Neurodegenerative diseases (NDs) commonly present misfolded and aggregated proteins. Considerable research has been performed to unearth the molecular processes underpinning this pathological aggregation and develop therapeutic strategies targeting NDs. Fibrillary deposits of α-synuclein (α-Syn), a highly conserved and thermostable protein, are a critical feature in the development of NDs such as Alzheimer’s disease (AD), Lewy body disease (LBD), Parkinson’s disease (PD), and multiple system atrophy (MSA). Inhibition of α-Syn aggregation can thus serve as a potential approach for therapeutic intervention. The degradation of target proteins by small molecules has emerged as a new therapeutic modality, gaining the hotspot in pharmaceutical research. Additionally, interest is growing in the use of food-derived bioactive compounds as intervention agents against NDs via functional foods and dietary supplements. According to reports, dietary bioactive phospholipids may have cognition-enhancing and neuroprotective effects, owing to their abilities to influence cognition and mental health in vivo and in vitro.
- lysophospholipids
- neurodegenerative diseases
1. Overview
2. Effects of LPLs on Cognitive Decline
2.1. Dietary Sources of LPLs
2.2. Cognitive Function and LPLs
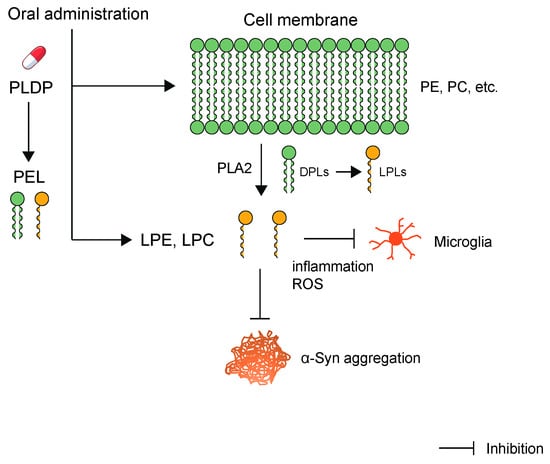
2.3. PLDP and LPLs
This entry is adapted from the peer-reviewed paper 10.3390/biomedicines10123126
References
- Bouvier-Müller, A.; Ducongé, F. Nucleic acid aptamers for neurodegenerative diseases. Biochimie 2018, 145, 73–83.
- Calabrese, G.; Molzahn, C.; Mayor, T. Protein interaction networks in neurodegenerative diseases: From physiological function to aggregation. J. Biol. Chem. 2022, 298, 102062.
- Moussaud, S.; Jones, D.R.; Moussaud-Lamodière, E.L.; Delenclos, M.; Ross, O.A.; McLean, P.J. Alpha-synuclein and tau: Teammates in neurodegeneration? Mol. Neurodegener. 2014, 9, 43.
- Cohen, A.S.; Calkins, E. Electron Microscopic Observations on a Fibrous Component in Amyloid of Diverse Origins. Nature 1959, 183, 1202–1203.
- Merker, H.-J.; Shibolet, S.; Sohar, E.; Gafni, J.; Heller, H. Periodic Cross-banding in Amyloid Filaments. Nature 1966, 211, 1401–1402.
- Seidler, P.M.; Boyer, D.R.; Rodriguez, J.A.; Sawaya, M.R.; Cascio, D.; Murray, K.; Gonen, T.; Eisenberg, D.S. Structure-based inhibitors of tau aggregation. Nat. Chem. 2018, 10, 170–176.
- Griner, S.L.; Seidler, P.; Bowler, J.; A Murray, K.; Yang, T.P.; Sahay, S.; Sawaya, M.R.; Cascio, D.; A Rodriguez, J.; Philipp, S.; et al. Structure-based inhibitors of amyloid beta core suggest a common interface with tau. eLife 2019, 8, e46924.
- Balasco, N.; Diaferia, C.; Morelli, G.; Vitagliano, L.; Accardo, A. Amyloid-Like Aggregation in Diseases and Biomaterials: Osmosis of Structural Information. Front. Bioeng. Biotechnol. 2021, 9, 641372.
