Your browser does not fully support modern features. Please upgrade for a smoother experience.
Please note this is an old version of this entry, which may differ significantly from the current revision.
Subjects:
Biology
Periostin (encoded by the Postn gene) was first recognized in 1993 from a mouse osteoblastic cell line, a secreted molecule containing no transmembrane domain, and was initially known as osteoblast-specific factor-2(OSF-2).
- periostin
- variants
- non-neoplastic diseases
1. Introduction
Periostin (encoded by the Postn gene) was first recognized in 1993 from a mouse osteoblastic cell line, a secreted molecule containing no transmembrane domain, and was initially known as osteoblast-specific factor-2(OSF-2) [1]. It was renamed periostin in 1999 due to its preferential expression in the periosteum and periodontal ligament in adult mice reported by Horiuchi et al. Periostin used as a regulator promotes the adhesion and differentiation of osteoblasts [2].
At approximately 90 kDa, periostin as an N-glycoprotein contains 23 exons exhibiting an NH2-terminal secretory signal peptide, accompanied by a highly conserved cysteine-rich EMI domain, which engages in the formation of multimers through cysteine disulfide bonds [3,4], four consecutive and homologous tandem Fasciclin I (FAS1) domains binding to integrins (αvβ1, αvβ3, αvβ5, αMβ2, α6β4, α5β1) and a COOH-terminal hydrophilic domain as an alternatively spliced region consisting of exons 15–23 (Figure 1) [5,6,7,8,9]. Periostin-integrin interactions lead to the activation of signaling pathways (Table 1; Figure 2). Apart from interacting with integrin receptors, it also binds other ECM proteins, for example, collagens, fibronectin, tenascin C, or heparin [4,10]. Periostin can form 11 splice variants (Figure 3). The expression pattern of periostin splicing variants has been reported in cerebral ischemia, asthma, MI, IPF, retinal ischemia, pIBD, joint, and serum (Table 2). Comparability of mouse and human periostin amino acid is 89.2% overall and 90.1% in a mature condition. Mouse and human periostin are respectively located on chromosome 3 and chromosome 13q.
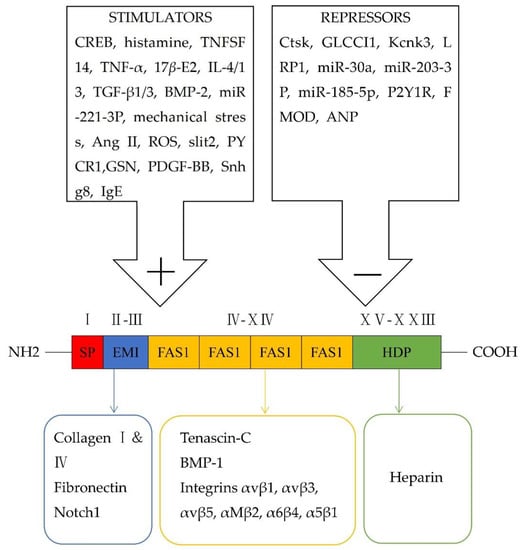
Figure 1. Schematic representation of modular structural domains of periostin and its interaction with different receptors, as well as its stimulators and repressors.
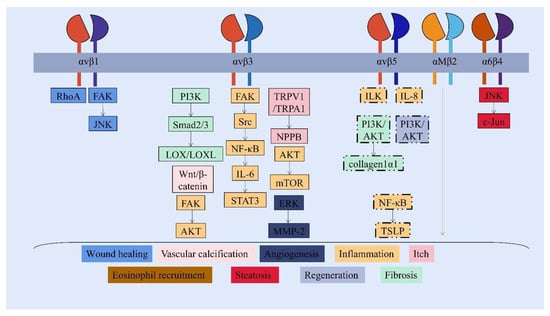
Figure 2. Periostin-integrins interaction and activation of downstream signaling pathways. The FAS1 domain interacts with integrin receptors to activate different and overlapping signaling pathways, which modulate the progression of non-neoplastic diseases under pathological status. (Shared signaling pathways of integrin αv are shown in the box with a dotted line.)
