neutralizes a large number of free radicals, and is regenerated to some extent [56]. Vitamin C inactivates free radicals and thus breaks the oxidative chain. Moreover, it strengthens the action and regeneration of α-tocopherol by reducing its radical formed after the reaction of tocopherol with free radical [42].
The level of vitamin C in the plasma decreases with age [57][58]. Clinical studies indicate a relationship between serum vit. C levels and the risk of cardiovascular disease (e.g. peripheral arterial disease or stroke). Patients with low vit. C level (27.8 µmol/L compared to control group 51.7 µmol/L; p <0.0001) have a significantly increased CRP (2.51 mg/L vs. control 4.80 mg/L; p <0.0001) and are at higher risk of developing fatal cardiovascular disease [59]. Also, serum vit. C levels are inversely associated with stroke incidence [60]. Similar observations apply to patients suffering from diabetes or hypertension, as their serum vitamin C levels are low [61]. On the other hand, studies show that daily vit. C intake does not reduce serious cardiovascular events, cancer outcomes, and cardiovascular mortality [62][63]. Nevertheless, it seems likely that population groups with low vit. C status may benefit from additional vitamin C intake [9]. It is especially visible in the case of seniors suffering from anorexia and therefore with an inadequate intake of vit. C; the high levels of oxidative damage should be observed in this group.
Moreover, vit. C is involved in the regulation of gene expression [64]. The authors show that ascorbate deficiency reduces the expression of the TET1-dependent (methylcytosine oxidase ten-eleven translocation proteins) genes crucial for germline development. Reproductive cells lacking vit. C have a different gene expression profile than controls because vit. C is a cofactor of TET hydroxylases (involved in the demethylation of DNA). Reduced expression of TET genes accompanies many types of cancers [65], which indicates the role of TET as tumor suppressors [66]. Vitamin C seems to be required for DNA demethylation and thus, proper epigenetic regulation. Studies indicate ascorbic acid as a DNA protector—a group of healthy subjects shows a significant decrease in the level of 8-oxodG (a marker of oxidative stress) in the plasma and urine after supplementation with 500 mg/day of vit. C [67]. Besides, gene expression analysis indicates that DNA repair processes are enhanced in cells treated with vitamin C which we discuss below [68].
3. Micronutrients, DNA damage, and repair
The human body changes with age. Aging is a set of complex biochemical and physiological phenomena determined by the molecular processes that occur at the cellular level. The aging process is influenced by numerous factors ranging from genetic predispositions to lifestyle choices (e.g. diet), performed kind of work and living conditions. It is a lifelong process constantly shaped by environmental factors which constitute more than 50% of overall human well-being [69]. Modern awareness about the diet as the major factor influencing our health goes as far as the XIXth century when the phrase “You are what you eat” originates. However, the changes occur at different times and intensities in various organs, and they reach the greatest intensity in old age.
According to one of the theories, aging is associated with the accumulation of oxidative changes in macromolecules and cellular structures [70]. Free radicals in physiological quantity play an important role in cell signaling [71][72] but in excess, they might damage the cell. Oxidation may impair the function of cell membranes, and proteins (e.g. enzymes and receptors), or may lead to the formation of DNA lesions. The brain is significantly affected by oxidative damage. Intensive metabolism, high content of fatty acids, and relatively low activity of antioxidant enzymes contribute to age-related neurodegenerative disease [73]. ROS are also generated in mitochondria as a part of the electron transport chain (ETC) when so-called electron leakage occurs. When radicals’ production is too high the function and structure of mitochondria are disrupted, including the integrity of the cristae and the inner membrane. Hindered mitochondrial function can induce a further increase in oxidative stress and subsequent DNA damage [74]. Moreover, the proximity of the respiratory chain may impair the integrity of mtDNA which leads to a decreased mitochondrial activity with age [75]. Maintaining stable concentrations of ROS in the cells is a major determinant of longevity and healthy aging. Cellular aging is also associated with extensive and irreversible DNA damage within telomeric and/or non-telomeric genome sequences [9][76][77].
Fundamental factors of aging include oxidative stress, cellular damage, low effectiveness of damage prevention, and inhibited DNA repair. The oxidative DNA lesions manifest as base modifications, strand breaks, or DNA adducts. The most common markers of oxidative DNA damage are 8-oxodG and 8-OHdG. Wolf et al. show that older rats have higher concentrations of 8-OHdG compared to the young ones [78]. The level of 8-OHdG increases with rats’ age in their heart, skeletal muscles, liver, peripheral blood, or brain. The most significant increase in 8-OHdG level is observed in the heart and peripheral blood lymphocytes (from 0.157 OD at 4 months to 0.370 OD at 12 months of age and 0.220 OD at 4 months to 0.550 OD at 24 months of age, respectively), which authors suggest is related to the DNA repair efficiency. The different study analyzes the level of 8-oxodG in human leukocytes in different age groups in correlation with the concentration of ascorbate in the plasma [79]. 8-oxodG level increases in leukocytes’ DNA with age. Authors conclude this may be related to the decrease in antioxidant defense with age (lower ascorbate level).
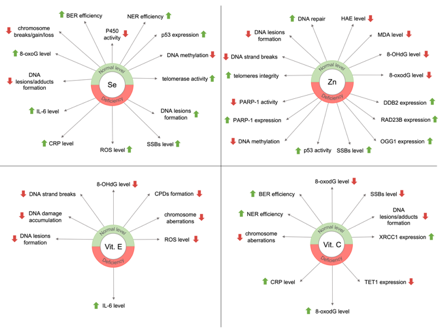
For chronic malnutrition the lack of antioxidants is pro-oxidative: it increases oxidative stress and impairs ROS neutralization via GSH [80]. Studies show that the consumption of antioxidative micronutrients reduces the level of DNA damage and/or improves DNA repair efficiency. Moreover, micronutrients are important for maintaining genome stability. Their deficiencies can lead to DNA damage formation similar to those resulting from radiation (DNA strands’ or chromosomes’ breaks) [81][82]. Figure 3 presents the main effects of selected micronutrients on genome stability concerning their deficiency or normal level in the body.
Figure 3. Effects of selected micronutrients on genome stability.
BER: base excision repair; NER: nucleotide excision repair; 8-oxoG: 8-oxo-7,8-dihydroguanine; 8-oxodG: 8-oxo-7,8-dihydro-2’-deoxyguanosine; 8-OHdG: 8-hydroxy-2´-deoxyguanosine; SSBs: single-strand breaks; P450: cytochrome P450; IL-6: interleukin 6; CRP: C-reactive protein; ROS: reactive oxygen species; CPDs: cyclobutane pyrimidine dimers; HAE: 4-hydroxyalkenals; MDA: malondialdehyde; PARP-1: poly[ADP-ribose] polymerase 1; OGG1: 8-oxoguanine glycosylase 1; DDB2: damage-specific DNA binding protein 2; RAD23B: RAD23 homolog B; XRCC1: X-ray repair cross-complementation group 1; TET1: methylcytosine oxidase ten-eleven translocation proteins
Selenium
Selenium is involved in the protection against the negative effects of ROS action. Se is a ROS scavenging agent and element of selenoproteins which catalyze reactions of ROS removal from the organism. However, its impact on genome stability is also known (Fig. 3). It may inhibit DNA adducts formation with e.g. carboplatin, polychlorinated biphenyl (PCB), or 7,12-dimethylbenz[α]anthracene (DMBA) and lower the number of chromosome breaks, gain, or loss resulting from carcinogens [83]. Moreover, selenium seems to support the repair of oxidative DNA damage. The potential of repair by the incision is significantly higher in protein extracts from cells pretreated with Se compared to the control (30% vs. 20% excision, respectively). Mice with Se deficit have upregulated genes induced by DNA damage, which suggests that Se deficiency may be a stress factor for the cell [84].
