Due to the confirmed therapeutic potential of resveratrol (Rv) for eye diseases, namely its powerful anti-angiogenic and antioxidant effects, this molecule must be studied more deeply. Nowadays, the pharmaceutic and pharmacokinetic available studies offer a troubling picture because of its low stability and bioavailability. To overcome this problem, researchers started to design and create different delivery systems that could improve the delivery amount of Rv. Therefore, this review aims to shed light on the proper and efficient techniques to isolate, purify and quantify the Rv molecule, and how this therapeutic molecule can be a part of a delivery system. The Rv great impact on aspects regarding its stability, bioavailability, and absorption are also debated here, based on the existent literature on in vitro and in vivo human and animal studies. Moreover, after its absorption, the Rv influence at the molecular level in ocular pathologies is described. In addition, the present review summarizes the available literature about Rv, hoping Rv will gain more attention to investigate its unexplored side.
- resveratrol
- delivery systems
- ocular pathologies
- antioxidant activity
- VEGF
- Sirtuins
1. Introduction
2. The Occurrence and Sources of Resveratrol
3. Biochemical Insight into Biosynthesis Pathways of Resveratrol and Its Derivatives
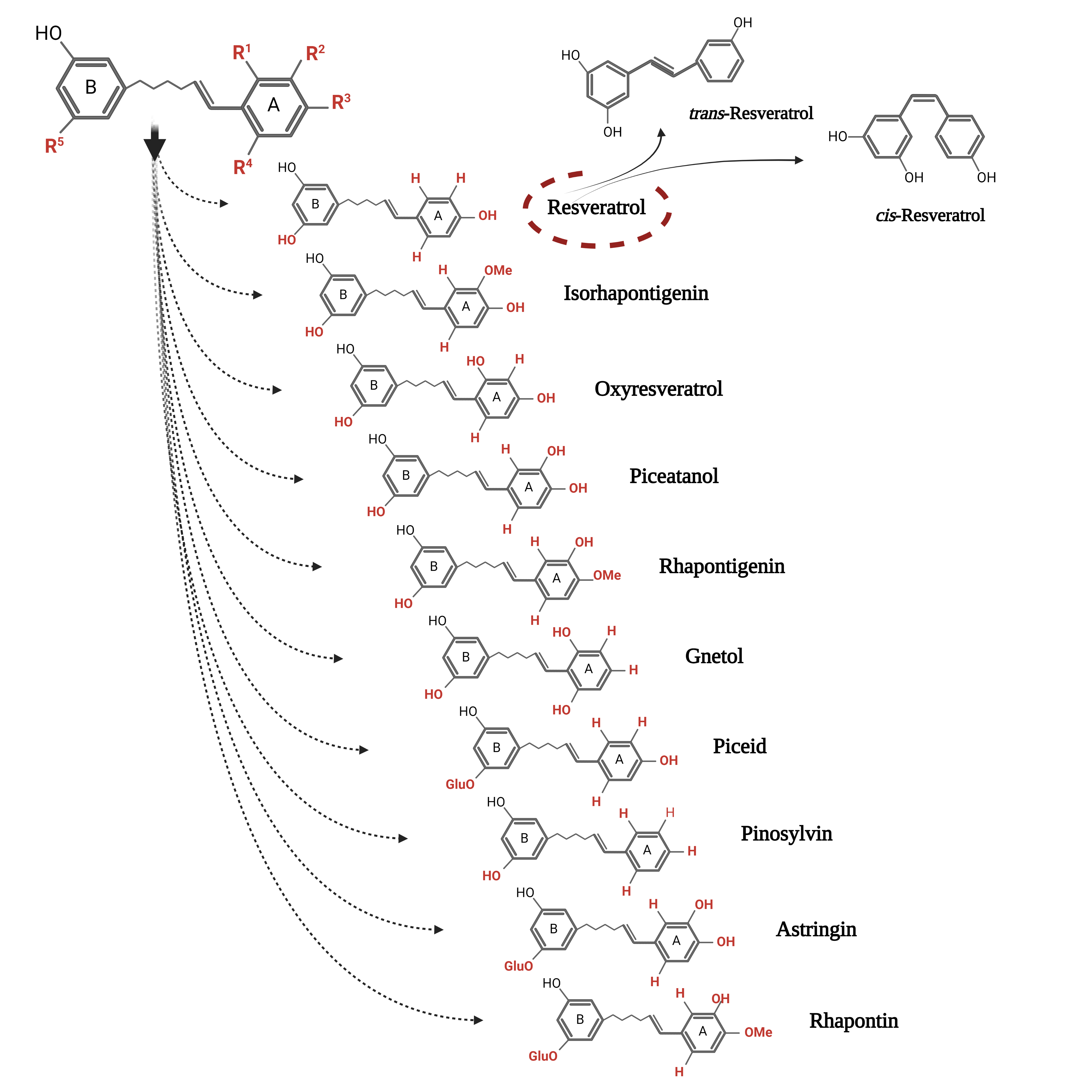 Figure 1. The chemical structure of the two conformations of resveratrol and its main monomeric derivatives.