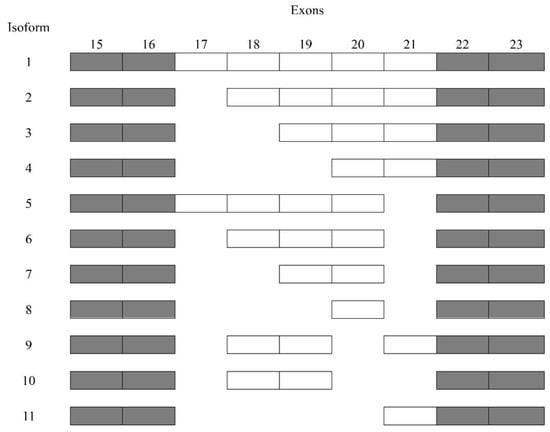
Figure 3. Sequencing of periostin splice variants.
Table 1. Expression (upregulation ↑ or downregulation ↓) and roles of periostin, the periostin-involved signaling pathways, therapies based on periostin, and potential disease biomarkers in disease progression.
| Tissues/ Diseases |
Expression of Periostin |
Roles of Periostin |
Reference | Periostin- Involved Downstream Signaling Pathways |
Reference | Therapies Based on Periostin |
Reference | Potential Disease Biomarkers |
Reference |
|---|---|---|---|---|---|---|---|---|---|
| EBI and cerebral ischemia | ↑ | BBB disruption | [11] | p38/ERK/MMP-9 | [11] | IAXO-102 Clarithromycin |
[12] [13] |
Serum periostin |
[14] |
| Ocular diseases | ↑ | Inflammation | [15] | Betamethasone; tacrolimus | [16] | Tear periostin | [17] | ||
| Tissue remodeling |
[18] | Serum periostin |
[19] | ||||||
| CRS and AR | ↑ | Inflammation | [20,21] | Omalizumab; mepolizumab; methylprednisolone; doxycycline | [22] | Serum periostin |
[23] | ||
| Tissue remodeling |
[21,24] | Src/AKT/mTOR | [24] | ||||||
| Protective role | [25] | Dupilumab | [26] | NLF periostin | [27] | ||||
| GCs | [28] | ||||||||
| AH | [29] | ||||||||
| AG-Ex | [30] | ||||||||
| Periodontitis | ↑ | Tissue remodeling |
[31] | GCF periostin | [32,33] | ||||
| Wound healing | [31] | β1/FAK/JNK; β1/RhoA | [31] | ||||||
| Bone formation | [34,35] | Saliva periostin | [33] | ||||||
| AD | ↑ | Inflammation | [36] | αv/NF-κB/TSLP; NF-κB/IL-6 | [36] | Dupilumab | [26] | ||
| Itch | [36] | αvβ3/TRPV1/TRPA1/NPPB; TSLP/TSLPR/JAK/STAT | [36] | ||||||
| Epidermal barrier malfunction | [37] | IL-24/STAT3 | [37] | Cinnamaldehyde | [38] | ||||
| Scleroderma | ↑ | Skin fibrosis | [39] | αv/PI3K/AKT/collagen1α1 | [39] | Crenolanib | [40] | Serum periostin |
[40] |
| EE | ↑ | Inflammation | [41] | αM | [41] | Dupilumab | [26] | Serum periostin |
[42] |
| Asthma | ↑ | Inflammation | [43] | Tralokinumab; dupilumab | [44,45] | Serum periostin |
[46] | ||
| Tissue remodeling |
[47] | Omalizumab | [48] | Plasma periostin |
[49] | ||||
| Hydroprednisone | [50] | Saliva periostin | [49] | ||||||
| Protective roles | [51,52] | Clarithromycin | [53] | EBC periostin | [54] | ||||
| Sputum periostin |
[55] | ||||||||
| Cardiovascular diseases (MI, heart failure, VHD, hypertension, and vascular calcification) | ↑ | Inflammation | [56] | Periostin/NLRP3/caspase-1 | [56] | Valsartan | [57] | Plasma periostin |
[58] |
| Tissue remodeling |
[59,60,61,62,63,64] | ||||||||
| Wound healing | [59,65] | Simvastatin | [63] | ||||||
| Cardiomyocytes apoptosis | [66] | ||||||||
| Myocardial regeneration |
[5,67,68] | αvβ1/αvβ3/αvβ5/PI3K/Akt; TNF-α/NF-κB; PI3K/AKT/cyclin D1 | [5,67,68] | Resveratrol | [69] | ||||
| Angiogenesis | [5] | ||||||||
| Vascular calcification | [70,71] | Crenolanib | [40] | ||||||
| Lung diseases (PF, EP, COVID-19, and PH) | ↑ | Inflammation | [72,73] | Serum monomeric periostin |
[74] | ||||
| Tissue remodeling |
[72,75,76,77] | EBC periostin | [74] | ||||||
| Angiogenesis | [78] | BALF periostin | [79,80] | ||||||
| Liver disease | ↑ | Liver steatosis | [81,82,83] | α6β4/JNK/c-Jun | [83] | Serum periostin |
[84] | ||
| Hepatic fibrosis | [82,85] | αvβ3/PI3K/Smad2/3/LOX/LOXL | [85] | ||||||
| Liver regeneration and angiogenesis |
[86] | ||||||||