Another study examines the influence of selenium on the level of DNA damage in a group of 43 people (age 50-75) [85]. Subjects with the initial Se level below the average of 100 ng/ml have higher levels of DNA damage in peripheral blood lymphocytes. The authors suggest that serum Se levels should be kept above 100 ng/ml as DNA damage prevention. Se supplementation is also beneficial in patients with a BRCA1 mutation [86]. BRCA1 is involved in the repair of DNA double-stranded breaks (DSBs), acts as a tumor suppressor, and maintains genome stability. Urine collected from the supplemented (300 µg/day of sodium selenite) BRCA1 mutation carriers contains a higher level of 8-oxo-7,8-dihydroguanine (8-oxoG, a product of BER system) in comparison to the non-supplemented group. The median of 8-oxoG in the urine samples of BRCA1 mutation carriers with supplementation reaches 7.75 nmol/mmol creatinine, compared to 5.77 nmol/mmol creatinine in the BRCA1 mutation carriers with no supplementation. Additionally, about 26% reduction of the 8-oxoG background level in cellular DNA is observed in supplemented patients. These results indicate that Se supplementation enhances the repair of oxidative damage. Selenium can influence gene expression and activate silenced genes (e.g. antioxidant enzymes or tumor suppressors) through epigenetic modulation of histones and DNA in prostate cancer cells (LNCaP). Xiang et al. show that selenite treatment causes partial demethylation of promoter DNA and re-expression of glutathione S-transferase, decreased overall DNA methylation, and restored expression of the tumor suppressor adenomatous polyposis coli (APC) and cellular stress response 1 (CSR1). The authors conclude that Se may play a role in the chemoprevention of prostate and other cancers through epigenetic regulation of anti-cancer genes [87].
Seleno-L-methionine (Se-Met) is a naturally occurring selenium-containing amino acid and it appears to selectively regulate the nucleotide excision repair (NER) pathway. Studies on human fibroblasts show that pretreatment with Se-Met (10 µL) protects the mouse embryonic fibroblast (MEF) cells from UV-induced DNA damage and induces p53-dependent DNA repair [88]. Similar results apply to human prostate adenocarcinoma cells [89]. Pretreating cell culture with 10 μM of Se-Met protects DNA against damage induced by UVA (50 J/cm) or H2O2 (200 μM). Studies conducted in hemodialysis patients with chronic kidney disease show that the subjects have a lower concentration of selenium than healthy people (40.6±10.4 ng/mL vs. 52.7±9.7 ng/mL; p<0.0001). Moreover, the number of DNA single-strand breaks (SSBs) in white blood cells is significantly higher (DNA damage expressed as the tail moment (0.73±0.84) as compared with the control group (0.25±0.24; p<0.01). After a 3-month supplementation (200 μg/day of selenium as Se-rich yeast), 2.6 times lower levels of oxidative damage were observed in hemodialysis patients compared to the control group [90].
Evidence that the older population needs higher selenium intake may also come from a study on primary human keratinocytes collected from normal skin biopsies [91]. Keratinocytes obtained from older subjects (age 60-70) are more susceptible to UVA-induced damage. Additionally, to inhibit the cytotoxic effect of radiation, the cells require 8 times higher doses of Se than those from younger people (age 20-30). Se supplementation may be beneficial for the elderly as it may activate telomerase and p53 expression [83]. Furthermore, selenium inhibits the cytochrome P450 system’s activity (phase I proteins). It converts chemical carcinogens into their reactive forms leading to the formation of various DNA lesions [92][93][94].
Interestingly, studies show that selenium deficiency can promote cancer development in humans [95][96]. While Se supplementation in the well-nourished population is rather modest, it may be healthful in older people with its low levels [97]. Se deficiency arises from many factors such as improper intake, lack of accompanying nutrients (e.g. methionine), bowel diseases impacting Se absorption, or variations of Se-related genes [83]. Despite plenty of studies about the influence of selenium on DNA damage, DNA repair, and aging, there are no clear guidelines for selenium daily supplementation as the adequate level of Se is highly individual [98]. Current intake recommendations vary in the range of 25-150 µg/day (depending on sex and recommending country) [83]. However, it must be taken into consideration that too much selenium may be as damaging as too little and it should be supplemented according to individual needs, especially in the elderly population.
Zinc
Zinc is a crucial element for the overall well-being of the human organism and its genome stability (Fig. 3). It is involved in apoptosis, cell proliferation, protection against free radicals, and DNA repair pathways. Zn plays a role as a cofactor of the antioxidant enzymes (e.g. Zn/Cu-SOD) and also of DNA repair-related enzymes such as 8-oxoguanine glycosylase 1 (OGG1), apurinic/apyrimidinic endonuclease (APE), or poly[ADP-ribose] polymerase 1 (PARP-1). Zinc is also a part of a tumor suppressor p53 protein which is responsible for the cell cycle arrest thus, allowing the DNA to repair before replication starts. Lack of Zn up-regulates p53 activity (however, 50% Zn depletion results in non-functional p53) and affects DNA repair response [81]. A different study indicates the important role of zinc in the activity of the PARP-1 protein [99]. PARP-1 functions in various repair mechanisms including BER, NER, and DSBs repair [100][101]. It contains the zinc finger motif and is crucial in DNA repair; it detects DNA damage and facilitates the selection of the repair path. Zn deficiency, characteristic for the older population, reduces PARP-1 activity, and thus reduces the effectiveness of BER [99]. Also, PARP-1 has a vital role in inflammatory processes that accompany aging organisms [102].
The shortage of Zn may lead to the accumulation of oxidative DNA lesions. OGG1 and PARP-1 expression levels are higher (p<0.05) in zinc-deficient cells, in addition to a significant increase in DNA strand breaks (p<0.05) [103]. Studies on primary human fibroblasts show that zinc deficiency induces oxidative stress and DNA damage (SSBs). It also modulates the expression of DNA repair enzymes [104]. Microarray analysis shows that lack of zinc affects genes involved in DNA damage, DNA repair, and oxidative stress: 2-fold up-regulation of damage-specific DNA binding protein 2 (DDB2) and 0.5-fold down-regulation of RAD23 homolog B is observed.
Dietary restriction and repletion of Zn affect DNA integrity. A study on healthy men (age 19-50) shows an increased number of DNA strand breaks during 6 weeks of Zn restriction. The study proves the importance of dietary Zn for genome stability as the level of DNA breaks drops after Zn repletion [105]. 6 weeks of low zinc consumption significantly increases DNA damage in peripheral blood cells (mean tail moment increased by 57%; p<0.05). Interestingly, zinc supplementation reduces DNA damage (mean tail moment decreased by 39.9%; p<0.01). Moreover, a clinical study on 200 patients (age 65-80) shows that Zn supplementation (20 mg/day) improves genome stability and telomeres integrity [106]. After 12 weeks of supplementation, the activity of Cu/Zn SOD in erythrocytes is significantly higher in the Zn group vs. control group (activity increased by 33.07% in the Zn group, while the placebo group showed only a 2.45% increase (relative to the initial value)). The study also shows a decrease in the micronuclei (MNi) and DNA damage formation compared to the non-supplemented group (MNi per 1000 binucleates: 6,930 vs. 11,125; p=0,001). Additionally, patients in the supplemented group have a lower level of 8-oxodG in telomeric regions (8-oxodG/kbp telomere: 6.820 vs. 9.937; p=0.291 respectively). The insufficient intake of Zn with the diet will result in an increased level of oxidative stress and subsequent lesions and it may affect cellular response to those lesions. The deficiency of zinc might increase the incidence of cancer which is especially dangerous for older people as they are already more prone to carcinogenesis. Cancer development and aging are also related to hypermethylation of CpG islands in DNA. Interestingly, studies show that Zn depletion leads to hypomethylation [107].
Similar to selenium [108], zinc doses should be selected individually, because the excess of Zn, just like the deficiency, is harmful and pro-oxidative [109]. Cases of poisoning by drinks containing 2500 mg/L of zinc were observed. In vitro study shows that optimal Zn concentration for DNA damage prevention ranges between 4-16 µM [103] while a study on healthy men shows that 11 mg/day of Zn helps to reduce DNA damage [105]. Too high levels of Zn may induce DSBs, bases oxidation, chromosomes’ instability [103]. However, recommendations are yet unclear, as human studies on Zn influence on the genome are still lacking. Many variables must be taken into consideration while planning supplementation plans for malnourished patients, especially older ones.