Figure 1. The chemical structure of the two conformations of resveratrol and its main monomeric derivatives.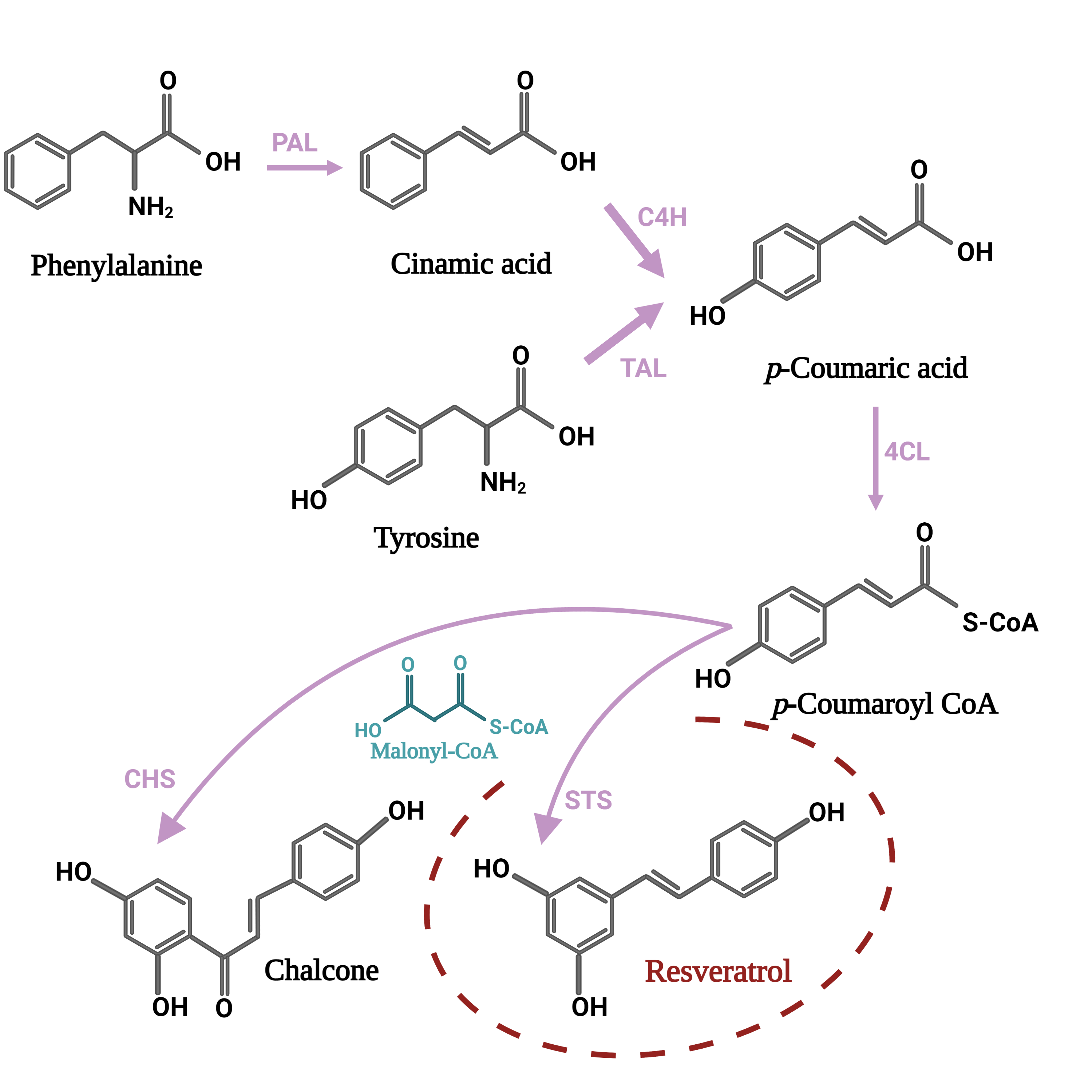
4. The Stability of Resveratrol
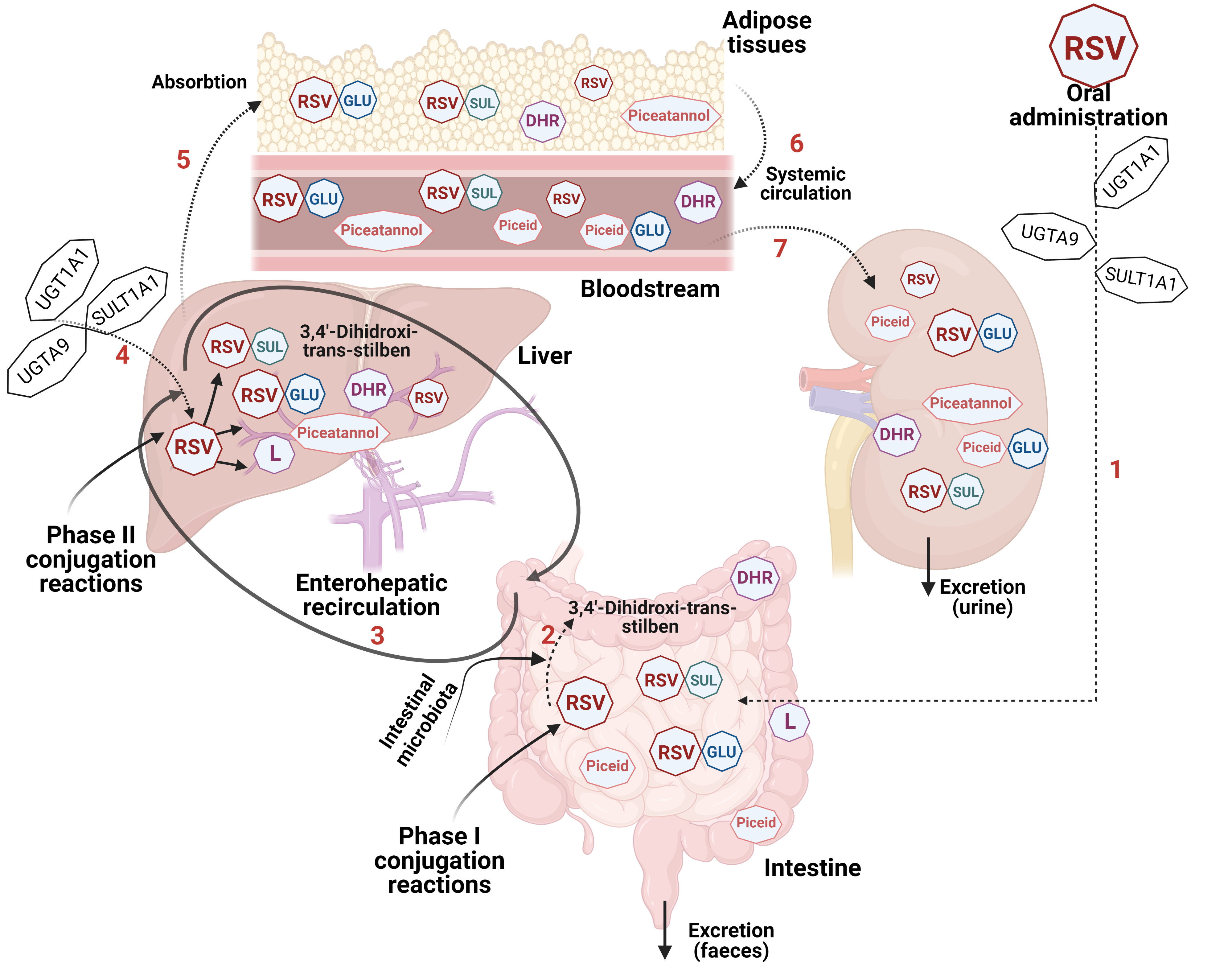
5. The Bioavailability and Safety of Resveratrol
6. The Anti-Angiogenic Modulatory Effect of Resveratrol
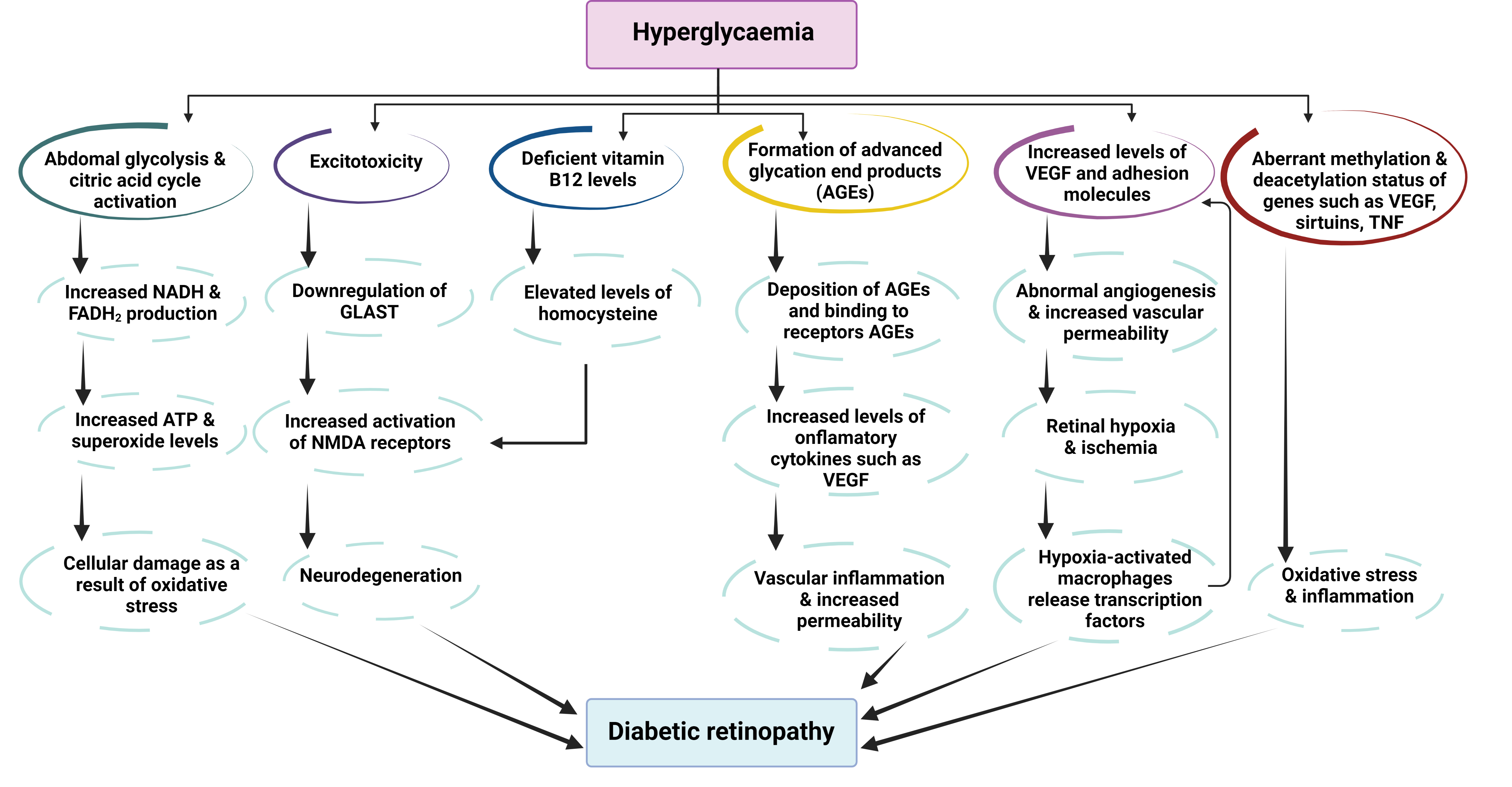 Figure 4. Pathophysiological pathways of diabetic retinopathy as a consequence of hyperglycemia. Abbreviations: AGEs—advance glycation end products; FADH2—redox cofactor flavin adenine dinucleotide; GLAST—glutamate transporter protein; NADH—reduced form of nicotinamide adenine dinucleotide; NMDA—N-methyl D-aspartate receptor; TNF—tumor necrosis factor; VEGF—vascular endothelium growth factor (adapted from [76]).
Figure 4. Pathophysiological pathways of diabetic retinopathy as a consequence of hyperglycemia. Abbreviations: AGEs—advance glycation end products; FADH2—redox cofactor flavin adenine dinucleotide; GLAST—glutamate transporter