- Soto, C.; Pritzkow, S. Protein misfolding, aggregation, and conformational strains in neurodegenerative diseases. Nat. Neurosci. 2018, 21, 1332–1340.
- Wells, C.; Brennan, S.; Keon, M.; Ooi, L. The role of amyloid oligomers in neurodegenerative pathologies. Int. J. Biol. Macromol. 2021, 181, 582–604.
- Currais, A.; Fischer, W.; Maher, P.; Schubert, D. Intraneuronal protein aggregation as a trigger for inflammation and neurodegeneration in the aging brain. FASEB J. 2017, 31, 5–10.
- Van Den Berge, N.; Ferreira, N.; Mikkelsen, T.W.; Alstrup, A.K.O.; Tamgüney, G.; Karlsson, P.; Terkelsen, A.J.; Nyengaard, J.R.; Jensen, P.H.; Borghammer, P. Ageing promotes pathological alpha-synuclein propagation and autonomic dysfunction in wild-type rats. Brain 2021, 144, 1853–1868.
- Su, X.; Maguire-Zeiss, K.A.; Giuliano, R.; Prifti, L.; Venkatesh, K.; Federoff, H.J. Synuclein activates microglia in a model of Parkinson’s disease. Neurobiol. Aging 2008, 29, 1690–1701.
- Zhang, W.; Wang, T.; Pei, Z.; Miller, D.S.; Wu, X.; Block, M.L.; Wilson, B.; Zhang, W.; Zhou, Y.; Hong, J.-S.; et al. Aggregated α-synuclein activates microglia: A process leading to disease progression in Parkinson’s disease. FASEB J. 2005, 19, 533–542.
- Chou, T.-W.; Chang, N.P.; Krishnagiri, M.; Patel, A.P.; Lindman, M.; Angel, J.P.; Kung, P.-L.; Atkins, C.; Daniels, B.P. Fibrillar α-synuclein induces neurotoxic astrocyte activation via RIP kinase signaling and NF-κB. Cell Death Dis. 2021, 12, 756.
- Minter, M.R.; Taylor, J.M.; Crack, P.J. The contribution of neuroinflammation to amyloid toxicity in Alzheimer’s disease. J. Neurochem. 2016, 136, 457–474.
- Tsukahara, T.; Sahara, Y.; Ribeiro, N.; Tsukahara, R.; Gotoh, M.; Sakamoto, S.; Handa, H.; Murakami-Murofushi, K. Adenine nucleotide translocase 2, a putative target protein for 2-carba cyclic phosphatidic acid in microglial cells. Cell. Signal. 2021, 82, 109951.
- Swerdlow, R.H. Mitochondria and Mitochondrial Cascades in Alzheimer’s Disease. J. Alzheimer’s Dis. 2018, 62, 1403–1416.
- Capiralla, H.; Vingtdeux, V.; Zhao, H.; Sankowski, R.; Al-Abed, Y.; Davies, P.; Marambaud, P. Resveratrol mitigates lipopolysaccharide- and Aβ-mediated microglial inflammation by inhibiting the TLR4/NF-κB/STAT signaling cascade. J. Neurochem. 2012, 120, 461–472.
- Qi, Y.; Shang, L.; Liao, Z.; Su, H.; Jing, H.; Wu, B.; Bi, K.; Jia, Y. Intracerebroventricular injection of resveratrol ameliorated Aβ-induced learning and cognitive decline in mice. Metab. Brain Dis. 2019, 34, 257–266.
- Huang, J.; Huang, N.; Xu, S.; Luo, Y.; Li, Y.; Jin, H.; Yu, C.; Shi, J.; Jin, F. Signaling mechanisms underlying inhibition of neuroinflammation by resveratrol in neurodegenerative diseases. J. Nutr. Biochem. 2021, 88, 108552.
- Matsuda, Y.; Haniu, H.; Tsukahara, T.; Uemura, T.; Inoue, T.; Sako, K.-I.; Kojima, J.; Mori, T.; Sato, K. Oral administration of porcine liver decomposition product for 4 weeks enhances visual memory and delayed recall in healthy adults over 40 years of age: A randomized, double-blind, placebo-controlled study. Exp. Gerontol. 2020, 141, 111064.
