Your browser does not fully support modern features. Please upgrade for a smoother experience.
Please note this is an old version of this entry, which may differ significantly from the current revision.
Subjects:
Biophysics
Osteosarcoma (OS) is a malignancy that is becoming increasingly common in adolescents. OS stem cells (OSCs) form a dynamic subset of OS cells that are responsible for malignant progression and chemoradiotherapy resistance. The unique properties of OSCs, including self-renewal, multilineage differentiation and metastatic potential, 149 depend closely on their tumor microenvironment.
- osteosarcoma
- cancer stem cell
- cell plastic
1. Introduction
Osteosarcoma (OS) is a malignancy that most commonly occurs in children and adolescents and is the second highest cause of cancer-related mortality in these groups [1,2,3]. There has been a rise in the annual incidence rate of OS to three cases per million individuals [4]. The majority of OS cases arises in the metaphyseal regions adjacent to the physis, including the distal femur, proximal tibia and the proximal humerus, with a strong capacity for proliferation [5]. Over the past 30 years, the treatment of OS has improved little, such that surgery accompanied with chemoradiotherapy remain as the main method of treatment [6]. Although novel clinical strategies such as gene editing, individualized treatment and novel molecular-targeted therapies, e.g., angiogenesis inhibitors, tyrosine kinase inhibitors and monoclonal antibodies, have all been deployed against OS, the outcomes for patients are poor, particularly those with more aggressive forms of the cancers [3,4,5,6,7]. Therefore, novel treatment strategies are in demand in clinical practice. In addition, the molecular mechanism underlying tumorigenesis and malignant metastasis needs to be studied in detail.
Based on the present research, OS is speculated to have two main origins, bone mesenchymal stem cells (BMSCs) and osteoblasts [8,9,10]. p53 as a classic cancer suppressor gene plays a key role in OS progression. The deficiency of p53 is an important reason leading to primary OS. In addition, retinoblastoma gene (Rb), cyclin dependent kinase inhibitor 2 (CDKN2), KRAS and c-Met also participate in the regulation of OS progression [8,9,10,11]. Within cancer tissues, there exist several dynamic subsets of cancer cells considered to be cancer stem cells (CSCs) or stem cell-like cancer cells [12,13]. CSCs have been frequently reported to exhibit stem cell properties and capabilities of long-term clonal proliferation, tumorigenicity, facilitating metastasis and promoting resistance to chemotherapy and radiotherapy [14,15]. Therefore, exploring the origins of cancer initiation and metastasis will likely facilitate the development of future therapies. In 1994, Lapidot et.al first reported that, in human acute myeloid leukemia, a rare population of CSCs exists [16]. Subsequently, an accumulating number of studies have also reported the existence of CSCs in other solid tumors, including prostate, glioblastoma, hepatoma, breast cancer and OS [17,18,19,20,21,22]. In fact, all types of malignant tumors consist of different subpopulations of tumor cells, leading to high degrees of heterogeneity.
The niche in which CSCs reside is the tumor microenvironment, where they co-exist with adjacent supporting cells, micro-vessels and the extracellular matrix [19,20]. In addition, the tumor microenvironment can contain soluble factors, such as chemokines and cytokines, whilst being under the influence of various mechanical factors, including matrix stiffness, solid stress and fluid stress [23,24]. In the OS microenvironment, OS stem cells (OSCs) are contained in a specialized niche that contains a unique bone microenvironment, which consists of various types of bone cells, such as osteoblasts or osteoclasts. OSCs are similar to other CSCs, in that they account for a proportion of cancer cells with tumorigenic and self-renewal capabilities. The existence of OSCs was first confirmed by Gibbs et al., who found that when primary human OS cells or the OS cell line MG63 were suspended in a serum-free medium with defined growth factors, 0.1% of the cells could form spheres with self-renewal capacity [17,18,19,20,21,22,23,24,25]. Subsequently, a series of studies have proven the existence of OSCs, in addition to revealing the phenotype and possible marker profile of OSCs. The recent studies on possible OSC markers and phenotypes (Table 1).
