Diabetic ketoacidosis (DKA) is characterized by uncontrolled hyperglycemia (>250 mg/dL), metabolic acidosis, and increased ketone concentration. DKA incidence increased at annual rate of 6.3% most notably in persons aged <45 years with significant increase in hospitalization rate between 2008 and 2018. Nevertheless, overall in-hospital mortality rates declined during the study period from 1.1% to 0.4%. DKA is a life-threatening, but preventable complication of type-1 diabetes with a thirty percent of the cases that could be attributed to type-2 diabetes. The major clinical difference between these two types of diabetes is a lesser degree of acidosis and absence of hyperkalemia in type 2 diabetes.
- diabetes mellitus
- DKA
- fluid management
- ketoacidosis
- euglycemic DKA
- special population
- SGLT2 inhibitors
1. Diabetic Ketoacidosis (DKA) in Pregnancy
2. DKA in Renal Replacement
3. DKA in Acute Pancreatitis and Islets Transplants
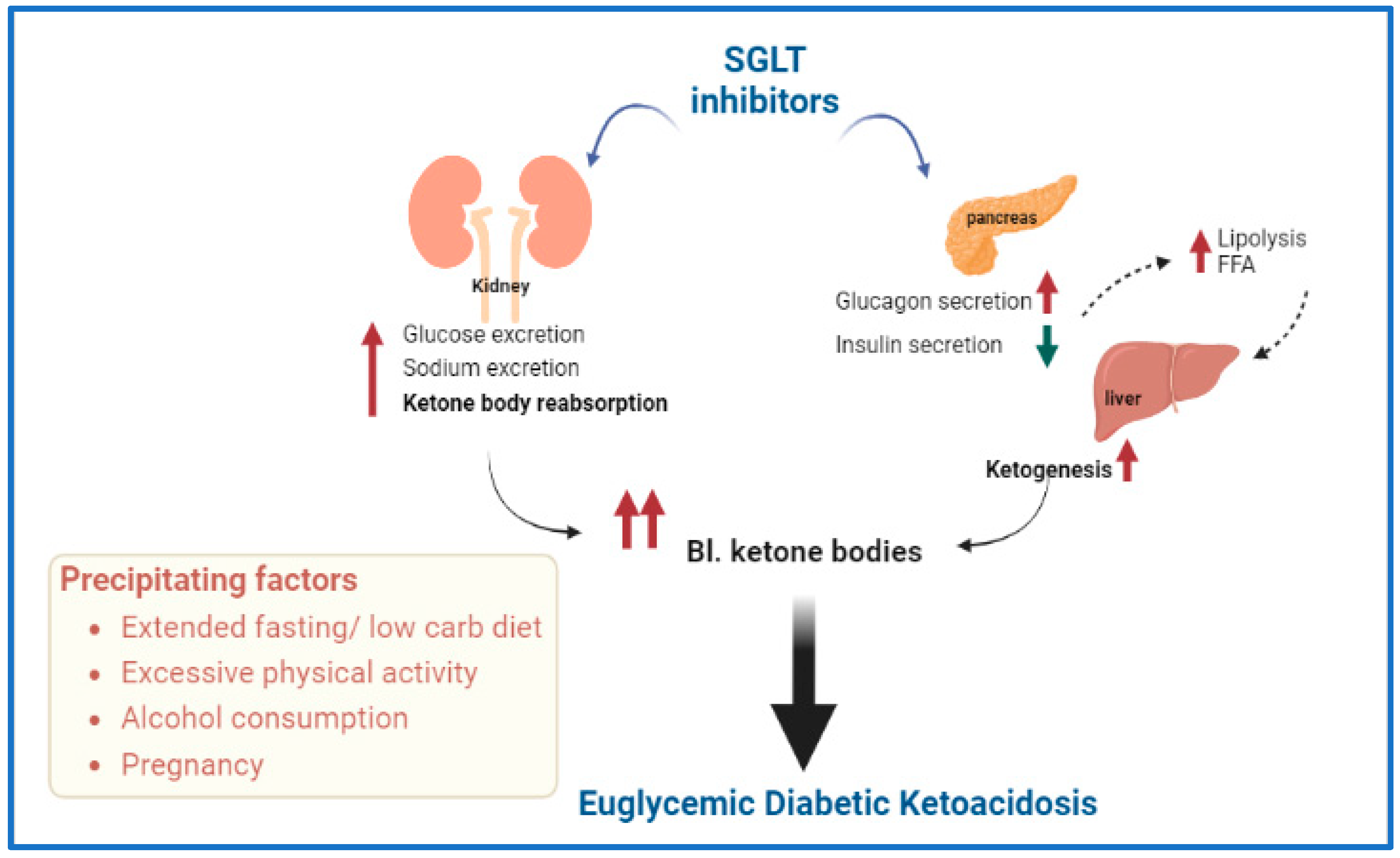
4. DKA and Sodium–Glucose Cotransporter (SGLT) Inhibitors
5. DKA in Patients with Congestive Heart Failure (CHF)
6. DKA and Insulin Pump Users
7. DKA in Patients with COVID-19
This entry is adapted from the peer-reviewed paper 10.3390/endocrines3040066
References
- Sibai, B.M.; Viteri, O.A. Diabetic ketoacidosis in pregnancy. Obstet. Gynecol. 2014, 123, 167–178.