| CKD | ↑ | Inflammation | [87,88] | αv/ILK; β3/FAK/AKT; αVβ3/AKT/mTOR | [5,88,89] | Losartan | [90] | Urine periostin | [91] |
| Renal fibrosis | [87,92,93] | FAK/p38/ERK; p38 MAPK | [92,93] | ||||||
| Vascular calcification |
[94] | αvβ3/Wnt/β-catenin | [94] | ||||||
| Renal repair | [95] | ||||||||
| Renal malfunction |
[96] | Serum periostin |
[97] | ||||||
| IBD | ↑ | Inflammation | [98] | NF-κB; αv/IL-8 | [98] | Plasma periostin |
[99] | ||
| Serum periostin |
[100] | ||||||||
| Osteoarthrosis (RA, OA, AS, osteoporosis, DDH, and IVD D) |
↓ in RA and osteoporosis | Bone formation | [101,102,103] | Inhibition of sclerostin/LRP5/Wnt, β-catenin; Wnt/β-catenin; ILK/Akt/GSK-3β; | [101,102,103] | Serum periostin |
[104] | ||
| ↑ in OA, AS, DDH, and IVDD | Inflammation | [105,106,107,108] | NF-κB/IL-6/8; Wnt/β-catenin/MMP-13/ADAMTS4; DDR1/Akt/Wnt/β-catenin/MMP-13; αvβ3/FAK/Src/NF-κB/IL-6/STAT3 | [105,106,107,108] | SF periostin | [109] | |||
| K-Postn | [110] | ||||||||
Table 2. The expression and roles of periostin isoforms in tissues/diseases.
| Tissues/Diseases | Certain Periostin Variants Expressed in Tissues/Diseases |
Roles of the Periostin Variants | Reference |
|---|---|---|---|
| Cerebral ischemia | Isoform 2 | Isoform 2 Minimizing the area of cerebral infarction via phosphorylation of Akt |
[111] |
| Asthma | Isoforms 6, 7, and 8 | Isoform 8 Promoting the eosinophil adhesion under IL-5 stimulation αMβ2] |
[112,113] |
| MI | Isoforms 1, 2, 5, and 6 | Isoform 1 Decreasing the attachment of fibroblasts and myocytes as well as facilitating myocytes death leading to ventricular dilation and tissue remodeling Isoform 6 Contributing to the migration of activated fibroblasts and healing of impaired tissue via the αv/FAK/AKT signaling pathway |
[114] |
| IPF | All periostin variants lacking exon 21 | - | [115] |
| Retinal ischemia | Isoforms 1, 2, and 5 | Isoforms 1, 2, and 5 Promoting preretinal pathological NV |
[116] |
| pIBD | Isoforms 2, 6, 7, and 8 | - | [99] |
| Joint | Articular chondrocytes highly expressed isoforms1 and 5, and anterior cruciate ligament(ACL) progenitor cells overexpressed isoforms 3, 4, 6, 7, and 8 | - | [117] |
| Serum | At least five isoforms, including 1 or 2, 3, 4, 5, 6 | At least five isoforms, including 1 or 2, 3, 4, 5, 6 Forming complex with IgA |
[118] |
Periostin is commonly overexpressed in human tissues during pathological processes. Periostin, as a matricellular protein and an ECM protein, exerts different roles in tissue development and progression of diseases, including brain injury, ocular diseases, chronic rhinosinusitis, allergic rhinitis, dental diseases, atopic dermatitis, scleroderma, eosinophilic esophagitis, asthma, cardiac diseases, lung diseases, liver diseases, chronic kidney diseases, inflammatory bowel disease, and osteoarthrosis. In a normal physiological situation, periostin is beneficial in mediating teeth development, maintaining the integrity of periodontal ligament (PDL) in postnatal teeth enamel formation, and mediating bone remodeling after orthodontic movement [119,120]; periostin promotes migration of mesenchymal cells in an αvβ3- and β1-based Rho/PI3K signaling mechanism during valve maturation [10]. Besides, during pathogenesis, the roles of periostin are more extensive, including tissue remodeling, fibrosis, inflammation, wound healing, repair, angiogenesis, tissue regeneration, bone formation, barrier, and vascular calcification; this makes it different from other ECM proteins.