Table 1. Putative OSC markers and phenotypes.
| Marker | Cell Origin | Phenotype |
|---|---|---|
| CD133 | Saos-2, MG-63, U2-OS, MNNG/HOS, 143B, HOS, Human primary cells | High stem cells gene expression, sphere formation, side population, increased cell proliferation [26,27,28,29,30]. |
| CD117/Stro-1 | K7M2, KHOS/NP, MNNG/HOS, 318–1, P932, BCOS | High stem cells gene expression, sphere formation, drug resistance, in vivo tumorigenicity and metastatic potential [17,18,19,20,21,22,23,24,25,26,27,28,29,30,31]. |
| CD271 | Human primary (FFPE), MNNG/HOS, U2-OS, Saos-2 | High stem cells gene expression, sphere formation, drug resistance, in vivo tumorigenicity [32]. |
| Aldehyde dehydrogenase | MG-63, OS99–1 Hu09, Saos-2 | High stem cells gene expression, sphere formation, drug resistance, increased cell proliferation [33,34]. |
| Stem cells antigen-1 | 4 Murine osteosarcoma cell lines | Sphere formation, in vivo tumorigenicity [35,36] |
| Fas apoptotic inhibitory molecule 2 | MNNG/HOS, U2-OS | Sphere formation, drug resistance, in vivo tumorigenicity [37]. |
| Side population | OS2000, KIKU, NY, Huo9, HOS, U2OS, Saos-2, human primary |
High stem cells gene expression, Sphere formation, in vivo tumorigenicity, self-renewal, apoptosis resistant [38,39,40]. |
| Sphere formation | MG-63, MNNG/HOS, human primary | High stem cells gene expression, drug resistance, in vivo tumorigenicity [17,18,19,20,21,22,23,24,25,26,27,28,29,30,31,32,33,41,42]. |
2. Role of the Tumor Microenvironment in Regulating OSC Stemness
OSCs can interact with their microenvironment through complex and dynamic processes, including variation of oxygen, mechanical interactions, enzymatic modification of the extracellular matrix (ECM) structure and signaling cross-talk, all of which can influence the progression and the dissemination of OS cells (Figure 1)
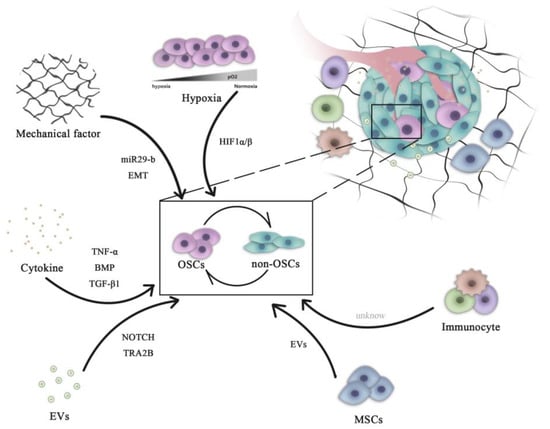
Figure 1. Role of microenvironment signaling in regulating OSC and non-OSC reversion. Complex pathways are necessary for the maintenance of the homeostasis of the OSC population. All microenvironment components, including cells (mesenchymal cells and immune cells) and non-cellular factors (hypoxia, cytokines and mechanical EVs), can influence the dynamic transition between OSCs and non-OSCs. OSCs, osteosarcoma stem cells; miR, microRNA; HIF, hypoxia-inducible factor; EVs, extracellular vesicles; EMT, epithelial-mesenchymal transition; BMP, bone morphogenetic protein; TNF, tumor necrosis factor; TGF, transforming growth factor; TRA2B, transformer 2β homolog.
2.1. Hypoxia
Common hallmarks of solid tumors include intra-tumoral hypoxia, necrosis, acidic environments and disturbed angiogenesis. Previous studies have shown that increased cancer cell stemness is associated with intra-tumoral hypoxia [54,55,56,57]. Our previous study demonstrated that a hypoxia microenvironment could induce non-OSCs dedifferentiation into OSCs by increasing the expression of TGF-β [58].
During the process of tumor formation, excessive proliferation of cancer cells consumes large quantities of oxygen in the microenvironment, resulting in the formation of a hypoxic zone in the central area of the tumor. In addition, aberrant secretion of angiogenetic factors, including vascular endothelial growth factor A and fibroblast growth factor 2 (FGF2), result in malformation and disorder in the neovascularization system [54,55]. This in turn causes the loss of oxygen supply, further aggravating hypoxia in the cancer tissue [59,60].
This hypoxic microenvironment induces the expression of hypoxia-inducible factor-1 (HIF-1), a vital member of the HIF family. By contrast, in the presence of oxygen, HIF-1 undergoes degradation by the von Hippel-Lindau protein, a tumor suppressor protein [61,62,63,64,65]. HIF-1 is a heterodimer that is ubiquitously expressed in human and mouse tissues. HIF-1 consists of two subunits, the hypoxia-inducible, oxygen-dependent subunit HIF-1α and the constitutively-expressed oxygen-independent subunit HIF-1β [66,67,68]. It is only when the oxygen concentration reaches <5% (such as when the volume of the tumor has grown to >300 mm2) that HIF-1α can exist stably. Activity of HIF-1 provides cancer cells with the ability to adapt to hypoxia and is closely associated with tumor metabolism, differentiation, angiogenesis, cell proliferation, metastasis and multidrug resistance. Of note, several studies have demonstrated that elevated expression of HIF-1α promoted the dedifferentiation of cancer cells into CSCs, whereas hypoxia is directly associated with poorer prognoses in patients with OS [69].