Vitamin E
Vitamin E is the most important agent scavenging lipid peroxyl radicals. It may inhibit lipid peroxidation, H2O2 action on DNA, and lower oxidative stress resulting from environmental mutagenic factors (e.g. smoke or food additives). The diet of rats enriched with vit. E (300 mg/kg for 6 months) significantly decreases the number of chromosomal aberrations in the bone marrow [110]. Other interesting studies were carried out on animals where α-tocopherol was administered prior to irradiation. In mice pretreated with 100 mg/kg/day of vitamin E, irradiated with 2 Gy, a statistically significant decrease in the incidence of MNi in polychromatic erythrocytes (PCE) is observed [111]. In the supplemented group (200 mg/kg/day of vit. E) statistically significant protection of the bone marrow against radiation is also detected (expressed as an increase in the PCE/(PCE+NCE) ratio compared to the positive control: 7.3% vs. 3.4% (p< 0.05)). These results suggest that vitamin E may have radioprotective effects. Vit. E is a well-known ROS scavenging agent that protects against UV-induced DNA damage [112]. It may prevent the formation of cyclobutane pyrimidine dimers (CPDs) developed as a result of UVA in human skin cells. Pre- or post-treatment with vitamin E (0.1 mM) results in lower oxidation and DNA damage. Moreover, studies show that α-tocopherol protects the DNA of liver cancer cells from oxidative lesions resulting from ionizing radiation [113]. The level of 8-OHdG increases after irradiation (5 cGy) but the effects are reversed by vit. E enrichment which indicates its genoprotective properties. The protective effect of α-tocopherol against neurodegeneration in prematurely aging mice is also described [114]. Xpg-/- mice mimic symptoms of Cockayne syndrome patients—they are highly sensitive to nutritional deficiencies. The authors suggest that vit. E supplementation inhibits the accumulation of DNA damage and oxidative stress in liver and brain tissues which both significantly deteriorate with age. Studies showthat vitamin E reduces the formation of DNA damage such as DNA strand breaks or modifications of 8-OHdG. A study on a group of 21 healthy non-smoking men (age 28.9±1.3) shows that an increase in vitamin E intake by an additional 80 mg/day in a high PUFA diet (15%) decreases DNAdamage. A high-fat diet makes lymphocytes more prone to DNA strand breakage. Increased vitamin E intake probably removes this effect [115].
Vitamin E supplementation for older people must be planned carefully as the proper intake depends on many factors such as background level of vit. E, its supplemented form, duration of treatment, and possible genetic variations (which may alter vitamin absorption or metabolism) [114]. Different forms of vit. E may have a different impact on oxidative status in older adults as the study on 71 patients (age 50-55) shows [116]. Tocotrienol rich fraction and α-tocopherol were administered for 6 months. DNA damage level drops for tocotrienol rich fraction in female subjects after 6 months. The study shows that form of vitamin E matters and may have a different effect, which is also dependent on sex. Interestingly, vit. E (100 µM) provesto be potentially beneficial for oncological patients due to its antioxidant action and lack of interference with camptothecin (chemotherapeutic) [117]. Nonetheless, other data show no beneficial effect of vitamin E supplementation on cellular DNA damage [118][119]. A positive correlation is described between serum vitamin E levels and the level of 8-OHdG in peripheral blood lymphocytes in premenopausal non-smoking women (age 45-50) [120]. In another study, healthy men (age 50-70) received 500 mg of vit. E, but there was no effect observed on micronucleus formation after 8 weeks of supplementation [118].
While in vitro studies prove the likely positive effect of vit. E on DNA damage (Fig. 3) human studies do not fully confirm it. However, lower levels of circulating α-tocopherol are associated with reduced immune function (increased levels of inflammatory markers) and QoL in the elderly [121]. Among 69 elderly subjects (mean age 78.9) the elevated level of IL-6 was observed and accompanied by a decreased concentration of vitamin E (R -(0.277); p<0.01). Results correlate with poor physical and mental health. The authors suggest that insufficient intake of antioxidants (including vit. E)leads to reduced QoL and increases the risk of age-related diseases. The influence of α-tocopherol on DNA damage is yet to be confirmed to recommend adequate doses for supplementation in the older population.
Vitamin C
Vitamin C is a crucial antioxidative agent with the ability to enhance genome stability (Fig. 3). Studies show that its adequate intake may lower the number of chromosome aberrations, DNA adducts, and strand breakage [122]. Insufficient consumption of vitamin C leads to an increase in the level of oxidative DNA lesions [123]. Fraga et al. show that low/poor dietary supply of vit. C results in a 2-fold increase in the level of 8-oxodG in the sperm DNA. A different study on 112 patients with coronary artery disease shows that low levels of ascorbate and GSH in peripheral blood lymphocytes are accompanied by more frequent chromosomal aberrations [124].
An interesting study conducted on a group of 139 subjects examines the correlation of air pollution, markers of oxidative DNA damage (including 8-oxodG), and DNA repair gene expression [125]. Authors selected genes coding enzymes involved in the repair of 8-oxodG in BER and non-homologous end-joining (NHEJ): human 8-oxoguanine glycosylase 1 (hOGG1), apurinic/apyrimidinic endodeoxyribonuclease 1 (APEX1), X-ray repair cross-complementation group 1 (XRCC1), XRCC4, XRCC5, XRCC6 and DNA ligase 4 (LIG4). The group of subjects living in a more polluted environment has a lower concentration of 8-oxodG in urine (4.16 vs. 4.99 nmol/mmol creatinine) with simultaneously higher plasma ascorbate levels (11.8 vs. 8.3 mg/L) compared to subjects living in relatively cleaner regions. The authors speculate that higher plasma levels of ascorbate result in higher XRCC1 expression in some people and a later increase in BER efficiency (resulting in lower levels of 8-oxodG in urine), thereby protecting the body from oxidative DNA damage. The different study tested 340 healthy Norwegians for dietary and genetic factors influencing DNA damage and repair capacity [126]. Subjects did not undergo a special diet nor supplementation–The Food Frequency Questionnaire and tests of fasting blood samples were used. The levels of DNA strand breaks, oxidized lesions, the activity of BER and NER were measured. Results show a significant correlation between diet and level of DNA damage. The quantity of strand breaks and oxidized lesions of purines and pyrimidines is higher when subjects consume fewer vegetables and fruit. The study also shows that female subjects who consume less fruit have approximately 20% higher level of DNA damage. Authors observe, among other things, that NER efficiency is 0,141-fold higher for subjects with a higher level of ascorbate and total DNA damage is 0,037-fold lower.
It seems that antioxidant supplementation is especially important for poorly nourished people [127]. Guarnieri et al. show the basic level of DNA repair by an incision in mononuclear blood cells is significantly lower in poorly nourished patients compared to well-nourished ones. At the same time, poorly nourished people have higher levels of oxidized guanine. Vitamin C supplementation is potentially beneficial as an increase in DNA repair incision capacity is observed, which is not seen in well-nourished subjects. It is possible that the influence of vit. C on DNA depends on the preexisting level of this vitamin (the protective effect is observed for >50 µmol/L vit. C in plasma) and individual level of oxidative stress (resulting from environmental factors such as smoking or exposure to mutagenic chemicals). Moreover, vitamin C should be supplemented for longer periods and together with other antioxidants (e.g. vitamin E) to observe possible positive effects [122].
Oxidative stress is widely recognized as the epigenetic factor of aging. Antioxidative micronutrients play a key role in reducing the inflammatory response associated with poor health outcomes in the elderly population. Also, the enzymatic capacity of cellular antioxidants declines with age, therefore older people with lower peripheral antioxidant parameters and reduced antioxidant capacity are more susceptible to age-related diseases, disability, weakness, and higher mortality throughout a 5-year follow-up [128][129]. These factors explain the increasing trend towards researching the effects of antioxidants on aging and preventing age-related disease.
4. Conclusions
Micronutrients are an important part of the antioxidant defense mechanisms. Oxidative metabolism inevitably leads to the production of ROS which may cause further oxidation, particularly of cell membranes and nucleic acids. The cell can counteract oxidative damage with endo- and exogenous antioxidants and repair systems. The damaging potential of free radicals is directly inhibited by the action of ascorbate, tocopherols, or enzyme systems—Zn/Cu-SOD and GPX (dependent on selenium). Therefore, vitamins and trace elements (e.g. selenium or zinc) supplied with the diet are crucial for the proper function of the antioxidant enzymes [9]. In older people, the level of oxidative damage increases, thereby disrupting healthyaging at the molecular level. Shortage of microelements, such as vitamin C, E, zinc, and selenium makes DNA more susceptible to oxidation. One of the most interesting examples are telomeres, which shorten with age. DNA lesions in their sequence (e.g. guanine oxidation) may result in SSBs or DSBs [130], which increases the risk of age-related diseases such as cancer, cardiovascular, neurodegenerative diseases (e.g. dementia), and diabetes [9]. Anorexia of aging is a state of severe deficiencies of microelements involved in antioxidant protection and maintaining genome stability. It accelerates and aggravates the course of the aging process and seriously disrupts the integrity of genetic information, causing SSBs, DSBs, and oxidative DNA damage. These micronutrient deficiencies can be as harmful as DNA lesions resulting from UV rays and chemical agents’ activity.