protein; NADH—reduced form of nicotinamide adenine dinucleotide; NMDA—N-methyl D-aspartate receptor; TNF—tumor necrosis factor; VEGF—vascular endothelium growth factor (adapted from [76]).7. The Antioxidant Role of Resveratrol in Eye Diseases
8. The Relationship between Resveratrol and Sirtuins
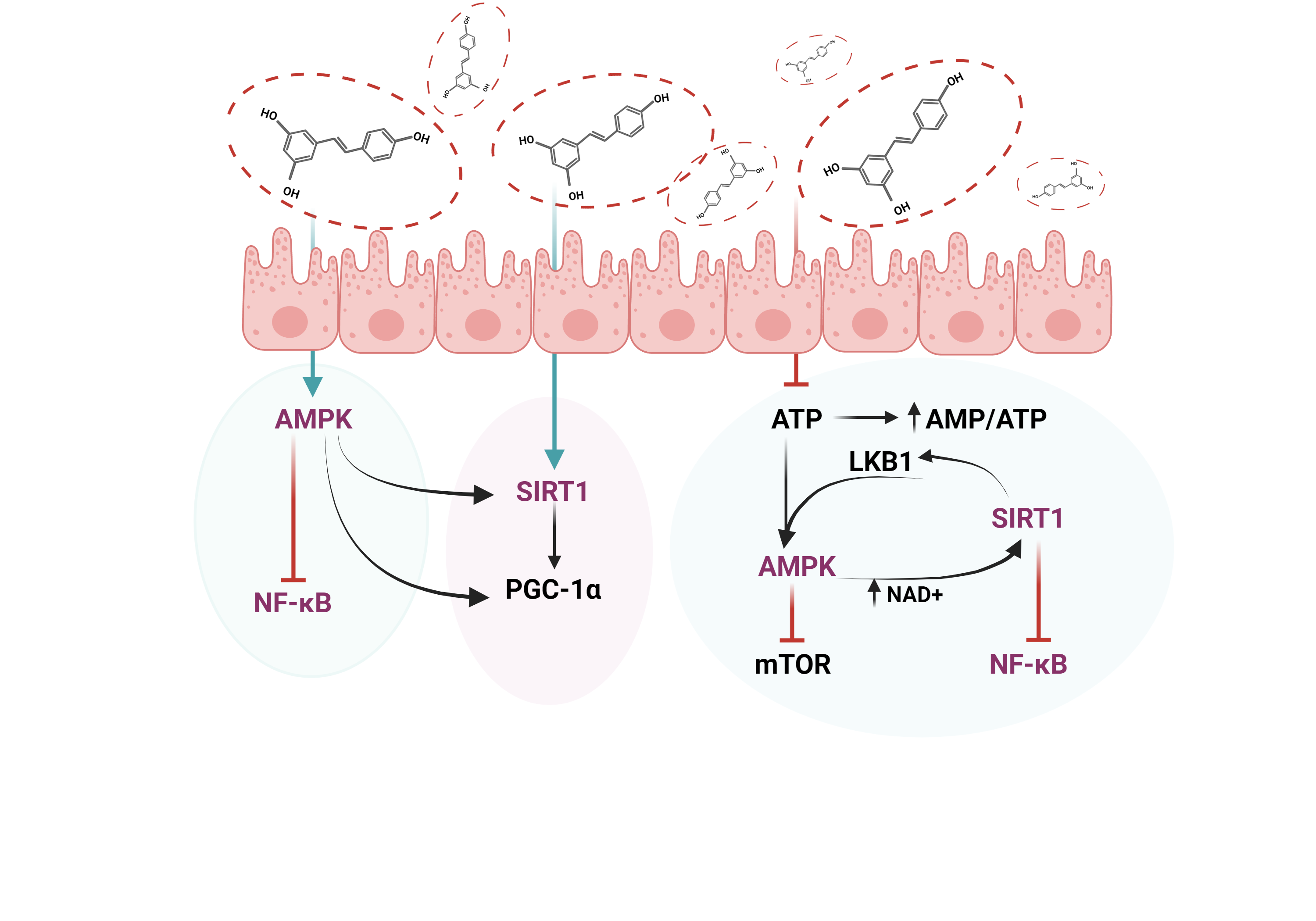 Figure 5. The interplay between key molecules implicated in the management of diabetic retinopathy. Abbreviations: AMPK—AMP-activated protein kinase, SIRT1—Sirtuin 1, NF-kB—Nuclear factor-kB, PGC-1α—Peroxisome proliferator-activated receptor-gamma coactivator, mTOR—Mammalian target of rapamycin, LKB1—liver kinase B1, AMP—Adenosine monophosphate, ATP—Adenosine 5′-triphosphate, NAD+—Nicotinamide adenine dinucleotide.
Figure 5. The interplay between key molecules implicated in the management of diabetic retinopathy. Abbreviations: AMPK—AMP-activated protein kinase, SIRT1—Sirtuin 1, NF-kB—Nuclear factor-kB, PGC-1α—Peroxisome proliferator-activated receptor-gamma