Numerous studies have demonstrated that hypoxia can promote the expression of the stem cell marker CD133 to maintain stemness and drug resistance in the Saos-2 OS cell line [70]. Lin et al. previously reported that hypoxia can increase the expression of embryonic stem cell markers, including Oct3/4 and Nanog, in the MNNG/HOS OS cell line [71]. Zhang et al. showed that a hypoxic microenvironment stabilized HIF-1α in OS cells, such that HIF-1 promoted the expression of microRNA (miR or miRNA)-210, which then induced and accelerated the dedifferentiation of OS cells into OSCs [58,59,60,61,62,63,64,65,66,67,68,69,70,71,72]. These observations aforementioned suggest that HIF-1 and subsequent hypoxia signaling pathways can regulate the differentiation of CSCs and the dedifferentiation of non-stem cells in tumors [73,74].
In addition to HIF, hypoxia can also cause integrin-linked kinase dysfunction, triggering CSCs formation [75]. Hypoxia has been previously found to promote breast cancer stemness by HIF-dependent and AlkB homolog 5-mediated N6-methyladenosine (m6A)-demethylation of Nanog mRNA [76]. Shi et al. used the evolutionary theory to identify the hypoxic adaptation-associated gene YTH N6-methyladenosine RNA binding protein 1 (YTHDF1). As a member of the N6-methyladenosine (m6A)-modified RNA-binding protein family, YTHDF1 may interplay with other m6A modifiers and serve a pivotal role in the self-renewal and differentiation of stem cells [77]. Under hypoxia, AKT will accumulate in the mitochondria of tumor cells, whereby 3-phosphoinositide-dependent protein kinase 1 is phosphorylated at special sites. This pathway shifts the tumor metabolic program to glycolysis, which antagonizes apoptosis and autophagy and inhibits oxidative stress. This in turn maintains the survival and proliferation capabilities of tumor cells, as evidenced by the sphere-forming ability of cells in 3D cultures under severe hypoxia [78].
These aforementioned findings suggest that hypoxia may contribute to the creation of a microenvironment rich in tumor stem cells, where this unique hypoxic microenvironment may provide essential cellular interactions and environmental signals for the maintenance of CSCs [70,71,72,73,74,75,76,77,78,79]. By contrast, the hypoxia microenvironment can also regulate non-CSC dedifferentiation by regulating the activities of other pathways, including epithelial-mesenchymal transition (EMT), metabolic reprogramming, DNA hypermethylation and apoptotic resistance. Additionally, the hypoxic microenvironment can mediate the resistance of CSCs against drugs through drug transporters [80]. The majority of CSCs express the ATP-binding cassette (ABC) family of membrane transporters at high levels, including multidrug resistance gene 1, breast cancer resistance protein and multidrug resistance-associated protein. These proteins can transport metabolites, drugs and other substances, allowing CSCs to become highly resistant to chemotherapy. The relationship between hypoxia and ABC proteins was previously documented to have a strong association with mediating tumor drug resistance [81].
In conclusion, the hypoxic microenvironment with the activation of hypoxic signaling can serve key roles in the dedifferentiation of OS cells into OSCs. Therefore, it is important to study the molecular mechanism underlying OS dedifferentiation, which is expected to hold important clinical significance for improving the efficacy of therapeutic strategies. However, the molecular mechanism of how exactly the hypoxic microenvironment can regulate OSC physiology biology requires additional experimental evidence for validation. In particular, HIF-1 is a key molecule of the hypoxia signaling pathway, the downstream molecules of which are expected to become important markers and potential molecular targets of OSCs.
2.2. Biomechanical Force
Under physiological conditions, most if not all organisms experience complex biomechanical forces, including shear stress, matrix stiffness, tension and compression pressure [82,83,84,85,86]. Biomechanical forces experienced by solid tumors have different profiles compare with those in the surrounding or healthy tissue [87]. Throughout the process of cancer development, excessive cell proliferation will lead to the abnormal development of the biomechanical microenvironment, including solid stress, increased matrix stiffness (decrease in OS due to osteolysis) and abnormal interstitial fluid pressure [88,89,90].