- Diguisto, C.; Strachan, M.W.J.; Churchill, D.; Ayman, G.; Knight, M. A study of diabetic ketoacidosis in the pregnant population in the United Kingdom: Investigating the incidence, aetiology, management and outcomes. Diabet. Med. 2022, 39, e14743.
- ohan, M.; Baagar, K.A.M.; Lindow, S. Management of diabetic ketoacidosis in pregnancy. Obstet. Gynaecol. 2017, 19, 55–62.
- Koye, D.N.; Magliano, D.J.; Nelson, R.G.; Pavkov, M.E. The Global Epidemiology of Diabetes and Kidney Disease. Adv. Chronic Kidney Dis. 2018, 25, 121–132.
- Seddik, A.A.; Bashier, A.; Alhadari, A.K.; AlAlawi, F.; Alnour, H.H.; Bin Hussain, A.A.; Frankel, A.; Railey, M.J. Challenges in management of diabetic ketoacidosis in hemodialysis patients, case presentation and review of literature. Diabetes Metab. Syndr. 2019, 13, 2481–2487.
- Tzamaloukas, A.H.; Ing, T.S.; Siamopoulos, K.C.; Rohrscheib, M.; Elisaf, M.S.; Raj, D.S.; Murata, G.H. Body fluid abnormalities in severe hyperglycemia in patients on chronic dialysis: Review of published reports. J. Diabetes Its Complicat. 2008, 22, 29–37.
- Galindo, R.J.; Pasquel, F.J.; Fayfman, M.; Tsegka, K.; Dhruv, N.; Cardona, S.; Wang, H.; Vellanki, P.; Umpierrez, G.E. Clinical characteristics and outcomes of patients with end-stage renal disease hospitalized with diabetes ketoacidosis. BMJ Open Diabetes Res. Care 2020, 8, e000763.
- Tzamaloukas, A.H.; Ing, T.S.; Siamopoulos, K.C.; Raj, D.S.; Elisaf, M.S.; Rohrscheib, M.; Murata, G.H. Pathophysiology and management of fluid and electrolyte disturbances in patients on chronic dialysis with severe hyperglycemia. Semin. Dial. 2008, 21, 431–439.
- Mathuram Thiyagarajan, U.; Ponnuswamy, A.; Chung, A. An enigmatic triad of acute pancreatitis, diabetic ketoacidosis and hypertriglyceridaemia: Who is the culprit? BMJ Case Rep. 2019, 12, e217272.
- Scherer, J.; Singh, V.P.; Pitchumoni, C.S.; Yadav, D. Issues in hypertriglyceridemic pancreatitis: An update. J. Clin. Gastroenterol. 2014, 48, 195–203.
- Wang, Y.; Attar, B.M.; Hinami, K.; Jaiswal, P.; Yap, J.E.; Jaiswal, R.; Devani, K.; Simons-Linares, C.R.; Demetria, M.V. Concurrent Diabetic Ketoacidosis in Hypertriglyceridemia-Induced Pancreatitis: How Does It Affect the Clinical Course and Severity Scores? Pancreas 2017, 46, 1336–1340.
- Nair, S.; Yadav, D.; Pitchumoni, C.S. Association of diabetic ketoacidosis and acute pancreatitis: Observations in 100 consecutive episodes of DKA. Am. J. Gastroenterol. 2000, 95, 2795–2800.
- Singla, A.A.; Ting, F.; Singla, A. Acute pancreatitis secondary to diabetic ketoacidosis induced hypertriglyceridemia in a young adult with undiagnosed type 2 diabetes. JOP. J. Pancreas 2015, 16, 201–204.
- Ravindran, D.; Mugunthan, R.S.R.; Shruthi, T.K.; Jayaraman, D. Severe diabetic ketoacidosis and autoimmune pancreatitis with SIRS in an adolescent with LRBA deficiency—A rare complication of a common primary immunodeficiency disease. J. Fam. Med. Prim. Care 2022, 11, 1552–1554.