coactivator, mTOR—Mammalian target of rapamycin, LKB1—liver kinase B1, AMP—Adenosine monophosphate, ATP—Adenosine 5′-triphosphate, NAD+—Nicotinamide adenine dinucleotide.9. Resveratrol Delivery Systems
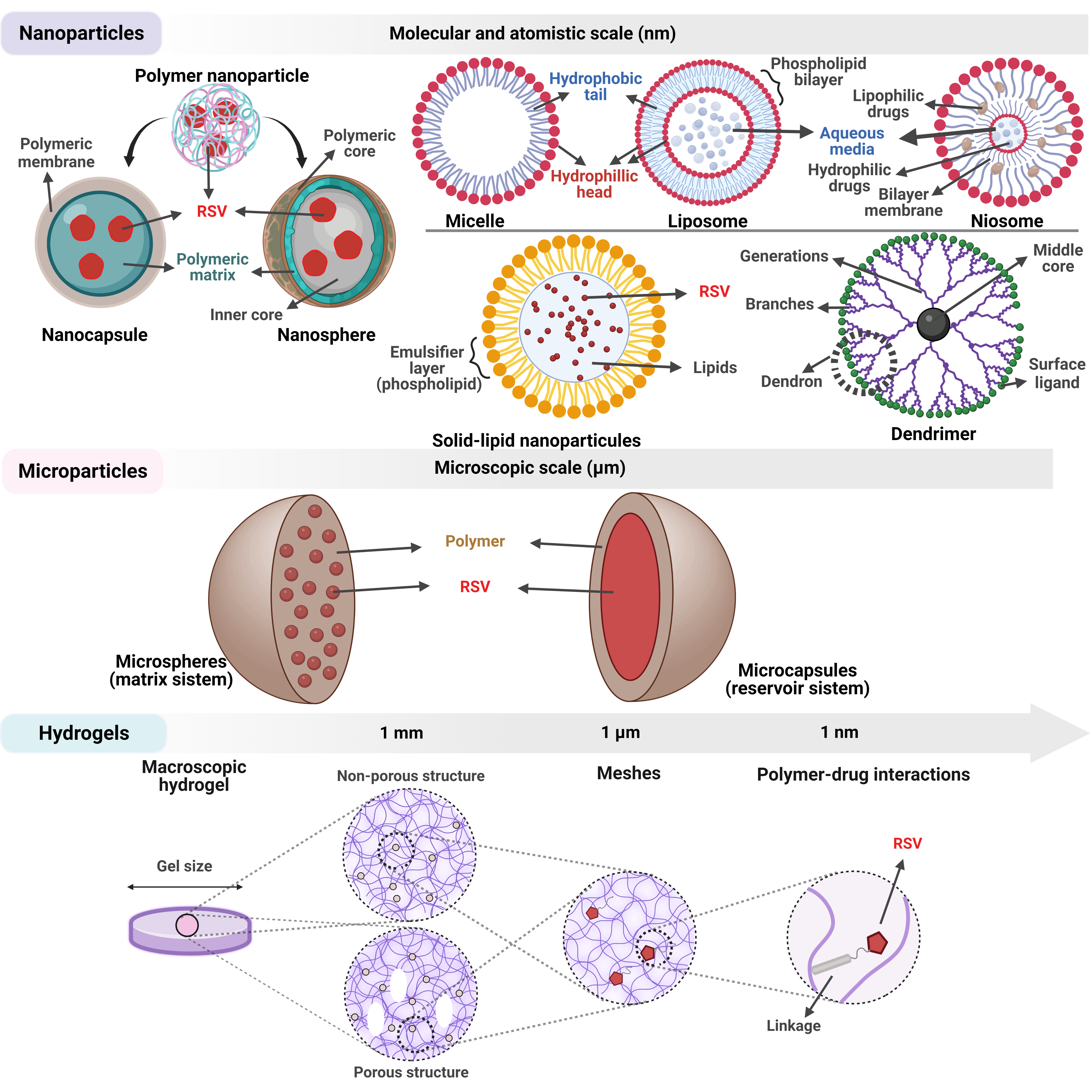 Figure 6. Structural aspects regarding the main resveratrol delivery systems successfully used to date.
Figure 6. Structural aspects regarding the main resveratrol delivery systems successfully used to date.10. Conclusions and Future Perspectives
This entry is adapted from the peer-reviewed paper 10.3390/antiox11122447
References
- Jiang, T.; Gu, J.; Chen, W.; Chang, Q. Resveratrol inhibits high-glucose-induced inflammatory “metabolic memory” in human retinal vascular endothelial cells through SIRT1-dependent signaling. Can. J. Physiol. Pharmacol. 2019, 97, 1141–1151.
- Anekonda, T.S.; Adamus, G. Resveratrol prevents antibody-induced apoptotic death of retinal cells through upregulation of Sirt1 and Ku70. BMC Res. Notes 2008, 1, 122.