- Takamori, S.; Holt, M.; Stenius, K.; Lemke, E.A.; Grønborg, M.; Riedel, D.; Urlaub, H.; Schenck, S.; Brügger, B.; Ringler, P.; et al. Molecular Anatomy of a Trafficking Organelle. Cell 2006, 127, 831–846.
- Tsukahara, T.; Haniu, H.; Uemura, T.; Matsuda, Y. Therapeutic Potential of Porcine Liver Decomposition Product: New Insights and Perspectives for Microglia-Mediated Neuroinflammation in Neurodegenerative Diseases. Biomedicines 2020, 8, 446.
- Tsukahara, T.; Hara, H.; Haniu, H.; Matsuda, Y. The Combined Effects of Lysophospholipids against Lipopolysaccharide-induced Inflammation and Oxidative Stress in Microglial Cells. J. Oleo Sci. 2021, 70, 947–954.
- Grzelczyk, A.; Gendaszewska-Darmach, E. Novel bioactive glycerol-based lysophospholipids: New data—New insight into their function. Biochimie 2013, 95, 667–679.
- Hao, Y.; Guo, M.; Feng, Y.; Dong, Q.; Cui, M. Lysophospholipids and Their G-Coupled Protein Signaling in Alzheimer’s Disease: From Physiological Performance to Pathological Impairment. Front. Mol. Neurosci. 2020, 13, 58.
- Rosell-Valle, C.; Pedraza, C.; Manuel, I.; Moreno-Rodríguez, M.; Rodríguez-Puertas, R.; Castilla-Ortega, E.; Caramés, J.M.; Conde, A.I.G.; Zambrana-Infantes, E.; Ortega-Pinazo, J.; et al. Chronic central modulation of LPA/LPA receptors-signaling pathway in the mouse brain regulates cognition, emotion, and hippocampal neurogenesis. Prog. Neuro-Psychopharmacol. Biol. Psychiatry 2021, 108, 110156.
- Birgbauer, E. Lysophosphatidic Acid Signalling in Nervous System Development and Function. NeuroMolecular Med. 2021, 23, 68–85.
- Shah, A.K.M.A.; Nagao, T.; Kurihara, H.; Takahashi, K. Production of a Health-Beneficial Food Emulsifier by Enzymatic Partial Hydrolysis of Phospholipids Obtained from the Head of Autumn Chum Salmon. J. Oleo Sci. 2017, 66, 147–155.
- D’Arrigo, P.; Servi, S. Synthesis of Lysophospholipids. Molecules 2010, 15, 1354–1377.
- Zhang, M.; Bai, H.; Zhao, Y.; Wang, R.; Li, G.; Zhang, G.; Zhang, Y. Effects of Dietary Lysophospholipid Inclusion on the Growth Performance, Nutrient Digestibility, Nitrogen Utilization, and Blood Metabolites of Finishing Beef Cattle. Antioxidants 2022, 11, 1486.
- Zhao, P.; Li, H.; Hossain, M.; Kim, I. Effect of emulsifier (lysophospholipids) on growth performance, nutrient digestibility and blood profile in weanling pigs. Anim. Feed Sci. Technol. 2015, 207, 190–195.
- Hui, D.Y. Intestinal phospholipid and lysophospholipid metabolism in cardiometabolic disease. Curr. Opin. Infect. Dis. 2016, 27, 507–512.
- Takatera, A.; Takeuchi, A.; Saiki, K.; Morisawa, T.; Yokoyama, N.; Matsuo, M. Quantification of lysophosphatidylcholines and phosphatidylcholines using liquid chromatography–tandem mass spectrometry in neonatal serum. J. Chromatogr. B 2006, 838, 31–36.
- Guillermo, R.B.; Yang, P.; Vickers, M.H.; McJarrow, P.; Guan, J. Supplementation with complex milk lipids during brain development promotes neuroplasticity without altering myelination or vascular density. Food Nutr. Res. 2015, 59, 25765.