Understanding the influence of nutrition on cellular and molecular pathways will enable the development of nutritional strategies to maintain health and possibly to treat and prevent diseases triggered by dietary deficiencies. The elderly population is particularly vulnerable to various deficits due to reduced intake of food rich in vitamins, micro- and macroelements. Micronutrients are essential for the maintenance of physical and cognitive functions in the aging body. Their insufficient consumption might lead to deterioration of health and general QoL. Anorexia of aging additionally worsens the health condition of older patients, as it exacerbates the natural decrease in micronutrient levels that occur with age [131]. Nevertheless, oral supplementation should be recommended with caution in the elderly population, only for patients with diagnosed deficiencies, under medical supervision, and for a finite time. A well-balanced diet rich in vegetables and fruit should be the most important part of prophylaxis of age-related diseases such as cardiovascular disease, neurodegenerative diseases, or age-related anorexia and a way to promote healthy aging with a high QoL.
This entry is adapted from the peer-reviewed paper 10.3390/nu12113364
References
- World Health Organization. Comprehensive Implementation Plan on Maternal, Infant and Young Child Nutritio; World Health Organization, 2014; WHO Reference Number: WHO/NMH/NHD/14.1 . World Health Organization. Retrieved 2020-11-18
- Giorgia Sebastiani; Ana Herranz Barbero; Cristina Borràs-Novell; Miguel Alsina; Victoria Aldecoa-Bilbao; Vicente Andreu-Fernández; Mireia Pascual Tutusaus; Silvia Ferrero Martínez; Maria Dolores Gomezroig; Oscar Garcia-Algar; et al. The Effects of Vegetarian and Vegan Diet during Pregnancy on the Health of Mothers and Offspring. Nutrients 2019, 11, 557, 10.3390/nu11030557.
- Lorenzo Maria Donini; Eleonora Poggiogalle; Maria Piredda; Alessandro Pinto; Mario Barbagallo; Domenico Cucinotta; Giuseppe Sergi; Anorexia and Eating Patterns in the Elderly. PLOS ONE 2013, 8, e63539, 10.1371/journal.pone.0063539.
- Francesco Landi; R. Liperoti; F. Lattanzio; A. Russo; M. Tosato; C. Barillaro; R. Bernabei; G. Onder; Effects of anorexia on mortality among older adults receiving home care: An observational study. The journal of nutrition, health & aging 2011, 16, 79-83, 10.1007/s12603-011-0064-y.
- Adam Wysokiński; Tomasz Sobów; Iwona Kłoszewska; Tomasz Kostka; Mechanisms of the anorexia of aging—a review. AGE 2015, 37, 1-14, 10.1007/s11357-015-9821-x.
- W B Ershler; Interleukin-6: a cytokine for gerontologists.. Journal of the American Geriatrics Society 1993, 41, 176–181, .
- Umberto Fagiolo; Andrea Cossarizza; Enrico Scala; Emanuele Fanales-Belasio; Claudio Ortolani; Emanuele Cozzi; Daniela Monti; Claudio Franceschi; Roberto Paganelli; Increased cytokine production in mononuclear cells of healthy elderly people. European Journal of Immunology 1993, 23, 2375-2378, 10.1002/eji.1830230950.
- Philipp Schuetz; Martina Bally; Zeno Stanga; Ulrich Keller; Loss of appetite in acutely ill medical inpatients: physiological response or therapeutic target?. Swiss Medical Weekly 2014, 144, 13957, 10.4414/smw.2014.13957.
- Siepelmeyer, A.; Micka, A.; Simm, A.; Bernhardt, J. . Molecular Basis of Nutrition and Aging; Academic Press: : Cambridge, MA, USA, 2016; pp. 109–120.
- Zewen Liu; Zhangpin Ren; Jun Zhang; Chia-Chen Chuang; Eswar Kandaswamy; Tingyang Zhou; Li Zuo; Role of ROS and Nutritional Antioxidants in Human Diseases. Frontiers in Physiology 2018, 9, 477, 10.3389/fphys.2018.00477.
- Karwowski, B.T.; 5’8-cyklo-deoksyadenozyna. Podwójne uszkodzenie w obre ̨bie pojedynczego nukleozydu/nukleotydu. Wiadomości chemiczne 2010, 64, 1013–1048, .
- Xueqing Ba; Istvan Boldogh; 8-Oxoguanine DNA glycosylase 1: Beyond repair of the oxidatively modified base lesions. Redox Biology 2018, 14, 669-678, 10.1016/j.redox.2017.11.008.
- Lynnette R. Ferguson; Martin Philpott; Nutrition and Mutagenesis. Annual Review of Nutrition 2008, 28, 313-329, 10.1146/annurev.nutr.28.061807.155449.
- Anthony P. Winston; The clinical biochemistry of anorexia nervosa.. Annals of Clinical Biochemistry: An international journal of biochemistry and laboratory medicine 2012, 49, 132-143, 10.1258/acb.2011.011185.
- Jon Arcelus; Alex J. Mitchell; Jackie Wales; Søren Nielsen; Mortality Rates in Patients With Anorexia Nervosa and Other Eating Disorders. Archives of General Psychiatry 2011, 68, 724-731, 10.1001/archgenpsychiatry.2011.74.
- Klecha, B.; Bukowska, B.; Selen w organizmie człowieka—charakterystyka pierwiastka i potencjalne zastosowanie terapeutyczne. Bromatologia i Chemia Toksykologiczna 2016, 4, 818–829, .
- Bradley A. Carlson; Min-Hyuk Yoo; Rajeev K. Shrimali; Robert Irons; Vadim N. Gladyshev; Dolph L. Hatfield; Jin Mo Park; Role of selenium-containing proteins in T-cell and macrophage function. Proceedings of the Nutrition Society 2010, 69, 300-310, 10.1017/s002966511000176x.
- Marta Wołonciej; Elżbieta Milewska; Wiesława Roszkowska-Jakimiec; Trace elements as an activator of antioxidant enzymes. Postępy Higieny i Medycyny Doświadczalnej 2016, 70, 1483-1498, 10.5604/17322693.1229074.
- Youcef Mehdi; Jean-Luc Hornick; Louis Istasse; Isabelle Dufrasne; Selenium in the Environment, Metabolism and Involvement in Body Functions. Molecules 2013, 18, 3292-3311, 10.3390/molecules18033292.
- María Jesús Oliveras-López; Inmaculada Ruiz-Prieto; Patricia Bolaños-Ríos; Francisco De La Cerda; Franz Martín; Ignacio Jáuregui-Lobera; Antioxidant Activity and Nutritional Status in Anorexia Nervosa: Effects of Weight Recovery. Nutrients 2015, 7, 2193-2208, 10.3390/nu7042193.
- Peter M Abuja; Riccardo Albertini; Methods for monitoring oxidative stress, lipid peroxidation and oxidation resistance of lipoproteins. Clinica Chimica Acta 2001, 306, 1-17, 10.1016/s0009-8981(01)00393-x.
- Margaret P Rayman; The importance of selenium to human health. The Lancet 2000, 356, 233-241, 10.1016/s0140-6736(00)02490-9.
- Sandra Torres; Marina Prista Guerra; Leonor Lencastre; Kylee Miller; Filipa Mucha Vieira; António Roma-Torres; Isabel Brandão; Patrício Costa; Alexithymia in anorexia nervosa: The mediating role of depression. Psychiatry Research 2015, 225, 99-107, 10.1016/j.psychres.2014.10.023.
- Ghania Hammad; Yona Legrain; Zahia Touat-Hamici; Stéphane Duhieu; David Cornu; Anne-Laure Bulteau; Laurent Chavatte; Interplay between Selenium Levels and Replicative Senescence in WI-38 Human Fibroblasts: A Proteomic Approach. Antioxidants 2018, 7, 19, 10.3390/antiox7010019.