- Yilmaz-Yalcin, Y.; Bascil-Tutuncu, N.; Baysan-Cebi, H.P.; Verdi, H.; Erol, S.; Kaymaz, F.F.; Atac, F.B. A Lipotoxic Medium Decreases the Number of Lipid Droplets in beta Cells: One Possible Explanation of the beta-Cell Failure in Patients with Hyperlipidemia Receiving Tacrolimus. Exp. Clin. Transplant. epub before print. 2022.
- Porrini, E.L.; Diaz, J.M.; Moreso, F.; Delgado Mallen, P.I.; Silva Torres, I.; Ibernon, M.; Bayes-Genis, B.; Benitez-Ruiz, R.; Lampreabe, I.; Lauzurrica, R.; et al. Clinical evolution of post-transplant diabetes mellitus. Nephrol. Dial. Transplant. 2016, 31, 495–505.
- Mazumder, M.A.; Gulati, S.; Narula, A.S.; Shehwar, D.; Mir, I.M. Tacrolimus-induced acute pancreatitis and diabetic ketoacidosis (DKA) in pediatric kidney transplant recipient. Pediatr. Transplant. 2022, 26, e14194.
- Rickels, M.R.; Robertson, R.P. Pancreatic Islet Transplantation in Humans: Recent Progress and Future Directions. Endocr. Rev. 2019, 40, 631–668.
- Rickels, M.R.; Stock, P.G.; de Koning, E.J.P.; Piemonti, L.; Pratschke, J.; Alejandro, R.; Bellin, M.D.; Berney, T.; Choudhary, P.; Johnson, P.R.; et al. Defining outcomes for beta-cell replacement therapy in the treatment of diabetes: A consensus report on the Igls criteria from the IPITA/EPITA opinion leaders workshop. Transpl. Int. 2018, 31, 343–352.
- Hering, B.J.; Clarke, W.R.; Bridges, N.D.; Eggerman, T.L.; Alejandro, R.; Bellin, M.D.; Chaloner, K.; Czarniecki, C.W.; Goldstein, J.S.; Hunsicker, L.G.; et al. Phase 3 Trial of Transplantation of Human Islets in Type 1 Diabetes Complicated by Severe Hypoglycemia. Diabetes Care 2016, 39, 1230–1240.
- Lyons, S.K.; Hermann, J.M.; Miller, K.M.; Hofer, S.E.; Foster, N.C.; Rami-Merhar, B.M.; Aleppo, G.; Seufert, J.; DiMeglio, L.A.; Danne, T.; et al. Use of Adjuvant Pharmacotherapy in Type 1 Diabetes: International Comparison of 49,996 Individuals in the Prospective Diabetes Follow-up and T1D Exchange Registries. Diabetes Care 2017, 40, e139–e140.
- Sarafidis, P.; Loutradis, C.; Ferro, C.J.; Ortiz, A. SGLT-2 Inhibitors to Treat Hyponatremia Associated with SIADH: A Novel Indication? Am. J. Nephrol. 2020, 51, 553–555.
- Loutradis, C.; Papadopoulou, E.; Angeloudi, E.; Karagiannis, A.; Sarafidis, P. The Beneficial Hemodynamic Actions of SGLT-2 Inhibitors beyond the Management of Hyperglycemia. Curr. Med. Chem. 2020, 27, 6682–6702.
- Refardt, J.; Imber, C.; Sailer, C.O.; Jeanloz, N.; Potasso, L.; Kutz, A.; Widmer, A.; Urwyler, S.A.; Ebrahimi, F.; Vogt, D.R.; et al. A Randomized Trial of Empagliflozin to Increase Plasma Sodium Levels in Patients with the Syndrome of Inappropriate Antidiuresis. J. Am. Soc. Nephrol. 2020, 31, 615–624.
- Yeoh, S.E.; Docherty, K.F.; Jhund, P.S.; Petrie, M.C.; Inzucchi, S.E.; Kober, L.; Kosiborod, M.N.; Martinez, F.A.; Ponikowski, P.; Sabatine, M.S.; et al. Relationship of Dapagliflozin with Serum Sodium: Findings From the DAPA-HF Trial. JACC Heart Fail. 2022, 10, 306–318.
- Dhanasekaran, M.; Narayanan, S.; Mastoris, I.; Mehta, S. Canagliflozin-associated severe hyponatremia: A rare and potentially adverse effect? Endocrinol. Diabetes Metab. Case Rep. 2022, 2022, 21-0035.
- Branco, A.; Fatima, R.; Liblik, K.; Jackson, R.; Payne, D.; El-Diasty, M. Euglycemic Diabetic Ketoacidosis Associated with Sodium-Glucose Cotransporter-2 Inhibitors After Cardiac Surgery: A Review of Current Literature. J. Cardiothorac. Vasc. Anesthesia 2022, 36, 3877–3886.