- Singh, G. Resveratrol: Nanocarrier-based delivery systems to enhance its therapeutic potential. Nanomedicine 2020, 15, 2801–2817.
- Manojlovic, V.; Rajic, N.; Djonlagic, J.; Obradovic, B.; Nedovic, V.; Bugarski, B. Application of Electrostatic Extrusion—Flavour Encapsulation and Controlled Release. Sensors 2008, 8, 1488–1496.
- Davidov-Pardo, G.; McClements, D.J. Resveratrol encapsulation: Designing delivery systems to overcome solubility, stability and bioavailability issues. Trends Food Sci. Technol. 2014, 38, 88–103.
- Tapeinos, C.; Battaglini, M.; Ciofani, G. Advances in the design of solid lipid nanoparticles and nanostructured lipid carriers for targeting brain diseases. J. Control. Release 2017, 264, 306–332.
- Neves, A.R.; Lúcio, M.; Martins, S.; Lima, J.L.C.; Reis, S. Novel resveratrol nanodelivery systems based on lipid nanoparticles to enhance its oral bioavailability. Int. J. Nanomed. 2013, 8, 177–187.
- Mohseni, R.; ArabSadeghabadi, Z.; Ziamajidi, N.; Abbasalipourkabir, R.; RezaeiFarimani, A. Oral Administration of Resveratrol-Loaded Solid Lipid Nanoparticle Improves Insulin Resistance Through Targeting Expression of SNARE Proteins in Adipose and Muscle Tissue in Rats with Type 2 Diabetes. Nanoscale Res. Lett. 2019, 14, 227.
- Li, Z.; Tan, S.; Li, S.; Shen, Q.; Wang, K. Cancer drug delivery in the nano era: An overview and perspectives (Review). Oncol. Rep. 2017, 38, 611–624.
- Zhao, Y.N.; Cao, Y.N.; Sun, J.; Liang, Z.; Wu, Q.; Cui, S.H.; Zhi, D.F.; Guo, S.T.; Zhen, Y.H.; Zhang, S.B. Anti-breast cancer activity of resveratrol encapsulated in liposomes. J. Mater. Chem. B 2020, 8, 27–37.
- Jhaveri, A.; Deshpande, P.; Pattni, B.; Torchilin, V. Transferrin-targeted, resveratrol-loaded liposomes for the treatment of glioblastoma. J. Control. Release 2018, 277, 89–101.
- Jagwani, S.; Jalalpure, S.; Dhamecha, D.; Jadhav, K.; Bohara, R. Pharmacokinetic and Pharmacodynamic Evaluation of Resveratrol Loaded Cationic Liposomes for Targeting Hepatocellular Carcinoma. ACS Biomater. Sci. Eng. 2020, 6, 4969–4984.