- Legrain Yona; Touat-Hamici Zahia; Laurent Chavatte; Interplay between selenium levels, selenoprotein expression, and replicative senescence in WI-38 human fibroblasts. Free Radical Biology and Medicine 2014, 75, S25, 10.1016/j.freeradbiomed.2014.10.744.
- Min Liu; Huijuan Jing; Jianjun Zhang; Gen Che; Meng Zhou; Zheng Gao; Shangshang Li; Zhenzhen Ren; Long Hao; Yu Liu; et al. Optimization of Mycelia Selenium Polysaccharide Extraction from Agrocybe cylindracea SL-02 and Assessment of their Antioxidant and Anti-Ageing Activities. PLOS ONE 2016, 11, e0160799, 10.1371/journal.pone.0160799.
- Yumiko Yamashita; Takeshi Yabu; Michiaki Yamashita; Discovery of the strong antioxidant selenoneine in tuna and selenium redox metabolism. World Journal of Biological Chemistry 2010, 1, 144-150, 10.4331/wjbc.v1.i5.144.
- Barbara Klapcinska; Stanislaw Poprzecki; Alojzy Danch; Andrzej Sobczak; Katarzyna Kempa; Selenium Levels in Blood of Upper Silesian Population: Evidence of Suboptimal Selenium Status in a Significant Percentage of the Population. Biological Trace Element Research 2005, 108, 001-016, 10.1385/bter:108:1-3:001.
- G. Forte; M. Deiana; S. Pasella; A. Baralla; P. Occhineri; I. Mura; R. Madeddu; E. Muresu; S. Sotgia; A. Zinellu; et al. Metals in plasma of nonagenarians and centenarians living in a key area of longevity. Experimental Gerontology 2014, 60, 197-206, 10.1016/j.exger.2014.10.016.
- Silvia Giovannini; G. Onder; F. Lattanzio; S. Bustacchini; G. Di Stefano; R. Moresi; A. Russo; R. Bernabei; F. Landi; Selenium Concentrations and Mortality Among Community-Dwelling Older Adults: Results from ilSIRENTE Study. The journal of nutrition, health & aging 2018, 22, 608-612, 10.1007/s12603-018-1021-9.
- G. F. Combs; M. L. Scott; Nutritional Interrelationships of Vitamin E and Selenium. BioScience 1977, 27, 467-473, 10.2307/1297528.
- Jennifer K. MacFarquhar; Acute Selenium Toxicity Associated With a Dietary Supplement. Archives of Internal Medicine 2010, 170, 256-261, 10.1001/archinternmed.2009.495.
- A S Prasad; Clinical, endocrinologic, and biochemical effects of zinc deficiency.. Special topics in endocrinology and metabolism 1985, 7, 45-76, .
- Ananda S. Prasad; Effects of Zinc Deficiency on Th1 and Th2 Cytokine Shifts. Journal of Infectious Diseases 2000, 182, S62-S68, 10.1086/315916.
- Paula DePasquale-Jardieu; Pamela J. Fraker; Interference in the Development of a Secondary Immune Response in Mice by Zinc Deprivation: Persistence of Effects. The Journal of Nutrition 1984, 114, 1762-1769, 10.1093/jn/114.10.1762.
- Matteo Cassandri; Artem Smirnov; Flavia Novelli; Consuelo Pitolli; Massimiliano Agostini; Michal Malewicz; Gerry Melino; Giuseppe Raschellà; Zinc-finger proteins in health and disease. Cell Death Discovery 2017, 3, 17071-17071, 10.1038/cddiscovery.2017.71.
- Stephen R Hennigar; Alyssa M Kelley; James P McClung; Metallothionein and Zinc Transporter Expression in Circulating Human Blood Cells as Biomarkers of Zinc Status: a Systematic Review. Advances in Nutrition 2016, 7, 735-746, 10.3945/an.116.012518.
- Yongsong Guan; Comments to metallothionein as an anti-inflammatory mediator.. Mediators of Inflammation 2009, 2009, 426214-2, 10.1155/2009/426214.
- Wolfgang Maret; Metallothionein/disulfide interactions, oxidative stress, and the mobilization of cellular zinc. Neurochemistry International 1995, 27, 111-117, 10.1016/0197-0186(94)00173-r.
- Xiaoping Yang; Thomas A. Doser; Cindy X. Fang; Jennifer M. Nunn; Rajiv Janardhanan; Meijun Zhu; Nair Sreejayan; Mark T. Quirin; Jun Ren; Mark T. Quinn; et al. Metallothionein prolongs survival and antagonizes senescence‐associated cardiomyocyte diastolic dysfunction: role of oxidative stress. The FASEB Journal 2006, 20, 1024-1026, 10.1096/fj.05-5288fje.
- Ananda S. Prasad; Frances W J Beck; Bin Bao; James T Fitzgerald; Diane C Snell; Joel D Steinberg; Lavoisier J Cardozo; Zinc supplementation decreases incidence of infections in the elderly: effect of zinc on generation of cytokines and oxidative stress. The American Journal of Clinical Nutrition 2007, 85, 837-844, 10.1093/ajcn/85.3.837.
- Marisol Izquierdo; David Domínguez; Juan Ignacio Jiménez; Reda Saleh; Carmen Maria Hernández-Cruz; Maria Jesus Zamorano; Kristin Hamre; Interaction between taurine, vitamin E and vitamin C in microdiets for gilthead seabream (Sparus aurata) larvae. Aquaculture 2019, 498, 246-253, 10.1016/j.aquaculture.2018.07.010.
- Maria Wallert; Melanie Ziegler; Xiaowei Wang; Ana Maluenda; Xiaoqiu Xu; May Lin Yap; Roman Witt; Corey Giles; Stefan Kluge; Marcus Hortmann; et al. α-Tocopherol preserves cardiac function by reducing oxidative stress and inflammation in ischemia/reperfusion injury. Redox Biology 2019, 26, 101292, 10.1016/j.redox.2019.101292.
- Zielin ́ska,A.; Nowak,I.; Tokoferole i tokotrienole jako witamina E. Chemik 2014, 68, 585–591, .
- Areej Elkamil; Krisztina K. Johansen; Jan Aasly; Ataxia with Vitamin E Deficiency in Norway. Journal of Movement Disorders 2015, 8, 33-36, 10.14802/jmd.14030.
- Dolores Moyano; Cristina Sierra; Nuria Brandi; Rafael Artuch; Aurea Mira; Antioxidant status in anorexia nervosa. International Journal of Eating Disorders 1999, 25, 99-103, 10.1002/(sici)1098-108x(199901)25:1<99::aid-eat12>3.0.co;2-n.
- Marcus, J.B. Aging, Nutrition and Taste; Academic Press: Cambridge, MA, USA, 2019; pp. 254.
- Anna Skowrońska; Klaudia Sójta; Dominik Strzelecki; Refeeding syndrome as treatment complication of anorexia nervosa.. Psychiatria Polska 2019, 53, 1113-1123, 10.12740/PP/OnlineFirst/90275.
- Hercberg, S.; Galan, P.; Preziosi, P.; Bertrais, S.; Mennen, L.; Malvy, D.; Briançon, S.; The SU.VI.MAX Study: A Randomized, Placebo-Controlled Trial of the Health Effects of Antioxidant Vitamins and Minerals—Correction. Archives of Internal Medicine 2005, 165, 286, 10.1001/archinte.165.3.286.
- S Czernichow; P Galan; Serge Hercberg; Antioxidant supplements for prevention of gastrointestinal cancers.. The Lancet 2005, 365, 470-471, 10.1016/S0140-6736(05)17857-X.
- W J Blot; J Y Li; P R Taylor; W Guo; S Dawsey; G Q Wang; C S Yang; S F Zheng; M Gail; Guangyao Li; et al. Nutrition intervention trials in Linxian, China: supplementation with specific vitamin/mineral combinations, cancer incidence, and disease-specific mortality in the general population.. JNCI: Journal of the National Cancer Institute 1993, 85, 1483–1491, .
- Saliha Rizvi; Syed T. Raza; Faizal Ahmed; Absar Ahmad; Shania Abbas; Farzana Mahdi; The Role of Vitamin E in Human Health and Some Diseases. Sultan Qaboos University Medical Journal 2014, 14, e157-e165, .
