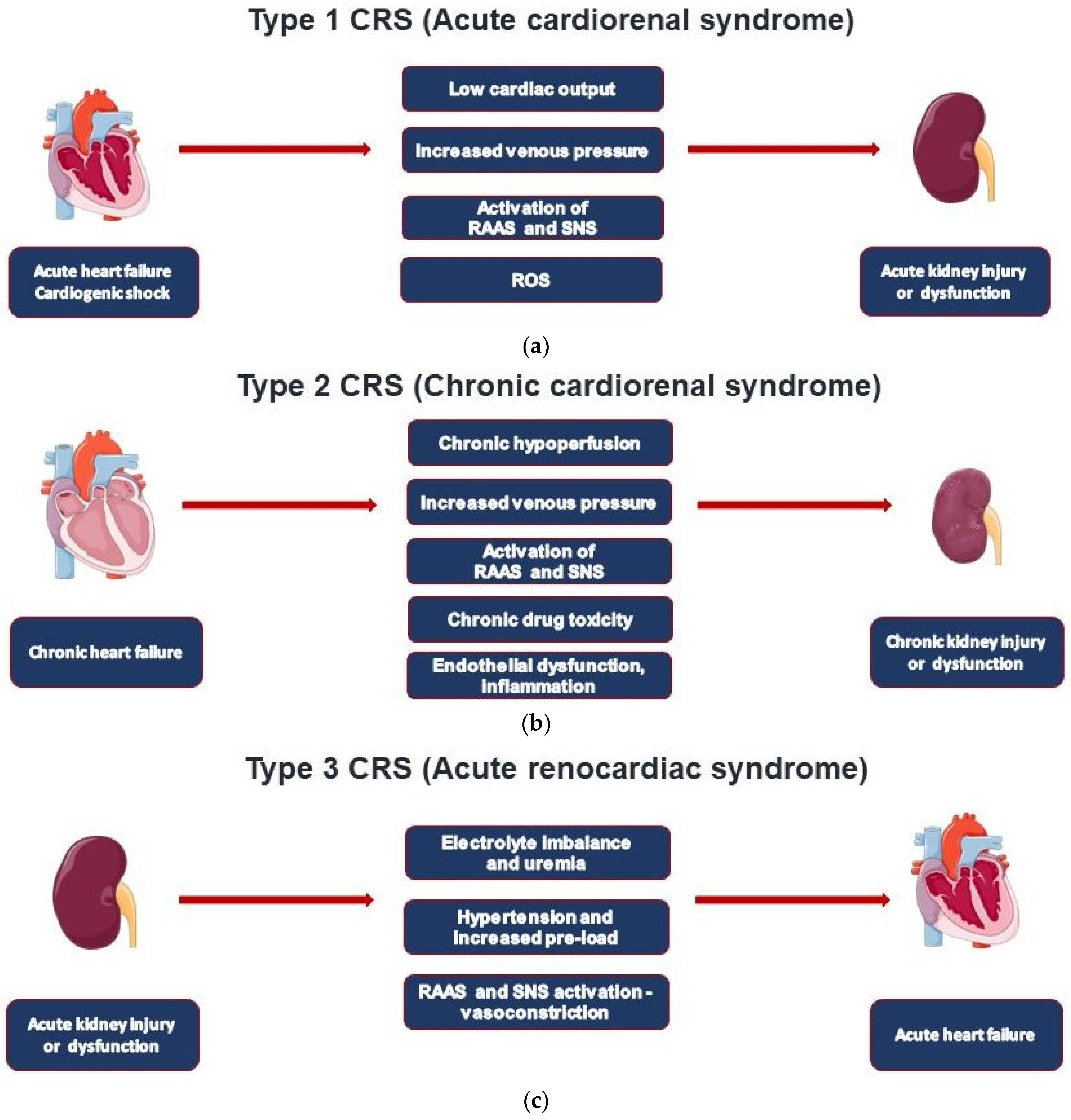
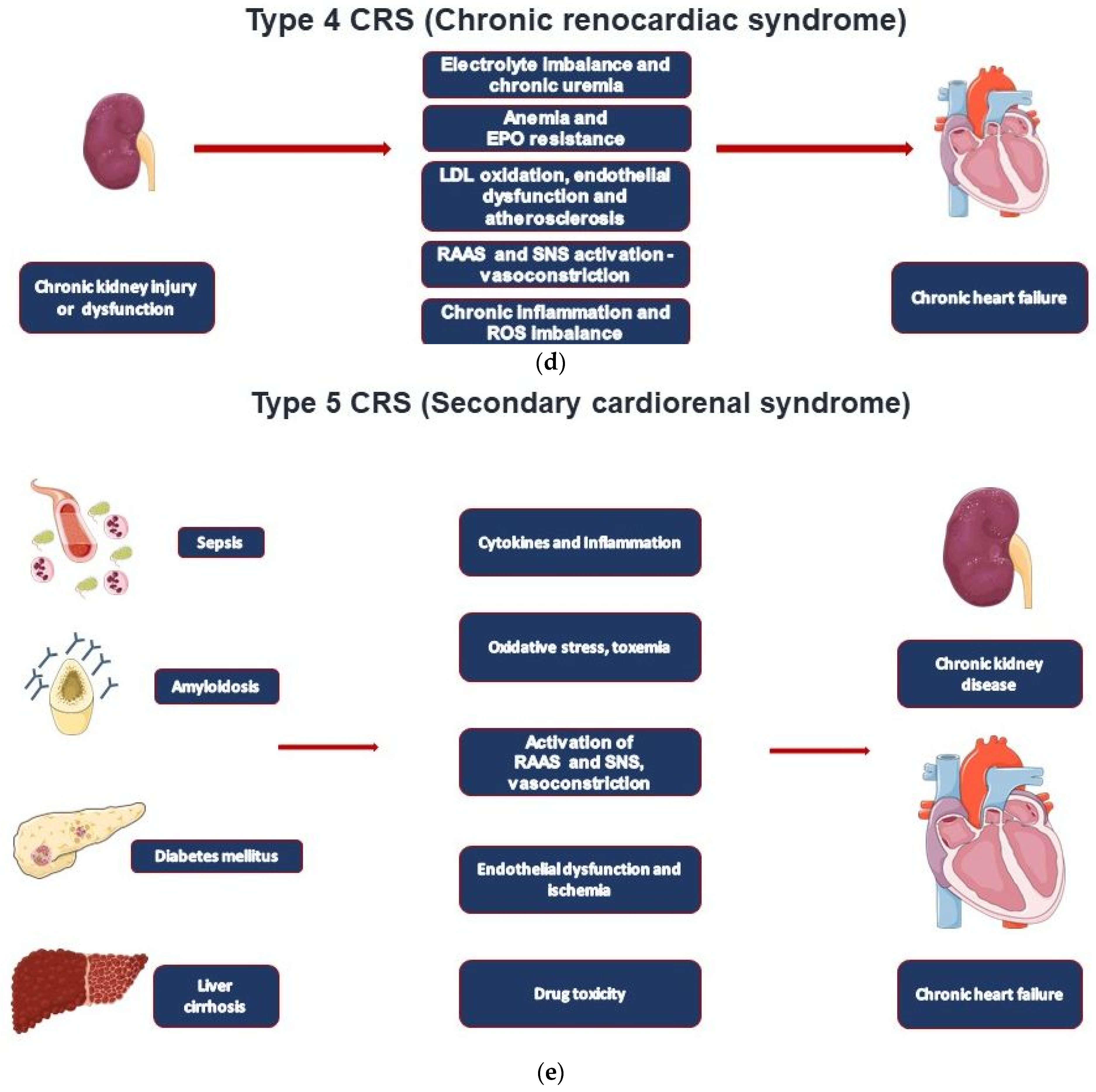
In cardiorenal syndrome (CRS), heart failure and renal failure are pathophysiologically closely intertwined by the reciprocal relationship between cardiac and renal injury. Type 1 CRS is most common and associated with acute heart failure. A preexistent chronic kidney disease (CKD) is common and contributes to acute kidney injury (AKI) in CRS type 1 patients (acute cardiorenal syndrome). The remaining CRS types are found in patients with chronic heart failure (type 2), acute and chronic kidney diseases (types 3 and 4), and systemic diseases that affect both the heart and the kidney (type 5). Establishing the diagnosis of CRS requires various tools based on the type of CRS, including non-invasive imaging modalities such as TTE, CT, and MRI, adjuvant volume measurement techniques, invasive hemodynamic monitoring, and biomarkers.