Periostin assists in modulating the ECM network [9]. Periostin/BMP-1/LOX cascade assisted in collagen cross-linking [121]. During abnormal scar formation, periostin stimulated the secretion of TGF-β1 via the RhoA/ROCK signaling pathway in human dermal fibroblasts (HDFs), yielding a vicious circle [122].
2. Early Brain Injury (EBI) and Cerebral Ischemia
The periostin was upregulated in neurons and capillary endothelial cells in the cerebral cortex at 24 h post-subarachnoid hemorrhage (SAH) and initiated BBB disruption, possibly via p38/ERK/MMP-9 signaling pathways and induction of tenascin-C [11].
Following transient cerebral ischemia, isoform 2 minimized the area of cerebral infarction displaying a neuroprotective role with phosphorylation of Akt [111]. Greater serum periostin levels were related to a larger cerebral infarction area and more serious neurological defects at 6-28 days following ischemia [14]. Toll-like receptor 4 (TLR4) selective blockade-IAXO-102 and clarithromycin inhibited BBB disruption and periostin expression [12,13].
3. Cardiovascular Diseases
3.1. Myocardial Infarction (MI)
Ang II evidently increased periostin through Ras/p38 MAPK (mitogen-activated protein kinase)/CREB and ERK/TGF-β1 pathways in myocytes and fibroblasts [123]. Detection of human tissue specimens reflected prominently high periostin expression in ischemic and reperfused tissue, as well as no expression in healthy myocardium [59]. The lineage analyses of mice verified that periostin-expressing CFs mainly derived from a mass of TCF21+ cells [124]. After MI, TGF-β1, mechanical pressure, and Cyclic AMP response element-binding protein 1 (CREB) stimulated cardiac fibroblasts, thereby augmenting ECM deposition, development of collagenous scar and cardiac remodeling, and release of periostin [125]. TGF-β1 upregulated periostin levels in CFs and vascular smooth muscle cells (VSMCs) employing Smad signaling pathways [126,127]. Periostin showed minimal levels under miR-203-3p-binding circumstances restricting cardiomyocytes apoptosis. However, the complex of periostin, miR-203-3p, and small nucleolar RNA host gene 8 (Snhg8) mediated neonatal mouse cardiomyocytes (NMCMs) apoptosis after hypoxia-treated NMCMs, contributing to acute myocardial infarction [66]. Treatment of MI with cardiac mesenchymal stem cells (MSCs) marked by Nestin demonstrated a greater effect on cardiac healing than bone marrow-derived MSCs (NesbmMSCs), which results from part involvement of periostin-induced M2 macrophage polarization [65]. In a rat MI model, Yoshiaki Taniyama et al. discovered four periostin isoforms, including isoforms 1, 2, 5, and 6. Isoform 1 decreased the attachment of fibroblasts and myocytes as well as facilitated myocyte death, leading to ventricular dilation and tissue remodeling. Blockade of exon 17 as prior target assists in protesting cardiac remodeling, diminishing fibrosis, ameliorating ejection fraction, and cardiac function eight weeks after MI [114]. Isoform 6 can mediate the migration of activated fibroblasts and the healing of impaired tissue by αv/FAK/AKT cascade [59]. The inhibition of periostin by valsartan might have an improved effect on cardiac remodeling after MI [57].