- Roberto Bei; Effects of Vitamin C on health: a review of evidence. Frontiers in Bioscience 2013, 18, 1017, 10.2741/4160.
- Bartosz, G.. Druga Twarz Tlenu. Wolne Rodniki w Przyrodzie; Wydawnictwo Naukowe PWN: Warszawa, Poland, 2006; pp. 1.
- Juan Du; Joseph J. Cullen; Garry R. Buettner; Ascorbic acid: Chemistry, biology and the treatment of cancer. Biochimica et Biophysica Acta (BBA) - Reviews on Cancer 2012, 1826, 443-457, 10.1016/j.bbcan.2012.06.003.
- O. Arrigoni; Mario C. De Tullio; Ascorbic acid: much more than just an antioxidant. Biochimica et Biophysica Acta (BBA) - General Subjects 2002, 1569, 1-9, 10.1016/s0304-4165(01)00235-5.
- Kevin J Lenton; Hélène Therriault; André M Cantin; Tamàs Fülöp; Hélène Payette; J Richard Wagner; Direct correlation of glutathione and ascorbate and their dependence on age and season in human lymphocytes. The American Journal of Clinical Nutrition 2000, 71, 1194-1200, 10.1093/ajcn/71.5.1194.
- Andrea Mezzetti; Domenico Lapenna; Ferdinando Romano; Fabrizio Costantini; Sante D. Pierdomenico; Domenico De Cesare; Franco Cuccurullo; Giuseppe Riario-Sforza; Giovanni Zuliani; Renato Fellin; et al. Systemic Oxidative Stress and Its Relationship with Age and Illness. Journal of the American Geriatrics Society 1996, 44, 823-827, 10.1111/j.1532-5415.1996.tb03741.x.
- Michel Langlois; Daniel Duprez; Joris Delanghe; Marc De Buyzere; Denis L. Clement; Serum Vitamin C Concentration Is Low in Peripheral Arterial Disease and Is Associated With Inflammation and Severity of Atherosclerosis. Circulation 2001, 103, 1863-1868, 10.1161/01.cir.103.14.1863.
- T Yokoyama; C Date; Y Kokubo; N Yoshiike; Y Matsumura; H Tanaka; Serum vitamin C concentration was inversely associated with subsequent 20-year incidence of stroke in a Japanese rural community. The Shibata study.. Stroke 2000, 31, 2287–2294, .
- J A Simon; Vitamin C and cardiovascular disease: a review.. Journal of the American College of Nutrition 1992, 11, 107–125, .
- L. Jackson Roberts; Maret G. Traber; Balz Frei; Vitamins E and C in the prevention of cardiovascular disease and cancer in men. Free Radical Biology and Medicine 2009, 46, 1558, 10.1016/j.freeradbiomed.2009.03.002.
- Lukas Schwingshackl; Heiner Boeing; Marta Stelmach-Mardas; Marion Gottschald; Stefan Dietrich; Georg Hoffmann; Anna Chaimani; Dietary Supplements and Risk of Cause-Specific Death, Cardiovascular Disease, and Cancer: A Systematic Review and Meta-Analysis of Primary Prevention Trials. Advances in Nutrition 2017, 8, 27-39, 10.3945/an.116.013516.
- Stephanie P. DiTroia; Michelle Percharde; Marie-Justine Guerquin; Estelle Wall; Evelyne Collignon; Kevin T. Ebata; Kathryn Mesh; Swetha Mahesula; Michalis Agathocleous; Diana J. Laird; et al. Maternal vitamin C regulates reprogramming of DNA methylation and germline development. Nature 2019, 573, 271-275, 10.1038/s41586-019-1536-1.
- Ciesielski, P.; Józwiak, P.; Krzeslak, A.; Białka TET a modyfikacje epigenetyczne w nowotworach. Postępy Higieny i Medycyny Doświadczalnej 2015, 69, 1371–1383, .
- Hua-Lin Fu; Yue Ma; Lun-Gen Lu; Peng Hou; Bao-Jie Li; Wei-Lin Jin; Da-Xiang Cui; TET1 Exerts Its Tumor Suppressor Function by Interacting with p53-EZH2 Pathway in Gastric Cancer. Journal of Biomedical Nanotechnology 2014, 10, 1217-1230, 10.1166/jbn.2014.1861.
- Marcus S. Cooke; Mark D Evans; Ian D Podmore; Karl E Herbert; Nalini Mistry; Pratibha Mistry; Peter T Hickenbotham; Amina Hussieni; Helen R Griffiths; Joseph Lunec; et al. Novel repair action of vitamin C upon in vivo oxidative DNA damage. FEBS Letters 1998, 439, 363-367, 10.1016/s0014-5793(98)01403-3.
- Bevan, R.J.; Mistry, N.; Patel, P.R.; Halligan, E.P.; Dove, R.; Lunec, J.; Can vitamin C induce nucleotide excision repair? Support from in vitro evidence. British Journal of Nutrition 2009, 103, 686–695, .
- Lalonde, M; A new perspective on the health of Canadians. AARN news letter 1976, 32, 1–5, .
- D. Harman; Aging: A Theory Based on Free Radical and Radiation Chemistry. Journal of Gerontology 1956, 11, 298-300, 10.1093/geronj/11.3.298.
- Aurel Popa-Wagner; Smaranda Mitran; Senthilkumar Sivanesan; Edwin Chang; Ana-Maria Buga; ROS and Brain Diseases: The Good, the Bad, and the Ugly. Oxidative Medicine and Cellular Longevity 2013, 2013, 1-14, 10.1155/2013/963520.
- Helmut Sies; Dean P. Jones; Reactive oxygen species (ROS) as pleiotropic physiological signalling agents. Nature Reviews Molecular Cell Biology 2020, 21, 363-383, 10.1038/s41580-020-0230-3.
- Hércules Rezende Freitas; Gustavo Da Costa Ferreira; Isis Hara Trevenzoli; Karen De Jesus Oliveira; Ricardo Augusto De Melo Reis; Fatty Acids, Antioxidants and Physical Activity in Brain Aging. Nutrients 2017, 9, 1263, 10.3390/nu9111263.
- A Terman; B Gustafsson; U T Brunk; Autophagy, organelles and ageing. The Journal of Pathology 2007, 211, 134-143, 10.1002/path.2094.
- Nuo Sun; Richard J. Youle; Toren Finkel; The Mitochondrial Basis of Aging. Molecular Cell 2016, 61, 654-666, 10.1016/j.molcel.2016.01.028.
- Hewitt, G.; Jurk, D.; Marques, F.D.M.; Correia-Melo, C.; Hardy, T.; Gackowska, A.; Telomeres are favored targets of a persistent DNA damage response in aging and stress-induced senescence. Nature Communications 2012, 3, 708, .
- Jorge Pinto Soares; António Cortinhas; Teresa Bento; José Carlos Leitão; Andrew R. Collins; Isabel Gaivão; Maria Paula Mota; Aging and DNA damage in humans: a meta-analysis study. Aging 2014, 6, 432-439, 10.18632/aging.100667.
- Federica I Wolf; Silvia Fasanella; Beatrice Tedesco; Gabriella Cavallini; Alessio Donati; Ettore Bergamini; Achille Cittadini; Peripheral lymphocyte 8-OHdG levels correlate with age-associated increase of tissue oxidative DNA damage in Sprague?Dawley rats. Protective effects of caloric restriction. Experimental Gerontology 2005, 40, 181-188, 10.1016/j.exger.2004.11.002.
- Ryszard Olinski; Agnieszka Siomek; Rafal Rozalski; Daniel Gackowski; Marek Foksinski; Jolanta Guz; Tomasz Dziaman; Anna Szpila; Barbara Tudek; Oxidative damage to DNA and antioxidant status in aging and age-related diseases.. Acta Biochimica Polonica 2007, 54, 11–26, .
- Zenger, F.; Russmann, S.; Junker, E.; Wuthrich, C.; Bui, M.H.; Lauterburg, B.H.; Decreased glutathione in patients with anorexia nervosa. Risk factor for toxic liver injury?. European Journal of Clinical Nutrition 2004, 58, 238–243, .
- Bruce N. Ames; Micronutrient Deficiencies: A Major Cause of DNA Damage. Annals of the New York Academy of Sciences 1999, 889, 87-106, 10.1111/j.1749-6632.1999.tb08727.x.