- Senior, S.L.; Ninkina, N.; Deacon, R.; Bannerman, D.; Buchman, V.L.; Cragg, S.J.; Wade-Martins, R. Increased striatal dopamine release and hyperdopaminergic-like behaviour in mice lacking both alpha-synuclein and gamma-synuclein. Eur. J. Neurosci. 2008, 27, 947–957.
- Petersen, R.C.; Smith, G.E.; Waring, S.C.; Ivnik, R.J.; Tangalos, E.G.; Kokmen, E. Mild Cognitive Impairment: Clinical characterization and outcome. Arch. Neurol. 1999, 56, 303–308.
- Petersen, R.C. Early Diagnosis of Alzheimers Disease: Is MCI Too Late? Curr. Alzheimer Res. 2009, 6, 324–330.
- Foltynie, T.; Brayne, C.R.; Robbins, T.W.; Barker, R.A. The cognitive ability of an incident cohort of Parkinson’s patients in the UK. The CamPaIGN study. Brain 2004, 127, 550–560.
- Muslimović, D.; Post, B.; Speelman, J.D.; Schmand, B. Cognitive profile of patients with newly diagnosed Parkinson disease. Neurology 2005, 65, 1239–1245.
- Hassin-Baer, S.; Cohen, O.S.; Israeli-Korn, S.; Yahalom, G.; Benizri, S.; Sand, D.; Issachar, G.; Geva, A.B.; Shani-Hershkovich, R.; Peremen, Z. Identification of an early-stage Parkinson’s disease neuromarker using event-related potentials, brain network analytics and machine-learning. PLoS ONE 2022, 17, e0261947.
- Facal, D.; Spuch, C.; Valladares-Rodriguez, S. New Trends in Cognitive Aging and Mild Cognitive Impairment. Geriatrics 2022, 7, 80.
- Campbell, N.L.; Unverzagt, F.; LaMantia, M.A.; Khan, B.A.; Boustani, M.A. Risk Factors for the Progression of Mild Cognitive Impairment to Dementia. Clin. Geriatr. Med. 2013, 29, 873–893.
- Godos, J.; Currenti, W.; Angelino, D.; Mena, P.; Castellano, S.; Caraci, F.; Galvano, F.; Del Rio, D.; Ferri, R.; Grosso, G. Diet and Mental Health: Review of the Recent Updates on Molecular Mechanisms. Antioxidants 2020, 9, 346.
- Caruso, G.; Godos, J.; Privitera, A.; Lanza, G.; Castellano, S.; Chillemi, A.; Bruni, O.; Ferri, R.; Caraci, F.; Grosso, G. Phenolic Acids and Prevention of Cognitive Decline: Polyphenols with a Neuroprotective Role in Cognitive Disorders and Alzheimer’s Disease. Nutrients 2022, 14, 819.
- Guiné, R.P.F.; Florença, S.G.; Barroca, M.J.; Anjos, O. The Link between the Consumer and the Innovations in Food Product Development. Foods 2020, 9, 1317.
- Gómez-Pinilla, F. Brain foods: The effects of nutrients on brain function. Nat. Rev. Neurosci. 2008, 9, 568–578.
- Chianese, R.; Coccurello, R.; Viggiano, A.; Scafuro, M.; Fiore, M.; Coppola, G.; Operto, F.F.; Fasano, S.; Laye, S.; Pierantoni, R.; et al. Impact of Dietary Fats on Brain Functions. Curr. Neuropharmacol. 2018, 16, 1059–1085.
- Dadhania, V.P.; Trivedi, P.P.; Vikram, A.; Tripathi, D.N. Nutraceuticals against Neurodegeneration: A Mechanistic Insight. Curr. Neuropharmacol. 2016, 14, 627–640.
- Yuyama, K.; Igarashi, Y. Linking glycosphingolipids to Alzheimer’s amyloid-ß: Extracellular vesicles and functional plant materials. Glycoconj. J. 2022, 39, 613–618.
- Yuyama, K.; Takahashi, K.; Usuki, S.; Mikami, D.; Sun, H.; Hanamatsu, H.; Furukawa, J.; Mukai, K.; Igarashi, Y. Plant sphingolipids promote extracellular vesicle release and alleviate amyloid-β pathologies in a mouse model of Alzheimer’s disease. Sci. Rep. 2019, 9, 16827.