- Ogawa, W.; Sakaguchi, K. Euglycemic diabetic ketoacidosis induced by SGLT2 inhibitors: Possible mechanism and contributing factors. J. Diabetes Investig. 2016, 7, 135–138.
- Teng, R.; Kurian, M.; Close, K.L.; Buse, J.B.; Peters, A.L.; Alexander, C.M. Comparison of Protocols to Reduce Diabetic Ketoacidosis in Patients with Type 1 Diabetes Prescribed a Sodium-Glucose Cotransporter 2 Inhibitor. Diabetes Spectr. 2021, 34, 42–51.
- Goldenberg, R.M.; Gilbert, J.D.; Hramiak, I.M.; Woo, V.C.; Zinman, B. Sodium-glucose co-transporter inhibitors, their role in type 1 diabetes treatment and a risk mitigation strategy for preventing diabetic ketoacidosis: The STOP DKA Protocol. Diabetes Obes. Metab. 2019, 21, 2192–2202.
- Klocker, A.A.; Phelan, H.; Twigg, S.M.; Craig, M.E. Blood beta-hydroxybutyrate vs. urine acetoacetate testing for the prevention and management of ketoacidosis in Type 1 diabetes: A systematic review. Diabet. Med. 2013, 30, 818–824.
- Islam, T.; Sherani, K.; Surani, S.; Vakil, A. Guidelines and controversies in the management of diabetic ketoacidosis—A mini-review. World J. Diabetes 2018, 9, 226–229.
- Weissberg-Benchell, J.; Antisdel-Lomaglio, J.; Seshadri, R. Insulin pump therapy: A meta-analysis. Diabetes Care 2003, 26, 1079–1087.
- Ramirez-Rincon, A.; Hincapie-Garcia, J.; Arango, C.M.; Aristizabal, N.; Castillo, E.; Hincapie, G.; Zapata, E.; Cuesta, D.P.; Delgado, M.; Abad, V.; et al. Clinical Outcomes after 1 Year of Augmented Insulin Pump Therapy in Patients with Diabetes in a Specialized Diabetes Center in Medellin, Colombia. Diabetes Technol. Ther. 2016, 18, 713–718.
- Alshami, A.; Purewal, T.; Douedi, S.; Alazzawi, M.; Hossain, M.A.; Ong, R.; Sen, S.; Cheng, J.; Patel, S. Effect of Insulin Pump Use on Diabetic Ketoacidosis in Type 1 Diabetes Mellitus: A Matched Cohort Study. J. Clin. Med. 2021, 10, 898.
- Evans, K. Diabetic ketoacidosis: Update on management. Clin. Med. 2019, 19, 396–398.
- Chee, Y.J.; Ng, S.J.H.; Yeoh, E. Diabetic ketoacidosis precipitated by COVID-19 in a patient with newly diagnosed diabetes mellitus. Diabetes Res. Clin. Pract. 2020, 164, 108166.
- Kazakou, P.; Paschou, S.A.; Psaltopoulou, T.; Gavriatopoulou, M.; Korompoki, E.; Stefanaki, K.; Kanouta, F.; Kassi, G.N.; Dimopoulos, M.A.; Mitrakou, A. Early and late endocrine complications of COVID-19. Endocr. Connect. 2021, 10, R229–R239.
- Li, J.; Wang, X.; Chen, J.; Zuo, X.; Zhang, H.; Deng, A. COVID-19 infection may cause ketosis and ketoacidosis. Diabetes Obes. Metab. 2020, 22, 1935–1941.
- Alhumaid, S.; Al Mutair, A.; Al Alawi, Z.; Rabaan, A.A.; Alomari, M.A.; Al Salman, S.A.; Al-Alawi, A.S.; Al Hassan, M.H.; Alhamad, H.; Al-Kamees, M.A.; et al. Diabetic ketoacidosis in patients with SARS-CoV-2: A systematic review and meta-analysis. Diabetol. Metab. Syndr. 2021, 13, 120.
- Chen, X.; Zhao, B.; Qu, Y.; Chen, Y.; Xiong, J.; Feng, Y.; Men, D.; Huang, Q.; Liu, Y.; Yang, B.; et al. Detectable Serum Severe Acute Respiratory Syndrome Coronavirus 2 Viral Load (RNAemia) Is Closely Correlated with Drastically Elevated Interleukin 6 Level in Critically Ill Patients with Coronavirus Disease 2019. Clin. Infect. Dis. 2020, 71, 1937–1942.
- Stentz, F.B.; Umpierrez, G.E.; Cuervo, R.; Kitabchi, A.E. Proinflammatory cytokines, markers of cardiovascular risks, oxidative stress, and lipid peroxidation in patients with hyperglycemic crises. Diabetes 2004, 53, 2079–2086.