- cardiorenal syndrome
- heart failure
- Epidemiological Data
1. Introduction
| CRS Types | Mechanisms | Clinical Conditions |
|---|---|---|
| Type 1—Acute cardiorenal syndrome | AHF leading to AKI | AHF, ACS, cardiogenic shock |
| Type 2—Chronic cardiorenal syndrome | CHF leading to CKD | CHF regardless of cause |
| Type 3—Acute renocardiac syndrome | AKI leading to AHF | Volume overload, uremic metabolic disturbances, and inflammatory eruption |
| Type 4—Chronic renocardiac syndrome | CKD leading to CHF | CKD-induced cardiomyopathy resulting in cardiac remodeling and heart failure |
| Type 5—Secondary cardiorenal syndrome | Systemic disorder leading to cardiorenal dysfunction | Sepsis, diabetes, liver cirrhosis, amyloidosis, M. Fabry |
2. CRS Types and Epidemiological Data
This entry is adapted from the peer-reviewed paper 10.3390/jcm11237041
References
- Vaidya, V.S.; Ramirez, V.; Ichimura, T.; Bobadilla, N.A.; Bonventre, J.V. Urinary kidney injury molecule-1: A sensitive quantitative biomarker for early detection of kidney tubular injury. Am. J. Physiol. Renal. Physiol. 2006, 290, F517–F529.
- Parikh, C.R.; Abraham, E.; Ancukiewicz, M.; Edelstein, C.L. Urine IL-18 is an early diagnostic marker for acute kidney injury and predicts mortality in the intensive care unit. J. Am. Soc. Nephrol. 2005, 16, 3046–3052.
- Liangos, O.; Perianayagam, M.C.; Vaidya, V.S.; Han, W.K.; Wald, R.; Tighiouart, H.; MacKinnon, R.W.; Li, L.; Balakrishnan, V.S.; Pereira, B.J.; et al. Urinary N-acetyl-beta-(D)-glucosaminidase activity and kidney injury molecule-1 level are associated with adverse outcomes in acute renal failure. J. Am. Soc. Nephrol. 2007, 18, 904–912.
- Kjeldsen, L.; Johnsen, A.H.; Sengelov, H.; Borregaard, N. Isolation and primary structure of NGAL, a novel protein associated with human neutrophil gelatinase. J. Biol. Chem. 1993, 268, 10425–10432.
- Mishra, J.; Dent, C.; Tarabishi, R.; Mitsnefes, M.M.; Ma, Q.; Kelly, C.; Ruff, S.M.; Zahedi, K.; Shao, M.; Bean, J.; et al. Neutrophil gelatinase-associated lipocalin (NGAL) as a biomarker for acute renal injury after cardiac surgery. Lancet 2005, 365, 1231–1238.
- Haase, M.; Bellomo, R.; Devarajan, P.; Schlattmann, P.; Haase-Fielitz, A.; NGAL Meta-Analysis Investigator Group. Accuracy of neutrophil gelatinase-associated lipocalin (NGAL) in diagnosis and prognosis in acute kidney injury: A systematic review and meta-analysis. Am. J. Kidney Dis. 2009, 54, 1012–1024.
- Maisel, A.S.; Mueller, C.; Fitzgerald, R.; Brikhan, R.; Hiestand, B.C.; Iqbal, N.; Clopton, P.; van Veldhuisen, D.J. Prognostic utility of plasma neutrophil gelatinase-associated lipocalin in patients with acute heart failure: The NGAL EvaLuation Along with B-type NaTriuretic Peptide in acutely decompensated heart failure (GALLANT) trial. Eur. J. Heart Fail. 2011, 13, 846–851.