3.2. Cardiomyocyte Regeneration
Release of periostin facilitated cardiomyocyte regeneration and angiogenesis by interacting with αvβ1, αvβ3, or αvβ5 integrins on myocytes and vascular endothelial cells to activate the PI3K-Akt pathways after MI. The treatment of animals with periostin patches (lacking the N-terminal signal peptide and C-terminal region) not only perfected cardiac fraction and ejection fraction but also contained fibrosis after MI [5]. Periostin eased inflammation and induced reentry of the cardiomyocytes cycle via TNF-α/NF-κB signaling transduction in conjunction with a declining caspase 7 activity [67]. Periostin ablation hindered myocardial regeneration by suppressing the PI3K/AKT/cyclin D1 transmission [68]. Another work in a mouse model of overexpressed full-length periostin indicated that periostin did not speed up the DNA synthesis of cardiomyocytes [128]. Further studies are needed to clarify these issues.
3.3. Heart Failure
In diabetic rat hearts, periostin is noticeably overexpressed relative to healthy controls [79]. In the experimental autoimmune myocarditis (EAM) rats model, periostin was spotted in macrophages and fibroblasts. It elicited cardiac fibrosis, likely by recruiting immune cells [60]. A recent examination of atrial appendages from atrial fibrillation (AF) patients suggested a clear association between periostin levels of atrial tissues and deteriorated heart failure, as well as lessened ejection fraction [129]. MiR-30a and fibromodulin (FMOD) tempted the descent of periostin levels and the decrease of atrial fibrosis [61]. GSN, silencing P2Y1R, and slit2-Robo1 pathways inversely initiated periostin release, tempting fibrosis [62]. Periostin prompted pyroptosis by triggering the NLRP3/caspase-1 pathway during myocardial ischemia-reperfusion injury (MIRI) [56].
Valsartan and simvastatin (SIM) hindered periostin expression and alleviated pathologic remodeling [63,79]. Targeting of diabetic animals with the antioxidant resveratrol limited myofibroblast activation and downregulated the expression of periostin via suppressing ERK/TGF-β signaling [69].
3.4. Valvular Heart Disease (VHD)
Periostin expression intensively goes up in valvular interstitial cells (VICs) of the mitral valve, compared to wild-type mice. The mitral valve biopsies of male patients going through prosthetic surgery detected a pronounced enhancement in periostin in the ventricular [130]. Besides, periostin was firmly upregulated in the infiltrated inflammatory cells and myofibroblasts within patients with atherosclerotic or rheumatic valves. Meanwhile, massive periostin in the valve leaflet brings about extensive production of matrix metalloproteinase-2 (MMP-2) and MMP-9, leading to severe fibrosis in atherosclerotic and rheumatic VHD [64]. Periostin also prompted the osteogenic potential of aortic valve calcification [131].
3.5. Hypertension and Vascular Calcification
Atrial natriuretic peptide (ANP) inhibited periostin expression in the VSMCs and cardiac fibroblasts [70], but oxidative stress contributes to periostin production [132]. The increase in periostin augmented the differentiation and migration of VSMCs [133].
In a hyperlipidemia-associated model of rats, periostin upregulation caused calcium deposits through the successive inhibition of p53 and SLC7A11 in VSMCs [71]. Additionally, plasma periostin levels were positively connected with the Agatston score in patients with coronary artery calcification (CAC). Periostin promoted glycolysis and mitochondrial malfunction as well as contained peroxisome proliferation-activated receptor γ(PPARγ) in VSMCs, thereby provoking arterial calcification [58].
This entry is adapted from the peer-reviewed paper 10.3390/cells12010050
This entry is offline, you can click here to edit this entry!