- Ozawa, H.; Miyazawa, T.; Miyazawa, T. Effects of Dietary Food Components on Cognitive Functions in Older Adults. Nutrients 2021, 13, 2804.
- Kinney, J.W.; BeMiller, S.M.; Murtishaw, A.S.; Leisgang, A.M.; Salazar, A.M.; Lamb, B.T. Inflammation as a central mechanism in Alzheimer’s disease. Alzheimer’s Dement. 2018, 4, 575–590.
- Garcia, P.; Jürgens-Wemheuer, W.; Huarte, O.U.; Michelucci, A.; Masuch, A.; Brioschi, S.; Weihofen, A.; Koncina, E.; Coowar, D.; Heurtaux, T.; et al. Neurodegeneration and neuroinflammation are linked, but independent of alpha-synuclein inclusions, in a seeding/spreading mouse model of Parkinson’s disease. Glia 2022, 70, 935–960.
- Hammond, T.R.; Marsh, S.E.; Stevens, B. Immune Signaling in Neurodegeneration. Immunity 2019, 50, 955–974.
- Crotti, A.; Ransohoff, R.M. Microglial Physiology and Pathophysiology: Insights from Genome-wide Transcriptional Profiling. Immunity 2016, 44, 505–515.
- Guzman-Martinez, L.; Maccioni, R.B.; Andrade, V.; Navarrete, L.P.; Pastor, M.G.; Ramos-Escobar, N. Neuroinflammation as a Common Feature of Neurodegenerative Disorders. Front. Pharmacol. 2019, 10, 1008.
- Janvin, C.C.; Larsen, J.P.; Aarsland, D.; Hugdahl, K. Subtypes of mild cognitive impairment in parkinson’s disease: Progression to dementia. Mov. Disord. 2006, 21, 1343–1349.
- Kamioka, H.; Tsutani, K.; Origasa, H.; Yoshizaki, T.; Kitayuguchi, J.; Shimada, M.; Wada, Y.; Takano-Ohmuro, H. Quality of Systematic Reviews of the Foods with Function Claims in Japan: Comparative Before- and After-Evaluation of Verification Reports by the Consumer Affairs Agency. Nutrients 2019, 11, 1583.
- Alqahtani, M.S.; Kazi, M.; Alsenaidy, M.A.; Ahmad, M.Z. Advances in Oral Drug Delivery. Front. Pharmacol. 2021, 12, 618411.
- Tsukahara, T.; Haniu, H.; Uemura, T.; Matsuda, Y. Porcine liver decomposition product-derived lysophospholipids promote microglial activation in vitro. Sci. Rep. 2020, 10, 3748.
- Gotoh, M.; Sano-Maeda, K.; Murofushi, H.; Murakami-Murofushi, K. Protection of Neuroblastoma Neuro2A Cells from Hypoxia-Induced Apoptosis by Cyclic Phosphatidic Acid (cPA). PLoS ONE 2012, 7, e51093.
- Sheikh, A.M.; Michikawa, M.; Kim, S.; Nagai, A. Lysophosphatidylcholine increases the neurotoxicity of Alzheimer’s amyloid β1-42 peptide: Role of oligomer formation. Neuroscience 2015, 292, 159–169.
- Mulder, C.; Wahlund, L.-O.; Teerlink, T.; Blomberg, M.; Veerhuis, R.; Van Kamp, G.J.; Scheltens, P.; Scheffer, P.G. Decreased lysophosphatidylcholine/phosphatidylcholine ratio in cerebrospinal fluid in Alzheimer’s disease. J. Neural Transm. 2003, 110, 949–955.
- Sugasini, D.; Thomas, R.; Yalagala, P.C.R.; Tai, L.M.; Subbaiah, P.V. Dietary docosahexaenoic acid (DHA) as lysophosphatidylcholine, but not as free acid, enriches brain DHA and improves memory in adult mice. Sci. Rep. 2017, 7, 11263.