- Noiri, E.; Doi, K.; Negishi, K.; Tanaka, T.; Hamasaki, Y.; Fujita, T.; Portilla, D.; Sugaya, T. Urinary fatty acid-binding protein 1: An early predictive biomarker of kidney injury. Am. J. Physiol. Renal. Physiol. 2009, 296, F669–F679.
- Niizeki, T.; Takeishi, Y.; Arimoto, T.; Nozaki, N.; Hirono, O.; Watanabe, T.; Nitobe, J.; Miyashita, T.; Miyamoto, T.; Koyama, Y.; et al. Persistently increased serum concentration of heart-type fatty acid-binding protein predicts adverse clinical outcomes in patients with chronic heart failure. Circ. J. 2008, 72, 109–114.
- Kashani, K.; Al-Khafaji, A.; Ardiles, T.; Artigas, A.; Bagshaw, S.M.; Bell, M.; Bihorac, A.; Birkhahn, R.; Cely, C.M.; Chawla, L.S.; et al. Discovery and validation of cell cycle arrest biomarkers in human acute kidney injury. Crit. Care 2013, 17, R25.
- Zarbock, A.; Kullmar, M.; Ostermann, M.; Lucchese, G.; Baig, K.; Cennamo, A.; Rajani, R.; McCorkell, S.; Arndt, C.; Wulf, H.; et al. Prevention of Cardiac Surgery-Associated Acute Kidney Injury by Implementing the KDIGO Guidelines in High-Risk Patients Identified by Biomarkers: The PrevAKI-Multicenter Randomized Controlled Trial. Anesth. Analg. 2021, 133, 292–302.
- Ronco, C.; Haapio, M.; House, A.A.; Anavekar, N.; Bellomo, R. Cardiorenal syndrome. J. Am. Coll. Cardiol. 2008, 52, 1527–1539.
- Ronco, C.; Di Lullo, L. Cardiorenal syndrome. Heart Fail. Clin. 2014, 10, 251–280.
- Patel, K.P.; Katsurada, K.; Zheng, H. Cardiorenal Syndrome: The Role of Neural Connections Between the Heart and the Kidneys. Circ. Res. 2022, 130, 1601–1617.
- Bright, R. Cases and Observations Illustrative of Renal Disease, Accompanied with the Secretion of Albuminous Urine. Med. Chir. Rev. 1836, 25, 23–35.
- U.S. Department of Health & Human Services. Cardio-Renal Connections in Heart Failure and Cardiovascular Disease. 2004. Available online: https://www.nhlbi.nih.gov/events/2004/cardio-renal-connections-heart-failure-and-cardiovascular-disease (accessed on 25 March 2022).
- Ronco, C.; McCullough, P.; Anker, S.D.; Anand, I.; Aspromonte, N.; Bagshaw, S.M.; Bellomo, R.; Berl, T.; Bobek, I.; Cruz, D.N.; et al. Acute Dialysis Quality Initiative consensus g. Cardio-renal syndromes: Report from the consensus conference of the acute dialysis quality initiative. Eur. Heart J. 2010, 31, 703–711.
- George, S.M.; Kalantarinia, K. The role of imaging in the management of cardiorenal syndrome. Int. J. Nephrol. 2011, 2011, 245241.
- Padeletti, L.; Innocenti, L.; Paoletti Perini, A.; Gronda, E. Arrhythmic complication in cardiorenal syndrome. Heart Fail. Rev. 2011, 16, 569–573.
- Lin, L.; Chen, Y.; Han, D.; Yang, A.; Wang, A.Y.; Qi, W. Cardiorenal Syndrome in COVID-19 Patients: A Systematic Review. Front. Cardiovasc. Med. 2022, 9, 915533.
- Uduman, J. Epidemiology of Cardiorenal Syndrome. Adv. Chronic Kidney Dis. 2018, 25, 391–399.