- Michael Fenech; Nutritional treatment of genome instability: a paradigm shift in disease prevention and in the setting of recommended dietary allowances. Nutrition Research Reviews 2003, 16, 109-122, 10.1079/nrr200359.
- Lynnette R Ferguson; Nishi Karunasinghe; Shuotun Zhu; Alice H. Wang; Selenium and its’ role in the maintenance of genomic stability. Mutation Research/Fundamental and Molecular Mechanisms of Mutagenesis 2012, 733, 100-110, 10.1016/j.mrfmmm.2011.12.011.
- Lin Rao; Birgit Puschner; Tomas A. Prolla; Gene Expression Profiling of Low Selenium Status in the Mouse Intestine: Transcriptional Activation of Genes Linked to DNA Damage, Cell Cycle Control and Oxidative Stress. The Journal of Nutrition 2001, 131, 3175-3181, 10.1093/jn/131.12.3175.
- Nishi Karunasinghe; Jacqueline Ryan; John Tuckey; Jonathan Masters; Michael Jamieson; Larry C Clarke; James R Marshall; Lynnette R. Ferguson; DNA stability and serum selenium levels in a high-risk group for prostate cancer.. Cancer Epidemiology Biomarkers & Prevention 2004, 13, 391–397, .
- Tomasz Dziaman; Tomasz Huzarski; Daniel Gackowski; Rafal Rozalski; Agnieszka Siomek; Anna Szpila; Jolanta Guz; Jan Lubiński; Wojciech Wasowicz; Krzysztof Roszkowski; et al. Selenium Supplementation Reduced Oxidative DNA Damage in Adnexectomized BRCA1 Mutations Carriers. Cancer Epidemiology Biomarkers & Prevention 2009, 18, 2923-2928, 10.1158/1055-9965.epi-09-0529.
- Nong Xiang; Rui Zhao; Guoqing Song; Weixiong Zhong; Selenite reactivates silenced genes by modifying DNA methylation and histones in prostate cancer cells. Carcinogenesis 2008, 29, 2175-2181, 10.1093/carcin/bgn179.
- Joshua L Fischer; Jody K Lancia; Anita Mathur; Martin L Smith; Selenium protection from DNA damage involves a Ref1/p53/Brca1 protein complex.. Anticancer Research 2006, 26, 899-904, .
- Viviana De Rosa; Pinar Erkekoğ Lu; Anne Forestier; Alain Favier; Filiz Hincal; Alan M. Diamond; Thierry Douki; Walid Rachidi; Low doses of selenium specifically stimulate the repair of oxidative DNA damage in LNCaP prostate cancer cells. Free Radical Research 2012, 46, 105-116, 10.3109/10715762.2011.647009.
- Bronislaw A. Zachara; Jolanta Gromadzinska; Jadwiga Palus; Zbigniew Zbrog; Rafal Swiech; Ewa Twardowska; Wojciech Wasowicz; The Effect of Selenium Supplementation in the Prevention of DNA Damage in White Blood Cells of Hemodialyzed Patients: A Pilot Study. Biological Trace Element Research 2010, 142, 274-283, 10.1007/s12011-010-8776-0.
- C. Favrot; D. Beal; E. Blouin; M. T. Leccia; A. M. Roussel; W. Rachidi; Age-Dependent Protective Effect of Selenium against UVA Irradiation in Primary Human Keratinocytes and the Associated DNA Repair Signature. Oxidative Medicine and Cellular Longevity 2018, 2018, 1-9, 10.1155/2018/5895439.
- Tsutomu Shimada; K El-Bayoumy; P Upadhyaya; T R Sutter; F P Guengerich; H Yamazaki; Inhibition of human cytochrome P450-catalyzed oxidations of xenobiotics and procarcinogens by synthetic organoselenium compounds.. Cancer Research 1997, 57, 4757–4764, .
- Yimei Cong; Qianru Chi; Xiaohua Teng; Shu Li; The Protection of Selenium Against Cadmium-Induced Mitochondrial Damage via the Cytochrome P450 in the Livers of Chicken. Biological Trace Element Research 2018, 190, 484-492, 10.1007/s12011-018-1557-x.
- Lv-Hui Sun; Niya Zhang; Ming-Kun Zhu; Ling Zhao; Ji-Chang Zhou; De-Sheng Qi; Prevention of Aflatoxin B1 Hepatoxicity by Dietary Selenium Is Associated with Inhibition of Cytochrome P450 Isozymes and Up-Regulation of 6 Selenoprotein Genes in Chick Liver. The Journal of Nutrition 2015, 146, 655-661, 10.3945/jn.115.224626.
- W C Willett; B F Polk; J S Morris; M J Stampfer; S Pressel; B Rosner; J O Taylor; K Schneider; C G Hames; Prediagnostic serum selenium and risk of cancer.. The Lancet 1983, 2, 130–134, .
- Jarmo Virtamo; Esko Valkeila; Georg Alfthan; Sven Punsar; Jussi K. Huttunen; Martti J. Karvonen; Serum selenium and risk of cancer. A prospective follow-up of nine years. Cancer 1987, 60, 145-148, 10.1002/1097-0142(19870715)60:2<145::aid-cncr2820600203>3.0.co;2-u.
- Vasileios Pagmantidis; Catherine Méplan; Evert M Van Schothorst; Jaap Keijer; John E Hesketh; Supplementation of healthy volunteers with nutritionally relevant amounts of selenium increases the expression of lymphocyte protein biosynthesis genes. The American Journal of Clinical Nutrition 2008, 87, 181-189, 10.1093/ajcn/87.1.181.
- Yildiz, A.; Kaya, Y.; Tanriverdi, O.; EffectoftheInteractionBetweenSeleniumandZinconDNARepairin Association with Cancer Prevention. Journal of Cancer Prevention 2019, 24, 146–154, .
- Kunzmann,A.; Dedoussis,G.; Jajte,J.; Malavolta,M.; Mocchegiani,E.; Bürkle,A.; Effectofzinconcellular poly(ADP-ribosyl)ation capacity. Experimental Gerontology 2008, 43, 409–414, .
- Ronson, G.E.; Piberger, A.L.; Higgs, M.R.; Olsen, A.L.; Stewart, G.S.; McHugh, P.J.; Lakin, N.D.; PARP1 and PARP2 stabilize replication forks at base excision repair intermediates through Fbh1-dependent Rad51 regulation. Nature Communications 2018, 9, 1-12, .
- A. (Alex) Pines; M.G. (Mischa ) Vrouwe; J.A. (Jurgen) Marteijn; D. (Dimitris) Typas; M.S. (Martijn) Luijsterburg; M. (Medine) Cansoy; P. (Paul) Hensbergen; A.M. (André) Deelder; A. (Anton) De Groot; S. (Syota) Matsumoto; et al. PARP1 promotes nucleotide excision repair through DDB2 stabilization and recruitment of ALC1. Journal of Cell Biology 2012, 199, 235-249, 10.1083/jcb.201112132.
- Aswin Mangerich; Alexander Bürkle; Pleiotropic Cellular Functions of PARP1 in Longevity and Aging: Genome Maintenance Meets Inflammation. Oxidative Medicine and Cellular Longevity 2012, 2012, 1-19, 10.1155/2012/321653.
- Razinah Sharif; Philip Thomas; Peter Zalewski; Robin D. Graham; Michael Fenech; The effect of zinc sulphate and zinc carnosine on genome stability and cytotoxicity in the WIL2-NS human lymphoblastoid cell line. Mutation Research/Genetic Toxicology and Environmental Mutagenesis 2011, 720, 22-33, 10.1016/j.mrgentox.2010.12.004.
- Emily Ho; Chantal Courtemanche; Bruce N. Ames; Zinc Deficiency Induces Oxidative DNA Damage and Increases P53 Expression in Human Lung Fibroblasts. The Journal of Nutrition 2003, 133, 2543-2548, 10.1093/jn/133.8.2543.
- Yang Song; Carolyn S Chung; Richard S Bruno; Maret G Traber; Kenneth H Brown; Janet C King; Emily Ho; Dietary zinc restriction and repletion affects DNA integrity in healthy men.. The American Journal of Clinical Nutrition 2009, 90, 321-8, 10.3945/ajcn.2008.27300.