- Wang, M.; Li, L.; Zhang, X.; Liu, Y.; Zhu, R.; Liu, L.; Fang, Y.; Gao, Z.; Gao, D. Magnetic Resveratrol Liposomes as a New Theranostic Platform for Magnetic Resonance Imaging Guided Parkinson’s Disease Targeting Therapy. ACS Sustain. Chem. Eng. 2018, 6, 17124–17133.
- Yücel, Ç.; Karatoprak, G.; Aktaş, Y. Nanoliposomal Resveratrol as a Novel Approach to Treatment of Diabetes Mellitus. J. Nanosci. Nanotechnol. 2018, 18, 3856–3864.
- Machado, N.D.; Fernandez, M.A.; Diaz, D.D. Recent Strategies in Resveratrol Delivery Systems. Chempluschem 2019, 84, 951–973.
- Muzzalupo, R.; Tavano, L. Niosomal drug delivery for transdermal targeting: Recent advances. Res. Rep. Transdermal Drug Deliv. 2015, 4, 23–33.
- El-Haddad, M.E.; Hussien, A.A.; Saeed, H.M.; Farid, R.M. Down regulation of inflammatory cytokines by the bioactive resveratrol-loaded chitoniosomes in induced ocular inflammation model. J. Drug Deliv. Sci. Technol. 2021, 66, 102787.
- Pircalabioru, G.G.; Bleotu, C.; Curutiu, C.; Mihaescu, G.; Chifiriuc, M.-C. Chapter 2—Nanodrug delivery systems in cancer. In Biomedical Applications of Nanoparticles; Grumezescu, A.M., Ed.; William Andrew Publishing: Norwich, NY, USA, 2019; pp. 31–62.
- Ahmadi, Z.; Mohammadinejad, R.; Ashrafizadeh, M. Drug delivery systems for resveratrol, a non-flavonoid polyphenol: Emerging evidence in last decades. J. Drug Deliv. Sci. Technol. 2019, 51, 591–604.
- Devi, P.; Sharma, P.; Rathore, C.; Negi, P. Novel Drug Delivery Systems of Resveratrol to Bioavailability and Therapeutic Effects. In Resveratrol—Adding Life to Years, Not Adding Years to Life; IntechOpen: London, UK, 2019.
- Li, M.; Zhang, L.; Li, R.; Yan, M. New resveratrol micelle formulation for ocular delivery: Characterization and in vitro/in vivo evaluation. Drug Dev. Ind. Pharm. 2020, 46, 1960–1970.
- Selvarajan, V.; Obuobi, S.; Ee, P.L.R. Silica Nanoparticles—A Versatile Tool for the Treatment of Bacterial Infections. Front. Chem. 2020, 8, 602.
- Kumar, A.; Ma, H.; Zhang, X.; Huang, K.; Jin, S.; Liu, J.; Wei, T.; Cao, W.; Zou, G.; Liang, X.-J. Gold nanoparticles functionalized with therapeutic and targeted peptides for cancer treatment. Biomaterials 2012, 33, 1180–1189.
- Dong, Y.; Wan, G.; Yan, P.; Qian, C.; Li, F.; Peng, G. Fabrication of resveratrol coated gold nanoparticles and investigation of their effect on diabetic retinopathy in streptozotocin induced diabetic rats. J. Photochem. Photobiol. B Biol. 2019, 195, 51–57.
- Sarma, S.; Agarwal, S.; Bhuyan, P.; Hazarika, J.; Ganguly, M. Resveratrol-loaded chitosan-pectin core-shell nanoparticles as novel drug delivery vehicle for sustained release and improved antioxidant activities. R. Soc. Open Sci. 2022, 9, 210784.
- Soldati, P.P.; Polonini, H.C.; Paes, C.Q.; Restrepob, J.A.S.; Creczynksi-Pasa, T.B.; Chaves, M.G.A.M.; Brandão, M.A.F.; Pittella, F.; Raposo, N.R.B. Controlled release of resveratrol from lipid nanoparticles improves antioxidant effect. IFAC Pap. 2018, 51, 16–21.
- Ha, E.-S.; Park, H.; Lee, S.-K.; Sim, W.-Y.; Jeong, J.-S.; Baek, I.-h.; Kim, M.-S. Pure Trans-Resveratrol Nanoparticles Prepared by a Supercritical Antisolvent Process Using Alcohol and Dichloromethane Mixtures: Effect of Particle Size on Dissolution and Bioavailability in Rats. Antioxidants 2020, 9, 342.
- Natesan, S.; Pandian, S.; Ponnusamy, C.; Palanichamy, R.; Muthusamy, S.; Kandasamy, R. Co-encapsulated resveratrol and quercetin in chitosan and peg modified chitosan nanoparticles: For efficient intra ocular pressure reduction. Int. J. Biol. Macromol. 2017, 104, 1837–1845.
- Bhatt, P.; Fnu, G.; Bhatia, D.; Shahid, A.; Sutariya, V. Nanodelivery of Resveratrol-Loaded PLGA Nanoparticles for Age-Related Macular Degeneration. AAPS PharmSciTech 2020, 21, 291.