These complex mechanical systems are essential for the maintenance of the homeostasis of the CSC population. Indeed, previous studies have demonstrated that non-CSCs can be transformed into CSCs by receiving mechanical signals from the surrounding microenvironment, such as increased matrix stiffness [91,92,93,94] and/or fluid shear stress [95,96,97,98]. In OS, soft substrate (7 kPa) has been reported to preserve OS stemness, mainly through miR-29b/Spin 1-dependent signaling. Manipulation of cancer niche stiffness and miR-29b expression may therefore be potentially novel drug targets in OS [99]. Previous studies have shown that EMT can promote the progression and invasion of tumors [100]. EMT have been observed to serve as a direct link between non-CSCs and the gain of CSC properties [101]. Matrix stiffness in the tumor microenvironment can actively regulate EMT and migration of OS cells through cytoskeletal remodeling and the translocation of myocardin related transcription factor A, which may contribute to cancer progression [102]. Although the aforementioned studies revealed that mechanical factors are at least partially associated with the dynamic conversion between non-CSCs and CSCs, further research into the association between mechanical signaling and OSC stemness is warranted.
Mechanical receptors on the cell surface, such as integrins, CD44 and ion channels, can sense the changes in ECM and activate key downstream molecules, including focal adhesion kinase, integrin-linked kinase, RhoA and yes-associated protein. Several of the signals induce non-CSC reprogramming and transform them into CSCs by increasing the expression of sex determining region Y-box 2 (Sox2), octamer-binding transcription factor (Oct)-4 and Nanog [91,92,93,94,95,96,97,98,99,100,101,102,103,104]. CSCs and normal stem cells frequently share similar surface markers and signaling pathways, which would restrict the design of treatment regimens [105]. The abnormal mechanical system in OS microenvironments, which rarely occur in the harmonious microenvironments of normal stem cells, may provide novel insights for designing CSC-targeted treatment methods. As such, discovering the relationship between biomechanical factors and CSCs will greatly enable the generation of novel research strategies to investigate the occurrence, development, and recurrence of cancers.
2.3. Growth Factors
Growth factors are pivotal in maintaining the physiological behavior of healthy individuals. Cells in the tumor microenvironment can secrete growth factors to regulate processes of tumor development [106,107]. When OS arise in the bone, OS cells secrete factors that direct osteoclast-mediated bone destruction. In addition, matrix-derived growth factors, especially transforming growth factor β1 (TGF-β1), are released from bone matrix. In addition, OS cells can release TGF-β1 directly, where increased TGF-β1 expression is associated with high-grade metastases of OS [108]. TGF-β1 is a multi-function cytokine that serves as a mediator in the tumor to facilitate further tumor expansion, metastasis and cytokine production [109]. Wang et.al previously reported that TGF-β1 can switch the OSC chemoresistance through the miR-499a/SHKBP1 axis [110]. In another study, TGF-β1 signaling and a hypoxic environment were found to induce the transformation of non-OSCs into OSCs dynamically, which promoted the acquisition of chemoresistance, tumorigenicity, neovasculo-genicity and metastatic potential. Furthermore, blocking the TGF-β1 signaling pathway was reported to inhibit this switch from non-OSCs to OSCs, inhibit OSC self-renewal and suppress hypoxia-mediated dedifferentiation [58]. In the bone microenvironment, TGF-β1 signaling is responsible for OSC generation and critical to chemoresistance in vivo. In addition to OS, TGF-β1 can also regulate the dynamic switching between stem cells and non-stem cells to influence the progression of tumors from different tissue origins [111,112]. In conclusion, TGF-β1 serves a key role in regulating the dynamic plasticity of OSC, which can lead to non-stem cells adopting OSC characteristics to promote tumorigenesis and chemoresistance, highlighting TGF-β1 as a potential therapeutic target.
Bone morphogenetic proteins (BMPs) are members of the TGF-β superfamily and serve important roles in the activity of various tissues. In OS, BMP-2 suppresses tumor growth by reducing the expression of oncogenes whilst promoting the differentiation of OSCs [113]. Histological examination and gene expression analysis of OS tissues revealed that fibrotic remodeling of the tumor microenvironment favors tumorigenesis. Zhang et al. previously demonstrated that fibrotic reprogramming in the lung induced by OSCs is critical for OS pulmonary metastasis, with FGF-FGF receptor 2 (FGFR2) signaling being responsible for this important process [114]. In OS, the tumor necrosis factor-α/miR-155 axis has been found to induce OSC transformation between non-OSCs and OSCs through the extracellular signal-regulated protein kinase signaling pathway [115]. Melatonin, one of the hormones secreted by the pineal gland of the brain, has been shown to significantly inhibit sphere formation by OSCs through the key transcription factor Sox-9 [116]. Although all of the aforementioned growth factors have shown the potential to target OSCs, the underlying mechanism require further exploration.
This entry is adapted from the peer-reviewed paper 10.3390/ijms232416155
This entry is offline, you can click here to edit this entry!