- Razinah Sharif; Philip Thomas; Peter Zalewski; Michael Fenech; Zinc supplementation influences genomic stability biomarkers, antioxidant activity, and zinc transporter genes in an elderly Australian population with low zinc status. Molecular Nutrition & Food Research 2015, 59, 1200-1212, 10.1002/mnfr.201400784.
- Razinah Sharif; Philip Thomas; Peter Zalewski; Michael Fenech; The role of zinc in genomic stability. Mutation Research/Fundamental and Molecular Mechanisms of Mutagenesis 2012, 733, 111-121, 10.1016/j.mrfmmm.2011.08.009.
- Lucia Letavayová; Viera Vlčková; Jela Brozmanová; Selenium: From cancer prevention to DNA damage. Toxicology 2006, 227, 1-14, 10.1016/j.tox.2006.07.017.
- Wolfgang Maret; The redox biology of redox-inert zinc ions. Free Radical Biology and Medicine 2019, 134, 311-326, 10.1016/j.freeradbiomed.2019.01.006.
- Soheir M. El-Nahas; Fathy E. Mattar; Amal A. Mohamed; Radioprotective effect of vitamins C and E. Mutation Research Letters 1993, 301, 143-147, 10.1016/0165-7992(93)90037-v.
- Maria Konopacka; Maria Widel; Joanna Rzeszowska-Wolny; Modifying effect of vitamins C, E and beta-carotene against gamma-ray-induced DNA damage in mouse cells. Mutation Research/Genetic Toxicology and Environmental Mutagenesis 1998, 417, 85-94, 10.1016/s1383-5718(98)00095-3.
- George J. Delinasios; Mahsa Karbaschi; Marcus S. Cooke; Antony R. Young; Vitamin E inhibits the UVAI induction of “light” and “dark” cyclobutane pyrimidine dimers, and oxidatively generated DNA damage, in keratinocytes. Scientific Reports 2018, 8, 1-12, 10.1038/s41598-017-18924-4.
- Ornella Fantappiè; Maura Lodovici; Paola Fabrizio; Serena Marchetti; Valentina Fabbroni; Michela Solazzo; Nadia Lasagna; Pietro Pantaleo; Roberto Mazzanti; Vitamin E Protects DNA from Oxidative Damage in Human Hepatocellular Carcinoma Cell Lines. Free Radical Research 2004, 38, 751-759, 10.1080/10715760410001705140.
- G. La Fata; N. Van Vliet; S. Barnhoorn; R.M.C. Brandt; S. Etheve; E. Chenal; C. Grunenwald; N. Seifert; P. Weber; J.H.J. Hoeijmakers; et al. Vitamin E Supplementation Reduces Cellular Loss in the Brain of a Premature Aging Mouse Model.. The journal of prevention of Alzheimer's disease 2017, 4, 226-235, 10.14283/jpad.2017.30.
- A. McE. Jenkinson; A. R Collins; S. J. Duthie; K W. J. Wahle; G. G. Duthie; The effect of increased intakes of polyunsaturated fatty acids and vitamin E on DNA damage in human lymphocytes. The FASEB Journal 1999, 13, 2138-2142, 10.1096/fasebj.13.15.2138.
- Jo Aan Goon; Nor Helwa Ezzah Nor Azman; Siti Madiani Abdul Ghani; Zalina Hamid; Wan Zurinah Wan Ngah; Comparing palm oil tocotrienol rich fraction with α-tocopherol supplementation on oxidative stress in healthy older adults. Clinical Nutrition ESPEN 2017, 21, 1-12, 10.1016/j.clnesp.2017.07.004.
- Mustansir Bhori; Kanchanlata Singh; Marar Thankamani; C. Murali Krishna; Exploring the effect of vitamin E in cancer chemotherapy-A biochemical and biophysical insight. Journal of Biophotonics 2018, 11, e201800104, 10.1002/jbio.201800104.
- M Fenech; I Dreosti; C Aitken; Vitamin-E supplements and their effect on vitamin-E status in blood and genetic damage rate in peripheral blood lymphocytes. Carcinogenesis 1997, 18, 359-364, 10.1093/carcin/18.2.359.
- S. Astley; A. Langrish-Smith; S. Southon; M. Sampson; Vitamin E supplementation and oxidative damage to DNA and plasma LDL in type 1 diabetes. Diabetes Care 1999, 22, 1626-1631, 10.2337/diacare.22.10.1626.
- Franca Bianchini; Sölve Elmståhl; Carmen Martinez-Garciá; Anne-Linda Van Kappel; Thierry Douki; Jean Cadet; Hiroshi Ohshima; Elio Riboli; Rudolf Kaaks; Oxidative DNA damage in human lymphocytes: correlations with plasma levels of alpha-tocopherol and carotenoids. Carcinogenesis 2000, 21, 321-324, 10.1093/carcin/21.2.321.
- Lucile Capuron; Aurélie Moranis; Nicole Combe; Florence Cousson-Gélie; Dietmar Fuchs; Véronique De Smedt-Peyrusse; Pascale Barberger-Gateau; Sophie Layé; Vitamin E status and quality of life in the elderly: influence of inflammatory processes. British Journal of Nutrition 2009, 102, 1390-1394, 10.1017/s0007114509990493.
- Radim J. Sram; Blanka Binkova; Pavel Rossner; Vitamin C for DNA damage prevention. Mutation Research/Fundamental and Molecular Mechanisms of Mutagenesis 2012, 733, 39-49, 10.1016/j.mrfmmm.2011.12.001.
- C. G. Fraga; P. A. Motchnik; M. K. Shigenaga; H. J. Helbock; R. A. Jacob; B. N. Ames; Ascorbic acid protects against endogenous oxidative DNA damage in human sperm.. Proceedings of the National Academy of Sciences 1991, 88, 11003-11006, 10.1073/pnas.88.24.11003.
- A Supriya Simon; V Chithra; Anoop Vijayan; Roy D Dinesh; T Vijayakumar; Altered DNA repair, oxidative stress and antioxidant status in coronary artery disease. Journal of Biosciences 2013, 38, 385-389, 10.1007/s12038-013-9313-z.
- Pavel Rossner; Katerina Uhlirova; Olena Beskid; Andrea Rossnerova; Vlasta Svecova; Radim J. Sram; Expression of XRCC5 in peripheral blood lymphocytes is upregulated in subjects from a heavily polluted region in the Czech Republic. Mutation Research/Fundamental and Molecular Mechanisms of Mutagenesis 2011, 713, 76-82, 10.1016/j.mrfmmm.2011.06.001.
- Jana Slyskova; Yolanda Lorenzo; Anette Karlsen; Monica H. Carlsen; Vendula Novosadova; Rune Blomhoff; Pavel Vodicka; Andrew R. Collins; Both genetic and dietary factors underlie individual differences in DNA damage levels and DNA repair capacity. DNA Repair 2014, 16, 66-73, 10.1016/j.dnarep.2014.01.016.
- Serena Guarnieri; Steffen Loft; Patrizia Riso; Marisa Porrini; Lotte Risom; Henrik E. Poulsen; Lars O. Dragsted; Peter Møller; DNA repair phenotype and dietary antioxidant supplementation. British Journal of Nutrition 2008, 99, 1018-1024, 10.1017/s0007114507842796.
- Jeremy Walston; Q. Xue; R. D. Semba; L. Ferrucci; A. R. Cappola; M. Ricks; J. Guralnik; L. P. Fried; Serum Antioxidants, Inflammation, and Total Mortality in Older Women. American Journal of Epidemiology 2005, 163, 18-26, 10.1093/aje/kwj007.
- Pinar Soysal; Ahmet Turan Isik; Andre F. Carvalho; Brisa S. Fernandes; Marco Solmi; Patricia Schofield; Nicola Veronese; Brendon Stubbs; Oxidative stress and frailty: A systematic review and synthesis of the best evidence. Maturitas 2017, 99, 66-72, 10.1016/j.maturitas.2017.01.006.
- Thomas Von Zglinicki; Rita Pilger; Nicolle Sitte; Accumulation of single-strand breaks is the major cause of telomere shortening in human fibroblasts. Free Radical Biology and Medicine 2000, 28, 64-74, 10.1016/s0891-5849(99)00207-5.
- Neva L. Crogan; Nutritional Problems Affecting Older Adults. Nursing Clinics of North America 2017, 52, 433-445, 10.1016/j.cnur.2017.04.005.