- Saha, M.; Saha, D.R.; Ulhosna, T.; Sharker, S.M.; Shohag, M.H.; Islam, M.S.; Ray, S.K.; Rahman, G.M.S.; Reza, H.M. QbD based development of resveratrol-loaded mucoadhesive lecithin/chitosan nanoparticles for prolonged ocular drug delivery. J. Drug Deliv. Sci. Technol. 2021, 63, 102480.
- Razzacki, S.Z.; Thwar, P.K.; Yang, M.; Ugaz, V.M.; Burns, M.A. Integrated microsystems for controlled drug delivery. Adv. Drug Deliv. Rev. 2004, 56, 185–198.
- Rabanel, J.M.; Banquy, X.; Zouaoui, H.; Mokhtar, M.; Hildgen, P. Progress technology in microencapsulation methods for cell therapy. Biotechnol. Prog. 2009, 25, 946–963.
- Lengyel, M.; Kállai-Szabó, N.; Antal, V.; Laki, A.J.; Antal, I. Microparticles, Microspheres, and Microcapsules for Advanced Drug Delivery. Sci. Pharm. 2019, 87, 20.
- Kalaycioglu, G.D.; Aydogan, N. Layer-by-layer coated microcapsules with lipid nanodomains for dual-drug delivery. Colloids Surf. A Physicochem. Eng. Asp. 2020, 584, 124037.
- Sun, Z.; Wu, B.; Ren, Y.; Wang, Z.; Zhao, C.X.; Hai, M.; Weitz, D.A.; Chen, D. Diverse Particle Carriers Prepared by Co-Precipitation and Phase Separation: Formation and Applications. Chempluschem 2021, 86, 49–58.
- Cano-Higuita, D.M.; Malacrida, C.R.; Telis, V.R.N. Stability of Curcumin Microencapsulated by Spray and Freeze Drying in Binary and Ternary Matrices of Maltodextrin, Gum Arabic and Modified Starch. J. Food Process. Preserv. 2015, 39, 2049–2060.
- de Moura, S.C.S.R.; Berling, C.L.; Germer, S.P.M.; Alvim, I.D.; Hubinger, M.D. Encapsulating anthocyanins from Hibiscus sabdariffa L. calyces by ionic gelation: Pigment stability during storage of microparticles. Food Chem. 2018, 241, 317–327.
- Ren, X.; Hou, T.; Liang, Q.; Zhang, X.; Hu, D.; Xu, B.; Chen, X.; Chalamaiah, M.; Ma, H. Effects of frequency ultrasound on the properties of zein-chitosan complex coacervation for resveratrol encapsulation. Food Chem. 2019, 279, 223–230.
- Wang, L.; Lai, C.; Li, D.; Luo, Z.; Liu, L.; Jiang, Y.; Li, L. Lecithin-Polysaccharide Self-Assembled Microspheres for Resveratrol Delivery. Antioxidants 2022, 11, 1666.
- Gartziandia, O.; Lasa, A.; Pedraz, J.L.; Miranda, J.; Portillo, M.P.; Igartua, M.; Hernandez, R.M. Preparation and Characterization of Resveratrol Loaded Pectin/Alginate Blend Gastro-Resistant Microparticles. Molecules 2018, 23, 1886.
- Ruginǎ, D.; Ghiman, R.; Focșan, M.; Tăbăran, F.; Copaciu, F.; Suciu, M.; Pintea, A.; Aștilean, S. Resveratrol-delivery vehicle with anti-VEGF activity carried to human retinal pigmented epithelial cells exposed to high-glucose induced conditions. Colloids Surf. B Biointerfaces 2019, 181, 66–75.
- Stoia, D.; Pop, R.; Campu, A.; Nistor, M.; Astilean, S.; Pintea, A.; Suciu, M.; Rugina, D.; Focsan, M. Hybrid polymeric therapeutic microcarriers for thermoplasmonic-triggered release of resveratrol. Colloids Surf. B Biointerfaces 2022, 220, 112915.
- Harrison, I.P.; Spada, F. Hydrogels for Atopic Dermatitis and Wound Management: A Superior Drug Delivery Vehicle. Pharmaceutics 2018, 10, 71.
- Ulijn, R.V.; Bibi, N.; Jayawarna, V.; Thornton, P.D.; Todd, S.J.; Mart, R.J.; Smith, A.M.; Gough, J.E. Bioresponsive hydrogels. Mater. Today 2007, 10, 40–48.
- Buosi, F.S.; Alaimo, A.; Di Santo, M.C.; Elías, F.; García Liñares, G.; Acebedo, S.L.; Castañeda Cataña, M.A.; Spagnuolo, C.C.; Lizarraga, L.; Martínez, K.D.; et al. Resveratrol encapsulation in high molecular weight chitosan-based nanogels for applications in ocular treatments: Impact on human ARPE-19 culture cells. Int. J. Biol. Macromol. 2020, 165, 804–821.
